Occipitoatlantal Dislocation: Understanding this Fatal Craniovertebral Injury
Introduction & Epidemiology
Occipitoatlantal dislocation (OAD), also known as craniovertebral dissociation (CVD), represents a catastrophic and inherently unstable injury to the craniocervical junction (CCJ). This injury involves the disruption of the ligamentous and bony connections between the occiput and the atlas (C1), leading to a loss of articulation between the occipital condyles and the superior articular facets of C1. Historically, OAD was primarily identified in postmortem studies, where it consistently ranked as the leading cause of death in high-energy trauma, particularly motor vehicle accidents (MVAs). The vast majority of individuals sustaining OAD succumb to the injury at the scene or shortly thereafter due to profound brainstem and spinal cord compromise.
For the rare survivors, the neurological sequelae are devastating. These can range from complete C1 flaccid quadriplegia, reflecting severe disruption of the upper cervical spinal cord and brainstem, to mixed incomplete syndromes such as Brown-Séquard syndrome, which signifies hemi-cord involvement. The high mortality and morbidity underscore the critical importance of prompt recognition and emergent management, though diagnosis in the acute setting remains a formidable challenge.
Epidemiologically, OAD demonstrates a bimodal distribution but is notably
twice as common in children
compared to adults. This increased susceptibility in the pediatric population is attributed to several anatomical and biomechanical factors unique to the developing skeleton. These include:
1.
Inclination of the condyles:
Pediatric occipital condyles tend to be flatter and more horizontally oriented, offering less inherent bony stability compared to the more vertically oriented adult condyles.
2.
Ligamentous laxity:
Children possess greater ligamentous laxity throughout the spine, which translates to a reduced stabilizing capacity of the critical craniovertebral ligaments.
3.
Larger head-to-body ratio:
The relatively larger head size and weaker neck musculature in children increase the leverage on the CCJ during accelerative or decelerative trauma, predisposing them to hyperflexion, hyperextension, or distraction injuries.
4.
Underdeveloped musculature:
Weaker paraspinal and neck musculature provides less dynamic stability against translational or rotational forces.
The mechanism of injury is invariably a high-energy event, most commonly resulting from significant blunt trauma, such as high-speed MVAs, falls from height, or pedestrian versus vehicle collisions. The forces involved typically combine hyperextension, distraction, and rotation at the craniocervical junction. These complex forces overwhelm the inherent bony and ligamentous stability of the occipitoatlantal articulation, leading to the catastrophic disruption.
Associated injuries are frequent and can complicate both diagnosis and management. These include:
*
Submental lacerations:
Indicating a hyperflexion-distraction component.
*
Mandibular fractures:
Often seen in conjunction with significant facial and head trauma.
*
Posterior pharyngeal wall lacerations:
Resulting from direct impact or bony displacement against the pharynx, portending significant airway management challenges.
*
Cranial nerve injuries:
Predominantly affecting the lower cranial nerves due to their proximity to the craniocervical junction. The abducens (CN VI) and hypoglossal (CN XII) nerves are most commonly implicated, manifesting as diplopia, gaze palsies, or tongue weakness/deviation. Other cranial nerves, including glossopharyngeal (CN IX), vagus (CN X), and accessory (CN XI), can also be involved.
*
First three cervical nerves:
C1, C2, and C3 nerve roots can be directly stretched, contused, or avulsed, leading to specific motor and sensory deficits, including compromised diaphragmatic function (phrenic nerve, primarily C3-C5 but with C3 contribution).
*
Vertebral artery injury:
The vertebral arteries traverse the transverse foramina of C1 and C2 before entering the skull, making them highly susceptible to stretching, compression, dissection, or occlusion during OAD. This can result in posterior circulation strokes, even in survivors.
The diagnosis of OAD is frequently missed in the initial evaluation of polytrauma patients due to competing life-threatening injuries, severe head trauma obfuscating neurological assessment, and the often subtle radiographic findings on plain radiographs. However, advancements in computed tomography (CT) and magnetic resonance imaging (MRI) have significantly improved diagnostic accuracy.
Surgical Anatomy & Biomechanics
A thorough understanding of the intricate surgical anatomy and biomechanics of the craniocervical junction is paramount for addressing occipitoatlantal dislocation. This region, spanning the occiput, atlas (C1), and axis (C2), is a complex interplay of bony structures, ligaments, and neural and vascular elements.
