Management of Forearm Compartment Syndrome and Volkmann Ischemic Contracture
Key Takeaway
Acute forearm compartment syndrome is a surgical emergency requiring immediate fasciotomy to prevent irreversible ischemia and Volkmann contracture. Diagnosis relies on clinical suspicion and intracompartmental pressure monitoring, with a delta pressure of less than 30 mm Hg indicating impending ischemia. This guide details the step-by-step surgical execution of volar and dorsal forearm fasciotomies, intrinsic hand muscle releases, and the reconstructive management of established ischemic contractures.
Management of Acute Compartment Syndrome of the Forearm
Acute compartment syndrome (ACS) of the forearm is a devastating orthopedic emergency characterized by elevated intracompartmental pressures that compromise local tissue perfusion. If left untreated, the resulting ischemia leads to irreversible muscle and nerve necrosis, culminating in Volkmann ischemic contracture.
Diagnostic Thresholds and Indications for Fasciotomy
Impending tissue ischemia must be assumed when the intracompartmental tissue pressure reaches between 30 mm Hg and 10 mm Hg below the diastolic blood pressure (a concept known as the delta pressure, or ΔP). A delta pressure of less than 30 mm Hg is a strong, absolute indication that emergent fasciotomy should be recommended.
Clinical Pearl: In a hypotensive patient, the acceptable absolute compartment pressure is significantly lower due to decreased systemic perfusion pressure. The delta pressure (Diastolic BP - Compartment Pressure) remains the most reliable metric.
Mubarak, Owen, and Hargens established definitive criteria recommending that fasciotomy be performed in the following scenarios:
1. Normotensive patients with positive clinical findings, compartment pressures greater than 30 mm Hg, and a duration of increased pressure that is unknown or estimated to be longer than 8 hours.
2. Uncooperative or unconscious patients with an absolute compartment pressure greater than 30 mm Hg.
3. Hypotensive patients with a compartment pressure greater than 20 mm Hg.
As a cardinal rule in orthopedic traumatology: when in doubt, the compartment should be released. If a fasciotomy proves later to have been unnecessary, the patient is left with only a surgical scar. However, if a fasciotomy is indicated but withheld, catastrophic loss of muscle tissue, permanent neuropathy, and limb dysfunction will result.
McQueen, Gaston, and Court-Brown have extensively demonstrated that a delay in diagnosis is the single most important determining factor for a poor functional outcome. They strongly recommend continuous monitoring of compartment pressures in high-risk demographics, particularly young patients with high-energy injuries to the forearm diaphysis or distal radius, or in patients with significant soft-tissue crush injuries complicated by a bleeding diathesis.
Surgical Anatomy and Intervals
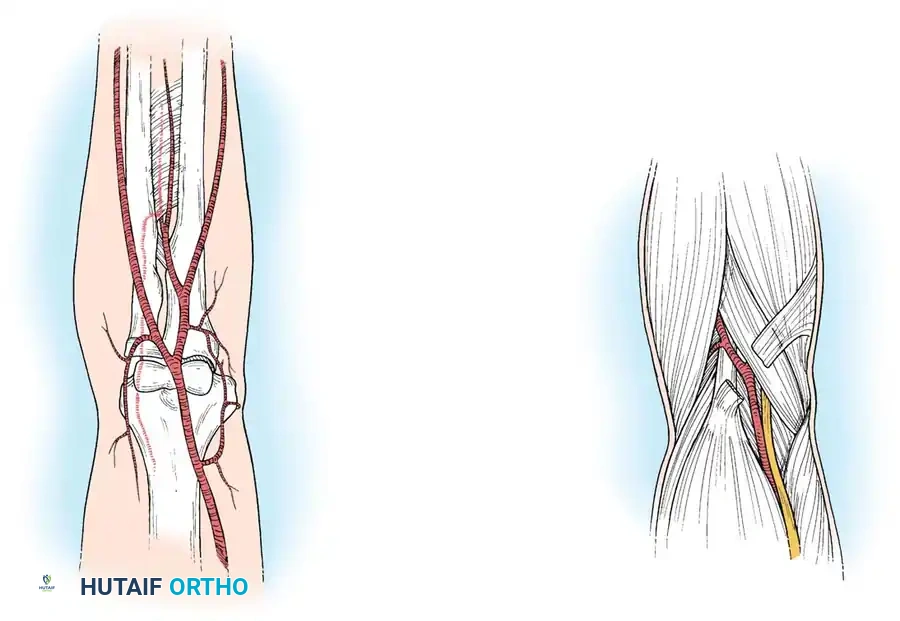
When performing a volar fasciotomy, a volar curvilinear incision is utilized. This extensile approach allows for the proximal release of the lacertus fibrosus (bicipital aponeurosis) and the distal release of the carpal tunnel.
