Elbow Collateral Ligament Instability: Epidemiology, Anatomy, and Biomechanics
Key Takeaway
Elbow collateral ligament instability involves injuries to the medial (UCL) and lateral (LCL) complexes, critical for elbow function. UCL injuries often affect overhead athletes due to valgus stress, while LCL injuries typically cause posterolateral rotatory instability from trauma like FOOSH. Understanding their distinct anatomy and biomechanics is paramount for diagnosis and treatment.
Introduction & Epidemiology
Elbow collateral ligament instability represents a significant spectrum of pathology, ranging from acute, isolated sprains to chronic, complex rotatory instabilities, profoundly impacting elbow function and stability. The two primary collateral ligament complexes, the medial (ulnar) collateral ligament (UCL) and the lateral collateral ligament (LCL), are critical static stabilizers of the ulnohumeral and radiohumeral joints. Injuries to these structures are common, particularly in athletic populations, but also occur due to trauma in the general population.
Epidemiologically, UCL injuries are overwhelmingly associated with overhead throwing athletes, with baseball pitchers being the most frequently affected cohort. The repetitive high-velocity valgus stress generated during the late cocking and early acceleration phases of throwing can lead to microtrauma, degeneration, and eventual macroscopic failure of the anterior bundle of the UCL. The incidence of UCL reconstruction in professional baseball players has risen substantially over the last two decades, reflecting increased athletic demands, improved diagnostics, and wider acceptance of surgical intervention. While less common, acute UCL ruptures can also occur from single traumatic events, such as elbow dislocations or direct blows.
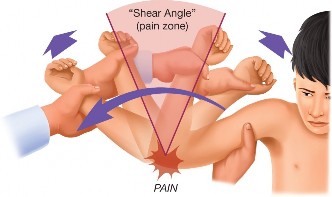
Lateral collateral ligament complex injuries most commonly manifest as posterolateral rotatory instability (PLRI) of the elbow. PLRI typically results from a fall onto an outstretched hand (FOOSH) with a supinated forearm and axial compression, or from recurrent elbow dislocations. It can also be iatrogenic following lateral epicondylar releases for epicondylitis. Unlike UCL injuries, LCL injuries are less frequently linked to specific sports, though they can occur in contact sports or activities involving repetitive varus stress or hyperextension. The prevalence of symptomatic PLRI is likely underestimated due to its often subtle and episodic nature, making diagnosis challenging.
Clinical presentation varies widely based on the specific ligament involved, the acuteness of the injury, and the degree of instability. Patients may report pain, a sense of apprehension or "giving way," clicking, locking, or frank dislocation. A thorough understanding of the distinct anatomy, biomechanics, and injury patterns of each collateral ligament complex is paramount for accurate diagnosis and effective management.
Surgical Anatomy & Biomechanics
A precise understanding of the elbow's intricate ligamentous anatomy and its contribution to joint stability is fundamental for any surgeon addressing collateral ligament pathology. The elbow joint is a complex hinge joint stabilized by bony congruity, a strong capsule, and distinct medial and lateral ligamentous complexes.
Medial (Ulnar) Collateral Ligament (UCL) Complex
The UCL complex is the primary static stabilizer against valgus stress at the elbow. It consists of three main components:
*
Anterior Bundle:
This is the strongest and most critical component, originating from the anterior-inferior aspect of the medial epicondyle and inserting onto the sublime tubercle of the coronoid process. It is considered nearly isometric, maintaining relatively constant tension throughout the arc of motion. The anterior bundle is the primary restraint to valgus stress from approximately 20° to 120° of flexion. It further subdivides into an anterior and posterior band, with the anterior band taut in extension and the posterior band taut in flexion.
*
Posterior Bundle:
This fan-shaped thickening of the posterior capsule originates from the medial epicondyle and inserts onto the medial olecranon. It becomes taut in extreme flexion (>90°) and forms the floor of the cubital tunnel. Its role in valgus stability is secondary to the anterior bundle.