Bony Anatomy
The CCJ comprises three primary bony components:
1.
Occipital bone:
Specifically, the occipital condyles, which articulate with the superior articular facets of the atlas.
2.
Atlas (C1):
A ring-shaped vertebra lacking a body and spinous process. It consists of two lateral masses, connected by an anterior and posterior arch. Its superior articular facets are kidney-shaped and concave, designed to accept the convex occipital condyles.
3.
Axis (C2):
Characterized by the dens (odontoid process), which projects superiorly from its body, articulating with the anterior arch of C1.
Ligamentous Anatomy
The stability of the CCJ is overwhelmingly reliant on its robust ligamentous apparatus. These ligaments prevent excessive translation, rotation, and distraction:
Primary Stabilizers:
- Alar Ligaments: Paired, strong, cord-like ligaments extending from the superolateral aspects of the dens to the medial surfaces of the occipital condyles. They limit axial rotation (especially contralateral rotation), lateral bending, and to a lesser extent, flexion-extension.
- Tectorial Membrane: A broad, strong ligament that is the superior continuation of the posterior longitudinal ligament. It extends from the posterior surface of the C2 body/dens to the clivus on the anterior rim of the foramen magnum. It provides significant resistance to flexion and distraction.
- Apical Ligament: A small, delicate ligament connecting the apex of the dens to the anterior rim of the foramen magnum (clivus). Its contribution to stability is minor compared to the alar ligaments and tectorial membrane.
Secondary Stabilizers:
- Anterior Atlanto-Occipital Membrane: Connects the anterior arch of C1 to the anterior rim of the foramen magnum, blending with the anterior longitudinal ligament. Limits extension.
- Posterior Atlanto-Occipital Membrane: Connects the posterior arch of C1 to the posterior rim of the foramen magnum. Limits flexion.
- Cruciform Ligament: Composed of the transverse atlantal ligament (TAL) and superior/inferior longitudinal bands. While primarily stabilizing the C1-C2 articulation, severe OAD often involves disruption of the TAL due to the extensive nature of the trauma. The TAL is the primary stabilizer against anterior translation of C1 on C2.

Vascular Anatomy
The vertebral arteries (VAs) are of critical concern. They typically enter the transverse foramen of C6 and ascend through C1. After exiting the transverse foramen of C1, they curve posteriorly within the sulcus arteriosus on the superior surface of the C1 posterior arch, then turn superiorly to pierce the posterior atlanto-occipital membrane and dura mater, entering the foramen magnum. This tortuous course, particularly around C1-C2, renders them highly susceptible to injury (stretching, dissection, rupture, or occlusion) during the violent displacement associated with OAD. Bilateral VA injury can be immediately fatal due to brainstem ischemia.
Neural Anatomy
The craniocervical junction houses the lower brainstem (medulla oblongata) and the uppermost cervical spinal cord, making it a region of immense neurological vulnerability.
*
Brainstem:
Contains vital centers for respiration, cardiac function, and consciousness. Compression or transection here is lethal.
*
Cranial Nerves:
As mentioned, CN VI (abducens) and CN XII (hypoglossal) are frequently affected due to their close proximity to the C1-C2 complex and the anterior aspect of the brainstem. CN VI exits the brainstem near the pontomedullary junction and traverses a long intracranial course, making it susceptible to stretch injury. CN XII exits the medulla lateral to the pyramid and travels distally, vulnerable to direct compression or avulsion. Other lower cranial nerves (IX, X, XI) also exit the brainstem in this region and can be injured.
*
Cervical Nerves:
The C1, C2, and C3 nerve roots emerge from the spinal cord in this region and can be directly damaged. The C3, C4, and C5 segments contribute to the phrenic nerve, meaning injury at these levels can compromise diaphragmatic function and respiration.
*
Cervicomedullary Syndromes:
The seed content accurately highlights these more unusual forms of incomplete spinal cord injury, which reflect the specific anatomy of the spinal tracts at the junction of the brainstem and spinal cord:
*
Cruciate Paralysis (Bell's Cruciate Paralysis):
This syndrome involves disproportionate weakness, often more profound proximally than distally, primarily affecting the upper extremities (classically, both arms) while sparing or minimally affecting the lower extremities. It results from a lesion at the cervicomedullary junction affecting the decussating fibers of the corticospinal tracts destined for the upper extremities (located anteriorly and medially in the pyramidal decussation), while sparing the more laterally and posteriorly located fibers for the lower extremities. It can be clinically similar to central cord syndrome but with a distinct anatomical basis.
