PRINCIPLES OF MUSCLE AND MUSCULOCUTANEOUS FREE FLAPS
The advent of microvascular surgery has revolutionized the orthopaedic approach to complex extremity reconstruction. Muscle and musculocutaneous free flaps serve two primary, indispensable roles in modern reconstructive microsurgery. First, they are widely applied for the robust coverage of extensive soft tissue defects in the upper and lower extremities, particularly when exposed bone, tendon, or orthopaedic hardware necessitates highly vascularized tissue to promote healing and combat infection. They are equally valuable in reconstructing contour deficits in the head, neck, and trunk.
Second, these flaps are critical in the transfer of functioning neuromuscular units to replace paralyzed or irreparably damaged muscular units in the extremities and face (Functional Muscle Transfer).
When selecting a flap, the surgeon must consider the long-term biomechanical and physiological behavior of the transferred tissue. Muscle flaps that are not reinnervated will predictably undergo denervation atrophy over time, significantly diminishing in bulk. This characteristic can be advantageous when initial bulk is required to obliterate dead space, but a more contoured, less prominent long-term result is desired. Furthermore, donor site functional deficits following the harvest of workhorse muscle flaps (such as the latissimus dorsi or rectus abdominis) are usually insignificant due to the synergistic compensation of adjacent musculature.
Clinical Pearl: Unless the restoration of local sensibility is strictly required (e.g., weight-bearing heel reconstruction), a muscle flap covered with a split-thickness skin graft is often preferable to a bulky cutaneous flap for free tissue transfer, as it contours better to extremity defects and provides superior vascularity to ischemic or infected recipient beds.
THE LATISSIMUS DORSI TRANSFER
Building on the foundational 1896 reports of Tansini—who first utilized the latissimus dorsi muscle pedicle flap for breast reconstruction—and the subsequent extensive use of the latissimus dorsi as a pedicled flap for trunk, head, and neck reconstruction, pioneers such as Baudet et al., Harii et al., and Maxwell et al. demonstrated the successful transfer of the latissimus dorsi muscle as a microvascular free flap.
Today, this myocutaneous free flap is utilized extensively for formidable soft tissue coverage problems. Its enduring popularity stems from its massive surface area, consistent anatomy, and a long, highly reliable vascular pedicle with an adequate vessel diameter that facilitates straightforward microvascular anastomosis.
Vascular Anatomy and Innervation
The latissimus dorsi is classified as a Type V muscle flap according to the Mathes and Nahai classification, meaning it possesses one dominant vascular pedicle and secondary segmental vascular pedicles.
Arising from the thoracolumbar fascia, the posterior iliac crest, and the lower three or four ribs, the latissimus dorsi muscle covers the majority of the lower posterior trunk. Its fibers converge as they pass superolaterally to insert on the inferior portion of the bicipital groove of the humerus.
The principal (dominant) vascular supply to the latissimus dorsi is the thoracodorsal artery, a direct continuation of the subscapular artery, accompanied by its venae comitantes. The subscapular artery originates from the third part of the axillary artery. As the thoracodorsal artery courses distally, it runs just deep to the anterior margin of the latissimus dorsi and enters the muscle on its deep surface, typically 8 to 12 cm distal to the humeral insertion.