*
Transverse Ligament (Cooper's Ligament):
This small, inconstant fibrous band connects the coronoid and olecranon without contributing significantly to elbow stability.
The ulnar nerve courses approximately 5-10 mm posterior and inferior to the medial epicondyle, making it vulnerable during medial elbow surgery. The flexor-pronator mass (originating from the medial epicondyle) overlies the UCL and must be navigated carefully during surgical exposure.
Lateral Collateral Ligament (LCL) Complex
The LCL complex is the primary static stabilizer against varus stress and posterolateral rotatory instability (PLRI). It originates from the lateral epicondyle and consists of four main components:
*
Lateral Ulnar Collateral Ligament (LUCL):
This is the most crucial component for elbow stability, originating from the lateral epicondyle, blending with the annular ligament, and inserting onto the supinator crest of the ulna. It acts as the primary restraint to posterolateral rotation of the ulna relative to the humerus. Injury to the LUCL is the hallmark of PLRI.
*
Radial Collateral Ligament (RCL):
Originates from the lateral epicondyle and inserts into the annular ligament. It does not directly attach to the radius or ulna, contributing less to elbow stability.
*
Annular Ligament:
Encircles the radial head, maintaining its articulation with the ulna.
*
Accessory Lateral Collateral Ligament (ALCL):
Extends from the annular ligament to the supinator crest of the ulna.
The superficial branch of the radial nerve and the posterior interosseous nerve (PIN) are critical structures on the lateral side. The PIN pierces the supinator muscle, approximately 3-4 cm distal to the radiohumeral joint, and must be protected during lateral approaches. The common extensor origin overlies the LCL complex.
Biomechanically, the LUCL, along with the coronoid process, resists external rotation and posterolateral subluxation of the ulna and radial head relative to the humerus. When the LUCL is disrupted, the radius and ulna can posterolaterally subluxate with respect to the humerus, particularly with a varus stress applied to a supinated forearm. This leads to the characteristic "pivot shift" phenomenon observed in PLRI.
Indications & Contraindications
The decision-making process for operative versus non-operative management of elbow collateral ligament injuries is multifaceted, influenced by patient factors, injury characteristics, and functional demands.
General Considerations
- Acute vs. Chronic: Acute injuries may be amenable to direct repair, especially if stable and involving good tissue quality. Chronic injuries, particularly with significant tissue attenuation or retraction, typically necessitate reconstruction.
- Partial vs. Complete Tear: Partial tears, especially those without gross instability, often respond to conservative management. Complete tears, particularly in high-demand individuals, frequently require surgical intervention.
- Associated Injuries: Fractures, dislocations, or other soft tissue injuries may influence the timing and type of intervention.
- Patient Expectations & Compliance: Critical for successful rehabilitation outcomes, particularly in athletes.
Indications
Operative Indications
| Condition | Operative Indication |
|---|---|
| UCL Injury | |
| Acute Complete Rupture |
- High-level throwing athletes or overhead athletes seeking to return to sport.
- Acute traumatic elbow dislocation with persistent valgus instability after reduction. - Associated medial epicondyle avulsion fracture with displacement >2mm. |
| Chronic Partial Tear with Instability |
- Symptomatic valgus instability (pain, "dead arm" syndrome, decreased velocity/control) refractory to a dedicated 3-6 month conservative management program.
- Imaging (MRI, stress radiography) confirming significant injury and instability. - High-demand individuals (e.g., throwing athletes, manual laborers). |
| Chronic Complete Rupture |
- Symptomatic valgus instability in active individuals.
- Failure of prior non-operative management. |
| LCL Injury (PLRI) | |
| Acute Dislocation with Instability |
- Acute traumatic elbow dislocation with persistent posterolateral rotatory instability (positive pivot shift test) after reduction, especially if non-reducible or recurrent.
