Patient Presentation & History
A 25-year-old otherwise healthy male presented to the emergency department with acute onset of severe pain and inability to bear weight in his left thigh following a ground-level fall. He reported a sudden "pop" sensation in his mid-thigh.
Mechanism of Injury: The fall was low-energy, involving a slip on a wet surface. This mechanism, combined with the acute severe pain and functional limitation, immediately raised suspicion for a pathological fracture rather than a simple traumatic event.
History of Present Illness: The patient reported a 6-month history of a vague, dull ache in his left mid-thigh, which he attributed to muscle strain from his job, which involves prolonged sitting. The pain was intermittent, non-radiating, and minimally responsive to over-the-counter analgesics. He denied any fevers, chills, night sweats, or unintentional weight loss. There was no history of previous fractures, bone deformities, or systemic symptoms indicative of endocrine dysfunction or dermatological manifestations.
Demographics: A 25-year-old male of average build, previously active and without significant limitations.
Comorbidities: The patient had no known medical comorbidities, was not on any chronic medications, and denied any history of substance abuse. His surgical history was limited to an appendectomy in childhood. Family history was negative for known skeletal dysplasias, metabolic bone diseases, or malignancy. Review of systems was otherwise negative.
Clinical Examination
Upon examination, the patient was alert, oriented, and in acute distress, requiring significant opioid analgesia for pain control.
Local Examination (Left Thigh):
*
Inspection:
A gross deformity of the left mid-femur was evident, characterized by angular deviation and approximately 2 cm of shortening compared to the contralateral limb. Significant swelling was present circumferentially around the mid-thigh, with overlying ecchymosis extending proximally and distally. The skin integrity was intact, with no open wounds, signs of tenting, or impending compromise.
*
Palpation:
Marked tenderness was elicited over the mid-diaphyseal region of the left femur. Palpable crepitus was noted with gentle manipulation. There was localized warmth, but no discrete pulsatile mass. No regional lymphadenopathy was identified.
*
Range of Motion (ROM):
Active and passive range of motion at the left hip and knee was severely restricted and exquisitely painful due to the fracture.
*
Neurological Assessment:
Distal neurological function was intact. The patient was able to dorsiflex and plantarflex his foot, and extend his great toe, indicating normal motor strength (5/5) in the L2-S1 distribution. Sensation to light touch was intact throughout the dermatomes of the foot and leg.
*
Vascular Assessment:
Peripheral pulses (femoral, popliteal, dorsalis pedis, posterior tibial) were all readily palpable (2+/2+ bilateral). Capillary refill in the toes was brisk (<2 seconds). No signs of compartment syndrome were present.
Systemic Examination:
General physical examination revealed no other bony tenderness, joint effusions, or skin lesions. There was no café-au-lait pigmentation, which would suggest McCune-Albright syndrome. The remainder of the systemic examination was unremarkable.
Imaging & Diagnostics
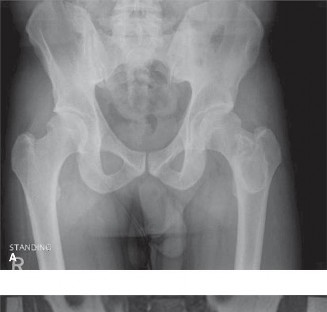
Initial Radiographs (Left Femur):
Anterior-posterior (AP) and lateral views of the left femur were obtained. These revealed a displaced, comminuted, transverse fracture of the mid-diaphyseal femur. Critically, associated with the fracture was a large, expansile, lytic lesion occupying nearly the entire mid-diaphyseal segment. The lesion exhibited the classic "ground-glass" matrix appearance, which is highly characteristic of fibrous dysplasia. Significant cortical thinning and endosteal scalloping were evident along the length of the lesion. There was no overt periosteal reaction indicative of aggressive malignant change, nor was there clear evidence of osteoid or chondroid matrix production within the lesion itself, apart from the ground-glass pattern. Cortical breach was clearly visible at the fracture site. No obvious extraosseous soft tissue mass was identified on plain films.