*
Hemiplegia Cruciata (Wallenberg's Hemiplegia Cruciata):
This rare syndrome involves ipsilateral arm weakness and contralateral leg weakness. It occurs due to a specific lesion in the pyramidal decussation affecting the corticospinal tracts before their full decussation. For instance, a lesion affecting the ipsilateral arm fibers and the contralateral leg fibers within the decussation could produce this presentation.
The biomechanics of OAD involve the failure of these ligamentous stabilizers under high-energy loads. The integrity of the alar ligaments and tectorial membrane is particularly crucial. Disruption of these structures leads to gross instability, allowing for anterior, posterior, or vertical translation of the occiput relative to C1.
Indications & Contraindications
Given the inherent and profound instability of occipitoatlantal dislocation, surgical stabilization is almost always indicated in hemodynamically stable patients. The primary goals are spinal cord decompression (if compromised), immediate stabilization of the craniocervical junction, and prevention of further neurological injury.
Indications for Surgical Stabilization
- Confirmed Occipitoatlantal Dislocation: Any definitive diagnosis of OAD, irrespective of neurological status (unless the patient is unsalvageable), necessitates surgical stabilization due to the inherent severe instability.
- Neurological Deficit: Presence of any neurological deficit attributable to the OAD, ranging from subtle cranial nerve palsies to profound quadriplegia, is a strong indication. The surgery aims to prevent worsening and optimize conditions for recovery.
- Progressive Instability: Even in rare instances where a patient might initially present without gross displacement or severe neurological deficits (e.g., partial ligamentous injury), any radiographic or clinical evidence of progressive instability mandates surgical intervention.
- Failure of Conservative Management: While conservative management is almost never appropriate for true OAD (except for temporary stabilization prior to surgery), any failure to maintain reduction or stability in a non-operative setting would lead to surgical intervention.
- Imaging Evidence of Severe Ligamentous Disruption: MRI findings of complete disruption of the alar ligaments and tectorial membrane, along with CT evidence of significant malalignment (e.g., Powers ratio >1.0, Basion-Dens Interval (BDI) >10-12mm, Basion-Axial Interval (BAI) >12mm), confirm the need for surgical fusion.
Contraindications
Absolute contraindications for surgical intervention are rare and generally relate to the overall physiological state of the patient:
- Irreversible Brain Death/Moribund Patient: If the patient is determined to be brain dead or is in a moribund state with no possibility of neurological recovery, aggressive surgical intervention may not be appropriate.
- Uncorrectable Coagulopathy: Severe, uncorrectable coagulopathy that precludes safe surgical hemostasis.
- Severe Comorbidities: Extremely severe cardiopulmonary or other medical comorbidities that render the patient unfit for any surgical procedure or general anesthesia.
- Active Infection: An active, uncontrolled local or systemic infection that would significantly increase the risk of surgical site infection and hardware failure. In such cases, infection control would take precedence, with a delayed stabilization if feasible.
Operative vs. Non-Operative Indications
| Feature | Operative Indications | Non-Operative Indications |
|---|---|---|
| Instability | All cases of OAD are inherently unstable and require fusion. | None. |
| Neurological Deficit | Present or impending neurological compromise. | None, unless patient is unsalvageable with brain death. |
| Imaging Findings | Gross occipitoatlantal dissociation, significant ligamentous disruption (MRI), severe bony malalignment (CT). | None, as OAD implies severe instability. |
| Patient Status | Hemodynamically stable enough to tolerate surgical procedure. | Moribund, irreversible brain death, severe uncorrectable coagulopathy, severe physiological instability precluding surgery. |
| Goal of Management | Spinal cord decompression, immediate and permanent stabilization (arthrodesis). | Palliation, comfort care in cases of unsalvageable injury. |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning and careful patient positioning are paramount in managing OAD to optimize outcomes and minimize further neurological injury. This phase begins immediately upon suspicion of OAD and extends through surgical preparation.
Initial Resuscitation & Spinal Immobilization
Upon arrival, a standard ATLS protocol is initiated:
1.
Airway, Breathing, Circulation (ABCs):
Critical, especially considering potential brainstem injury affecting respiratory drive. Airway management is particularly challenging due to associated injuries (mandibular fractures, pharyngeal lacerations) and the risk of exacerbating spinal cord injury during intubation.