- Associated fractures (e.g., radial head, coronoid) requiring surgical stabilization where LCL repair aids overall stability. - LCL avulsion from lateral epicondyle. |
| Chronic Symptomatic PLRI |
- Recurrent elbow dislocations or subluxations.
- Persistent subjective instability ("giving way") and functional limitations despite a dedicated 3-6 month conservative program. - Objective findings of PLRI (e.g., positive posterolateral rotatory instability test). - Iatrogenic PLRI following lateral epicondylar release. |
Non-Operative Indications
| Condition | Non-Operative Indication |
|---|---|
| UCL Injury | |
| Acute Partial Tear |
- Minimal or no objective valgus instability.
- Low-demand individuals or those not involved in overhead sports. - Initial management for most partial tears in athletes, involving rest, NSAIDs, physical therapy focusing on periscapular and core strengthening, and kinetic chain optimization. |
| Chronic Partial Tear without Instability |
- Asymptomatic or mildly symptomatic without functional limitation.
- Low-demand individuals. |
| Chronic Complete Rupture |
- Sedentary individuals or those with low functional demands who do not experience instability symptoms.
- Unwillingness or inability to undergo surgery or comply with extensive rehabilitation. |
| LCL Injury (PLRI) | |
| Acute Dislocation without Instability |
- After successful closed reduction of a simple elbow dislocation that is stable through a full range of motion.
- Isolated LCL sprain without gross instability. - Initial immobilization and controlled mobilization with focus on strengthening dynamic stabilizers. |
| Chronic Asymptomatic PLRI |
- Incidental findings on imaging.
- Patients with a positive PLRI test on examination but no subjective symptoms or functional limitations. - Unwillingness or inability to undergo surgery or comply with rehabilitation. |
Contraindications
-
Absolute Contraindications:
- Active infection (local or systemic).
- Severe elbow osteoarthritis or diffuse degenerative joint disease (address primary pathology first).
- Poor skin quality or compromised soft tissue envelope.
- Significant medical comorbidities precluding safe anesthesia and surgery.
- Unrealistic patient expectations or inability to adhere to rigorous post-operative rehabilitation.
-
Relative Contraindications:
- Severe osteoporosis or poor bone stock for anchor/tunnel fixation.
- Peripheral neuropathy or other neurological deficits that might compromise post-operative function.
- Inability to protect the operative extremity.
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is essential for a successful outcome in collateral ligament surgery, encompassing detailed patient evaluation, advanced imaging, and meticulous surgical setup.
Pre-Operative Evaluation & Imaging
- Clinical History and Physical Examination: Reconfirm the diagnosis and the specific ligament involved. Document the presence of any neurological symptoms (e.g., ulnar nerve paraesthesias for UCL, radial nerve symptoms for LCL). Assess pre-operative range of motion, stability, and pain levels.
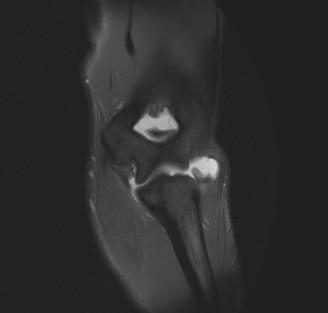
- Plain Radiographs: Standard AP, lateral, and oblique views. Stress radiographs (valgus for UCL, varus for LCL) are invaluable for quantifying instability. Look for osteophytes, loose bodies, medial epicondyle avulsion fractures, or lateral epicondylar changes.
- Magnetic Resonance Imaging (MRI): The gold standard for visualizing ligamentous integrity. MRI with contrast can further delineate partial tears and assess associated soft tissue injuries (e.g., flexor-pronator mass tears, common extensor origin pathology). Dynamic MRI can provide functional assessment.
- Computed Tomography (CT) Scan: Primarily useful for evaluating bony architecture, particularly in cases with associated fractures (e.g., coronoid, radial head) or for templating tunnel placement in complex reconstructions.
