Patient Presentation & History
A 22-year-old male presented to our clinic with an 18-month history of progressive right knee pain. Initially described as a dull ache, the pain gradually intensified, becoming more constant and less responsive to over-the-counter analgesics. Unlike typical osteoid osteoma, the pain did not exhibit a profound nocturnal predominance, nor was it consistently relieved by NSAIDs, though some temporary symptomatic improvement was noted. The patient reported increasing difficulty with weight-bearing activities, including prolonged standing and ambulation, due to localized discomfort in the distal femur. There was no history of acute trauma, fever, chills, or night sweats. He denied any referred pain, paresthesias, or motor weakness. His past medical history was unremarkable, with no known allergies or significant comorbidities. He was a non-smoker and consumed alcohol occasionally. Family history was negative for bone tumors or sarcomas.
Clinical Examination
Upon inspection of the right lower extremity, a subtle, localized fullness was noted over the distal metaphysis of the femur, slightly proximal to the lateral femoral condyle. There was no visible erythema, skin changes, or obvious gross deformity. Muscle bulk appeared symmetrical, without evidence of significant atrophy.
Palpation revealed a localized, firm, and tender area over the distal lateral femur. The warmth of the overlying skin was symmetrical to the contralateral limb. There was no palpable mass distinct from the bone itself, suggesting an intraosseous or juxtacortical lesion.
Range of motion (ROM) of the right knee was minimally restricted due to pain at the extremes of flexion (0-120 degrees of flexion, compared to 0-140 degrees on the contralateral side). Extension was full. Joint line tenderness was absent, and collateral and cruciate ligaments were stable to stress testing, ruling out primary ligamentous pathology.
A thorough neurological examination of the right lower extremity was performed. Motor strength was graded 5/5 in all major muscle groups (hip flexion, extension, abduction, adduction; knee flexion, extension; ankle dorsiflexion, plantarflexion, inversion, eversion). Sensory examination to light touch and pinprick was intact in all dermatomes (L2-S1). Deep tendon reflexes (patellar, Achilles) were symmetrical and 2+ bilaterally. Peripheral pulses (femoral, popliteal, dorsalis pedis, posterior tibial) were strong and palpable bilaterally, indicating intact vascular supply. There was no evidence of lymphadenopathy in the inguinal region.
Imaging & Diagnostics
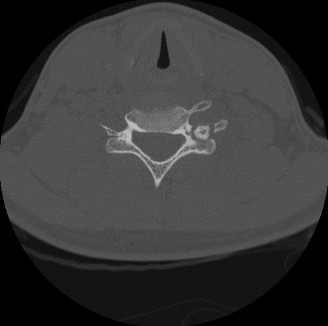
Initial radiographic evaluation of the right knee revealed a well-circumscribed, expansile lytic lesion in the distal lateral metaphysis of the femur. The lesion measured approximately 3.5 x 2.8 cm, demonstrating a distinct sclerotic rim and mild cortical thickening without overt cortical breach or aggressive periosteal reaction. No definite matrix mineralization was observed within the lesion. The joint space was preserved, and there were no findings suggestive of degenerative arthritis.

Computed Tomography (CT) Scan: A CT scan of the distal femur was subsequently performed to better characterize the lesion's bony architecture and delineate its precise extent. The CT confirmed an intramedullary, eccentric lesion with a well-defined sclerotic border, originating in the metaphyseal region. It demonstrated significant cortical expansion and thinning of the lateral femoral cortex, but without frank breach. The nidus, characteristic of osteoblastoma, was hyperdense and showed heterogeneous attenuation. Critically, the CT provided invaluable detail regarding the three-dimensional morphology, cortical integrity, and proximity to the physis (which was closed in this 22-year-old), informing surgical planning for potential intralesional curettage or marginal excision.
Magnetic Resonance Imaging (MRI): An MRI of the right knee, with and without gadolinium contrast, was performed to assess soft tissue involvement, bone marrow edema, and neurovascular proximity. The MRI revealed a T1 hypointense and T2 hyperintense lesion, consistent with a benign bone tumor. There was significant perilesional bone marrow edema and surrounding soft tissue edema, correlating with the patient's pain. Post-gadolinium sequences demonstrated avid, heterogeneous enhancement within the lesion. Crucially, the MRI confirmed no extension into the knee joint, nor any encasement or direct invasion of the adjacent neurovascular bundle (femoral artery/vein, sciatic nerve branches), although the lesion was in close proximity to the superior genicular artery. This information was vital for surgical planning to minimize iatrogenic neurovascular injury.
