Unraveling Your Painful TKR: Diagnosing Infection & Loosening
Introduction & Epidemiology
Persistent pain following total knee replacement (TKR) is a challenging clinical entity, affecting approximately 10-20% of patients. While TKR is a highly successful procedure for end-stage knee arthritis, a significant subset of patients report dissatisfaction due to ongoing pain, functional limitations, or perceived surgical failure. The differential diagnosis for a painful TKR is broad, encompassing both implant-related and non-implant-related etiologies. Key among the implant-related causes, and often the most concerning, are periprosthetic joint infection (PJI) and aseptic loosening. Distinguishing between these, and other causes, is paramount for appropriate management and often necessitates a methodical diagnostic algorithm.
Periprosthetic joint infection (PJI) is a devastating complication, with an incidence ranging from 0.5% to 2% following primary TKR, but significantly higher (up to 10-20%) in revision scenarios. It is associated with considerable morbidity, functional decline, and substantial healthcare costs. The most common causative organisms are coagulase-negative staphylococci and Staphylococcus aureus , often forming biofilms that render eradication challenging. PJI can manifest acutely (within weeks to months post-surgery) or chronically (months to years later), with varying clinical presentations.
Aseptic loosening, in contrast, refers to the failure of the implant-bone interface in the absence of infection. Its incidence varies depending on implant design, surgical technique, and patient factors, but it remains a leading cause for revision TKR, alongside PJI. Aseptic loosening typically presents years after primary arthroplasty, often due to mechanical wear of the polyethylene bearing leading to osteolysis, or direct mechanical failure of the cement or uncemented fixation interface. The biological response to wear debris (e.g., polyethylene particles) can incite an inflammatory cascade, resulting in progressive bone loss and implant micromotion. Understanding the distinct diagnostic pathways for PJI and aseptic loosening is critical, as their management strategies diverge significantly.
Surgical Anatomy & Biomechanics in the Context of Failure
A comprehensive understanding of knee arthroplasty design, surgical anatomy, and joint biomechanics is foundational when evaluating a painful TKR, particularly concerning potential infection or loosening.
Component Design and Fixation
Modern TKRs typically comprise femoral, tibial, and patellar components. The femoral component is commonly cobalt-chromium alloy, fixed with cement. The tibial component consists of a metallic tray (cobalt-chromium or titanium alloy) affixed to the resected tibia, often with a modular polyethylene bearing insert. Cemented fixation remains the gold standard for most primary TKRs, providing immediate stability. Uncemented components rely on porous coatings for biologic ingrowth, requiring initial press-fit stability. Hybrid fixation, with a cemented femoral component and uncemented tibial component, is also employed.
Loosening can occur at any component interface:
*
Femoral Component:
Loosening often manifests as radiolucent lines at the cement-bone or cement-implant interface, particularly in stress-bearing areas.
*
Tibial Component:
Tibial loosening is more common than femoral loosening and can involve the tibial tray-cement or cement-bone interface. Polyethylene wear, a major driver of aseptic loosening, generates particles that trigger an osteolytic response, predominantly affecting the tibial side.
*
Patellar Component:
Patellar loosening is less frequent but can cause anterior knee pain.
Biomechanics of Aseptic Loosening
The knee joint experiences complex loading patterns during daily activities. Excessive shear or tensile forces at the implant-bone interface, often exacerbated by malalignment, inappropriate component sizing, or poor cement technique, can lead to initial micromotion. If this micromotion exceeds the critical threshold for bone ingrowth (typically 50-150 µm), fibrous tissue formation occurs instead of bone integration, leading to progressive loosening.
*
Polyethylene Wear:
The most common biomechanical driver for aseptic loosening. Cumulative stress and motion cause polyethylene particles to abrade from the bearing surface. Macrophages engulf these particles, releasing pro-inflammatory cytokines that stimulate osteoclasts, leading to periprosthetic osteolysis and subsequent implant migration.
*
Stress Shielding:
In uncemented designs, excessive stiffness of the implant can shield the underlying bone from physiological stress, leading to bone resorption (Wolff's Law) and potential loosening.
*
Material Fatigue:
Over extended periods, implants, particularly those with modular junctions, can experience material fatigue, leading to fracture or loss of integrity, which can mimic or directly cause loosening.