The critical surgical interval for releasing both the deep and superficial volar compartments lies between the flexor carpi ulnaris (FCU) and the flexor digitorum sublimis (FDS). For the dorsal forearm, the fascia is released through the interval between the extensor carpi radialis brevis (ECRB) and the extensor digitorum communis (EDC). Cadaveric dissections by Ronel et al. have reaffirmed the anatomical safety and surgical advantages of utilizing these specific internervous and intermuscular planes.

Forearm Fasciotomy and Arterial Exploration (Technique 71-1)
The objective of this procedure is the complete decompression of all involved forearm compartments, exploration of the neurovascular bundles, and restoration of distal perfusion.
Volar Approach and Decompression
- Incision Planning: Make an anterior curvilinear incision similar to McConnell’s combined exposure of the median and ulnar neurovascular bundles, as popularized by Henry. Begin medial to the biceps tendon, crossing the elbow flexion crease at an oblique angle to prevent postoperative flexion contractures.
- Distal Extension: Carry the incision distally into the palm to allow for a complete carpal tunnel release. Strictly avoid crossing the wrist flexion crease at a right angle to prevent bowstringing and hypertrophic scarring.
- Proximal Release: Identify and divide the lacertus fibrosus proximally. Evacuate any underlying hematoma that may be compressing the brachial artery.
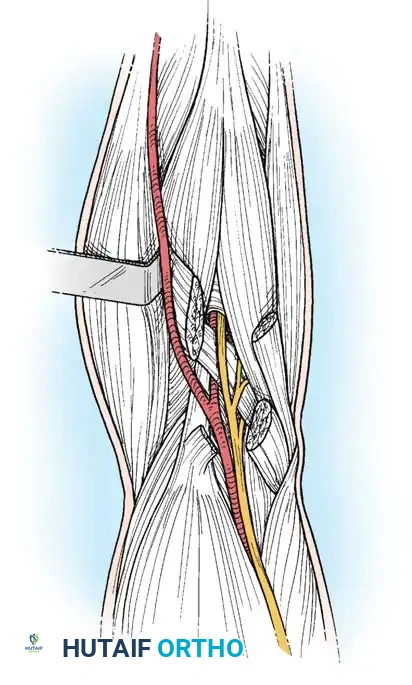
Arterial Exploration
- Vascular Assessment: In patients with a suspected brachial artery injury (e.g., associated with a supracondylar humerus fracture), expose the brachial artery and assess for free blood flow.
- Adventitial Resection: If flow is unsatisfactory, perform a careful arteriotomy or remove the adventitia to expose any underlying clot, arterial spasm, or intimal tear. Resect the damaged adventitia if necessary. If the vessel is irreparably damaged, proceed with primary anastomosis or an interposition vein graft.
Deep and Superficial Compartment Release
- Superficial Release: Release the superficial volar compartment throughout its entire length using open scissors, meticulously freeing the fascia overlying the superficial flexor musculature.
- Deep Compartment Exposure: Identify the FCU. Retract the FCU and its underlying ulnar neurovascular bundle medially. Simultaneously, retract the FDS and the median nerve laterally. This maneuver exposes the flexor digitorum profundus (FDP) within its deep compartment.
- Epimysiotomy: Inspect the overlying fascia and epimysium of the FDP. If it is tight, incise it longitudinally.
Surgical Warning: If the muscle appears gray, dusky, or non-contractile, the prognosis for functional recovery is guarded. However, borderline muscle may still be viable and should be allowed time to perfuse. Do not aggressively debride muscle during the index fasciotomy unless it is frankly necrotic and friable.
- Carpal Tunnel Release: Continue the dissection distally by incising the transverse carpal ligament along the ulnar border of the palmaris longus tendon and median nerve.
