Introduction and Epidemiology
Skeletal muscle comprises approximately 40% of total human body mass and is the primary driver of locomotion, joint stability, and metabolic homeostasis. Within the orthopedic and surgical disciplines, a profound understanding of skeletal muscle physiology—specifically the differentiation, function, and pathology of Type II (fast-twitch) muscle fibers—is paramount. Type II fibers are optimized for rapid, high-force generation but are highly susceptible to fatigue, atrophy, and specific injury patterns.
Type II muscle fibers are subdivided into Type IIa (fast oxidative-glycolytic) and Type IIx/b (fast glycolytic). Epidemiologically, muscles with a high proportion of Type II fibers, such as the rectus femoris, hamstrings, and gastrocnemius, represent the most frequent sites of acute myotendinous strain injuries in athletic populations. Furthermore, Type II fibers are disproportionately affected by age-related sarcopenia, immobilization atrophy following orthopedic trauma, and denervation injuries.
Understanding the basic science of these contractile elements is not merely an academic exercise; it directly informs surgical decision-making in tendon transfers, the management of compartment syndrome, the mitigation of malignant hyperthermia, and the optimization of postoperative rehabilitation protocols. This comprehensive review synthesizes the cellular anatomy, biomechanics, and surgical implications of Type II skeletal muscle fibers.
Surgical Anatomy and Biomechanics
Cellular Anatomy and the Sarcolemma
The foundational unit of skeletal muscle is the myofiber, a multinucleated syncytium surrounded by a specialized plasma membrane known as the sarcolemma. The multiple nuclei are typically located peripherally, directly adjacent to the sarcolemma, which allows the central volume of the cell to be dedicated entirely to contractile machinery.

The sarcolemma extends deep into the intracellular environment, surrounding individual myofibrils to form the transverse tubules (T-tubules). This invagination is critical for the rapid propagation of action potentials from the neuromuscular junction to the deep interior of the muscle fiber, ensuring synchronous contraction of the entire cell.
The Sarcoplasmic Reticulum and Excitation Contraction Coupling
Adjacent to the T-tubules lies the Sarcoplasmic Reticulum (SR), a highly specialized smooth endoplasmic reticulum that surrounds the individual myofibrils. The primary function of the SR is the storage and rapid release of calcium ions ($Ca^{2+}$) in intracellular membrane-bound channels.
The physical and functional connection between the sarcolemma-derived T-tubules and the SR is mediated by Ryanodine receptors (specifically RYR-1 in skeletal muscle). These receptors regulate the release of calcium from the SR into the sarcoplasm. When an action potential travels down the T-tubule, it induces a conformational change in the dihydropyridine (DHP) receptor, which mechanically opens the RYR-1 channel, flooding the sarcoplasm with calcium and initiating the contractile cascade.

Abnormalities in the RYR-1 receptor are directly implicated in patients susceptible to malignant hyperthermia, a life-threatening hypermetabolic response to volatile anesthetic gases and depolarizing neuromuscular blockers (e.g., succinylcholine). In these patients, the RYR-1 channel remains open, leading to unregulated calcium release, sustained tetanic contraction, massive oxygen consumption, and rhabdomyolysis. The pharmacological antidote, dantrolene, functions by directly inhibiting the RYR-1 receptor, thereby decreasing the pathological loss of calcium from the SR.
Contractile Elements and Sarcomere Organization
The basic functional unit of muscle contraction is the sarcomere. Myofibrils consist of a set of sarcomeres arranged in series, parallel to the longitudinal axis of the cell. Individual myofibrils are typically 1 to 3 µm in diameter and can be up to 1 to 2 cm long. The highly organized, overlapping arrangement of these sarcomeres causes the characteristic banding pattern (striations) observed in skeletal muscle under light microscopy.