Anatomical Considerations for Diagnosis
- Synovial Fluid Aspiration: Understanding the knee capsule and suprapatellar pouch is critical for effective aspiration. The suprapatellar approach (just superior to the patella, lateral to the quadriceps tendon) or a medial/lateral mid-patellar approach are common.
- Periprosthetic Membranes/Biofilms: Infected implants are often encased in a glycocalyx biofilm, making bacterial detection challenging. Biopsy of the periprosthetic membrane during revision surgery is crucial for cultures and histology.
- Bone Loss Patterns: Aseptic loosening often leads to characteristic patterns of bone loss related to wear debris or mechanical stress. PJI-related bone loss can be more aggressive and diffuse. Identifying these patterns pre-operatively helps in surgical planning for bone stock management.
Indications & Contraindications
The decision-making process for a painful TKR, specifically regarding workup for PJI or aseptic loosening and potential revision surgery, requires careful consideration of indications and contraindications.
Indications for Diagnostic Workup
- Persistent or new-onset pain: Unexplained, worsening, or recalcitrant pain.
- Stiffness or decreased range of motion: Progressive loss of flexion or extension.
- Swelling or effusion: Persistent or recurrent joint effusion, especially with warmth or erythema.
- Systemic symptoms: Fever, chills, fatigue (highly suggestive of PJI).
- Laboratory abnormalities: Elevated ESR, CRP without clear non-infectious etiology.
- Radiographic changes: Progressive radiolucent lines (>2mm), component migration, osteolysis, endosteal scalloping.
- Sinus tract: Pathognomonic for PJI, even without systemic symptoms.
- Instability: New or progressive instability, especially rotational.
- Mechanical symptoms: Locking, catching, giving way, clunking.
Indications for Operative Management (Revision Arthroplasty or Surgical Debridement)
-
Confirmed PJI:
Based on established criteria (e.g., MSIS, ICM 2018).
- Acute PJI (symptoms <3 weeks, well-fixed components): Debridement, antibiotics, and implant retention (DAIR) with polyethylene exchange.
- Chronic PJI (symptoms >3 weeks) or failed DAIR: Two-stage exchange arthroplasty (gold standard) or one-stage exchange arthroplasty in selected cases.
- Presence of a sinus tract communicating with the prosthesis.
-
Symptomatic Aseptic Loosening:
- Progressive pain unresponsive to conservative measures.
- Radiographic evidence of significant component migration (>5mm), subsidence, or wide radiolucent lines (>2mm in multiple zones).
- Periprosthetic osteolysis compromising bone stock or implant stability.
- Component fracture (e.g., polyethylene insert wear-through, tibial tray fracture).
- Mechanical Instability: Persistent symptomatic instability despite bracing and rehabilitation.
- Arthrofibrosis: Severe, debilitating stiffness unresponsive to manipulation under anesthesia.
- Periprosthetic Fracture: Displaced or unstable fractures involving the implant-bone interface.
Contraindications to Revision Surgery
- Uncontrolled Systemic Infection: Active infection at a remote site must be controlled prior to elective revision.
- Severe Comorbidities: Medical comorbidities that render the patient an unacceptable anesthetic or surgical risk. A thorough pre-operative medical evaluation is crucial.
- Non-Ambulatory Status: Patients with severely limited functional potential who are unlikely to benefit significantly from a complex revision procedure.
- Non-Compliance: Patients unwilling or unable to adhere to post-operative rehabilitation protocols or antibiotic regimens (in the case of PJI).
- Inadequate Bone Stock: Severe, unmanageable bone loss that precludes stable implant fixation, making arthrodesis or amputation more viable options.
- Severe Extensor Mechanism Deficiency: Irreparable damage to the quadriceps mechanism can severely limit functional outcomes regardless of TKR revision.
- Unrealistic Patient Expectations: Mismatched expectations regarding pain relief and functional improvement.