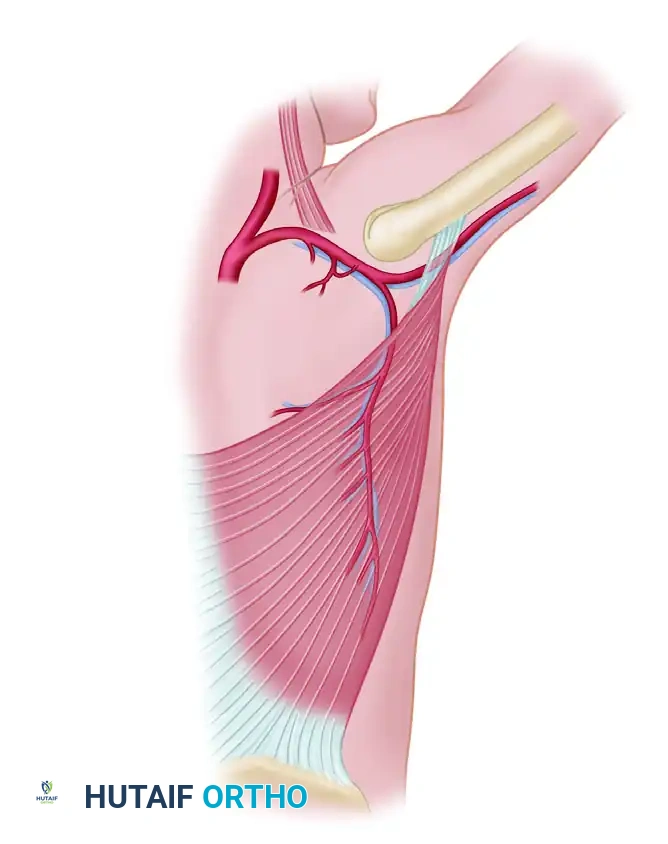
If the thoracodorsal artery is harvested below the takeoff of the circumflex scapular branch, it yields a vessel diameter of 1.5 to 3.0 mm, which is ideal for microvascular anastomosis. The pedicle length can reach up to 10 to 15 cm if dissected to the axillary artery.
Secondary vascular pedicles enter the muscle medially and inferiorly; these are the perforating branches of the lumbar and posterior intercostal arteries. While these secondary pedicles can support the muscle in pedicled reverse-turnover flaps, they are divided during free flap harvest.
The motor innervation is supplied by the thoracodorsal nerve (C6, C7, C8), a branch of the posterior cord of the brachial plexus. The nerve closely follows the thoracodorsal artery and typically consists of two to three fascicles with a diameter of approximately 2 mm.
Surgical Warning: When harvesting the latissimus dorsi for Functional Muscle Transfer (FMT), preserving the maximal length of the thoracodorsal nerve is paramount. The nerve should be dissected proximally into the axilla to ensure tension-free coaptation at the recipient site.
All of the muscle, along with most of its overlying skin, can be transferred safely based on the thoracodorsal neurovascular bundle. Skin paddles of varying sizes and orientations (transverse, oblique, or vertical) may be designed over the muscle as dictated by the recipient site defect.
Surgical Technique 63-17: Latissimus Dorsi Harvest
The harvest of the latissimus dorsi requires meticulous positioning and a systematic approach to the axillary anatomy.
Positioning and Preparation:
* Place the patient in the lateral decubitus position, maintaining this posture securely with a beanbag, sandbags, and kidney rests. Ensure an axillary roll is placed under the contralateral axilla to protect the dependent brachial plexus.
* Prepare and drape the patient widely, leaving the entire shoulder, axilla, and thorax exposed anteriorly and posteriorly. The ipsilateral arm should be draped free to allow for passive manipulation during axillary dissection.
Incision and Exposure:
* Design the skin paddle (if a myocutaneous flap is planned) over the central or anterior-superior portion of the muscle, where the musculocutaneous perforators are most dense.
* Make an incision along the anterior border of the latissimus dorsi, extending from the axilla down toward the iliac crest.
* Elevate the skin and subcutaneous tissue off the latissimus fascia posteriorly and anteriorly, taking care not to shear the skin paddle off the underlying muscle.
Pedicle Dissection:
* Retract the anterior edge of the latissimus dorsi laterally and posteriorly to expose the areolar tissue between the latissimus and the serratus anterior muscle.
* Identify the thoracodorsal neurovascular bundle on the deep surface of the muscle.
* Trace the pedicle proximally toward the axilla. Identify and ligate the branch to the serratus anterior.
* Continue proximal dissection to the junction with the circumflex scapular artery. If maximum pedicle length is required, the circumflex scapular artery can be ligated, allowing the pedicle to be traced up to the subscapular artery's origin from the axillary artery.
Muscle Elevation and Harvest:
* Divide the posterior and inferior attachments of the latissimus dorsi from the thoracolumbar fascia and iliac crest using electrocautery.
* Elevate the muscle from distal to proximal, separating it from the underlying ribs and serratus anterior.
* Divide the humeral insertion of the muscle near the bicipital groove.
* Once the flap is isolated entirely on its neurovascular pedicle, allow it to perfuse for 15 to 20 minutes before ligating the thoracodorsal artery and vein.