Computed Tomography (CT) Scan of the Left Femur:
A CT scan was performed to further characterize the lesion's internal matrix, cortical integrity, and precise extent, especially in relation to the fracture. The CT confirmed the expansile nature of the lesion, demonstrating diffuse areas of hazy, ground-glass attenuation interspersed with more lucent areas, consistent with the fibrous and immature osseous components of fibrous dysplasia. Extensive cortical thinning, endosteal scalloping, and focal cortical defects were noted. There was no definitive evidence of aggressive periosteal reaction or cortical destruction extending into soft tissues, which would be highly concerning for a high-grade sarcoma. However, an ill-defined adjacent soft tissue component was noted, likely representing hemorrhage and edema from the fracture, but initially raising a subtle red flag for an aggressive lesion. The fracture line was clearly delineated through the dysplastic bone.
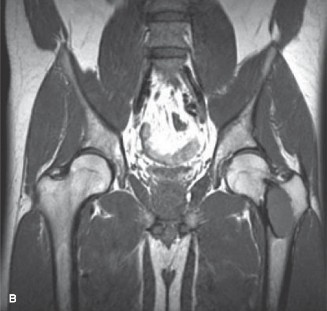
Magnetic Resonance Imaging (MRI) of the Left Femur:
An MRI was obtained to assess marrow involvement, soft tissue extension, and to rule out secondary aneurysmal bone cyst (ABC) or other features suggestive of malignant transformation.
*
T1-weighted images:
Demonstrated heterogeneous low-to-intermediate signal intensity within the lesion, reflecting its predominant fibrous and immature osseous components.
*
T2-weighted images:
Showed areas of high signal intensity, consistent with fibrous stroma, areas of fluid, and potentially hemorrhage associated with the fracture. Importantly, no definitive fluid-fluid levels were identified, which would have been highly suggestive of an ABC.
*
Post-contrast T1-weighted images with fat saturation:
Revealed heterogeneous enhancement, primarily peripheral, with some internal septal enhancement.
Crucially, the MRI did not show overt aggressive soft tissue invasion, extensive peritumoral edema, or solid enhancing nodules typically associated with high-grade sarcomas. There was no evidence of skip lesions within the medullary canal. The findings were largely consistent with fibrous dysplasia, but the expansile nature and subtle soft tissue signal changes necessitated a careful differential diagnosis, particularly from an oncological perspective.
Whole-Body Bone Scan (Technetium-99m MDP):
Given the diagnosis of fibrous dysplasia, a whole-body bone scan was performed to evaluate for polyostotic involvement. This is an important step to differentiate monostotic from polyostotic disease and to rule out other synchronously active skeletal lesions. The scan demonstrated markedly increased tracer uptake in the left mid-femoral diaphysis, corresponding to the known lesion and fracture site. No other areas of abnormal increased uptake were identified throughout the axial or appendicular skeleton, confirming monostotic fibrous dysplasia.
Laboratory Investigations:
*
Routine blood work:
Complete blood count, erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) were all within normal limits, arguing against an infectious, inflammatory, or highly aggressive malignant process.
*
Metabolic panel:
Serum calcium, phosphate, and alkaline phosphatase (ALP) levels were within normal ranges, effectively ruling out hyperparathyroidism or other metabolic bone diseases that can present with lytic lesions (e.g., brown tumor). Parathyroid hormone (PTH) levels were also normal.
*
Tumor markers:
While not specific for fibrous dysplasia, tumor markers such as LDH are often included in an initial oncology workup for lytic bone lesions, and these were also unremarkable in this case.