Awake fiberoptic intubation
is often the preferred method to maintain cervical spine stability and avoid manipulation.
2.
Cervical Spine Immobilization:
The patient must be maintained in strict cervical spine immobilization from the outset. This typically involves a rigid cervical collar, sandbags, and tape to the backboard or gurney.
Skeletal traction (e.g., Gardner-Wells tongs)
may be cautiously applied
for reduction purposes
but is not a definitive long-term immobilization strategy for OAD given its inherent instability and potential for distraction. Traction should only be applied with minimal weight (e.g., 5-10 lbs initially), under constant radiographic and neurological monitoring, and only if deemed necessary for achieving reduction prior to fixation.
Diagnostic Imaging
Comprehensive imaging is crucial for definitive diagnosis and surgical planning:
1.
Computed Tomography (CT) Scan:
The gold standard for assessing bony anatomy and craniocervical alignment. Key measurements include:
*
Basion-Dens Interval (BDI):
Distance from the tip of the dens to the inferior margin of the basion. Normal <10 mm in adults (pediatric values vary, but typically <5-6 mm). Increased BDI indicates distraction.
*
Basion-Axial Interval (BAI):
Distance from the basion to a line drawn along the posterior cortex of the C2 body. Normal <12 mm.
*
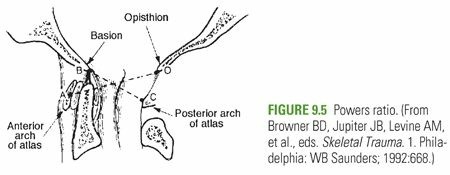
Powers Ratio:
Ratio of BC (basion to posterior arch of C1) to OA (opisthion to anterior arch of C1). Normal <1.0; a ratio >1.0 suggests anterior OAD, while <0.7 suggests posterior OAD. However, it can be unreliable in cases of significant rotation or C1 fracture.
*
Condylar-C1 Asymmetry:
Any significant gapping or displacement of the occipital condyles relative to C1 facets.
2.
Magnetic Resonance Imaging (MRI):
Essential for evaluating soft tissue structures, including the tectorial membrane, alar ligaments, and spinal cord parenchyma. It can detect cord edema, contusion, hemorrhage, and discern the extent of ligamentous disruption, which is critical for understanding the injury pattern. MRI also helps identify any pre-existing conditions that might influence surgical planning.
3.
Computed Tomography Angiography (CTA) or Magnetic Resonance Angiography (MRA):
Crucial to assess for vertebral artery injury (dissection, occlusion, pseudoaneurysm). Given the high incidence of VA injury in OAD, these studies are often performed pre-operatively. If injury is found, endovascular intervention (e.g., stenting) may be required before or concurrently with spinal stabilization, or surgical strategy may need modification.
Neurological Assessment
Repeated and thorough neurological examinations are vital to establish baseline deficits and detect any changes. This includes assessment of cranial nerves, motor and sensory function, and deep tendon reflexes. Intraoperative neuromonitoring (SSEP, MEP) should be planned for the surgical procedure.
Anesthesia Considerations
Close collaboration with the anesthesiology team is essential. Anesthetic plans must account for potential blood loss, hemodynamic instability, and challenges with airway management and patient positioning. Arterial and central venous access are typically required.
Patient Positioning
The patient is typically placed in the
prone position
on a radiolucent operating table.
1.
Head Fixation:
The head is secured using a
Mayfield cranial clamp
or similar three-point fixation device. This allows for precise control of head position and facilitates intraoperative reduction and stabilization. The pins should be placed carefully to avoid major vessels and nerves.
2.
Support and Padding:
Adequate padding is crucial to prevent pressure sores (especially elbows, knees, heels), nerve palsies (ulnar, peroneal), and eye injuries. The chest and pelvis are supported to allow for free abdominal movement, reducing intra-abdominal pressure and epidural venous bleeding.
3.
Neutral Alignment:
The head and neck should be positioned in neutral alignment, or with slight flexion (up to 10-15 degrees) to facilitate posterior exposure and potentially aid in reducing the OAD. Extreme extension should be avoided as it can cause further neurological compromise.
4.
Fluoroscopy/X-ray Access:
Ensure unrestricted access for intraoperative fluoroscopy or X-ray imaging to confirm reduction and hardware placement.