Median Nerve Assessment and Fracture Management
- Nerve Decompression: In cases presenting with median nerve palsy or paresthesias, trace the median nerve along the entire zone of injury. Ensure it is not severed, contused, or entrapped between the ulnar and humeral heads of the pronator teres. If entrapment is identified, a partial pronator tenotomy is mandatory.
- Skeletal Stabilization: In a patient with an unstable fracture (e.g., supracondylar fracture), reduce the fracture and stabilize it (typically with Kirschner wires or rigid internal fixation) to prevent further soft-tissue trauma and control osseous bleeding.
- Flap Coverage: If the median nerve is exposed within the distal forearm, suture a distally based radial forearm flap loosely over the nerve to prevent desiccation.
Dorsal Compartment Release
- Reassessment: Check the dorsal compartments clinically or repeat intracompartmental pressure measurements. While volar fasciotomy often decompresses the dorsal musculature sufficiently due to fascial continuity, persistent dorsal tension mandates a dorsal release.
- Dorsal Incision: Make a longitudinal incision distal to the lateral epicondyle, utilizing the interval between the EDC and ECRB, extending approximately 10 cm distally.
- Fascial Release: Gently undermine the subcutaneous tissue and release the fascia overlying the mobile wad of Henry and the extensor retinaculum.
Postoperative Aftertreatment
Do not attempt primary skin closure. The arm is elevated for 24 to 48 hours postoperatively. If delayed primary closure is not possible within 5 days due to persistent edema, a split-thickness skin graft should be applied.
Alternatively, gradual closure of fasciotomy wounds can be achieved using progressive tension with the vessel loop shoelace technique. Vessel loops are woven through the skin edges and tightened progressively during bedside dressing changes. Wound closure by this method can usually be accomplished within 2 weeks.
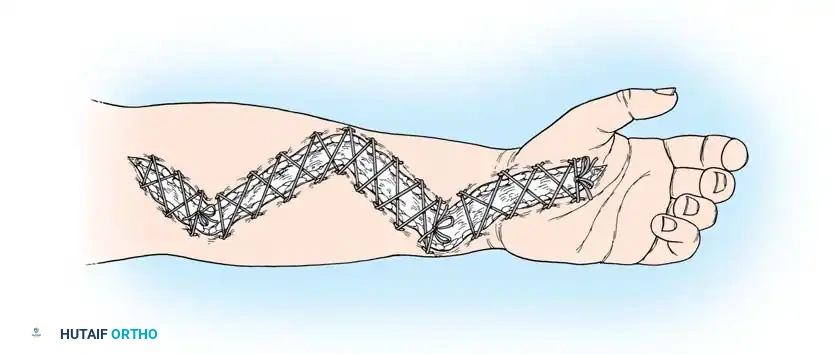
Fig. 71-6 Vessel loop shoelace technique for fasciotomy closure. (Redrawn from Asgari MM, Spinelli HM: The vessel loop shoelace technique for closure of fasciotomy wounds, Ann Plast Surg 44:225, 2000.)
Apply a sterile, non-adherent moist dressing and a long-arm splint. The elbow should not be immobilized in flexion beyond 90 degrees to prevent vascular kinking.
Release of Acute Ischemic Contracture of the Intrinsic Musculature (Technique 71-2)
Severe swelling of the hand with delayed capillary refill necessitates immediate decompression of the intrinsic compartments to prevent intrinsic-plus contractures.