Differential Diagnosis
The clinical presentation of a pathological fracture through an expansile lytic lesion with a ground-glass matrix in a young adult mandates a thorough differential diagnosis, emphasizing distinctions between aggressive benign lesions and low-grade malignancies.
| Feature | Fibrous Dysplasia (Monostotic) | Low-Grade Chondrosarcoma (Grade 1 / Atypical Cartilaginous Tumor) | Aneurysmal Bone Cyst (ABC) |
|---|---|---|---|
| Pathogenesis | Developmental anomaly from GNAS1 somatic mutation, leading to abnormal osteoblastic differentiation. | Malignant cartilaginous tumor; can be primary or secondary to enchondroma. | Benign, destructive lesion of blood-filled spaces, often secondary to other lesions (e.g., FD, GCT, non-ossifying fibroma). |
| Age Group | Adolescents and young adults (10-30s) | Adults (30-60s) | Children and young adults (<20s) |
| Typical Location | Metaphysis/Diaphysis of long bones (femur, tibia), ribs, craniofacial bones. | Pelvis, femur (metaphysis/diaphysis), humerus, ribs. | Metaphysis of long bones, vertebrae, flat bones. |
| Clinical Presentation | Often asymptomatic or dull ache. Pathological fracture common. | Slow-growing pain, swelling; pathological fracture in advanced stages. | Rapid onset pain, swelling, pathological fracture. |
| Radiographic Features | Ground-glass matrix , expansile, cortical thinning, endosteal scalloping. No periosteal reaction. "Shepherd's crook" deformity in femur. | Endosteal scalloping >2/3 cortical thickness, focal cortical breakthrough, septations. Characteristic "rings and arcs" (popcorn) calcification. | Expansile, purely lytic, "bubbly" appearance, thin sclerotic rim, cortical thinning/expansion. No calcified matrix (unless secondary). |
| CT Features | Heterogeneous ground-glass attenuation, cortical expansion/thinning, indistinct trabeculae. | Calcified chondroid matrix (rings and arcs), significant endosteal erosion, cortical destruction/involvement. | Well-defined lytic lesion with multiple thin internal septa. Classic fluid-fluid levels due to blood sedimentation. |
| MRI Features | T1: Low-intermediate signal. T2: High signal (fibrous/cystic areas). Heterogeneous, predominantly peripheral enhancement. Absence of fluid-fluid levels (unless secondary ABC). | T1: Low-intermediate. T2: High signal. Peripheral and septal enhancement. May have lobular morphology. | T1/T2: Heterogeneous signal due to varying blood products. Key diagnostic feature: prominent fluid-fluid levels. Peripheral enhancement of septa. |
| Histopathology | Irregularly shaped trabeculae of immature woven bone without osteoblastic rimming, embedded in a loose fibrous stroma. "Chinese characters" pattern. | Lobules of hyaline cartilage with increased cellularity, nuclear atypia, myxoid change, sometimes permeation of marrow. | Blood-filled spaces separated by septa containing fibroblasts, osteoclast-like giant cells, and reactive woven bone. No epithelial lining. |
| Malignant Potential | Rare (0.4-4%), typically secondary osteosarcoma, often after radiation. | Inherent malignancy; metastatic potential, though low for Grade 1. | Benign. No malignant potential. |
| Key Differentiating Points | Ground-glass matrix on X-ray/CT. Histology with immature woven bone in fibrous stroma. | Rings and arcs calcification , aggressive cortical involvement, chondroid matrix on histology. | Fluid-fluid levels on MRI. Histology with blood-filled spaces. Often secondary. |
Surgical Decision Making & Classification
The presence of a displaced pathological fracture of the left mid-femur, causing significant pain and functional impairment, mandated urgent operative intervention. While the radiographic and MRI features strongly suggested fibrous dysplasia, the expansile nature and initial imaging nuances necessitated ruling out a low-grade malignancy. Therefore, the surgical strategy was carefully devised to achieve both fracture stabilization and definitive tissue diagnosis, adhering to oncological principles.
Surgical Goals:
1.
Fracture Stabilization:
To achieve anatomical reduction, restoration of length and rotation, and rigid internal fixation of the pathological fracture, allowing for early mobilization and union.
2.