5.
Neuromonitoring:
Electrodes for somatosensory evoked potentials (SSEPs) and motor evoked potentials (MEPs) are placed and baseline recordings obtained. This provides real-time feedback on spinal cord integrity during reduction maneuvers and instrumentation.
Detailed Surgical Approach / Technique
Surgical management of OAD typically involves a posterior approach for occipital-cervical fusion, most commonly to C2 (O-C2 fusion). The goal is anatomical reduction of the dislocation, robust internal fixation, and solid arthrodesis.
1. Incision and Exposure
- Positioning Confirmation: Once the patient is prone and positioned with the head in the Mayfield clamp, a final check of alignment and imaging capabilities is performed. Neuromonitoring is continuously active.
- Incision: A midline posterior cervical incision is made, extending from the inion (external occipital protuberance) superiorly to the spinous process of C3 or C4 inferiorly.
- Subperiosteal Dissection: The superficial fascia and trapezius muscle are incised in the midline. Subperiosteal dissection is then carried out laterally, raising the paraspinal muscles (semispinalis capitis, longissimus capitis, obliquus capitis superior/inferior, rectus capitis posterior major/minor) from the occipital squama, the posterior arch of C1, and the lamina/spinous process of C2.
-
Key Anatomical Landmarks:
- Occiput: Identify the external occipital protuberance and nuchal lines for occipital screw placement.
- C1: The posterior arch of C1 is often thin or absent in OAD. The C1-C2 interspace exposes the vertebral artery in its groove, requiring extreme caution. The C1 posterior tubercle may be palpable.
- C2: The large bifid spinous process of C2 is a reliable landmark. Dissect laterally to expose the C2 lamina and pars interarticularis. Care must be taken to identify and preserve the C2 ganglion, which emerges inferior to the C1-C2 articulation and can be injured during extensive lateral dissection.
2. Reduction of the Dislocation
- Assessment of Deformity: Intraoperative fluoroscopy or lateral radiographs are used to confirm the nature and degree of dislocation.
- Gentle Traction and Extension: Using the Mayfield clamp, controlled traction and slight extension of the head are typically applied. The goal is to gently reduce the occipital condyles back onto the C1 facets, or at least to a stable, near-anatomical alignment that allows for safe instrumentation and avoids spinal cord compression.
- Direct Manipulation: In some cases, gentle manual manipulation of the occiput relative to C1 and C2 may be required. This must be performed under direct visualization and with continuous neuromonitoring.
- Maintain Reduction: Once reduction is achieved, it is crucial to maintain it with skull traction or a temporary holding device while instrumentation is applied.
3. Instrumentation and Fixation (Occipital-C2 Fusion)
Occipital-C2 (O-C2) fusion is the standard for OAD stabilization.
*
Occipital Fixation:
*
Occipital Plate or Screws:
The most common method involves placing screws into the occipital bone. The preferred region for screw placement is typically 2-3 cm lateral to the midline, 1 cm inferior to the superior nuchal line, avoiding the midline sagittal sinus and lateral transverse sinuses. Bicortical purchase is generally preferred but requires careful depth measurement to avoid intracranial penetration.
* A pre-contoured occipital plate can be secured with multiple screws.
*
C2 Fixation:
C2 offers the most robust fixation points in the upper cervical spine.
*
C2 Pedicle Screws:
These provide the strongest and most rigid fixation. Pedicle screws are inserted through the lateral mass/pars of C2, angled medially and superiorly, aiming towards the C2 body. Pre-operative CT analysis is crucial to determine pedicle dimensions and trajectory to avoid the vertebral artery (which lies lateral to the pedicle) and spinal canal.
*
C2 Translaminar Screws:
An alternative to pedicle screws, especially if pedicles are small or compromised. These screws are placed from the dorsal aspect of the C2 lamina, directed anteriorly and medially into the contralateral lamina. They offer excellent biomechanical stability but require meticulous technique to avoid spinal cord injury.
*
C1 Lateral Mass Screws (Optional):
While O-C2 fusion is typically sufficient, if C1 is intact and robust, C1 lateral mass screws can be added to the construct, extending the fixation points and potentially enhancing stability. However, in OAD, C1 is often disrupted and may not offer reliable screw purchase.