Bone Scintigraphy: A technetium-99m bone scan showed intense focal uptake in the distal right femur, indicating increased osteoblastic activity, a common finding in benign bone tumors such as osteoblastoma and osteoid osteoma.
Biopsy: Given the clinical and radiographic findings, an image-guided core needle biopsy was performed under fluoroscopic guidance to obtain a definitive histological diagnosis. Histopathological examination revealed irregular trabeculae of woven bone lined by prominent, plump osteoblasts within a highly vascular fibrous stroma. There was an absence of significant cytological atypia, pleomorphism, or atypical mitotic figures, consistent with the diagnosis of osteoblastoma. Communication with the musculoskeletal pathologist confirmed the benign nature of the lesion, distinguishing it from osteosarcoma.
Differential Diagnosis
The presentation of a painful, expansile bone lesion in a young adult necessitates a thorough differential diagnosis, particularly given the imaging characteristics. Key differentials for osteoblastoma include:
| Feature | Osteoblastoma | Osteoid Osteoma | Aneurysmal Bone Cyst (ABC) | Osteosarcoma (Low-Grade Central/Parosteal) | Chronic Osteomyelitis |
|---|---|---|---|---|---|
| Age | 10-30 years (most common) | 10-25 years | <20 years (secondary ABC can occur at any age) | Any age, but peaks in adolescents/young adults (low-grade later) | Any age, often with prior trauma/surgery |
| Size | >2 cm (distinguishing feature from osteoid osteoma) | <1.5-2 cm (nidus) | Variable, often large and expansile | Variable, often large | Variable, localized bone destruction/sclerosis |
| Pain Characteristics | Dull, aching, progressive, less dramatic NSAID response, less nocturnal | Severe, nocturnal, characteristically relieved by NSAIDs (aspirin) | Localized pain, swelling, pathological fracture risk | Variable, often insidious, progressive, deep bone pain | Localized pain, tenderness, warmth, possibly systemic signs (fever, chills) |
| Radiology (X-ray/CT) | Lytic/sclerotic lesion with sclerotic rim, cortical expansion/thinning | Small lytic nidus (<1.5cm) with dense surrounding sclerosis | Expansile, multiloculated lytic lesion, fluid-fluid levels on MRI (classic) | Destructive, permeative, ill-defined margins, aggressive periosteal reaction (Codman's triangle, sunburst) | Lytic destruction, sequestra, involucrum, sinus tracts |
| MRI | T1 hypointense, T2 hyperintense, avid enhancement, significant edema | Nidus with surrounding intense edema (target sign) | Fluid-fluid levels (pathognomonic), extensive soft tissue component, T2 bright cystic spaces | Heterogeneous signal, soft tissue mass, marrow invasion, cortical destruction | Marrow edema, periosteal reaction, soft tissue phlegmon/abscess, sinus tracts |
| Location | Spine (posterior elements), long bones (diaphysis/metaphysis), small bones | Long bones (femur, tibia), spine (posterior elements), small bones | Metaphysis of long bones, vertebrae | Metaphysis of long bones, flat bones | Any bone, often at sites of trauma or compromised immunity |
| Histopathology | Irregular woven bone trabeculae, prominent osteoblasts, vascular stroma | Identical to osteoblastoma but smaller nidus | Blood-filled cystic spaces, septa with fibroblasts, giant cells, hemosiderin-laden macrophages | Atypical osteoblasts producing osteoid/chondroid matrix, malignant features | Inflammatory cells, necrosis, bacteria, sequestrum (dead bone) |
| Treatment | Marginal/wide excision; intralesional + adjuvant (curettage, cryoablation) | Radiofrequency ablation (RFA) or marginal excision | Curettage with adjuvant, marginal excision, selective embolization | En bloc wide excision, chemotherapy, radiation therapy (context-dependent) | Antibiotics, debridement, sequestrectomy, bone grafting (if needed) |
| Recurrence Potential | Moderate (10-20%) | Low (<5%) after complete ablation | Moderate (10-30%) | High if inadequate margins | High if inadequate debridement |
Surgical Decision Making & Classification
The decision for operative intervention in this case was driven by several factors:
1.