Definitive Biopsy:
To obtain sufficient, high-quality tissue for histopathological confirmation of the diagnosis, distinguishing fibrous dysplasia from its mimics, particularly low-grade chondrosarcoma or other primary bone tumors.
3.
Lesion Management:
To address the underlying fibrous dysplastic lesion within the intramedullary canal, aiming to reduce recurrence risk and provide structural reinforcement to the bone.
Classification of Pathological Fractures (Mirels' Scoring System):
The Mirels' scoring system is used to assess the risk of impending pathological fracture in bones affected by lytic lesions. Although a fracture was already present in this case, applying the score helps to contextualize the severity of bone compromise and reinforces the decision for aggressive stabilization.
*
Location:
Femur (3 points)
*
Pain:
Moderate (2 points, for chronic pain leading up to acute fracture)
*
Lesion Size:
>2/3 cortical diameter (3 points, as the lesion was expansive and involved most of the diaphyseal circumference)
*
Nature of Lesion:
Lytic (2 points)
*
Total Score:
3 + 2 + 3 + 2 =
10 points
.
A score of 10 points indicates a high risk of pathological fracture, corroborating the urgent need for operative stabilization and meticulous reconstruction.
Operative vs. Non-operative Management:
Non-operative management for a displaced femoral shaft pathological fracture in an adult is generally contraindicated. This approach carries an exceedingly high risk of non-union, malunion, limb shortening, significant pain, and poor functional outcomes, especially in the context of an underlying bone lesion that compromises healing. Given the patient's age and functional demands, operative stabilization was the unequivocal choice.
Specific Considerations for Fibrous Dysplasia in the Trauma Context:
*
Compromised Bone Quality:
Bone affected by fibrous dysplasia is inherently weak and prone to refracture. This necessitates robust fixation that can withstand physiological loads during the healing phase.
*
Non-union Risk:
Fractures through dysplastic bone have a significantly higher risk of non-union compared to fractures in healthy bone.
*
Recurrence:
Simple intralesional curettage of fibrous dysplasia can have a high recurrence rate, particularly in aggressive, expansile lesions.
*
Malignant Transformation:
While rare (incidence of 0.4-4%), malignant transformation (most commonly to osteosarcoma) is a known complication, particularly in polyostotic disease, previously irradiated lesions, or lesions with rapid growth and new pain. This potential, combined with the initial imaging features, made a definitive biopsy crucial to rule out a primary sarcoma or a transformed lesion.
Based on these considerations, the decision was made for open reduction and internal fixation of the femoral fracture using an intramedullary nail. This would be combined with thorough intralesional curettage of the dysplastic tissue, which would also serve as the biopsy. The curetted defect would then be augmented to provide structural support and reduce recurrence risk.
Surgical Technique / Intervention
The surgical procedure was performed under general anesthesia. The patient was positioned supine on a fracture table, allowing for dynamic traction, full range of motion of the hip and knee, and unhindered access for the image intensifier. The contralateral limb was abducted and flexed to allow for unencumbered access to the affected extremity.
- Preparation and Draping: The entire left lower extremity, from the iliac crest to the foot, was sterilely prepped and draped to allow for wide exposure and potential extension of the approach.
- Surgical Approach: A standard lateral approach to the proximal femur was employed, extending distally along the line of the intended nail insertion. A gluteal split was performed to access the piriformis fossa, which was chosen as the entry point for the intramedullary nail.
- Fracture Exposure and Provisional Reduction: A longitudinal incision was made directly over the palpable fracture site in the mid-diaphysis. Care was taken to minimize soft tissue stripping, preserving periosteal blood supply. The fracture hematoma was evacuated, and the fracture fragments, which appeared compromised and brittle due to the underlying dysplastic bone, were carefully assessed. Gentle traction and manipulation were used to achieve provisional reduction, ensuring restoration of length and alignment under fluoroscopic guidance.