Operative vs. Non-Operative Indications for Painful TKR Management
| Indication Category | Operative Management | Non-Operative Management |
|---|---|---|
| Infection (PJI) | Acute PJI (DAIR), Chronic PJI (2-stage revision), persistent sinus tract. | Not a primary option for definitive treatment; empiric antibiotics only if diagnostic workup in progress. |
| Aseptic Loosening | Symptomatic radiographic loosening, component fracture, significant osteolysis. | Asymptomatic loosening, minimal symptoms, stable implant despite radiographic findings (observation, activity modification). |
| Instability | Persistent symptomatic instability despite bracing/rehab, recurrent dislocations. | Mild, infrequent instability, effectively managed with physical therapy and bracing. |
| Component Malposition/Wear | Symptomatic malposition causing impingement/instability, severe polyethylene wear with osteolysis. | Asymptomatic malposition, mild wear without significant symptoms. |
| Arthrofibrosis | Severe motion restriction unresponsive to MUA or intensive PT. | Mild-moderate stiffness responsive to PT, manipulation under anesthesia. |
| Extensor Mechanism Issues | Patellar maltracking requiring realignment, patellar component loosening/fracture, quadriceps tendon rupture. | Mild patellofemoral pain, managed with bracing, PT, analgesia. |
| Periprosthetic Fracture | Displaced or unstable fracture, fracture around well-fixed components requiring fixation. | Stable, non-displaced fracture in well-fixed components (conservative management). |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is the cornerstone of successful management for a painful TKR, especially when considering revision for infection or aseptic loosening.
Diagnostic Workup for PJI
-
Inflammatory Markers:
- Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP): Elevated levels (e.g., CRP >10 mg/L, ESR >30 mm/hr) are highly suggestive of infection but lack specificity. Normal levels make PJI less likely.
-
Synovial Fluid Analysis:
This is the most critical diagnostic step.
- Leukocyte Count and Differential: A synovial fluid white blood cell (WBC) count >3,000 cells/µL or a polymorphonuclear neutrophil (PMN) percentage >80% are highly sensitive and specific for PJI.
- Culture: Aerobic and anaerobic cultures of synovial fluid are essential. Prolonged incubation (up to 14 days) and culturing in specific media can increase yield.
- Alpha-Defensin: A host-defense peptide elevated in synovial fluid during infection. Highly specific and sensitive for PJI, less affected by antibiotic use.
- Leukocyte Esterase (LE) Strip Test: A rapid point-of-care test, showing high correlation with PJI when positive (e.g., ++).
- Gram Stain: Can provide rapid preliminary information but has low sensitivity.
- Synovial Biopsy: Performed when synovial fluid is unobtainable or inconclusive. Can be done open or arthroscopically. Tissue samples are sent for histology (presence of acute inflammation) and culture.
- Imaging for PJI: While plain radiographs may show progressive lucencies or osteolysis that overlap with aseptic loosening, specific signs like sequestra or periosteal reaction can suggest infection. Labeled leukocyte scans (Indium-111 or Technetium-99m HMPAO-labeled WBC scans) combined with Technetium-99m sulfur colloid bone marrow scans can improve specificity in differentiating PJI from aseptic loosening, though their use is limited by availability and cost.
Diagnostic Workup for Aseptic Loosening
-
Radiographs:
- Standard Views: AP, lateral, Merchant (patellar) views, and full-length weight-bearing AP views of the knee and hip-to-ankle.
- Assessment: Look for progressive radiolucent lines (>2mm in multiple zones), component migration (>5mm translation or 2 degrees angulation), subsidence, periprosthetic osteolysis, or component fracture. Comparison with immediate post-operative films is crucial.
-
Advanced Imaging:
- Computed Tomography (CT) Scan: Excellent for assessing bone stock, particularly for planning augments or grafts in cases of significant osteolysis. Useful for evaluating component rotation and alignment, especially the patellofemoral joint. Artifacts from metal implants can be mitigated with specific protocols (e.g., MARS-MRI, but less common for loosening).
- Magnetic Resonance Imaging (MRI): Generally limited by significant metallic artifact in the presence of TKR components. Can be useful in specific cases, such as evaluating soft tissue pathology or extensor mechanism integrity.
- Bone Scintigraphy (Technetium-99m MDP): Increased uptake can indicate bone remodeling, stress, or loosening, but lacks specificity to differentiate aseptic loosening from infection or other causes of pain.
