Peripheral Nerve Topography and Regeneration: A Comprehensive Surgical Guide
Key Takeaway
Understanding the internal topography and regenerative pathophysiology of peripheral nerves is paramount for successful microsurgical neurorrhaphy. This guide details the transition from complex proximal fascicular networks to distinct distal arrangements, alongside the cellular mechanisms of Wallerian degeneration and axonal sprouting. Mastery of these principles dictates surgical timing, fascicular alignment techniques, and the management of nerve gaps to optimize functional recovery and prevent neuroma formation.
INTRODUCTION TO PERIPHERAL NERVE MICROANATOMY
The successful surgical management of peripheral nerve injuries demands an exhaustive understanding of both the microanatomical architecture and the complex cellular pathophysiology that dictates neuronal degeneration and regeneration. In the realm of operative orthopaedics and microsurgery, the surgeon must bridge the gap between basic neurobiology and meticulous surgical technique. The internal topography of a peripheral nerve is not a static cable of parallel wires; rather, it is a dynamic, plexiform network that changes continuously along its longitudinal axis. Recognizing these anatomical variations, alongside the predictable biological cascade of Wallerian degeneration and subsequent axonal sprouting, forms the foundation for critical decision-making regarding the timing of exploration, the choice of neurorrhaphy technique, and the management of critical nerve gaps.
INTERNAL TOPOGRAPHY OF PERIPHERAL NERVES
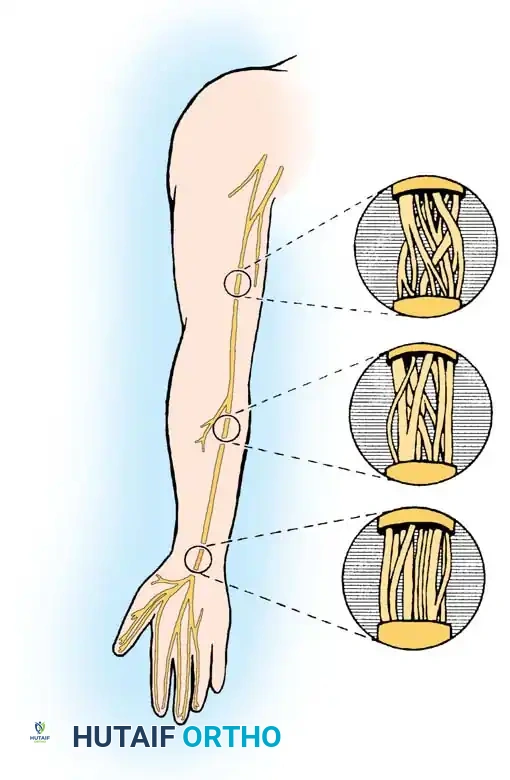
The internal topography, or fascicular arrangement, of major peripheral nerves—such as the radial, median, and ulnar nerves—was definitively described by Sunderland in 1945. He characterized the internal structure as a highly complex network of branching, dividing, and intermingling fascicles that constantly change their relative positions throughout the course of the nerve.
Proximal vs. Distal Fascicular Architecture
Extensive anatomical studies by Jabaley, Wallace, and Heckler; Williams and Jabaley; Terzis, Feeker, and Sismoue; and Brushart have elucidated a critical surgical principle: the complexity of fascicular intertwining is inversely proportional to the distance from the end organ.
In the proximal aspect of a peripheral nerve (e.g., the median nerve in the proximal arm), the fascicular arrangement is highly plexiform. Fascicles frequently divide and merge, making it nearly impossible to separate specific functional groups over any significant longitudinal distance without causing iatrogenic axonal damage.
Conversely, in the more distal portions of the nerve (e.g., the median nerve at the level of the distal forearm and carpal tunnel), the internal topography becomes markedly less complex. The fascicles organize into distinct, parallel groups corresponding to specific motor and sensory distributions (such as the recurrent motor branch or specific digital nerves).

Clinical Pearl: The diminished complexity of the distal internal topography is of paramount importance to the microsurgeon. Because distal fascicles run in parallel with minimal cross-connections, they can be safely dissected and neurolysed over long distances before merging occurs. This characteristic is the anatomical basis that allows for accurate group fascicular neurorrhaphy and precise intraneural dissection during the repair of distal nerve lacerations.
PATHOPHYSIOLOGY OF NEURONAL DEGENERATION
When a peripheral nerve is transected or severely crushed, a highly orchestrated sequence of degenerative changes occurs both distal and proximal to the zone of injury. Any part of a neuron that is physically detached from its parent cell body (located in the anterior horn of the spinal cord for motor neurons, or the dorsal root ganglion for sensory neurons) is destined to degenerate and be cleared by phagocytosis.