*
Rod Placement:
Once screws are placed, pre-contoured or malleable rods are connected between the occipital plate/screws and the C2 screws. The rods are carefully bent to match the normal cervicothoracic kyphosis or lordosis, maintaining the desired alignment. Locking nuts are applied and tightened to secure the construct.
*
Compression/Distraction:
Gentle compression or distraction can be applied to further enhance stability and facilitate reduction, depending on the injury pattern.
4. Bone Grafting
- Decortication: The posterior occipital squama, C1 posterior arch (if present and stable), and C2 lamina/lateral masses are carefully decorticated using a high-speed burr. This creates a bleeding bony bed for fusion.
- Graft Placement: Autogenous bone graft (typically harvested from the iliac crest) is the gold standard due to its osteoconductive, osteoinductive, and osteogenic properties. Allograft or bone graft substitutes may also be used. The graft is meticulously packed over the decorticated surfaces of the occiput, C1, and C2, ensuring maximal contact with the fusion bed and the instrumentation.
5. Closure
- Hemostasis: Thorough irrigation and meticulous hemostasis are performed. A drain is usually placed in the deep fascial layer to prevent hematoma formation.
- Layered Closure: The paraspinal muscles, fascia, subcutaneous tissue, and skin are closed in layers. Muscle reattachment to the spinous processes is important to minimize dead space and support fusion.
Throughout the procedure, intermittent fluoroscopy or X-rays are used to confirm hardware position and alignment. Intraoperative neuromonitoring alerts the surgeon to any potential compromise to the spinal cord or nerve roots, allowing for immediate corrective action.
Complications & Management
Occipitoatlantal dislocation surgery, while life-saving, is associated with a range of potential complications, given the critical anatomical region. Meticulous surgical technique and prompt recognition and management of complications are crucial for optimizing patient outcomes.
Intraoperative Complications
| Complication | Incidence | Salvage/Management Strategies |
|---|---|---|
| Vertebral Artery (VA) Injury | Rare (1-2%) | Immediate direct compression with Gelfoam/thrombin, local hemostatics. If bleeding persists, consider vessel ligation (only if contralateral VA is patent), endovascular stenting/coiling. Urgent neurovascular consultation. |
| Spinal Cord Injury (SCI) | Rare | Immediate intraoperative assessment via neuromonitoring (SSEP/MEP changes). Re-evaluate reduction/decompression. Remove/reposition offending hardware. Consider intraoperative MRI. Administer steroids (controversial). |
| Cranial Nerve (CN) Injury | Variable | Recognize CN XII (hypoglossal) and CN XI (accessory) vulnerability. Avoid aggressive lateral dissection around the jugular foramen. If identified, protect. Post-op: symptomatic management, ENT/neurology consultation. |
| Dural Tear / CSF Leak | 2-5% | Primary repair with suture (non-absorbable), dural substitute patch (fascia, synthetic), fibrin glue. Placement of a lumbar drain for 3-5 days. Strict bed rest post-op. |
| Inadequate Reduction | Variable | If recognized intraoperatively, adjust traction, re-manipulate, and reposition hardware. If stable but not anatomical, proceed with fusion to prevent further displacement. |
| Hardware Malposition | <1% | If detected by fluoroscopy/X-ray, remove and redirect screw. Careful trajectory planning is vital for C2 pedicle screws. |
| Excessive Blood Loss | Common | Pre-operative crossmatch, rapid transfusion protocols. Meticulous hemostasis, judicious use of bipolar cautery and hemostatic agents. |
Post-Operative Complications
| Complication | Incidence | Salvage/Management Strategies |
|---|---|---|
| Surgical Site Infection | 1-5% | Superficial: Oral/IV antibiotics, local wound care. Deep: Urgent surgical debridement, IV antibiotics, hardware retention if stable and fusion is progressing, or removal/revision if unstable/non-union. Vacuum-assisted closure (VAC) may be needed. |
| Pseudarthrosis / Non-union | 5-10% | Revision surgery with repeat decortication, generous autologous bone grafting, rigid hardware augmentation, and possibly biological adjuncts (e.g., BMP). |
| Hardware Failure | <5% | Revision surgery. May involve replacing broken hardware, extending fusion levels, or using stronger constructs. Addressing pseudarthrosis if present. |
| Persistent/Worsening Neurological Deficit | Variable | Urgent post-operative MRI to rule out hematoma, cord compression, or stroke (if VA injury suspected). Neurology/neurosurgery consultation. Consider revision if mechanical cause. |
| Dysphagia / Dysphonia | Common (transient) | Speech therapy, dietary modifications. Usually resolves over weeks to months. If persistent, ENT evaluation for vocal cord or swallowing dysfunction. |
| Neck Pain / Restricted ROM | Expected | Expected outcome of fusion. Manage with analgesia, physical therapy focusing on adjacent segments. |
| Post-operative Hematoma | <1% | Urgent surgical evacuation to prevent cord compression and neurological deterioration. Ensure adequate drain function. |
| Vertebral Artery Thrombosis/Stroke | Rare | Immediate neurological consultation, anti-coagulation (if not contraindicated by active bleeding), neurovascular intervention. Long-term anti-platelet therapy. |
General Principles of Complication Management
- Prevention: Meticulous pre-operative planning, precise surgical technique, thorough knowledge of anatomy, intraoperative neuromonitoring, and strict adherence to perioperative protocols are the best defenses.