- Intramedullary Nail Entry Portal and Reaming: The piriformis fossa was opened, and a guidewire was carefully inserted across the fracture site and down into the distal femoral segment. Sequential reaming was performed over the guidewire to prepare the intramedullary canal for the chosen nail size. The reaming process extended through the sclerotic and expanded portions of the dysplastic lesion. This reaming process helped to remove a significant portion of the dysplastic tissue.
- Intralesional Curettage and Biopsy: After reaming and achieving provisional fracture reduction, a meticulous intralesional curettage was performed using curettes and a high-speed burr. All macroscopic dysplastic tissue within the intramedullary canal and cortical defect was thoroughly removed. This debridement was crucial for both reducing the burden of dysplastic tissue and, importantly, served as the definitive incisional biopsy . Multiple representative tissue samples were harvested from various aspects of the lesion cavity and sent for histopathological examination. The cavity was thoroughly irrigated with pulsatile lavage.
- Augmentation of the Defect: Following comprehensive curettage, the large intramedullary defect and cortical windows created by the lesion and debridement required augmentation to restore structural integrity. In this case, a combination of allograft bone chips and Polymethyl Methacrylate (PMMA) bone cement was utilized. The allograft chips were first packed into irregular recesses of the cavity. Subsequently, PMMA bone cement, prepared to a doughy consistency, was carefully injected under pressure into the curetted cavity, ensuring maximal contact with the remaining cortical bone and robust filling of the defect. The PMMA provides immediate mechanical stability, acting as an internal cast, and its exothermic polymerization reaction is also thought to induce local necrosis in any residual dysplastic cells, potentially reducing recurrence rates.
- Intramedullary Nail Insertion: Once the PMMA had partially cured (to a firm but not brittle consistency), an appropriately sized locked intramedullary femoral nail (e.g., a T2 femoral nail system) was carefully inserted antegrade through the piriformis fossa. The nail was advanced past the fracture site and through the PMMA-augmented lesion, providing rigid internal fixation that bridged the entire dysplastic segment and the fracture.
- Distal Locking: Distal locking screws were inserted under precise image intensifier guidance to prevent shortening and rotation of the distal fragment.
- Proximal Locking: Proximal locking screws were then inserted to secure the nail proximally, completing the construct.
- Wound Closure: The surgical site was copiously irrigated. Meticulous hemostasis was achieved. Layered closure of the fascia, subcutaneous tissues, and skin was performed using absorbable sutures for deep layers and skin staples.
-
Final Imaging:
Intraoperative fluoroscopy confirmed satisfactory reduction, appropriate nail position, secure locking, and adequate filling of the dysplastic defect with PMMA and allograft.
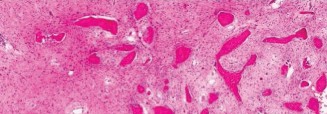
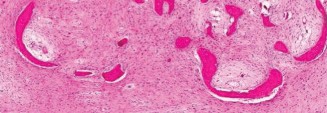
Pathology Report:
The histopathological examination of the curetted tissue samples definitively confirmed the diagnosis of
Monostotic Fibrous Dysplasia
. The specimens showed characteristic irregularly shaped trabeculae of immature woven bone without osteoblastic rimming, embedded within a loose, vascular fibrous stroma. There was no evidence of significant cellular atypia, pleomorphism, increased mitotic activity, or atypical cartilage formation, effectively ruling out a low-grade chondrosarcoma or malignant transformation. This pathological confirmation validated the chosen surgical approach of intralesional curettage and stabilization rather than a more aggressive en bloc resection.
Post-Operative Protocol & Rehabilitation
The post-operative management focused on effective pain control, early mobilization, and a structured, progressive rehabilitation program tailored to the patient's underlying bone pathology and surgical fixation.
Immediate Post-Operative Period (Day 0-3):
*
Pain Management:
A multimodal analgesia regimen was initiated, including a continuous peripheral nerve block (femoral nerve block), scheduled NSAIDs, acetaminophen, and opioid analgesics for breakthrough pain.