Wallerian (Secondary) Degeneration
The process of degeneration occurring distal to the point of injury is termed secondary, or Wallerian, degeneration. The timeline of this process varies between sensory and motor segments and is directly related to the cross-sectional size and the degree of myelinization of the specific nerve fiber.
- Acute Phase (Days 1 to 3): During the first 72 hours following injury, definite morphologic changes become apparent within the axon. Crucially for the surgeon, the distal nerve stump remains electrically excitable. A response to faradic stimulation can typically be obtained for periods ranging from 18 to 72 hours post-injury.
- Fragmentation Phase (Days 2 to 3): After 48 to 72 hours, the distal axonal segment becomes fragmented. As subsequent fluid loss occurs, these fragments begin to shrink, assuming a more oval or globular appearance. Concomitantly, the myelin sheath undergoes parallel fragmentation and shrinkage, breaking down into lipid droplets.
- Clearance Phase (Day 7 to Day 30): By the seventh day, blood-borne macrophages and resident phagocytic cells have infiltrated the area in massive numbers. The clearing of axonal and myelin debris is robust and is virtually complete between 15 to 30 days post-injury.
- Schwann Cell Proliferation: Simultaneously, Schwann cell division by mitosis becomes evident by day 7. These cells rapidly increase in number, aligning themselves longitudinally to fill the endoneurial tubes previously occupied by the intact axon and myelin sheath, forming the Bands of Büngner.
Primary (Traumatic or Retrograde) Degeneration
The reaction proximal to the point of detachment is termed primary, traumatic, or retrograde degeneration. This process proceeds proximally for at least one internode, or significantly more, depending on the severity and energy of the proximal insult. Histologically, retrograde degeneration is identical to Wallerian degeneration.
Cellular Body Response (Chromatolysis)
The parent cell body undergoes profound metabolic and morphologic changes, the severity of which depends on the type of cell and the proximity of the injury to the cell body (injuries closer to the neuraxis provoke a more severe response).
* Morphologic Changes: By day 7, the cell body exhibits chromatolysis—a dissolution of the Nissl bodies (rough endoplasmic reticulum). The cytoplasm swells, and the nucleus is displaced eccentrically to the periphery of the cell.
* Resolution: Death of the neuron or evidence of beginning recovery becomes apparent after 4 to 6 weeks. With recovery, cellular edema subsides, the nucleus migrates back toward the center of the cell, and Nissl substance begins to reaccumulate as the cell shifts its metabolic machinery from neurotransmitter production to structural protein synthesis for axonal repair.
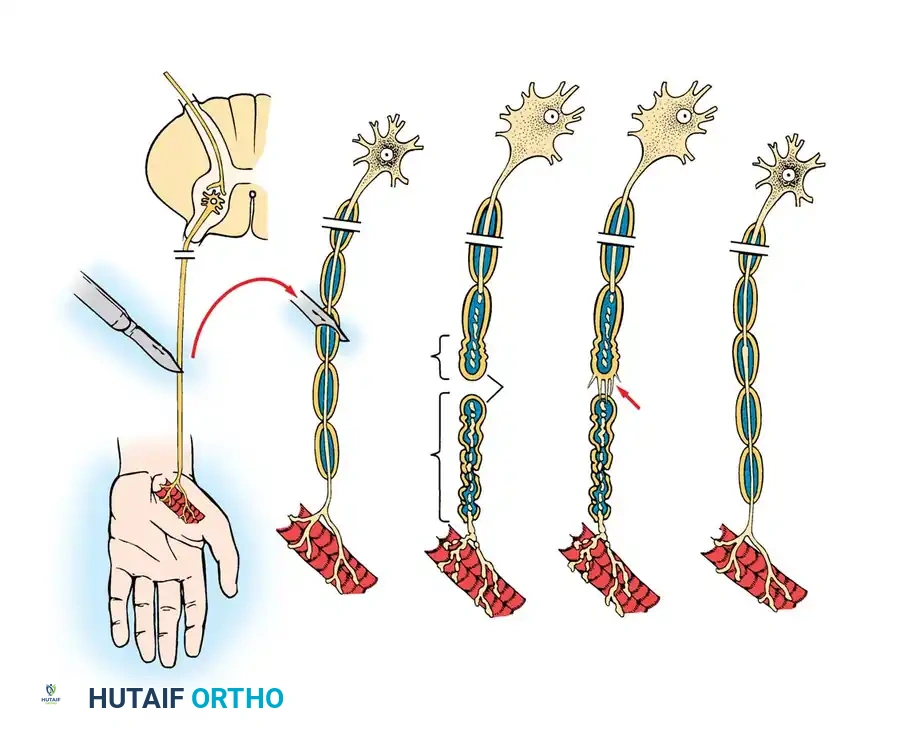
MECHANISMS OF NEURONAL REGENERATION
Following the clearance of debris, the distal nerve stump consists of empty endoneurial tubes filled with proliferating Schwann cells, primed to accept regenerating axonal sprouts from the proximal stump.