- Electrodiagnostic Studies (EMG/NCS): Indicated if pre-existing or suspected ulnar nerve neuropathy (for UCL) or posterior interosseous nerve (PIN) symptoms (for LCL) are present.
Graft Selection (for Reconstruction)
-
Autograft:
- Palmaris Longus: Most common choice for UCL reconstruction due to adequate length, appropriate diameter, and minimal donor site morbidity. Absent in 10-20% of the population.
- Gracilis or Semitendinosus: Excellent tensile strength and length, good for revision cases or when palmaris longus is absent/insufficient. Donor site morbidity includes potential for hamstring weakness.
- Toe Extensor Tendon: Alternative with low morbidity.
-
Allograft:
- Used in revision cases, multiligamentous injuries, or when autograft is limited/unavailable.
- Examples: Achilles tendon, tibialis anterior, semitendinosus.
- Advantages: No donor site morbidity, potentially faster operative time.
- Disadvantages: Risk of disease transmission (extremely low with current screening), slower incorporation, potentially higher cost.
Patient Positioning and Setup
- Anesthesia: General anesthesia is typical, often supplemented with a regional block (e.g., interscalene or supraclavicular brachial plexus block) for effective post-operative pain management.
-
Positioning:
- UCL Reconstruction: The patient is typically placed supine on the operating table. The affected arm is positioned on a padded arm board or hand table, allowing for full range of motion. Some surgeons prefer a lateral decubitus position with the arm draped free. The shoulder is abducted and the elbow flexed to approximately 90 degrees.
- LCL Reconstruction: A lateral decubitus position is often preferred, allowing easy access to the lateral aspect of the elbow. The affected arm is draped free over a chest bolster, permitting gravity-assisted varus stress. The elbow is typically flexed to 90 degrees and pronated. Alternatively, a supine position with the arm abducted and externally rotated on a hand table can be used. Prone positioning is another option but can be cumbersome.
- Tourniquet: A pneumatic tourniquet is routinely applied high on the arm to ensure a bloodless field, which is critical for identifying delicate neurovascular structures and the subtle anatomy of the ligaments.
- Sterile Prep and Drape: A wide sterile field is prepared, typically from the hand to the shoulder, to allow for graft harvest (if applicable) and full elbow mobility during the procedure.
Detailed Surgical Approach / Technique
Ulnar Collateral Ligament (UCL) Reconstruction (Modified Docking Technique)
This section focuses on the modified docking technique, a commonly employed and robust method for UCL reconstruction.
- Incision: A curvilinear or straight longitudinal incision is made on the medial aspect of the elbow, centered over the medial epicondyle, extending proximally for 5-7 cm and distally for 3-5 cm.
-
Ulnar Nerve Identification and Management:
- The medial antebrachial cutaneous nerve branches are identified and protected.
- The ulnar nerve is carefully identified within the cubital tunnel, just posterior to the medial epicondyle.
- It is meticulously freed from its fascial attachments and mobilized. The decision to perform anterior transposition (subcutaneous or submuscular) depends on pre-operative symptoms (e.g., neuropathy, subluxation) and surgeon preference. If the nerve is stable and asymptomatic, it can often be left in situ after adequate decompression. If transposed, it is often placed subcutaneously anterior to the medial epicondyle to minimize the risk of scarring into the graft.
-
Flexor-Pronator Mass Dissection:
- The superficial fascia overlying the flexor-pronator mass is incised.
- The flexor-pronator mass is split in line with its fibers, typically between the pronator teres and flexor carpi ulnaris origins, or through the common origin, to expose the underlying UCL. This split must be precise to avoid damaging the muscle belly unnecessarily.
- The remnants of the torn UCL are identified and debrided. The isometric point on the medial epicondyle (the origin) and the sublime tubercle (the insertion on the coronoid) are identified.
-
Graft Harvest:
- If using autograft, the palmaris longus tendon is typically harvested from the ipsilateral or contralateral forearm through a small transverse incision just proximal to the wrist crease.
