Operative Management of Articular Cartilage Injuries
Key Takeaway
Articular cartilage injuries present a profound challenge in orthopedic surgery due to the tissue's avascular nature and limited intrinsic healing capacity. This comprehensive guide details evidence-based operative interventions, including microfracture, osteochondral autograft transfer, allograft transplantation, and autologous chondrocyte implantation. By mastering precise patient selection, biomechanical principles, and meticulous surgical techniques, orthopedic surgeons can restore joint congruity, alleviate mechanical symptoms, and delay the onset of degenerative osteoarthritis in high-demand patients.
Fundamentals of Articular Cartilage Injuries
Articular cartilage is a highly specialized, complex connective tissue designed to withstand tremendous biomechanical forces over millions of cycles. Despite its remarkable durability, it possesses virtually no intrinsic ability to heal even after minor traumatic insults. This is primarily due to its avascular, aneural, and alymphatic nature. More than 250 years ago, William Hunter astutely observed, “Ulcerated cartilage is a troublesome thing, once destroyed is not repaired.”
The overarching health, kinematics, and function of a diarthrodial joint depend entirely on the viability of the articular cartilage; without it, progressive degenerative arthritis is inevitable. In young, high-demand athletic populations, articular cartilage defects are predominantly traumatic in origin (e.g., shear forces during ligamentous rupture or patellar dislocation). Conversely, in older populations, these changes represent the continuum of degenerative joint disease. Epidemiological data underscore the magnitude of this pathology: articular cartilage changes are radiographically evident in 4% to 10% of individuals between 15 and 24 years of age, escalating to 80% in those older than 55 years. In a landmark retrospective review of 31,516 knee arthroscopies, Curl et al. identified chondral lesions in 63% of cases, highlighting the pervasive nature of this clinical challenge.
Pathophysiology and Biomechanics
Hyaline cartilage is composed primarily of water (65-80%), Type II collagen, and proteoglycans (predominantly aggrecan). The structural integrity relies on the complex interaction between the swelling pressure of the hydrophilic glycosaminoglycans and the tensile restraint of the collagen network. When a traumatic shear or impaction force breaches the tidemark (the boundary between uncalcified and calcified cartilage), the mechanical disruption leads to chondrocyte apoptosis, proteoglycan depletion, and eventual structural collapse. Because the tissue lacks a vascular supply, the classic inflammatory cascade required for tissue regeneration cannot occur unless the subchondral bone is penetrated.
Classification of Articular Cartilage Lesions
Accurate classification is paramount for standardizing research, guiding surgical decision-making, and predicting clinical outcomes. Lesions are categorized by both their morphological configuration and their depth of penetration.
Configuration Classification (Bauer and Jackson)
- Type I: Linear
- Type II: Stellate
- Type III: Flap
- Type IV: Crater
- Type V: Fibrillation
- Type VI: Degrading
Severity and Depth Classification
The International Cartilage Repair Society (ICRS) and Modified Outerbridge systems are the gold standards for grading chondral defect severity:
- Grade 0: Normal, intact cartilage.
- Grade I: Softening, swelling, or blistering with an intact surface (ICRS: Superficial fissures and cracks).
- Grade II: Fragmentation and fissuring less than 0.5 inches in diameter (Outerbridge) or a lesion extending less than 50% of the depth of the articular cartilage (ICRS).
- Grade III: Deep ulceration, fibrillation, or chondral flap larger than 0.5 inches without exposed bone (Outerbridge), or a lesion extending more than 50% of the cartilage depth down to the calcified layer (ICRS).
- Grade IV: Full-thickness wear with exposed subchondral bone (Outerbridge/ICRS).
Clinical Pearl: Always probe the cartilage during diagnostic arthroscopy. Visual inspection alone frequently underestimates the true extent of chondromalacia, particularly in Grade I and II lesions where the surface may appear intact but lacks normal turgor.
Clinical Evaluation and Decision Making
Nonoperative management—including activity modification, unloader orthoses, physical therapy, NSAIDs, and intra-articular injections (corticosteroids, hyaluronic acid, or orthobiologics)—remains the first line of treatment, particularly for low-demand patients or those with diffuse degenerative changes.