Axonal Sprouting and Migration
Axonal sprouting from the proximal stump may commence within the first 24 hours after injury. Initially, all axonal sprouts are unmyelinated, regardless of whether they originate from a myelinated or an unmyelinated parent fiber. A single proximal axonal stump may generate up to 100 individual sprouts, which advance via specialized structures called growth cones.
- Intact Endoneurial Tubes (Neuropraxia/Axonotmesis): If the injury is a lesser grade (e.g., a crush injury where the endoneurial tube and its contained Schwann cells remain uninterrupted), the sprouts pass readily along their former anatomical courses. Following regeneration, the surviving cells accurately re-innervate their previous end organs, resulting in excellent or acceptable anatomical and functional recovery.
- Disrupted Endoneurial Tubes (Neurotmesis): If the injury is severe enough to completely interrupt the endoneurial architecture, the regenerating sprouts may migrate aimlessly. They can escape into the epineurial, perineurial, or adjacent extraneural soft tissues, resulting in the formation of a painful stump neuroma or a neuroma-in-continuity.
Surgical Warning: Migrating axonal sprouts that are barred from their native endoneurial tubes by dense scar tissue may inadvertently enter the empty endoneurial tubes of adjacent, unrelated funiculi. As demonstrated by Cabaud et al., these axons may successfully regenerate through newly formed endoneurial tubes, only to terminate in myotomal or dermatomal areas entirely foreign to their original function, leading to debilitating synkinesis or sensory cross-innervation.
Neurotrophism and Neurotropism
Axons do not regenerate blindly; they are heavily influenced by neurotrophic (survival-promoting) and neurotropic (directional-guidance) substances secreted by the distal nerve tissue and Schwann cells.
Experimental work by Mackinnon et al., Dellon, and Lundborg and Branemark utilizing primate models and inert silicone Y-chambers demonstrated that regenerating axons preferentially grow toward nerve tissue rather than muscle or tendon. Furthermore, Brushart demonstrated that a degree of end-organ specificity exists (motor axons preferentially seek motor pathways). However, this neurotropic effect is strictly distance-dependent; there is a critical gap distance (approximately 2 mm) beyond which this chemotactic gradient dissipates, rendering the neurotropic effect void.
SURGICAL PRINCIPLES AND OPERATIVE TECHNIQUES
The translation of these biological principles into surgical practice dictates the protocols for peripheral nerve exploration, preparation, and neurorrhaphy. Extensive injuries with complete disruption of the nerve, wide separation of the nerve ends, and extensive scar tissue obstruction will result in little to no functional return unless meticulously reconstructed.
Indications and Timing of Surgery
- Primary Repair (Immediate to 72 hours): Indicated for sharp, clean lacerations (e.g., glass or knife wounds) where there is minimal zone of injury. Immediate repair prevents retraction of the nerve ends and utilizes the optimal biological window before extensive scarring occurs.
- Delayed Primary Repair (1 to 3 weeks): Indicated for blunt transections, avulsions, or severe crush injuries. Delaying the repair allows the zone of traumatic retrograde degeneration to fully demarcate, ensuring that the surgeon resects back to truly healthy, viable fascicles.
- Secondary Reconstruction (Months): Indicated for late presentations or failed primary repairs, often requiring nerve grafting or nerve transfers.
Patient Positioning and Preparation
- Tourniquet Control: A pneumatic tourniquet is essential for maintaining a bloodless field, which is critical for identifying microscopic fascicular architecture. Exsanguination should be performed carefully.
- Magnification: Operating loupes (minimum 3.5x to 4.5x) are mandatory for initial exposure, while an operating microscope is highly recommended for the actual epineurial or fascicular neurorrhaphy.
- Positioning: The limb must be positioned to allow for full excursion of the joints, enabling the surgeon to assess tension on the repair during simulated range of motion.
Step-by-Step Surgical Approach
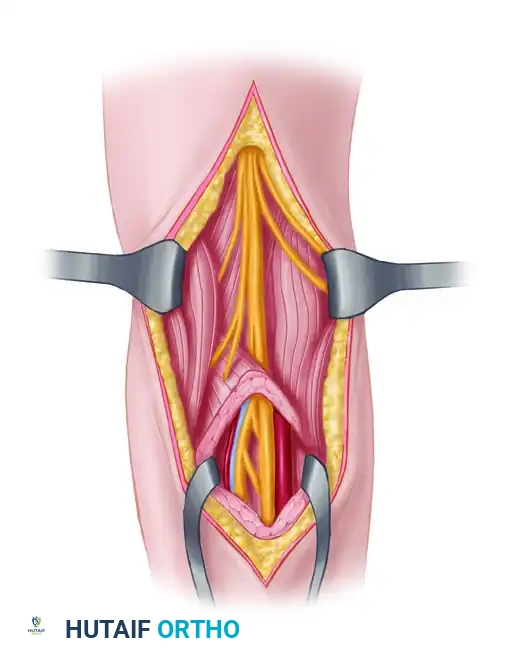
1. Exposure and Identification
Incision planning must allow for extensile exposure. Never dissect directly into the zone of injury initially.