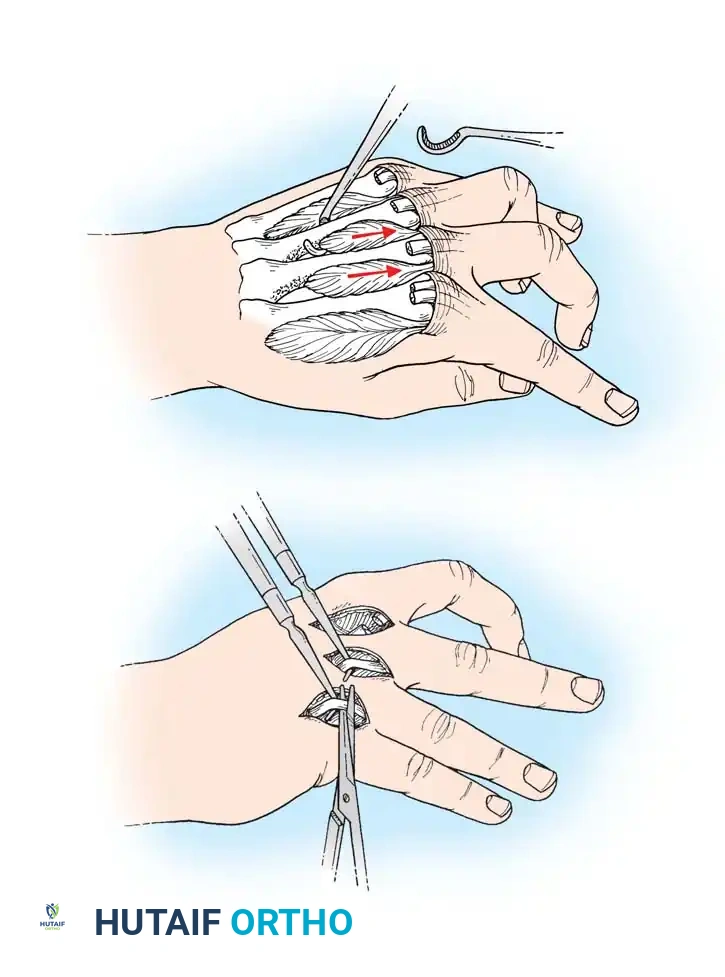
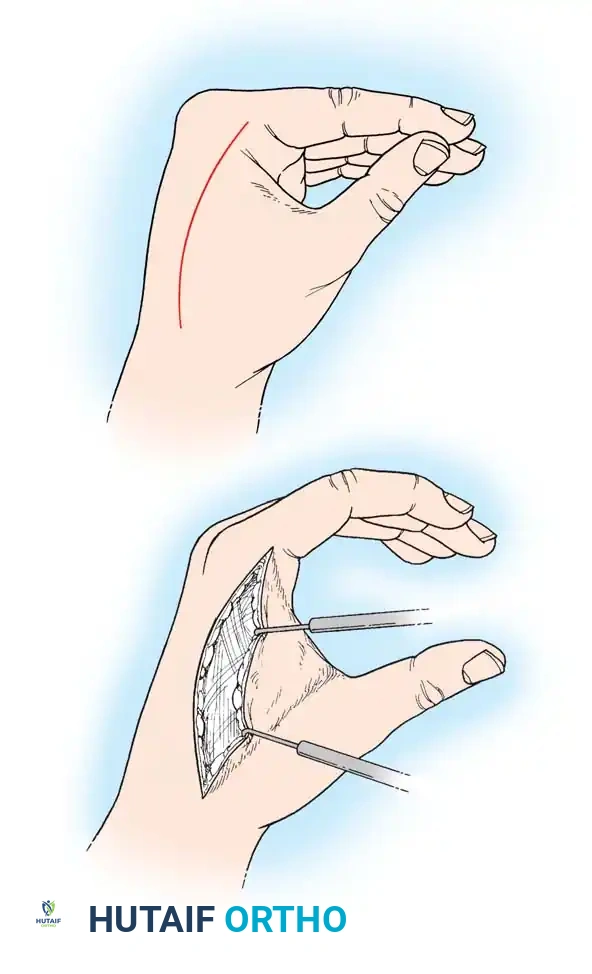
- Dorsal Incisions: Make two dorsal parallel longitudinal incisions through the skin overlying the second and fourth metacarpals. Begin at the level of the metacarpophalangeal (MCP) joints and extend just distal to the wrist. Carry each incision down to the musculofascial layer.
- Interosseous Decompression: Incise the dorsal interosseous fascia. Release the compression of the distended muscles, allowing them to extrude into the wound if necessary.
- Verification of Release: Identify each muscle belly individually. Passively flex the MCP joints and extend the proximal interphalangeal (PIP) joints to stretch the intrinsic muscles, ensuring that all compartments are adequately decompressed.