Operative intervention is strictly indicated when nonoperative modalities fail to relieve pain and mechanical symptoms (catching, locking, effusion) in the presence of a focal, symptomatic Grade III or IV defect. The surgical algorithm is dictated primarily by the size of the lesion and the patient's functional demands.
Surgical Treatment Algorithm by Lesion Size
- ≤ 1 cm²: Observation, Arthroscopic Debridement, Microfracture, or Osteochondral Autograft Transfer (OATS).
- 1 cm² - 2 cm²: Microfracture or OATS.
- 2 cm² - 3.5 cm²: Fresh Osteochondral Allograft or Autologous Chondrocyte Implantation (ACI).
- 3.5 cm² - 10 cm²: Autologous Chondrocyte Implantation (ACI) or Large Osteochondral Allograft.
- Multiple Lesions: Autologous Chondrocyte Implantation (ACI).
Surgical Warning: Cartilage restoration procedures will universally fail if concomitant pathologies are not addressed. Malalignment (varus/valgus), ligamentous instability (ACL/PCL deficiency), and meniscal loss must be corrected either prior to or concurrently with the cartilage procedure.
Operative Techniques
1. Arthroscopic Debridement and Lavage
For patients with minimal symptoms and small lesions (< 2 cm²) in non-weight-bearing areas, arthroscopic debridement provides palliative, short-term relief. The objective is to eliminate mechanical irritation by removing unstable chondral flaps, smoothing fibrillated edges, trimming meniscal tears, and excising inflamed synovium. Copious joint lavage mechanically clears inflammatory mediators, calcium phosphate crystals, and microscopic cartilage debris. While beneficial for older, low-demand patients, it does not alter the natural history of the disease.
2. Marrow Stimulation Techniques (Microfracture)
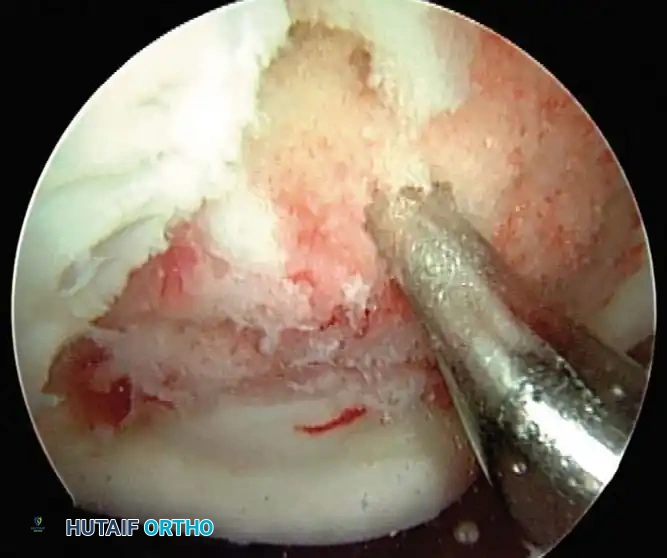
Microfracture relies on the principle of penetrating the avascular subchondral bone plate to access the highly vascularized cancellous bone marrow. This releases mesenchymal stem cells, growth factors, and a fibrin clot into the defect, which eventually differentiates into fibrocartilage (predominantly Type I collagen).
While fibrocartilage lacks the robust biomechanical properties of native hyaline cartilage, it provides a functional patch that improves symptoms in 60% to 70% of patients. However, studies by Kreuz et al. and Gobbi et al. demonstrate that clinical results often deteriorate after 18 to 72 months, particularly in patients over 40 years of age.

Fig. 43-153: Arthroscopic view of microfracture technique utilizing a specialized awl to penetrate the subchondral plate, allowing marrow elements to fill the articular cartilage lesion.
Surgical Technique:
1. Preparation: Debride the lesion to stable, vertical margins using a ring curette.
2. Calcified Layer Removal: Meticulously remove the calcified cartilage layer without damaging the underlying subchondral bone plate.