The sarcomere is structurally defined by the following elements:
* Z disc (or Z line): Represents the terminus of the sarcomere. It contains structural proteins including desmin, α-actinin, and filamin. The costamere complex connects the sarcomere to the sarcolemma at the Z disc, transmitting the force of contraction to the extracellular matrix.
* A-band (Dark band): Represents the thick filaments. These are primarily composed of myosin. The A-band also contains the H-band (the central region containing only thick filaments), M protein, C protein, titin (a massive elastic protein that provides passive tension), and creatine kinase.
* I-band (Light band): Represents the thin filaments. These are primarily composed of actin. The thin filaments also contain the regulatory proteins troponin (which possesses the binding site for calcium) and tropomyosin (which physically blocks the myosin-actin interaction site in the resting state). The thin filaments attach directly to the Z disc.
During contraction, calcium binds to troponin C, causing a conformational shift that moves tropomyosin away from the myosin-binding sites on the actin filament. Myosin heads then bind to actin, and through the hydrolysis of ATP, execute a "power stroke" that pulls the Z discs closer together, shortening the sarcomere.
Gross Anatomy and Connective Tissue Hierarchy
Skeletal muscle is organized into a hierarchical structure of connective tissue investments that facilitate force transmission, provide a conduit for neurovascular structures, and allow for independent sliding of muscle fascicles.
* Endomysium: A delicate layer of reticular fibers surrounding individual myofibers.
* Perimysium: Surrounds bundles of muscle fibers to form individual muscle fascicles (containing hundreds of muscle fibers).
* Epimysium: A more delicate connective tissue layer that surrounds the entire muscle belly, encompassing all the fascicles.
* Fascia: A tough, dense irregular connective tissue that covers the muscle, segregating it into compartments and allowing it to slide against adjacent structures.

Proprioception and Neuromuscular Control
Muscle function is tightly regulated by the central nervous system via specialized stretch receptors:
* Muscle Spindles: Located in parallel within the muscle belly. They detect changes in muscle length and the rate of change in length, transmitting this information to the CNS to control muscle stiffness and mediate the myotatic (stretch) reflex.
* Golgi Tendon Organs (GTOs): Located in series at the myotendinous junction. They detect changes in muscle tension. When tension becomes excessive, GTOs trigger an inhibitory interneuron in the spinal cord, causing the muscle to relax and protecting the tendon from avulsion or rupture.
Indications and Contraindications
Surgical intervention involving skeletal muscle—particularly Type II dominant muscles—is frequently indicated in the context of traumatic ruptures, compartment syndrome, and reconstructive tendon transfers. Understanding the physiological capacity of these muscles is critical for determining operative candidacy.
| Clinical Scenario | Operative Indications | Non-Operative Indications |
|---|---|---|
| Acute Myotendinous Rupture (e.g., Distal Biceps, Quadriceps) | Complete rupture with significant retraction; loss of functional strength; high-demand patient. | Partial tears; low-demand or medically unfit patient; minimal functional deficit. |
| Tendon Transfer Surgery | Irreversible nerve palsy (e.g., radial nerve palsy); massive irreparable rotator cuff tear; muscle must have MRC grade 4 or 5 strength. | Target muscle strength < MRC grade 4; fixed joint contracture; lack of passive ROM; progressive neurological disease. |
| Acute Compartment Syndrome | Intracompartmental pressure within 30 mmHg of diastolic blood pressure (Δp < 30); progressive neurological deficit. | Ambiguous clinical picture with normal continuous pressure monitoring; missed compartment syndrome (>24-48 hours) with established myonecrosis. |
| Muscle Biopsy for Myopathy | Suspected inflammatory myopathy, muscular dystrophy, or RYR-1 mutation confirmation. | Bleeding diathesis (relative); active local infection. |

Pre Operative Planning and Patient Positioning
Physiological Assessment and Templating
Preoperative planning for muscle-centric surgeries, particularly tendon transfers, requires meticulous assessment of muscle physiology. The donor muscle must possess adequate strength, amplitude (excursion), and a matching phase of activity if possible. Because Type II fibers are capable of generating high peak forces but fatigue rapidly, substituting a Type II dominant muscle (e.g., Latissimus dorsi) for a Type I dominant muscle (e.g., Infraspinatus) requires significant postoperative neuromuscular re-education to induce fiber-type shifting.
Electromyography (EMG) and Nerve Conduction Studies (NCS) are critical adjuncts in preoperative planning to confirm the denervation of the target muscle and the absolute viability of the proposed donor muscle.

Malignant Hyperthermia Precautions
Given the direct link between RYR-1 mutations and malignant hyperthermia, a thorough preoperative anesthetic history is mandatory. If a patient has a known history or strong family history of MH, the surgical facility must be equipped with a non-triggering anesthetic setup (Total Intravenous Anesthesia - TIVA) and an adequate supply of Dantrolene sodium.
Patient Positioning and Ischemia Time
Positioning must account for the susceptibility of large, Type II fiber-rich muscle groups (such as the gluteus maximus and quadriceps) to crush injury and rhabdomyolysis during prolonged surgical procedures. Bony prominences must be padded, and in cases requiring tourniquet use, ischemia time should be strictly monitored. Type II fibers, relying heavily on anaerobic glycolysis, tolerate ischemia slightly better than highly oxidative Type I fibers in the short term, but prolonged ischemia (>2 hours) leads to irreversible mitochondrial damage, depletion of ATP, failure of the SR calcium pumps, and subsequent myonecrosis.