Surgical Planning for Revision
- Component Removal Strategy: Plan for the sequence of component removal, considering the potential for cement adherence and bone loss. Specialized instruments for cement removal and osteotomes are essential.
- Bone Loss Management: Pre-operative imaging (especially CT) guides the anticipated bone defects (femoral, tibial, patellar). Plan for the use of augments (metal wedges), cones, sleeves, modular stems, or allograft bone.
- Implant Sizing: Use templates and consider contralateral knee measurements (if unaffected) for revision component sizing.
- Revision System Choice: Select a revision TKR system that can address anticipated bone loss and provide greater constraint if needed. Options range from posterior-stabilized revision components to fully constrained hinged implants.
- Antibiotic Regimen (for PJI): In PJI cases, a multi-disciplinary approach with infectious disease specialists is critical to determine the appropriate systemic antibiotic regimen before and after surgery.
Patient Positioning and Draping
- Supine Position: Standard for TKR procedures. Adequate padding of bony prominences, especially the heel, to prevent pressure sores.
- Tourniquet: Typically used to create a bloodless field, crucial for visualization during complex revision surgery. Its duration must be monitored.
- Positioning aids: A bump under the ipsilateral hip can help prevent external rotation of the limb. A foot support can be utilized to aid knee flexion and extension during surgery.
- Draping: Standard draping for a knee arthroplasty, ensuring adequate exposure for the planned incision and extensile options, such as a quadriceps snip or tibial tubercle osteotomy. Consider full limb draping to allow free manipulation of the extremity.
Detailed Surgical Approach / Technique
The surgical technique for diagnosing and treating PJI or aseptic loosening in a painful TKR is highly variable and depends on the specific pathology encountered.
Diagnostic Aspiration & Biopsy
- Aspiration Technique: Performed under strict aseptic conditions. The knee is typically flexed to 30-45 degrees. A common approach is the superolateral portal, directing a large-bore needle (e.g., 18-gauge) into the suprapatellar pouch, avoiding contact with the prosthetic components if possible.
- Sample Collection: Collect fluid into various tubes: plain tube for cell count and differential, sterile tubes for aerobic and anaerobic cultures, specialized tubes for alpha-defensin or leukocyte esterase.
- Synovial Biopsy: If aspiration is non-diagnostic or dry, a synovial biopsy may be necessary. This can be performed open through a small incision or arthroscopically, targeting areas of thickened or inflamed synovium. Multiple samples (at least 3-5) from different areas are critical for optimal yield for both histology and culture.
Surgical Management of Periprosthetic Joint Infection (PJI)
Debridement, Antibiotics, and Implant Retention (DAIR)
- Indication: Acute PJI (symptoms <3-6 weeks, prosthesis well-fixed, no sinus tract, susceptible organism).
- Approach: Typically through the existing incision.
-
Steps:
- Exposure: Re-expose the joint, ensuring adequate visualization.
- Synovectomy: Meticulous debridement and synovectomy of all inflamed and necrotic tissue. This is crucial for reducing bacterial load and removing biofilm.
- Polyethylene Exchange: Crucial step to remove biofilm from the largest component surface.
- Component Assessment: Ensure femoral and tibial components are well-fixed. If loosening is evident, DAIR is contraindicated.
- Pulsed Lavage: Copious irrigation with saline or antiseptic solutions.
- Antibiotic Delivery: Consider local delivery with antibiotic-laden cement beads or absorbable materials.
- Closure: Close over drains.
- Post-op: Long-term intravenous followed by oral antibiotics, guided by sensitivity profiles and infectious disease consultation.
Two-Stage Exchange Arthroplasty (Gold Standard for Chronic PJI)
Stage 1: Resection Arthroplasty and Spacer Placement
This stage focuses on complete infection eradication.
1.
Incision and Exposure:
Utilize the previous incision, extending it as needed. Often, extensile approaches are required, such as a quadriceps snip, V-Y quadriceps plasty, or tibial tubercle osteotomy, to facilitate implant removal and extensive debridement.
2.
Component Removal:
Carefully remove all components (femoral, tibial, patellar) and cement. Preserve as much bone stock as possible.