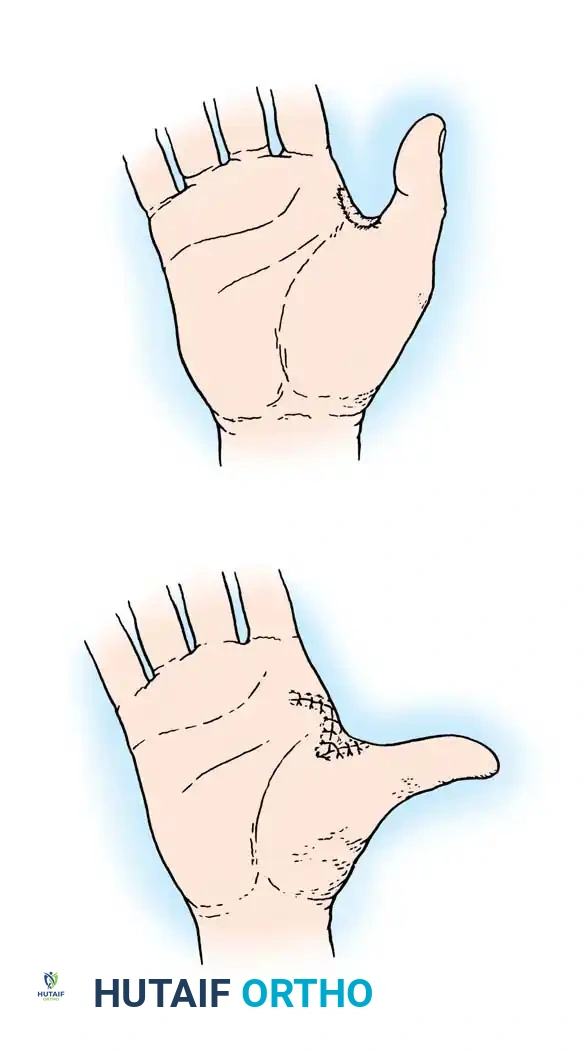
- Thenar and Hypothenar Release: If the thenar and hypothenar compartments are tense, make additional palmar radial (parallel to the thenar crease) and palmar ulnar incisions for separate decompression.
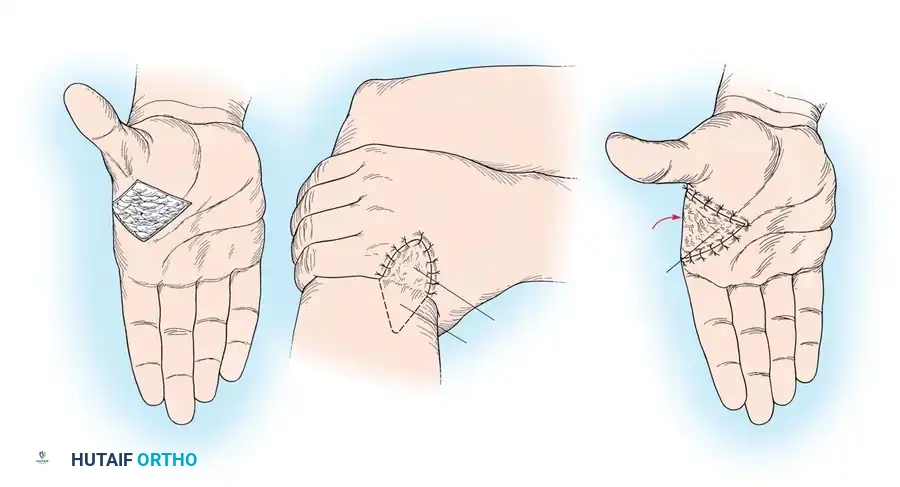
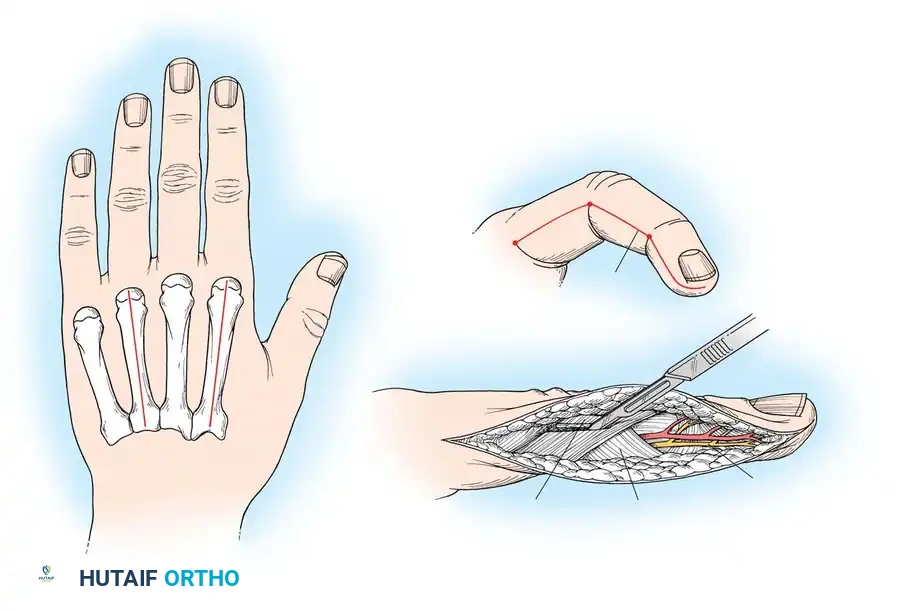
- Digital Fasciotomies: If the fingers remain tensely swollen with compromised perfusion, proceed with digital fasciotomies. Utilize midlateral incisions along the radial border of the ring and small fingers, and the ulnar border of the index and long fingers.
Surgical Pitfall: Do not attempt to debride the interosseous muscles during the acute release. Ischemic intrinsic muscles are highly susceptible to iatrogenic damage. Allow the wounds to granulate for secondary closure or skin grafting once edema subsides.
Management of Established Volkmann Contracture
If a compartment syndrome is unrecognized, untreated, or inadequately decompressed, compartment pressures will continue to rise until irreversible tissue ischemia and necrosis occur. The fibrotic replacement of necrotic muscle yields Volkmann ischemic contracture.
The ischemic infarct typically forms an ellipsoid shape, with the most profound necrosis occurring at the core of the deep compartment. Consequently, the earliest and most severe changes usually involve the flexor digitorum profundus (FDP) and the flexor pollicis longus (FPL) in the middle third of the forearm.