Detailed Surgical Approach and Technique
Surgical techniques involving skeletal muscle must respect the hierarchical connective tissue anatomy to optimize functional recovery and minimize iatrogenic denervation or devascularization.
Muscle Sparing Approaches and Internervous Planes
When exposing deep orthopedic structures, utilizing internervous planes is the gold standard. An internervous plane exploits the boundary between two muscles supplied by different peripheral nerves, ensuring that neither muscle is denervated during retraction.
For example, the classic anterior (Smith-Petersen) approach to the hip utilizes the internervous plane between the Sartorius (Femoral nerve) and the Tensor Fasciae Latae (Superior Gluteal nerve) superficially, and the Rectus Femoris (Femoral nerve) and Gluteus Medius (Superior Gluteal nerve) deeply.
During dissection, the surgeon must carefully incise the investing fascia and respect the epimysium. Aggressive stripping of the epimysium or violation of the perimysium leads to structural damage to the fascicles, subsequent fibrosis, and tethering that limits the functional excursion of the muscle postoperatively.

Principles of Tendon Transfer Surgery
When executing a tendon transfer, the biomechanical properties of the muscle—dictated heavily by its fiber type and sarcomere arrangement—must be meticulously managed.
- Mobilization: The donor muscle is mobilized by releasing its fascial attachments. The neurovascular pedicle must be identified and protected. The epimysium should remain intact to allow smooth gliding in the new subcutaneous or subfascial tunnel.
- Routing: The muscle-tendon unit is routed to its new insertion site. A straight line of pull is biomechanically superior.
- Tensioning: This is the most critical step, governed by the length-tension relationship of the sarcomere (the Blix curve). Maximum active force is generated at the muscle's resting length, where there is optimal overlap between the thick (myosin) and thin (actin) filaments.
- If tensioned too loosely, the sarcomeres are overly shortened, and the muscle cannot generate sufficient force.
- If tensioned too tightly, the sarcomeres are overstretched, minimizing cross-bridge overlap and drastically reducing active force generation.
- Fixation: The tendon is secured to the target bone or tendon using interference screws, suture anchors, or a Pulvertaft weave. Rigid fixation is required to allow for early protected mobilization, which is essential for preventing adhesions and optimizing the remodeling of Type II fibers.

Complications and Management
Surgical and pathological complications affecting skeletal muscle can be devastating, leading to permanent contracture, weakness, or systemic decompensation.
Malignant Hyperthermia
As previously discussed, MH is a hypermetabolic crisis triggered by anesthetic agents in genetically susceptible individuals (RYR-1 mutation).
* Signs: Unexplained tachycardia, rapid increase in end-tidal $CO_2$, muscle rigidity (especially masseter spasm), and hyperthermia (a late sign).
* Management: Immediately discontinue volatile anesthetics. Hyperventilate with 100% oxygen. Administer Dantrolene (2.5 mg/kg IV push, repeated as necessary). Initiate active cooling protocols and manage hyperkalemia and acidosis.

Delayed Onset Muscle Soreness and Rhabdomyolysis
Delayed-onset muscle soreness (DOMS) is primarily associated with eccentric loading of Type II muscle fibers, leading to ultrastructural damage at the Z-disc and an inflammatory cascade. While DOMS is a benign, self-limiting condition, severe eccentric overload or prolonged ischemic crush injury can precipitate rhabdomyolysis. The destruction of the sarcolemma releases myoglobin into the systemic circulation, which can precipitate in the renal tubules, leading to acute kidney injury. Management requires aggressive intravenous hydration, urine alkalinization, and monitoring of serum creatine kinase (CK) and electrolyte levels.
Hemophilic Arthropathy and Intramuscular Hemorrhage
Patients with bleeding disorders, such as hemophilia, present a unique challenge regarding muscle and joint pathology. Recurrent intra-articular and intramuscular bleeding severely compromises muscle function.

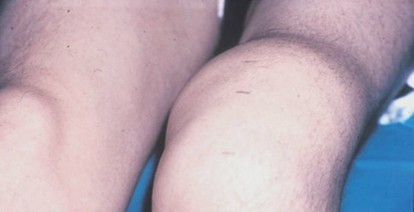
Recurrent knee effusions and synovitis are hallmarks of hemophilic arthropathy. The synovial proliferation observed in hemophilic arthropathy demonstrates phagocytic (type A) synovial cells laden with iron pigment (hemosiderin). Notably, histological examination typically reveals no giant cells, polymorphonuclear leukocytes, and only rare lymphocytes, distinguishing it from inflammatory or infectious arthritides.