- The graft is prepared by trimming excess muscle and fashioning a suitable size (e.g., 18-20 cm in length, folded to create a double-strand graft).
- The graft ends are whipstitched with non-absorbable suture.
-
Bone Tunnel Creation:
- Ulnar Tunnel: A single 3.5-4.5 mm drill hole is made from the posterior aspect of the sublime tubercle, exiting centrally on the anterior aspect. The drill hole is angled slightly anteriorly to optimize graft placement.
- Humeral Tunnels: Two divergent 3.5-4.5 mm drill holes are created on the medial epicondyle. The entry points are typically located ~18 mm proximal to the joint line and ~1.5 mm posterior to the midpoint of the medial epicondyle, and ~4 mm distal to the joint line and ~1.5 mm anterior to the midpoint. These tunnels converge centrally, forming a bony bridge. The goal is to mimic the native anatomical origin.
- Care is taken to avoid iatrogenic fracture of the medial epicondyle during tunnel creation.
-
Graft Passage and Fixation (Modified Docking):
- Two passing sutures are passed through the ulnar tunnel, retrieving the free ends of the prepared graft.
- The graft is pulled through the ulnar tunnel, creating a loop.
- Each free end of the graft is then passed through one of the two humeral tunnels.
- The elbow is flexed to 30-70 degrees (the optimal range for tensioning) and the forearm is pronated.
- The graft ends are tensioned firmly, usually with 20-25 lbs of tension, to restore valgus stability without overtightening.
- The graft ends are then secured to the bone bridge between the two humeral tunnels using strong non-absorbable sutures (e.g., FiberWire) in a crisscross or figure-of-8 fashion. Additional interference screws or suture anchors can be used for extra fixation.
-
Image 1:
 -
Image 3:
(This image could illustrate the humeral tunnels and the tensioning/suturing of the graft ends).
-
Closure:
- The flexor-pronator mass is meticulously repaired over the graft, ensuring it is not overtensioned.
- Subcutaneous tissues are closed, and skin is closed with sutures or staples.
- A sterile dressing and a posterior plaster splint or hinged elbow brace are applied in 90 degrees of flexion.
Lateral Ulnar Collateral Ligament (LUCL) Reconstruction (Anatomic Reconstruction)
This section describes an anatomical reconstruction technique for the LUCL.
- Incision: A posterior-lateral longitudinal incision is made, centered over the lateral epicondyle, extending proximally 4-5 cm and distally 5-6 cm.
-
Neurovascular Protection:
- The lateral antebrachial cutaneous nerve is identified and protected during skin incision and subcutaneous dissection.
- Deep dissection requires careful identification and protection of the posterior interosseous nerve (PIN), which lies anterior to the radial head, typically deep to the supinator. The safe zone for dissection is usually posterior to the radial head.
-
Extensor Origin Dissection:
- The common extensor origin is identified. It can either be detached subperiosteally from the lateral epicondyle or split in line with its fibers between the extensor carpi radialis brevis and extensor digitorum communis. The latter approach may minimize post-operative pain.
- The underlying capsule and the remnants of the torn LUCL are identified.
- The origin on the lateral epicondyle and the insertion on the supinator crest of the ulna are debrided and prepared.
-
Graft Harvest:
- A gracilis or semitendinosus autograft is typically harvested from the ipsilateral or contralateral lower extremity. Palmaris longus can also be used if adequate length and girth are available.
- The graft is prepared with whipstitches on both ends.
-
Bone Tunnel Creation:
- Humeral Tunnel: A 3.5-4.5 mm drill hole is made at the isometric origin of the LUCL on the lateral epicondyle, typically posterior and slightly proximal to the center of the epicondyle. This tunnel is oriented toward the center of the humerus.
- Ulnar Tunnel: A 3.5-4.5 mm drill hole is created obliquely from the posterior aspect of the supinator crest on the ulna, exiting on the anterior aspect of the crest. This tunnel should mimic the anatomical insertion.