Persistent and progressive pain:
The patient's symptoms significantly impacted his quality of life and functional capacity, despite conservative measures.
2.
Size and location of the lesion:
The osteoblastoma, measuring >3.5 cm, was larger than a typical osteoid osteoma, and its metaphyseal location in a weight-bearing bone posed an increasing risk for pathological fracture as cortical thinning progressed.
3.
Risk of recurrence with conservative management:
Unlike osteoid osteoma, osteoblastoma often does not spontaneously resolve and carries a higher propensity for recurrence with incomplete removal or conservative approaches.
4.
Definitive diagnosis:
Biopsy confirmed the benign nature, but the locally aggressive potential mandated surgical intervention for local control.
Surgical Classification:
While osteoblastoma is a benign tumor, its management often draws upon principles applicable to more aggressive lesions due to its potential for recurrence and local destruction. The
Enneking Staging System for Benign Musculoskeletal Tumors
is a critical framework:
*
Stage 1 (Latent):
Asymptomatic, stable lesion, minimal activity.
*
Stage 2 (Active):
Symptomatic, locally active, contained within bone or reactive rim. Most osteoblastomas fall into this category. Our patient's lesion, with progressive symptoms, cortical expansion, and significant surrounding edema, fit this description.
*
Stage 3 (Aggressive):
Locally aggressive, rapid growth, no containment, potential for cortical breach. Some osteoblastomas with very rapid growth or significant soft tissue extension might be classified here.
For this Stage 2 active osteoblastoma, the primary surgical goal was complete local eradication to prevent recurrence while preserving limb function and structural integrity. Given the metaphyseal location in a weight-bearing bone, a balance between aggressive tumor removal and bone stock preservation was paramount. The options considered were intralesional curettage with adjuvant therapy versus marginal en bloc excision. Due to the significant cortical thinning and the desire for a lower recurrence rate, marginal en bloc excision was deemed the most appropriate definitive treatment, accepting the need for subsequent reconstruction.
Surgical Technique / Intervention
Patient Positioning: The patient was positioned supine on a radiolucent operating table. A high thigh tourniquet was applied to the right lower extremity to facilitate a bloodless field. All bony prominences were adequately padded. C-arm fluoroscopy was prepared and draped for intraoperative imaging.
Approach: A longitudinal anterolateral incision, approximately 12 cm in length, was made over the distal lateral femur, centered over the lesion as localized by preoperative imaging and palpation. The fascia lata was incised, and the vastus lateralis muscle was split and retracted anteriorly. Care was taken to identify and protect the lateral superior genicular artery and nerve branches. The periosteum was then sharply incised and elevated off the lateral femoral cortex, revealing the cortical expansion.
Tumor Excision:
Under fluoroscopic guidance, the precise boundaries of the lesion were identified. A bone saw was used to create an osteotomy achieving a marginal en bloc excision of the cortical and medullary lesion. This involved excising a block of bone encompassing the tumor with a surrounding cuff of normal, uninvolved bone. The specimen was oriented and sent for histopathological analysis, including frozen sections to confirm clear margins if deemed necessary by the pathology team for critical anatomical sites, though for osteoblastoma, clean marginal excision with macroscopically visible normal tissue is generally sufficient.
Reconstruction of the Bony Defect:
The resulting segmental defect in the distal lateral femoral metaphysis was approximately 4 cm in length and involved a significant portion of the cortical circumference. To restore structural integrity and facilitate early weight-bearing, the defect was reconstructed.
1.
Preparation of the defect:
The bony bed was meticulously debrided to remove any residual tumor cells and ensure healthy, bleeding bone surfaces.
2.
Structural Allograft:
A precisely sized cortical strut allograft was fashioned to match the contour and dimensions of the resected segment. The allograft was then press-fit into the defect.
3.