Clinical Presentation
The classic clinical posture of an established Volkmann contracture includes:
* Elbow flexion
* Forearm pronation
* Wrist flexion
* Thumb adduction
* Metacarpophalangeal (MCP) joint extension
* Interphalangeal (IP) joint flexion
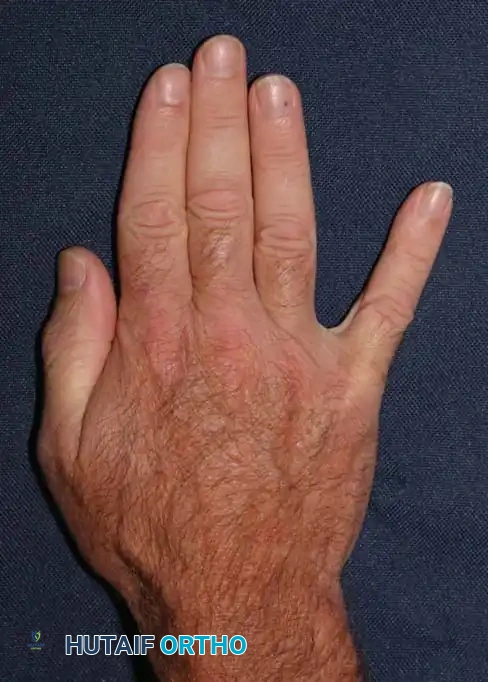
Fig. 71-9 Abduction contracture of fifth finger in a patient who developed fibrosis in the abductor digiti quinti, secondary to ischemic myositis.
Classification and Treatment Algorithms
Volkmann contractures are classified into three degrees of severity, dictating the reconstructive approach:
1. Mild (Localized) Contracture
Results from partial ischemia of the profundus mass. Flexion contractures usually involve only two or three fingers (typically the middle and ring fingers). Sensory changes are mild or absent, and intrinsic muscle contractures are not present.
* Early Stage: Dynamic splinting to prevent wrist contracture, functional training, and active mobilization.
* Late Stage (>3 months): The involved muscle-tendon units can be surgically released and lengthened. If multiple tendon units are involved, a muscle sliding operation is preferred over multiple individual tendon lengthenings.