- Early Recognition: High index of suspicion for potential complications, regular neurological assessments, and vigilant monitoring in the post-operative period.
- Multidisciplinary Approach: Management often requires the expertise of neurosurgeons, neurologists, intensivists, interventional radiologists, and rehabilitation specialists.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation following occipital-cervical fusion for OAD is a critical, multi-stage process designed to promote successful arthrodesis, manage pain, optimize neurological recovery, and facilitate functional independence. The protocols must be tailored to the individual patient's neurological status, overall health, and the extent of their injury.
Immediate Post-Operative Period (Weeks 0-6)
-
Spinal Immobilization:
- Rigid Cervical Collar: Patients typically remain in a rigid cervical collar (e.g., Miami J, Aspen) for 6 to 12 weeks post-surgery. This provides external support and helps protect the fusion site during the early healing phase.
- Halo Vest (Rarely Primary): While a halo vest offers greater immobilization, its use as a primary post-operative immobilization after O-C2 fusion for OAD is less common given the robust internal fixation. It may be considered in cases of compromised fixation, poor bone quality, or severe non-compliance, but its risks (pin site infection, dural penetration) must be weighed.
- Strict Spinal Precautions: Emphasize log-rolling for transfers, avoiding active neck flexion/extension/rotation, and limiting twisting or bending.
- Pain Management: Aggressive pain control with a multimodal approach (opioids, NSAIDs, muscle relaxants, neuropathic agents) is essential for comfort and to facilitate early mobilization.
- Wound Care: Regular monitoring of the surgical incision for signs of infection or CSF leak. Drain management per surgical protocol.
-
Early Mobilization:
- Out-of-bed activity: As soon as hemodynamically stable, patients are encouraged to sit up and ambulate with assistance, maintaining spinal precautions.
- Deep Vein Thrombosis (DVT) Prophylaxis: Pharmacological and mechanical prophylaxis (sequential compression devices) are vital, especially for patients with neurological deficits or prolonged bed rest.
- Respiratory Care: Especially important for patients with pre-existing or new respiratory compromise (e.g., phrenic nerve involvement). Incentive spirometry, chest physiotherapy, and close monitoring of respiratory mechanics.
Early Rehabilitation Phase (Weeks 6-12)
- Radiographic Assessment: Plain X-rays (lateral, AP, open-mouth odontoid) are obtained at regular intervals (e.g., 6 weeks, 12 weeks) to assess alignment and monitor early signs of fusion (trabecular bridging). A CT scan may be performed around 3-6 months post-op to definitively assess fusion.
-
Physical Therapy (PT) – Restricted:
- Focus on Trunk and Lower Extremity Strengthening: Exercises to improve core stability, balance, and lower extremity strength, avoiding any direct neck movements.
- Shoulder Girdle Strengthening: Gentle isometric exercises for the shoulder girdle and upper back to improve posture and muscle support.
- Range of Motion (ROM): Begin very gentle, passive range of motion of the shoulder and upper extremities, ensuring the cervical spine remains protected.
-
Occupational Therapy (OT):
- Activities of Daily Living (ADLs): Training on adaptive techniques for dressing, hygiene, and feeding while maintaining cervical precautions.
- Home Safety Assessment: Recommendations for home modifications to prevent falls and accommodate activity restrictions.
- Neurological Rehabilitation: For patients with neurological deficits, intensive physical and occupational therapy focused on functional recovery, gait training, fine motor skills, and addressing specific deficits (e.g., speech therapy for dysphagia/dysphonia, swallow therapy).