3. Penetration: Use arthroscopic awls (angled at 30°, 45°, or 90°) to create holes 3 to 4 mm deep.
4. Spacing: Holes must be spaced 1 to 2 mm apart.
Clinical Pearl (Gill's 5 Factors for Microfracture Success):
1. Complete removal of the calcified cartilage layer without ablating the subchondral bone.
2. Maintenance of a 1- to 2-mm bone bridge between penetrations to preserve structural integrity.
3. Immediate postoperative use of Continuous Passive Motion (CPM) to stimulate chondrogenic differentiation.
4. Strict adherence to protected weight-bearing protocols.
5. Correction of any mechanical axis abnormalities.
3. Osteochondral Autograft Transfer (OATS / Mosaicplasty)
For symptomatic lesions up to 2 cm² in high-demand patients, OATS provides a restorative solution by transferring mature, native hyaline cartilage. Cylindrical osteochondral plugs are harvested from relatively non-weight-bearing regions of the knee (e.g., the periphery of the lateral femoral trochlea or the intercondylar notch) and press-fit into the prepared defect.
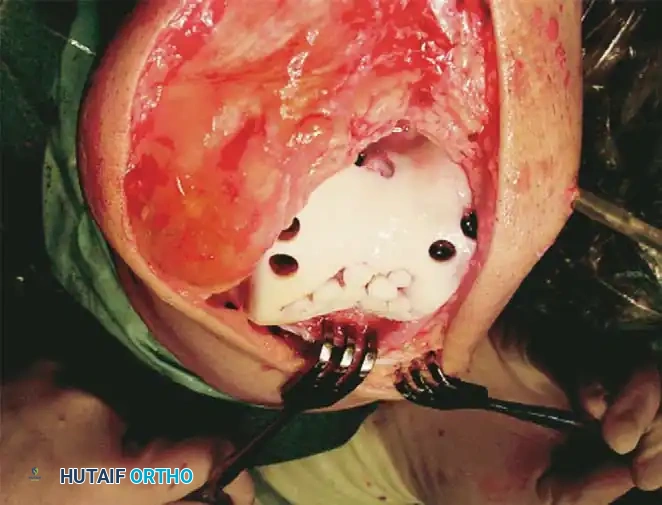
Fig. 43-154: (A) Miniarthrotomy mosaicplasty with the donor site accessed via knee extension. (B) Recipient area accessed via knee flexion. (C) Open mosaicplasty on the femoral trochlea.
Surgical Technique:
1. Recipient Site Preparation: The defect is sized, and a recipient harvester is used to drill perpendicular sockets to a depth of 15 mm.
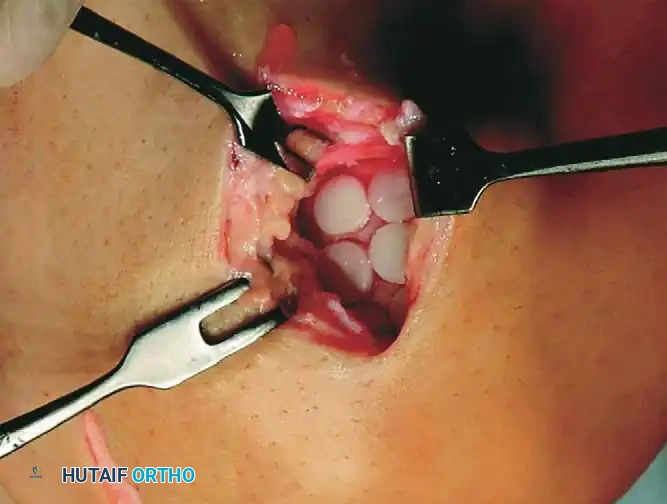
Recipient bed preparation demonstrating multiple drilled sockets ready to receive osteochondral autografts.
- Donor Harvesting: Plugs (typically 6 to 10 mm in diameter) are harvested perpendicularly from the donor site. The depth should match or slightly exceed the recipient socket to ensure a flush articular surface.
- Implantation: The plugs are gently tamped into the recipient sockets. A flush, congruent surface is critical; proud plugs will experience excessive shear forces and fail, while sunken plugs will not participate in load-bearing.

Osteochondral autograft plugs press-fit into the recipient bed, restoring the articular contour.