Internal Fixation:
To provide stable fixation for the allograft and protect the reconstructed segment, a pre-contoured locking plate (e.g., LCP Distal Lateral Femur Plate) was applied to the lateral aspect of the femur, bridging the defect and extending well into the proximal and distal healthy bone segments. Multiple locking screws were inserted bicortically into the native bone and monocortically through the allograft where appropriate, ensuring rigid fixation of the construct. The goal was to provide sufficient stability to allow for allograft incorporation and controlled rehabilitation.
4.
Bone Grafting (Supplemental):
Autogenous cancellous bone graft, harvested from the ipsilateral iliac crest, was packed around the interfaces of the allograft and host bone to promote osteointegration and accelerate healing. This was considered a critical step to enhance the biological integration of the allograft.
Closure: The tourniquet was deflated, and meticulous hemostasis was achieved. A suction drain was placed to prevent hematoma formation. The vastus lateralis muscle was repaired, the fascia lata closed, and the subcutaneous tissues and skin were closed in layers. A sterile dressing was applied.
Post-Operative Protocol & Rehabilitation
Immediate Post-Operative Period (Days 0-7):
*
Pain Management:
Multimodal analgesia regimen including opioids, NSAIDs (if not contraindicated), and acetaminophen. Regional nerve blocks were utilized intraoperatively and for the first 24-48 hours.
*
Wound Care:
Daily dressing changes, monitoring for signs of infection. Suction drain typically removed within 24-48 hours when output diminishes.
*
Deep Vein Thrombosis (DVT) Prophylaxis:
Low molecular weight heparin (LMWH) initiated immediately post-operatively and continued for 4-6 weeks, along with sequential compression devices.
*
Weight-bearing Status:
Strict non-weight-bearing on the operative leg for the initial 6 weeks to protect the allograft and internal fixation. Crutches or a walker were utilized.
*
Physical Therapy (PT):
Initiated on post-operative day 1. Focus on gentle, passive, and active-assisted range of motion (ROM) exercises for the knee within a pain-free arc (e.g., 0-90 degrees) to prevent stiffness. Isometric quadriceps and hamstring contractions to maintain muscle tone without stressing the reconstruction. Ankle pump exercises to reduce swelling and prevent DVT.
Early Rehabilitation (Weeks 1-6):
*
Weight-bearing:
Continued strict non-weight-bearing. Emphasis on transfers and mobility with assistive devices.
*
ROM:
Gradual progression of knee ROM exercises, aiming for full flexion and extension by 6 weeks, as tolerated.
*
Strengthening:
Progressive isometric strengthening exercises. Introduction of non-weight-bearing isotonic exercises for hip and ankle. Upper extremity strengthening to aid ambulation with crutches.
Intermediate Rehabilitation (Weeks 7-12):
*
Radiographic Assessment:
Plain radiographs at 6 weeks post-op to assess allograft position, fixation integrity, and early signs of healing/incorporation.
*
Weight-bearing:
Progressive protected weight-bearing initiated. Typically, 25% partial weight-bearing with crutches for 2 weeks, advancing to 50% for another 2 weeks, and then full weight-bearing by 12 weeks, as tolerated and guided by clinical and radiographic findings.
*
ROM:
Continue to work towards full, pain-free ROM.
*
Strengthening:
Progression to open-chain and closed-chain strengthening exercises (e.g., partial squats, leg press with light resistance) as weight-bearing increases. Proprioception and balance training initiated.
*
Scar Management:
Desensitization and massage for scar tissue.
Advanced Rehabilitation (Months 3-6):
*
Weight-bearing:
Full weight-bearing without assistive devices, as pain allows and radiographs confirm healing.
*
Strengthening:
Advanced strengthening program focusing on functional movements, sport-specific drills (if applicable). Gradual return to higher impact activities, but impact loading should be carefully introduced and monitored.
*
Endurance:
Cardiovascular conditioning.
Long-Term Follow-up:
*
Clinical and Radiological Assessment:
Regular follow-up appointments (e.g., every 3-6 months for the first 2 years, then annually) are crucial to monitor for recurrence and assess allograft incorporation. This typically involves clinical examination and plain radiographs. MRI may be utilized if recurrence is suspected or to assess allograft viability.