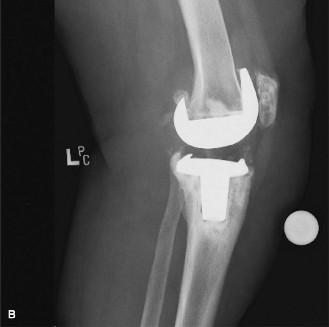
(Image depicting removal of components from an infected joint, highlighting bone loss and inflammatory tissue.)
3.
Debridement:
Aggressive debridement of all infected and necrotic soft tissues, including extensive synovectomy and removal of any infected capsule, bursae, or sinus tracts. All visible granulomatous tissue is excised.
4.
Tissue Samples:
Multiple tissue samples (at least 5-6) from different locations (synovium, joint capsule, interface membranes) are sent for aerobic, anaerobic, fungal, and atypical mycobacterial cultures, and histology (frozen sections intraoperatively for acute inflammation, and permanent sections for definitive diagnosis).
5.
Pulsed Lavage:
Copious irrigation with sterile saline.
6.
Antibiotic-Impregnated Cement Spacer:
A crucial step.
*
Types:
Can be static (non-articulating) or articulating. Articulating spacers (e.g., made with molds or custom-designed in theatre) allow for some knee motion and better soft tissue tension, aiding in rehabilitation and potentially making the second stage easier.
*
Antibiotics:
Typically vancomycin and an aminoglycoside (e.g., tobramycin) are incorporated into the cement, chosen based on local epidemiology or presumed pathogens.
7.
Closure:
Layered closure over suction drains.
8.
Post-op:
Intravenous antibiotics typically for 4-6 weeks, guided by intraoperative cultures and sensitivity. The knee is often immobilized or allowed limited weight-bearing with an articulating spacer.
Stage 2: Reimplantation
This stage proceeds after infection markers (ESR, CRP) have normalized, synovial fluid analysis (aspirated after an antibiotic holiday of 2-4 weeks) is negative, and the patient has completed the antibiotic course.
1.
Incision and Exposure:
Re-open the prior incision, potentially needing to mobilize scar tissue. Remove the cement spacer.
2.
Debridement and Fresh Frozen Sections:
Further debridement if necessary. Obtain fresh frozen sections of periprosthetic tissue; the absence of acute inflammatory cells is crucial confirmation of infection eradication.
3.
Bone Preparation:
Prepare the femoral and tibial bone for revision components, addressing any residual bone defects with augments, cones, or structural allografts.

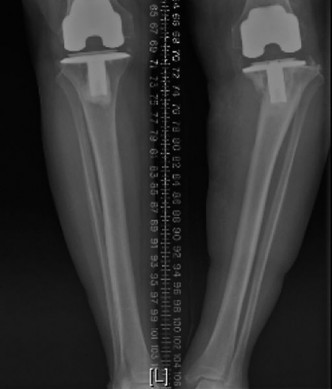
(Image showing prepared bone surfaces for revision, possibly with augments or stems in place, prior to final component implantation.)
4.
Component Implantation:
Implant the revision TKR components. These often include long stems for diaphyseal fixation, metal augments to fill bone defects, and constrained designs (e.g., constrained condylar knee or rotating hinge knee) to achieve stability if the collateral ligaments are compromised by previous infection or debridement. Cementation is typical for revision components.
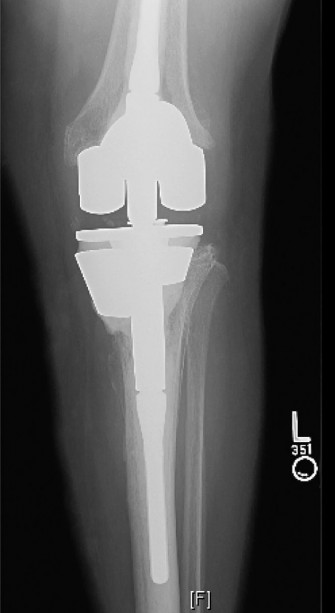
(Image of a revised TKR in situ, demonstrating the final reconstruction with potentially a constrained or stemmed implant.)
5.
Range of Motion and Stability:
Assess range of motion and stability.
6.
Closure:
Layered closure, often over drains.