4. Osteochondral Allograft Transplantation
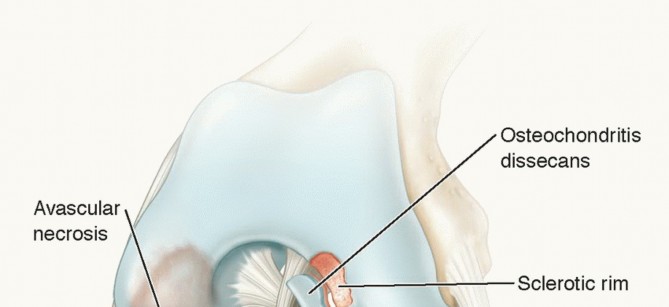
For massive defects (2 to 3.5 cm² or larger), particularly those involving significant subchondral bone loss (e.g., osteochondritis dissecans, avascular necrosis), fresh osteoarticular allografts are indicated. Fresh allografts (stored at 4°C for 14-28 days) are preferred over frozen grafts because they maintain chondrocyte viability of up to 80%. The primary logistical challenge is that patients must remain "on call" for immediate surgery once a size-matched graft becomes available.
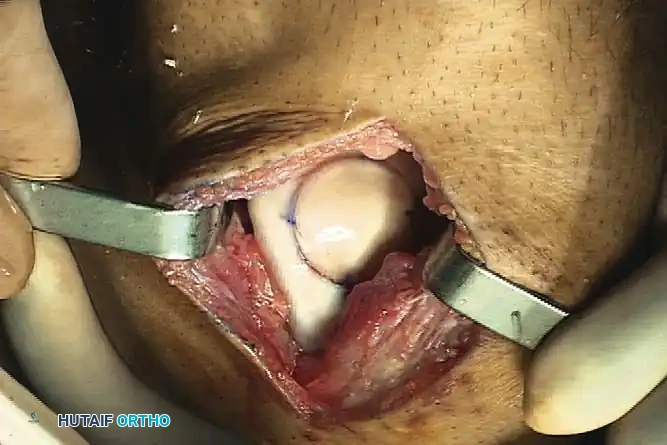
Fig. 43-155 (A & B): Preparation of the recipient bed for an osteochondral allograft. The defect is reamed to a precise depth to accommodate the donor plug.
Surgical Technique:
1. Exposure: A standard arthrotomy is performed to expose the lesion.
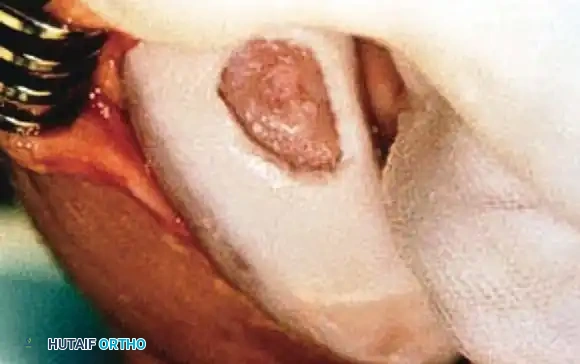
Open view of the meticulously prepared recipient bed, reamed to healthy, bleeding subchondral bone.
- Sizing and Reaming: A sizing guide determines the lesion diameter. A guide pin is placed centrally, and a cannulated reamer creates a perfectly cylindrical recipient socket (typically 6-8 mm deep).
-
Graft Preparation: The size-matched donor hemicondyle is secured in a workstation. A corresponding plug is cored out.
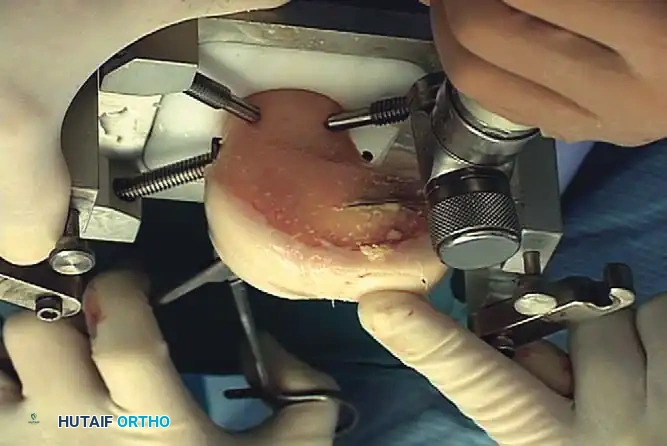
Fig. 43-155 (C & D): Removal of the size-matched graft from the donor bone and subsequent press-fit insertion into the recipient bed. -
Lavage: The donor bone is aggressively pulsatile-lavaged to remove marrow elements, significantly reducing the risk of immunogenic response and disease transmission.