Advanced Rehabilitation Phase (Months 3-6)
- Fusion Confirmation: Once radiographic evidence of solid fusion is confirmed (typically by 3-6 months), the cervical collar may be gradually weaned.
-
Progressive Physical Therapy:
- Gradual Neck Range of Motion: Once the collar is removed and fusion is solid, very gentle active and passive range of motion exercises for the non-fused segments can begin, carefully avoiding excessive stress on the fused segment.
- Cervical and Thoracic Strengthening: Progressive strengthening of the cervical and thoracic musculature, focusing on endurance and stability.
- Proprioception and Balance Training: Important to restore balance and coordination affected by loss of C0-C2 motion.
-
Return to Activity:
- Gradual return to light daily activities.
- Heavy lifting, strenuous physical activity, and contact sports are generally avoided indefinitely. Patients should be counseled on permanent restrictions to protect the fusion.
Long-Term Follow-up
- Regular clinical and radiographic follow-up to monitor fusion integrity, detect adjacent segment degeneration (though less common in O-C2 fusion than lower cervical fusions), and manage any long-term pain or neurological issues.
- Patient education regarding the implications of their fusion, body mechanics, and warning signs of complications.
- Psychological support may be necessary for patients coping with permanent disability or significant life changes.
Summary of Key Literature / Guidelines
The understanding and management of occipitoatlantal dislocation have evolved significantly from post-mortem discoveries to contemporary, advanced diagnostic and surgical protocols. Early literature highlighted the universally fatal nature of OAD, with survivors representing extremely rare cases. The advent of modern imaging modalities, particularly high-resolution CT and MRI, has revolutionized its diagnosis, allowing for antemortem identification and timely intervention.
Diagnostic Criteria: Historically, plain radiographs offered limited sensitivity. The development of radiographic criteria such as the Powers Ratio (BC/OA > 1.0 indicating anterior OAD) and the measurement of the Basion-Dens Interval (BDI) and Basion-Axial Interval (BAI) significantly improved diagnostic capabilities on lateral plain films and later, CT scans. Current consensus utilizes these measurements, along with direct visualization of articular displacement on CT, as definitive diagnostic indicators. MRI has further enhanced diagnosis by clearly demonstrating the extent of ligamentous disruption (alar ligaments, tectorial membrane) and direct assessment of spinal cord integrity and associated vascular injuries.
Treatment Evolution: The treatment paradigm for OAD has unequivocally shifted towards aggressive surgical stabilization . Historically, traction or external immobilization (e.g., halo vest) were attempted but almost universally failed due to the severe inherent instability. The high rate of pseudarthrosis and continued neurological decline associated with non-operative management, coupled with the improved safety and efficacy of modern spinal instrumentation, cemented surgical fusion as the gold standard.
Surgical Principles:
Current guidelines from leading organizations like the
AO Spine
and
North American Spine Society (NASS)
strongly endorse
posterior occipital-cervical fusion (typically O-C2)
as the definitive treatment for OAD. Key surgical principles emphasize:
1.
Anatomical or near-anatomical reduction:
Achieved through careful traction and intraoperative manipulation.
2.
Rigid internal fixation:
Utilizing strong screw-rod constructs that securely anchor the occiput to C2. C2 pedicle screws or translaminar screws are preferred for their superior biomechanical strength.
3.
Solid arthrodesis:
Achieved with meticulous decortication and generous autologous bone grafting to ensure long-term stability and prevent pseudarthrosis.
Multidisciplinary Approach: The management of OAD survivors necessitates a multidisciplinary approach involving trauma surgeons, neurosurgeons, orthopedic spine surgeons, intensivists, neurologists, interventional radiologists, and rehabilitation specialists. This integrated care model addresses not only the spinal injury but also associated head trauma, vascular injuries, and profound neurological deficits.
Prognosis: While survival rates have improved with early diagnosis and aggressive surgical intervention, the prognosis for functional recovery remains guarded due to the severe initial injury to the brainstem and spinal cord. Early recognition, prompt and accurate diagnosis, and timely surgical stabilization are critical factors in maximizing the potential for neurological recovery, however limited. Continued research focuses on improving diagnostic accuracy in the acute setting, refining surgical techniques, and enhancing rehabilitation strategies for these complex, often devastating, injuries.