Surgical Management of Aseptic Loosening
- Incision and Exposure: Similar to Stage 1 of a PJI revision, often requiring extensile exposures for adequate visualization and removal of well-fixed components.
-
Component Removal:
Systematically remove the femoral, tibial, and patellar components, along with all cement. Careful technique is paramount to avoid iatrogenic bone loss.
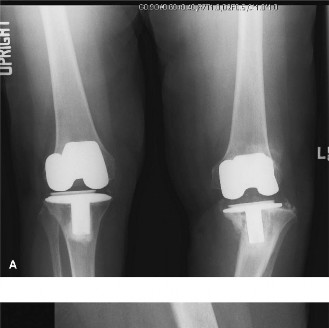
(Image illustrating radiographic evidence of aseptic loosening, potentially with osteolysis or migration, guiding the need for revision and highlighting the bone defects.) - Debridement: Remove any fibrous membrane or granulomatous tissue from the bone-implant interface. Send tissue for culture and histology to definitively rule out occult infection.
-
Bone Defect Management:
This is a critical step in revision for aseptic loosening. Bone defects are often classified using systems like the Anderson Orthopaedic Research Institute (AORI) classification.
- Minor Defects (AORI Type 1, 2A): Can often be managed with cement and impaction bone grafting.
- Moderate Defects (AORI Type 2B, 3): Require metal augments (wedges or blocks), cones, or sleeves to restore the joint line and provide stable support for the new components.
- Severe Defects (AORI Type 3): May necessitate structural allografts or custom implants.
-
Revision Component Implantation:
- Stems: Utilize modular stems (femoral and tibial) to bypass areas of bone loss and achieve diaphyseal fixation, improving implant stability.
- Augments: Position and secure metal augments to reconstruct bone deficiencies.
- Constraint: The degree of constraint in the revision implant is chosen based on soft tissue integrity and stability. This may range from posterior-stabilized to a constrained condylar knee (CCK) or rotating hinge knee (RHK) in cases of severe ligamentous compromise.
- Soft Tissue Balancing: Re-establish appropriate soft tissue tension and collateral ligament balance for optimal stability and range of motion.
- Patellar Tracking: Assess and address patellar tracking if a patellar component is revised or if patellofemoral issues contribute to pain.
-
Closure:
Layered closure over drains.
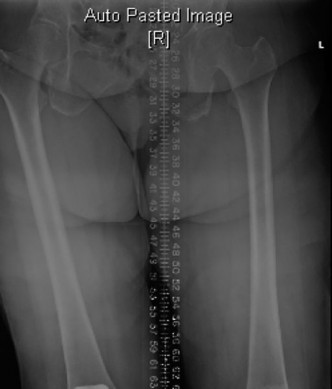
(Image depicting post-revision radiographs for aseptic loosening, showing well-fixed stemmed components and reconstructed bone defects.)
Complications & Management
Revision TKR, whether for infection or aseptic loosening, carries a significantly higher complication rate than primary TKR due to compromised bone stock, scarred soft tissues, and potentially resistant organisms.
Common Complications of Revision TKR, Incidence, and Salvage Strategies
| Complication | Incidence (Approx.) | Salvage Strategies |
|---|---|---|
| Persistent/Recurrent Infection | 5-15% | Repeat DAIR (if criteria met), prolonged antibiotics, repeat 2-stage revision, arthrodesis, amputation. |
| Aseptic Loosening (Recurrent) | 5-10% | Further revision with different fixation/component design, bone grafting, augments, custom implants, arthrodesis. |
| Instability | 5-15% | Component exchange (polyethylene, more constrained liner), soft tissue balancing, hinged knee prosthesis. |
| Periprosthetic Fracture | 1-5% | Open reduction and internal fixation (ORIF), revision with longer stems/different fixation, allograft-prosthesis composite. |
| Arthrofibrosis/Stiffness | 10-20% | Manipulation under anesthesia (MUA), arthroscopic or open lysis of adhesions, intensive physical therapy. |
| Extensor Mechanism Dysfunction | 1-5% | Quadricepsplasty, patellar tendon repair/reconstruction, extensor mechanism allograft, patellectomy (rare). |
| Neurovascular Injury | <1% | Intraoperative identification and repair, vascular surgery consultation, nerve grafting. |
| Deep Vein Thrombosis (DVT)/Pulmonary Embolism (PE) | 1-3% | Prophylactic anticoagulation, early mobilization, pharmacologic treatment, IVC filter in selected cases. |
| Limb Length Discrepancy | Variable | Shoe lift, contralateral knee revision, precise component sizing and bone resection. |
| Chronic Pain (unexplained) | 10-20% | Multidisciplinary pain management, psychological assessment, neuropathic pain medication, peripheral nerve blocks, advanced diagnostic workup. |
| Wound Complications | 5-10% | Local wound care, debridement, negative pressure wound therapy, rotational flaps for coverage. |
Management Considerations
- PJI Recurrence: The most feared complication. Requires aggressive re-evaluation and often repeat surgical intervention. Biofilm disruption remains a primary challenge.