*
Antibiotics:
Prophylactic intravenous antibiotics were continued for 24-48 hours post-operatively.
*
Thromboprophylaxis:
Mechanical thromboprophylaxis (sequential compression devices) was applied immediately, and chemical thromboprophylaxis (e.g., low molecular weight heparin or fondaparinux) was initiated post-operatively.
*
Mobilization:
On Post-Operative Day 1, physical therapy (PT) commenced with active ankle pumps, gentle knee flexion/extension within a comfortable range, and assisted transfers to a chair.
*
Weight-Bearing:
Touch-down weight-bearing (TDWB)
of 10-15 kg was permitted on the operative leg using crutches or a walker. Strict adherence to protected weight-bearing was emphasized due to the inherent weakness of the dysplastic bone and the healing fracture.
Early Rehabilitation Phase (Weeks 1-6):
*
Physical Therapy:
The patient transitioned to outpatient PT. Sessions focused on:
* Gradual increase in range of motion for the hip and knee, aiming for functional arcs.
* Isometric and progressing to isotonic strengthening exercises for quadriceps, hamstrings, and gluteal muscles.
* Gait training with continued protected weight-bearing, emphasizing proper technique and safety.
*
Weight-Bearing Progression:
Progress to
partial weight-bearing (PWB)
was initiated around 4-6 weeks post-operatively, contingent on clinical stability (reduced pain, no signs of implant loosening) and initial radiographic evidence of healing or stability of the PMMA construct. The immediate structural support provided by the intramedullary nail and PMMA typically allows for earlier weight-bearing progression compared to bone graft alone.
*
Radiographic Follow-up:
Scheduled X-rays at 2 weeks, 6 weeks, and 3 months were performed to assess fracture alignment, implant integrity, and the stability of the PMMA/allograft construct within the defect.
Intermediate Rehabilitation Phase (Months 2-6):
*
Weight-Bearing:
Progression to
full weight-bearing (FWB)
was permitted as tolerated and radiographically indicated, typically between 3-4 months post-operatively.
*
Strengthening and Functional Training:
Advanced strengthening exercises, proprioceptive training, and balance activities were incorporated. The goal was to restore full functional capacity and prepare for return to pre-injury activities.
*
Follow-up:
Clinical and radiographic follow-up was conducted at 3 months, 6 months, and 12 months post-surgery. This aimed to monitor for pain, functional recovery, implant-related complications, and any signs of local recurrence or progression of the dysplastic lesion.
Long-Term Follow-up:
Patients with fibrous dysplasia require long-term surveillance due to the potential for disease progression, local recurrence, or, albeit rare, malignant transformation. Annual clinical and radiographic review for several years is advisable, and further imaging should be obtained if any new symptoms (pain, swelling, deformity) or suspicious findings on routine radiographs emerge. Patient education regarding these possibilities is paramount.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
- Pathological Fracture Always Demands Etiology: A low-energy fracture in a young adult, especially with prodromal pain, must trigger a workup for underlying bone pathology. Never treat "just a fracture" without understanding the bone quality.
- Multidisciplinary Team Approach: Cases with suspected bone tumors, even those ultimately diagnosed as benign like fibrous dysplasia, necessitate discussion with musculoskeletal radiologists, pathologists, and orthopedic oncologists. This collaborative approach ensures optimal diagnostic and therapeutic pathways.
- Biopsy is King (with Oncological Principles): A definitive tissue diagnosis via biopsy is paramount. In a pathological fracture requiring urgent stabilization, an intraoperative incisional biopsy or meticulous curettage (providing ample tissue) adhering to oncological principles (e.g., careful soft tissue handling, planning future definitive resection if malignant) is acceptable.
- Radiographic "Ground-Glass" Appearance: This feature on plain radiographs and CT scans is highly characteristic of fibrous dysplasia, reflecting immature woven bone and fibrous stroma. Recognize this pattern, but be aware of its mimics.