*
Recurrence:
Osteoblastoma has a recurrence rate of approximately 10-20% even after marginal excision, emphasizing the need for diligent follow-up.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
*
Clinical Suspicion:
Always maintain a high index of suspicion for atypical, persistent bone pain in adolescents and young adults. While osteoid osteoma is more common, the lack of dramatic NSAID response or nocturnal pain should prompt consideration of osteoblastoma.
*
Imaging Hierarchy:
*
X-rays:
Initial assessment, often shows expansile lytic lesion with sclerotic rim.
*
CT:
Essential for detailed bony architecture, cortical involvement, nidus characterization, and pre-operative templating, especially for complex anatomical sites like the spine.
*
MRI:
Critical for assessing soft tissue extension, marrow edema, neurovascular proximity, and differentiating from other lesions (e.g., fluid-fluid levels in ABC).
*
Biopsy is Mandatory:
A definitive histological diagnosis via image-guided core needle biopsy is non-negotiable before definitive surgical intervention. Communicate closely with the musculoskeletal pathologist regarding the clinical and radiological context to avoid misdiagnosis, particularly distinguishing it from osteosarcoma.
*
Surgical Strategy for Local Control:
*
Marginal En Bloc Excision:
This is the preferred method for osteoblastoma, especially for large or aggressive lesions, to minimize recurrence rates.
*
Intralesional Curettage with Adjuvants:
If marginal excision is not feasible due to anatomical constraints (e.g., critical neurovascular structures, joint surfaces), thorough intralesional curettage combined with local adjuvants (e.g., high-speed burr, cryoablation, phenol, argon beam coagulation) can be considered, but recurrence rates are higher.
*
Spinal Lesions:
Osteoblastoma frequently affects the posterior elements of the spine. Surgical planning must include not only tumor excision but also meticulous neurological decompression and robust spinal stabilization/fusion, given the high risk of instability post-resection. The Weinstein-Boriani-Biagini (WBB) system is invaluable for assessing vertebral involvement and guiding surgical approach for these complex cases.
*
Reconstruction:
Anticipate and plan for significant bony defects post-excision. Utilize structural allografts, vascularized fibula grafts, or endoprosthetic reconstruction to restore biomechanical integrity, particularly in weight-bearing bones. Stable internal fixation is paramount.
*
Long-Term Follow-up:
Due to the risk of local recurrence, diligent clinical and radiological surveillance (plain films, MRI) for at least 5 years post-surgery is essential.
Pitfalls:
*
Misdiagnosis:
* Mistaking osteoblastoma for osteoid osteoma and treating with simple radiofrequency ablation without histological confirmation, leading to recurrence due to inadequate treatment of the larger, more aggressive lesion.
* Misinterpreting aggressive reactive bone formation or extensive edema as malignancy, leading to unnecessarily radical surgery without definitive biopsy.
* Inadequate biopsy technique (e.g., sampling reactive bone at the periphery instead of the nidus) resulting in non-diagnostic or misleading histological findings.
*
Incomplete Excision:
Undertaking simple curettage without adjuvant therapy or marginal margins, significantly increasing the risk of local recurrence and potentially requiring more extensive salvage surgery.
*
Neurological Complications:
In spinal lesions, failure to meticulously decompress neural elements or to provide adequate stabilization post-resection can lead to iatrogenic neurological deficits or progressive deformity.
*
Pathological Fracture:
Delay in diagnosis or inadequate protection of a weakened bone segment, leading to pathological fracture which complicates surgical management and increases morbidity.
*
Inadequate Reconstruction:
Failing to appreciate the biomechanical demands of the resected segment, resulting in reconstructive failure (e.g., non-union of allograft, hardware failure, malalignment) requiring further operative intervention.
*
Over-reliance on Conservative Management:
While benign, symptomatic osteoblastomas rarely resolve spontaneously and typically require intervention. Prolonged conservative management can lead to tumor growth, increased bone destruction, and more complex surgical challenges.
*
Ignoring Malignant Transformation:
Although rare, osteoblastomas can undergo malignant transformation, typically to osteosarcoma. Persistent pain or rapid growth in a previously stable lesion warrants re-evaluation and potentially re-biopsy.
Clinical & Radiographic Imaging