2. Moderate Contracture
Involves the long finger flexors, the FPL, and frequently the wrist flexors. Median and ulnar nerve sensory deficits are prominent, and intrinsic-minus deformities begin to appear due to nerve strangulation.
* Surgical Intervention: A muscle sliding operation combined with meticulous neurolysis of the median and ulnar nerves. Any fibrotic, non-functional muscle mass encountered must be excised.
* Tendon Transfers: If no useful movement of the finger flexors is retained, volar transfers of dorsal wrist extensors (e.g., brachioradialis to FPL, and extensor carpi radialis longus [ECRL] to FDP) are required, alongside a complete release of the wrist and finger flexors.
3. Severe Contracture
Involves both the flexor and extensor compartments of the forearm. Fractures, severe skin scarring, and profound sensory loss are present. The nerves are heavily strangulated by dense, scarred muscle.
* Surgical Intervention: Early, radical excision of all necrotic muscle combined with complete median and ulnar neurolysis to restore sensibility and salvage intrinsic function.
* Timing: Tsuge advocated for intervention between 3 to 12 months post-injury. However, modern consensus (Hovius, Ultee, Chuang) recommends early surgical intervention (within 3 weeks) to identify and remove nonviable muscle, thereby preventing progressive contracture.
* Reconstruction: Tendon transfers are performed as secondary procedures. If local motor units are unavailable, a free innervated muscle transfer (typically utilizing the gracilis muscle or a medial gastrocnemius myocutaneous flap) is indicated to restore finger flexion.
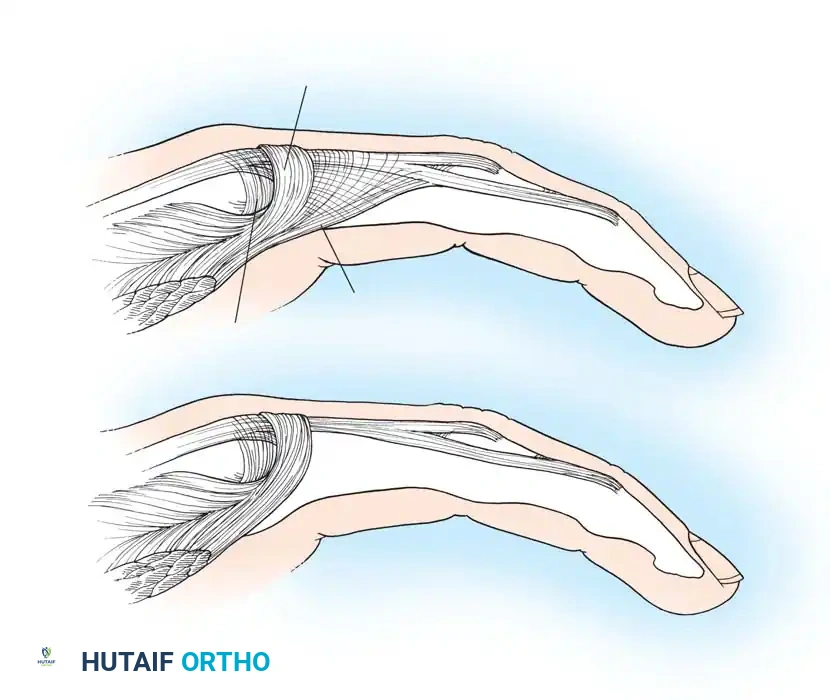
Fig. 71-10 Littler release of intrinsic contracture. Excision of the oblique fibers of the extensor aponeurosis allows proximal glide, relieving the intrinsic-plus posture.
The Muscle Sliding Operation
Originally described by Page in 1923 and endorsed by Scaglietti, the muscle sliding operation involves the proximal release of the flexor origin from the medial epicondyle, allowing the entire flexor-pronator mass to slide distally.
While historically used for Volkmann contractures, modern literature (supported by Hovius and Ultee) dictates that because the ischemic muscle is largely fibrotic and noncontractile, a muscle sliding operation alone is rarely satisfactory. It must be combined with radical infarct excision, neurolysis, and appropriate tendon transfers to achieve meaningful functional restoration.
You Might Also Like