- MRI for Soft Tissue and Aggressiveness: MRI is essential to assess soft tissue extension, marrow involvement, and to differentiate between benign and malignant features. The absence of aggressive soft tissue invasion or extensive peritumoral edema helps steer the differential away from high-grade sarcomas. Look for fluid-fluid levels, which strongly suggest an aneurysmal bone cyst (often secondary to FD).
- Mirels' Score as a Decision Aid: Utilize the Mirels' scoring system for assessing the risk of impending pathological fracture and for guiding the urgency and aggressiveness of prophylactic or definitive stabilization in known lesions.
-
Surgical Strategy for Fibrous Dysplasia:
- Thorough Intralesional Curettage & High-Speed Burring: Aggressive removal of dysplastic tissue is crucial to minimize recurrence.
- Augmentation with PMMA/Graft: Polymethyl methacrylate (PMMA) bone cement provides immediate structural support, allows for earlier weight-bearing, and its exothermic reaction may help devitalize residual dysplastic cells. It is often combined with autograft or allograft.
- Rigid Internal Fixation: Intramedullary nails are often preferred for long bone diaphyseal lesions due to their load-sharing capabilities and ability to provide robust stability through the compromised bone segment.
- Long-Term Surveillance: Fibrous dysplasia lesions can progress, recur, or, rarely, undergo malignant transformation. Long-term clinical and radiographic follow-up is essential for monitoring these possibilities.
- Differential Diagnosis Acuity: Be adept at distinguishing fibrous dysplasia from its critical mimics, such as low-grade chondrosarcoma (ACT/CS1), aneurysmal bone cyst, giant cell tumor, and even brown tumor of hyperparathyroidism, based on integrated clinical, radiographic, and histopathological features.
Pitfalls:
- Inadequate Biopsy leading to Misdiagnosis or Delay: Insufficient tissue or poorly planned biopsy can yield non-diagnostic results, leading to diagnostic uncertainty, inappropriate treatment, or delayed definitive management.
- Misinterpreting Aggressive-Looking Benign Lesions: The expansile nature and sometimes rapid growth of fibrous dysplasia can mimic malignancy on imaging. Over-interpreting benign aggressive features as malignant can lead to overly aggressive and unnecessary surgical procedures. Conversely, under-calling a true low-grade sarcoma as benign fibrous dysplasia is a grave error.
- Incomplete Curettage and High Recurrence Rates: Simple curettage without aggressive burring or augmentation (e.g., PMMA) often results in a high local recurrence rate, especially in younger patients with active lesions or polyostotic disease.
- Refracture due to Inadequate Fixation or Premature Loading: The inherent weakness of dysplastic bone makes it prone to refracture. Inadequate internal fixation (e.g., solely relying on a plate for a diaphyseal fracture without IM support) or premature unrestricted weight-bearing can lead to construct failure and refracture.
- Missing Malignant Transformation: Any new onset of rapidly increasing pain, swelling, or radiographic changes (e.g., cortical destruction, new soft tissue mass, aggressive periosteal reaction) in a known FD lesion should raise immediate suspicion for malignant transformation and necessitate urgent re-evaluation and biopsy. Do not dismiss new symptoms in a known "benign" lesion.
- Failure to Assess for Polyostotic Disease: Not performing a whole-body bone scan or other appropriate imaging to rule out polyostotic involvement. Polyostotic fibrous dysplasia (e.g., in McCune-Albright syndrome) has broader systemic implications and potentially higher complication rates.
- Ignoring Secondary Aneurysmal Bone Cysts: If an ABC is identified as secondary to fibrous dysplasia, simply treating the ABC without addressing the underlying FD can lead to recurrence of the ABC or continued issues from the FD.
- Lack of Adherence to Oncological Principles: Even when the lesion is suspected to be benign, adhering to tumor surgical principles (e.g., careful tissue planes, avoiding contamination, planning incision for potential wider resection) is crucial until definitive benign pathology is confirmed to prevent tumor seeding in case of unexpected malignancy.