- Bone Loss: Progressive or iatrogenic bone loss can limit future revision options. Meticulous surgical technique during component removal is crucial.
- Soft Tissue Coverage: Revision surgery, especially for infection, can compromise soft tissue envelopes, increasing the risk of wound dehiscence or non-healing. Plastic surgery consultation may be necessary for complex defects.
- Patient Expectations: Manage patient expectations carefully regarding pain relief, functional outcomes, and the potential for further interventions, particularly with PJI.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation following revision TKR is tailored to the specific surgical procedure, intraoperative findings (e.g., bone loss, implant stability, extensor mechanism repair), and the underlying etiology (PJI vs. aseptic loosening). The goal is to maximize functional recovery while protecting the surgical reconstruction.
General Principles
- Pain Management: Aggressive multimodal analgesia is essential for early mobilization.
- Early Mobilization: Unless specifically contraindicated (e.g., extensor mechanism repair, very fragile bone), early out-of-bed activity and weight-bearing are encouraged to prevent complications like DVT/PE and stiffness.
- Range of Motion (ROM): Gradual progression of active and passive ROM exercises is initiated, respecting tissue healing and component stability.
-
Weight-Bearing (WB):
- Aseptic Loosening Revision: Often immediate weight-bearing as tolerated (WBAT) with stemmed implants providing stable diaphyseal fixation. If significant bone grafting or an insecure construct is used, protected weight-bearing (e.g., toe-touch weight-bearing or partial weight-bearing) for 6-12 weeks may be indicated.
- PJI (After Stage 2 Reimplantation): Often more conservative. Protected weight-bearing or non-weight-bearing may be prescribed, especially if bone quality was compromised by infection or if a hinged implant was used.
- Muscle Strengthening: Progressive resistance exercises for quadriceps, hamstrings, and gluteal muscles.
- Proprioception and Balance: Training initiated once pain is controlled and weight-bearing permits.
Phase-Specific Rehabilitation
Phase I: Acute Post-Operative (Weeks 0-6)
- Goals: Pain and edema control, protection of surgical site, initiation of gentle ROM, basic mobility.
-
Interventions:
- Continuous Passive Motion (CPM) machine may be used, though its efficacy remains debated.
- Ankle pumps, quadriceps setting, gluteal sets.
- Assisted ROM exercises (flexion to 0-90 degrees, extension to 0 degrees).
- Transfers (bed to chair), gait training with appropriate assistive devices (walker, crutches) and weight-bearing status.
- Cryotherapy, elevation, wound care.
Phase II: Intermediate (Weeks 6-12)
- Goals: Restore full functional ROM, improve muscle strength and endurance, progress gait.
-
Interventions:
- Progressive resistive exercises: Closed-chain exercises (mini-squats, leg presses) and open-chain exercises (knee extension, hamstring curls).
- Stationary cycling.
- Balance and proprioceptive drills.
- Wean off assistive devices as strength and balance improve.
- Scar mobilization.
Phase III: Advanced & Return to Activity (Week 12+)
- Goals: Achieve optimal functional independence, return to desired activities (low-impact), long-term maintenance.
-
Interventions:
- Advanced strengthening, plyometrics (if appropriate, low impact).
- Sport-specific training (for those returning to recreational activities).
- Endurance training.
- Education on activity modification and long-term joint protection.