The toxic effect of iron on the articular cartilage leads to early, severe joint destruction. Radiographs of end-stage arthropathy demonstrate profound joint space narrowing, subchondral cyst formation, and squaring of the condyles. In the ankle, a bloody effusion may be the initial presentation in a teenager with a familial history of bleeding disorders. End-stage hemophilic arthropathy of the ankle demonstrates characteristic flattening of the talus.

Intramuscular hematomas in these patients can rapidly increase compartmental pressures, leading to focal muscle necrosis, fibrosis, and severe contractures (e.g., hemophilic pseudotumor or Volkmann's ischemic contracture). Surgical intervention must be meticulously coordinated with hematology for factor replacement therapy.
| Complication | Incidence | Clinical Presentation | Salvage Strategy / Management |
|---|---|---|---|
| Malignant Hyperthermia | 1:10,000 to 1:50,000 anesthetics | Elevated $ETCO_2$, rigidity, tachycardia. | Dantrolene IV, cooling, correct acidosis. |
| Acute Compartment Syndrome | Varies by fracture type (e.g., up to 10% in tibial shaft fx) | Pain out of proportion, pain with passive stretch, tense compartments. | Emergent four-compartment fasciotomy. |
| Tendon Transfer Rupture / Failure | 2% - 5% | Sudden loss of function, palpable gap, lack of active ROM. | Revision surgery, possible interposition graft, or joint arthrodesis. |
| Hemophilic Pseudotumor | 1% - 2% of severe hemophiliacs | Expanding, painless intramuscular mass with bony erosion. | Factor replacement, surgical excision vs. embolization. |

Post Operative Rehabilitation Protocols
Rehabilitation following muscle injury or surgical manipulation must be phased to respect the biological healing process while specifically targeting the recovery of Type II fiber function.
Phase I: Protection and Early Cellular Healing (Weeks 0-4)
During the initial postoperative phase, the focus is on protecting the surgical repair (e.g., tendon transfer or myotendinous repair). The inflammatory phase is followed by early fibroplasia. Uncontrolled tensile loading during this phase can lead to gap formation or catastrophic failure. Passive range of motion (ROM) is initiated within safe zones to prevent dense adhesion formation between the epimysium and surrounding fascia.
Phase II: Active Range of Motion and Motor Control (Weeks 4-8)
As the collagen matrix matures and cross-links, active ROM is initiated. For tendon transfers, this phase requires significant cortical remapping. Biofeedback and neuromuscular electrical stimulation (NMES) are frequently utilized to help the patient recruit the newly transferred muscle in its new functional phase.

Phase III: Strengthening and Type II Fiber Recruitment (Weeks 8-16+)
To maximize strength and reverse disuse atrophy, rehabilitation must specifically target Type II fibers. Because Type II fibers are recruited only during high-intensity or high-velocity contractions (following the Henneman Size Principle), low-resistance, high-repetition exercises are insufficient.
* Eccentric Loading: Eccentric contractions generate higher forces than concentric or isometric contractions and are highly effective at stimulating Type II fiber hypertrophy.
* Plyometrics: Rapid stretch-shortening cycles utilize the myotatic reflex (muscle spindles) to maximize motor unit recruitment and enhance the rate of force development.
Summary of Key Literature and Guidelines
The foundational understanding of skeletal muscle physiology in orthopedic surgery is derived from several key areas of research:
- Muscle Architecture and Tendon Transfers: The pioneering work of Paul Brand established the biomechanical principles of tendon transfer surgery. His research defined the critical importance of matching muscle excursion (proportional to fiber length) and force generation (proportional to physiological cross-sectional area) when selecting donor muscles.
- Malignant Hyperthermia Guidelines: The Malignant Hyperthermia Association of the United States (MHAUS) provides the definitive, evidence-based guidelines for the recognition and management of MH crises. The identification of the RYR-1 mutation as the primary pathophysiological driver has revolutionized preoperative screening and pharmacological management with Dantrolene.
- Hemophilic Arthropathy Management: Literature by Rodriguez-Merchan and others highlights the pathophysiology of iron-induced synovial proliferation and the necessity of aggressive, early factor prophylaxis to prevent the irreversible musculoskeletal complications of hemophilia, including end-stage arthropathy and intramuscular hematoma-induced contractures.

A rigorous comprehension of the cellular anatomy, contractile mechanisms, and pathological susceptibilities of skeletal muscle ensures that the orthopedic surgeon can optimize operative techniques, anticipate physiological complications, and maximize functional outcomes in the postoperative period.
Clinical & Radiographic Imaging