* Close the donor site over large-bore suction drains. If a large skin paddle was harvested (typically >10 cm in width), primary closure may not be possible, and a split-thickness skin graft over the muscle bed may be required.
THE LATERAL ARM FREE FLAP
While the latissimus dorsi is a premier muscle flap, orthopaedic reconstruction frequently requires thinner, pliable fasciocutaneous tissue. The lateral arm flap, based on the posterior radial collateral artery (PRCA), is a highly versatile septocutaneous free flap often utilized for moderate-sized defects of the hand, foot, and head/neck.
The lateral arm flap provides thin, sensate tissue and can be harvested as a composite flap including a segment of the humerus or triceps tendon.
Vascular Anatomy
The lateral arm flap is perfused by the posterior radial collateral artery (PRCA), a terminal branch of the profunda brachii artery. The PRCA courses within the lateral intermuscular septum of the arm, between the brachialis/brachioradialis anteriorly and the triceps posteriorly. The venous drainage is via the venae comitantes of the PRCA, which drain into the profunda brachii veins.
Sensory innervation to the flap is provided by the posterior cutaneous nerve of the arm and the posterior cutaneous nerve of the forearm, both branches of the radial nerve.
Surgical Technique: Lateral Arm Flap Harvest
Incision and Anterior Dissection:
* A line is drawn from the deltoid insertion to the lateral epicondyle, representing the lateral intermuscular septum. The skin paddle is designed centered over or slightly posterior to this axis.
* Incise the anterior margin of the flap. Elevate the skin and underlying deep fascia off the brachialis and brachioradialis muscles, dissecting posteriorly until the lateral intermuscular septum is reached.
Septal Dissection and Nerve Protection:
* Bluntly separate the septum from the triceps to its full depth. During this separation, carefully note the course of the posterior radial collateral artery within the septum.
* Identify and meticulously protect the radial nerve, which lies deep to the septum and anterior to the lateral head of the triceps.
* Identify the posterior cutaneous nerve of the arm, which enters the skin flap proximally and superficial to the triceps fascia. This nerve may be divided proximally and used for sensory reinnervation at the recipient site.
Pedicle Isolation:
* Continue the dissection proximally along the vascular pedicle into the interval between the triceps and the deltoid.
* Ligate and divide the posterior radial collateral artery and its venae comitantes at the distal margin of the flap, just above the lateral epicondyle.
* Proximally, identify and divide the anterior radial collateral artery.
* When adequate pedicle length has been gained, divide the profunda brachii artery and its venae comitantes.
* The posterior cutaneous nerve to the forearm is usually included within the flap to provide sensibility, although it can be preserved if a sensate flap is not required.
Donor Site Closure:
* Close the wound primarily over suction drains if the flap width is less than 6 cm.
* If the defect is wider, primary closure may cause compartment syndrome of the arm or unacceptable tension; in such cases, apply a split-thickness skin graft.
Pitfall: Over-aggressive primary closure of a lateral arm donor site wider than 6 cm can lead to radial nerve compression or delayed wound healing. Always have a low threshold to utilize a skin graft if tension is excessive.
POSTOPERATIVE MICROVASCULAR PROTOCOLS
The success of muscle and musculocutaneous free flaps relies heavily on rigorous postoperative monitoring and physiological optimization.
- Hemodynamics and Environment: The patient must be kept normothermic, well-hydrated, and adequately oxygenated. The patient's room should be kept warm (ambient temperature > 24°C) to prevent peripheral vasospasm.
- Positioning: The reconstructed extremity must be elevated to promote venous drainage, but extreme care must be taken to ensure the limb is positioned so that absolutely no pressure or torsion is applied to the microvascular pedicle.
- Flap Monitoring: Clinical monitoring remains the gold standard. The flap should be assessed every hour for the first 24 to 48 hours, evaluating color, capillary refill, tissue turgor, and temperature.
- Adjunctive Monitoring: Implantable venous Doppler probes, surface handheld Dopplers, and near-infrared tissue oximetry (NIRS) are highly recommended to detect early vascular compromise.
- Anticoagulation: While protocols vary by institution, most microvascular surgeons employ a regimen of intravenous hydration, low-dose aspirin (81 mg daily), and prophylactic subcutaneous heparin to prevent microvascular thrombosis without inducing hematoma formation at the surgical sites.