-
Specific Considerations:
- Extensor Mechanism Repair: Requires prolonged immobilization in extension and extremely cautious, delayed ROM progression to protect the repair.
- Hinged Prostheses: May have limitations on deep flexion and rotational activities.
Note: For patients undergoing two-stage revision for PJI, rehabilitation during the spacer phase (between stages) focuses on maintaining soft tissue mobility and preventing contractures, often with limited weight-bearing. Post-Stage 2 rehabilitation then follows a similar protocol to aseptic loosening revision but may be more conservative initially due to the history of infection and potentially compromised soft tissues.
Summary of Key Literature / Guidelines
The management of painful TKR, especially concerning PJI and aseptic loosening, is guided by robust literature and international consensus recommendations.
Periprosthetic Joint Infection (PJI)
- Diagnosis: The International Consensus Meeting (ICM) on Periprosthetic Joint Infection (2018) provides the most widely accepted diagnostic criteria. These include major criteria (e.g., sinus tract, pathogen isolation from multiple cultures) and minor criteria (e.g., elevated ESR/CRP, elevated synovial WBC/PMN%, positive alpha-defensin, positive leukocyte esterase, single positive culture). A diagnosis of PJI requires either two major criteria, or one major and at least three minor criteria.
- Alpha-Defensin: Multiple studies have validated the high sensitivity and specificity of synovial alpha-defensin for PJI diagnosis, even in patients who have recently received antibiotics. It is particularly useful when conventional markers are equivocal.
- Leukocyte Esterase: A rapid, point-of-care test, synovial leukocyte esterase has shown good correlation with PJI, particularly when combined with CRP and synovial fluid cell count.
- Metagenomic Sequencing: Emerging research suggests that next-generation sequencing techniques may improve pathogen detection rates, especially in culture-negative PJI, by identifying microbial DNA directly.
-
Management:
- DAIR: Indicated for acute PJI (<3-6 weeks symptoms), well-fixed components, and susceptible organisms. Success rates vary (40-70%) and are higher with early intervention and gram-positive organisms.
- Two-Stage Exchange Arthroplasty: Remains the gold standard for chronic PJI, with reported success rates for infection eradication ranging from 85-95%. A critical component is the antibiotic-impregnated cement spacer.
- One-Stage Exchange: Increasingly considered for highly selected cases, particularly with known, susceptible organisms and healthy soft tissues. Outcomes can be comparable to two-stage in experienced centers, but patient selection is paramount.
- Antibiotic Stewardship: Crucial for PJI management, requiring collaboration with infectious disease specialists to guide appropriate broad-spectrum empirical therapy, followed by pathogen-directed intravenous and oral regimens. The duration is typically 6-12 weeks post-surgery.
Aseptic Loosening
- Radiographic Criteria: The gold standard for initial diagnosis. Progressive radiolucent lines (>2mm in multiple zones), component migration, subsidence, and periprosthetic osteolysis are key indicators. Lucent lines at the cement-bone interface may be normal in some zones up to 1-2mm; it is the progression and extent that are critical.
- Polyethylene Wear: The mechanism of wear-induced osteolysis is well-documented, with studies demonstrating the role of particulate debris in activating macrophages and osteoclastogenesis, leading to bone resorption.
-
Revision Strategies:
- Bone Loss Management: AORI classification guides surgical planning for bone defects. The use of metal augments, cones, and sleeves combined with diaphyseal-engaging stems has significantly improved outcomes for managing significant bone loss in revision TKR.
- Stems: Long-stemmed components improve initial stability and distribute loads to healthier diaphyseal bone, especially in cases of compromised metadiaphyseal bone.
- Constraint: The choice of implant constraint (e.g., CCK, RHK) is dictated by residual ligamentous integrity and stability, with hinged prostheses reserved for severe instability or major ligamentous deficiency.
Overall Outcomes
Revision TKR for both PJI and aseptic loosening are complex procedures with less predictable outcomes than primary TKR. Patients should be counseled on higher complication rates, longer rehabilitation, and often less optimal functional results compared to their primary arthroplasty. Continuous research focuses on improving diagnostic accuracy, refining surgical techniques, and developing novel approaches for infection eradication and bone reconstruction.
Clinical & Radiographic Imaging