- Implantation: The graft is press-fit into the defect. If stability is questionable, bioabsorbable pins or headless compression screws may be utilized.
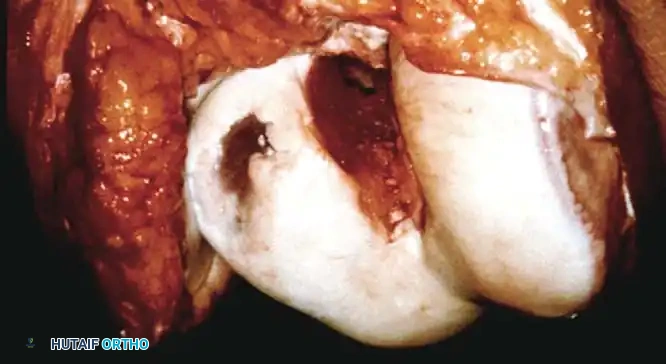
Final seating of the osteochondral allograft plug, demonstrating excellent articular congruity and stable fixation.
5. Autologous Chondrocyte Implantation (ACI)
ACI is a sophisticated, two-stage restorative procedure indicated for large (3.5 to 10 cm²) or multiple full-thickness chondral defects in high-demand patients. It aims to regenerate hyaline-like cartilage.
Stage 1: Arthroscopic Biopsy
A minor arthroscopic procedure is performed to harvest 200 to 300 mg of healthy articular cartilage from a minor load-bearing area (e.g., superior intercondylar notch). The tissue is sent to a laboratory where the chondrocytes are enzymatically isolated and expanded in vitro over 3 to 6 weeks, yielding 12 to 48 million cells.
Stage 2: Implantation
1. Arthrotomy and Debridement: The defect is exposed and debrided to stable vertical walls. The calcified layer is removed, taking care to maintain absolute hemostasis to prevent blood from inactivating the implanted chondrocytes.
2. Patch Fixation: Historically, a periosteal patch harvested from the proximal tibia was used. Today, bioabsorbable Type I/III collagen membranes (MACI) are preferred to avoid periosteal hypertrophy. The patch is sutured over the defect using 6-0 Vicryl, and the edges are sealed with fibrin glue to create a watertight compartment.
The periosteal or collagen patch meticulously sutured over the chondral defect, creating a watertight seal prior to cell injection.
- Cell Injection: The cultured chondrocytes are injected beneath the patch into the defect. The injection portal is then sealed with a final suture and fibrin glue.
Complications of ACI: The most common complications include articular adhesions (requiring subsequent arthroscopic lysis) and graft delamination or hypertrophy. Henderson et al. noted that patch-related problems dominate reoperations within the first 2 years, whereas cartilage-related failures occur later.
The "Sandwich" Technique (Peterson)
For deep osteochondral defects where significant bone loss accompanies the cartilage injury, Peterson advocated the "sandwich" technique.
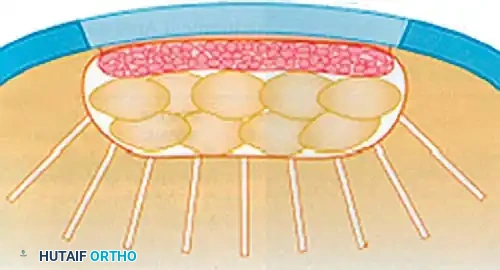
Diagrammatic representation of the ACI "Sandwich" technique. Cancellous bone graft fills the osseous void, separated from the chondrocyte suspension by an intermediate membrane.
Technique:
1. The osseous defect is packed with autologous cancellous bone graft.
2. A primary periosteal or collagen membrane is sutured over the bone graft at the level of the native subchondral plate (cambium layer facing the joint).
3. A secondary membrane is sutured to the articular surface rim (cambium layer facing the defect).
4. Chondrocytes are injected into the space between the two membranes, effectively reconstructing both the bone and cartilage layers simultaneously.
Postoperative Rehabilitation Protocols
The success of any cartilage restoration procedure is inextricably linked to strict adherence to postoperative rehabilitation. The protocol must balance the mechanical protection of the fragile healing tissue with the biological necessity of motion for chondrocyte nourishment.