* Identify the uninjured nerve proximally and distally in virgin tissue planes.
* Trace the healthy nerve meticulously toward the zone of injury, isolating the scarred or transected ends.
* Utilize vessel loops for gentle retraction, avoiding crush injury to the epineurium.
2. Preparation of the Nerve Stumps
The most critical step in neurorrhaphy is the adequate resection of the damaged nerve ends.
* Distal Stump: Resect the distal stump until healthy, distinct fascicles are visualized. The fascicles should "pout" or mushroom slightly from the epineurium.
* Proximal Stump: Resect the proximal neuroma or crushed tissue back to healthy tissue. The presence of robust, bleeding vasa nervorum and herniating axoplasm confirms adequate resection.
* Pitfall: Failure to resect back to healthy tissue guarantees that regenerating sprouts will be blocked by intraneural fibrosis, leading to a failed repair.
3. Fascicular Alignment
Accurate rotational alignment is paramount to prevent motor-to-sensory cross-innervation.
* Utilize longitudinal epineurial blood vessels as a primary landmark for rotational orientation.
* Match the cross-sectional fascicular topography. Remember that proximal topography is complex, while distal topography is grouped.
* Intraoperative nerve stimulation can be used in acute settings (within 72 hours) to identify distal motor fascicles before Wallerian degeneration renders them unexcitable.
4. Techniques of Neurorrhaphy
The repair must be absolutely tension-free. Tension induces ischemia, stimulates fibroblastic proliferation, and creates a dense scar barrier.
* Epineurial Repair: The standard for most nerve lacerations. Interrupted 8-0 or 9-0 non-absorbable monofilament sutures (e.g., nylon) are placed through the epineurium. This technique relies on the accurate alignment of the outer sheath to approximate the internal fascicles.
* Group Fascicular Repair: Indicated in distal nerves where distinct motor and sensory fascicular groups are easily identifiable (e.g., the ulnar nerve at the wrist). Sutures (10-0) are placed through the perineurium of matching fascicular groups.
5. Management of Nerve Gaps
If a tension-free primary repair cannot be achieved (even after extensive mobilization and joint positioning), a gap-spanning technique is mandatory.
* Autologous Nerve Grafting: The gold standard. The sural nerve or medial antebrachial cutaneous nerve is harvested and reversed (to prevent axonal escape through branching points). Multiple cables are used to match the cross-sectional area of the injured nerve.
* Nerve Conduits: Synthetic or biological tubes (e.g., collagen or polyglycolic acid) can be used for small, non-critical sensory nerve gaps (strictly less than 3 cm). They rely on the neurotropic and neurotrophic factors to guide axons across the gap.
* Allografts: Processed human nerve allografts are an alternative for intermediate gaps, providing a natural extracellular matrix scaffold without donor site morbidity.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The postoperative phase is as critical as the microsurgical repair itself. The delicate neurorrhaphy must be protected while the biological processes of Wallerian degeneration and axonal regeneration commence.
Immobilization Phase
- The affected limb is immobilized in a well-padded orthosis for 3 to 4 weeks.
- The joints are positioned to minimize tension on the repair site (e.g., slight wrist and elbow flexion for a volar median nerve repair).
- Warning: Extreme flexion postures should be avoided as they can cause joint contractures and secondary ischemic injury to the nerve.
Mobilization and Rehabilitation
- After 3 to 4 weeks, the orthosis is gradually modified to allow progressive, protected range of motion.
- Sensory Re-education: As regenerating axons reach the distal end organs, patients often experience altered or hypersensitive sensation. A structured sensory re-education program, initiated by a specialized hand therapist, is vital to help the cerebral cortex remap and interpret the new sensory input.
- Motor Monitoring: Axonal regeneration proceeds at an average rate of 1 mm per day (approximately 1 inch per month). Clinical progression is monitored via an advancing Tinel's sign and serial electromyography (EMG), which may show nascent motor unit potentials weeks before clinically detectable muscle contraction occurs.
In conclusion, the surgical management of peripheral nerve injuries is a profound exercise in applied neurobiology. By respecting the complex internal topography of the nerve and meticulously facilitating the natural processes of neuronal regeneration, the orthopaedic microsurgeon can optimize functional outcomes and restore quality of life to the patient.
You Might Also Like