- Phase I (0-6 Weeks):
- Weight-Bearing: Strict non-weight-bearing or touch-down weight-bearing (TDWB) for femoral condyle lesions. Patellofemoral lesions may allow weight-bearing in full extension (locked brace) to minimize shear forces.
- Motion: Immediate use of Continuous Passive Motion (CPM) for 6 to 8 hours daily. CPM enhances synovial fluid diffusion, promoting matrix synthesis and preventing intra-articular adhesions.
- Phase II (6-12 Weeks): Progressive partial to full weight-bearing. Initiation of closed-kinetic-chain exercises.
- Phase III (3-6 Months): Progression of strength and proprioception. Avoidance of high-impact activities.
- Phase IV (6-12+ Months): Gradual return to sport-specific activities. Full maturation of ACI grafts may take up to 18-24 months.
Future Directions and Investigational Therapies
The landscape of cartilage repair is rapidly evolving toward single-stage, cell-based therapies and advanced tissue engineering.
- Matrix-Induced ACI (MACI): Utilizing biodegradable collagen or hyaluronan-based scaffolds (e.g., Hyalograft C) seeded with chondrocytes. These "sticky" scaffolds can be implanted arthroscopically without the need for complex suturing, reducing morbidity.
- Minced Cartilage Techniques (CAIS): Lu et al. pioneered the use of minced donor cartilage placed on a bioabsorbable scaffold and stapled into the defect. This single-stage procedure eliminates the need for laboratory cell expansion, drastically reducing logistical complexities and costs.
- Gene Therapy: Investigational manipulation of the repair process at the cellular level. By transferring genetic information (e.g., genes encoding for TGF-β or BMPs) directly to the defect site, surgeons aim to supercharge the local environment, driving robust, durable hyaline cartilage regeneration.
Through meticulous patient selection, precise surgical execution, and rigorous postoperative rehabilitation, modern orthopedic surgery can successfully navigate the "troublesome" nature of articular cartilage injuries, restoring function and longevity to the joint.
📚 Medical References
- articular cartilage injuries: microfracture and OATS. Paper presented at the American Academy of Orthopaedic Surgeons Summer Institute, Seattle, Sept 1999.
- Casscells SW: The place of arthroscopy in the diagnosis and treatment of internal derangement of the knee: an analysis of 1000 cases, Clin Orthop Relat Res 151:135, 1980.
- Christen B, Jakob RP: Fractures associated with patellar ligament grafts in cruciate ligament surgery, J Bone Joint Surg 74B:617, 1992.
- Corrigan JP, Cashman WF, Brady MP: Proprioception in cruciate-defi cient knee, J Bone Joint Surg 74B:247, 1992.
- DeHaven KE, Collins HR: Diagnosis of internal derangements of the knee: the role of arthroscopy, J Bone Joint Surg 57A:802, 1975.
- DeLee JC, Riley MB, Rockwood CA Jr: Acute posterolateral rotatory instability of the knee, Am J Sports Med 11:199, 1983.
- DeLee JC, Riley MB, Rockwood CA Jr: Acute straight lateral instability of the knee, Am J Sports Med 11:404, 1983.
- Eriksson E: Comprehensive knee rehabilitation, Bull Hosp Jt Dis Orthop Inst 48:117, 1988.
- Ferrari DA, Ferrari JD, Coumas J: Posterolateral instability of the knee, J Bone Joint Surg 76B:187, 1994.
- Friedman MJ: Lateral knee anatomy, repairs, and reconstruction. Paper presented at the American Academy of Orthopaedic Surgeons Summer Institute, Seattle, Sept 1999.
- Fu FH, Harner CD, Johnson DL, et al: Biomechanics of knee ligaments, J Bone Joint Surg 75A:1716, 1993.
- Girgis FG, Marshall JL, Al Monajem ARS: The cruciate ligaments of the knee joint: anatomical, functional, and experimental analysis, Clin Orthop Relat Res 106:216, 1975.
- Grood ES, Noyes FR, Butler DL, et al: Ligamentous and capsular restraints preventing straight medial and lateral laxity in intact human cadaver knees, J Bone Joint Surg 63A:1257, 1981.
- Grood ES, Stowers SF, Noyes FR: Limits of movement in the human knee: effect of sectioning the
You Might Also Like