- Radial Tunnel (Optional): Some techniques involve a small radial tunnel for additional stability, but this is less common with anatomical LUCL reconstruction.
-
Graft Passage and Fixation:
- The graft is passed through the humeral tunnel. One limb of the graft is directed proximally, and the other distally, creating an "L" shape that mimics the LUCL.
- Both limbs are then passed through the ulnar tunnel.
- The elbow is positioned in approximately 60-90 degrees of flexion with the forearm in full pronation. This position is critical to tension the LUCL effectively and prevent post-operative stiffness or overtightening, which can lead to varus instability.
- The graft limbs are tensioned firmly and secured with an interference screw, suture anchors, or by tying the graft limbs to themselves over a cortical bridge on the ulna. The goal is to achieve stable fixation and eliminate posterolateral rotatory instability.
-
Image 2:
(This image could depict the lateral epicondyle, supinator crest, or the graft passage through the tunnels).
-
Closure:
- The common extensor origin is repaired over the reconstructed ligament.
- Subcutaneous tissues and skin are closed.
- A sterile dressing and a posterior plaster splint or hinged elbow brace are applied, typically with the forearm in pronation and the elbow flexed to 90 degrees.
Complications & Management
Elbow collateral ligament surgery, while generally successful, is not without potential complications. Awareness of these complications and their management strategies is crucial for optimal patient care.
| Complication | Incidence | Etiology | Management/Salvage Strategy |
|---|---|---|---|
| Ulnar Neuropathy | 5-15% (transient), <2% (persistent) |
- Direct injury during dissection.
- Entrapment/compression post-op (e.g., swelling, scar tissue). - Kinking after transposition. - Insufficient decompression. |
-
Transient:
Observation, rest, NSAIDs, nerve gliding exercises.
- Persistent/Severe: Surgical exploration, neurolysis, potential revision of transposition if necessary. |
| Graft Failure/Re-rupture | 2-10% (UCL), 5-15% (LCL) |
- Inadequate tensioning/fixation.
- Premature return to activity. - Graft material failure. - Persistent underlying biomechanical issues (e.g., valgus overload, varus torque). |
-
Minor symptomatic instability:
Intensive rehabilitation, bracing.
- Gross instability/functional failure: Revision reconstruction (often with allograft or a different autograft source), addressing original surgical deficiencies. |
| Elbow Stiffness/Loss of Motion | 10-25% (often minor) |
- Prolonged immobilization.
- Heterotopic ossification (HO). - Capsular contracture. - Overtightening of graft. |
- Early, aggressive, therapist-guided ROM exercises.
- Dynamic splinting. - Refractory stiffness: Manipulation under anesthesia (MUA), arthroscopic or open capsular release. - Prophylaxis for HO (NSAIDs, radiation) in high-risk patients. |
| Persistent Instability | 5-10% (UCL), 10-20% (LCL for PLRI) |
- Incomplete repair/reconstruction.
- Technical error (e.g., incorrect tunnel placement, insufficient tension). - Missed associated pathology (e.g., coronoid fracture for LCL). - Failure of graft integration. |
- Re-evaluation of diagnosis and surgical technique.
- Revision reconstruction with attention to anatomical landmarks and proper tensioning. Address associated bony instability if present. |
| Infection | <1% (superficial), <0.5% (deep) |
- Contamination during surgery.
- Compromised host immunity. |
-
Superficial:
Oral antibiotics, local wound care.
- Deep: Surgical debridement, intravenous antibiotics, irrigation, possible hardware removal, graft retention if stable, or staged reconstruction. |
| Heterotopic Ossification (HO) | 5-10% (may be asymptomatic) |
- Surgical trauma.
- Post-operative hematoma. - Prolonged immobilization. - Individual predisposition. |
- Prophylactic NSAIDs (Indomethacin) or low-dose radiation post-op in high-risk patients (e.g., traumatic brain injury, prior HO, complex fractures).
- Symptomatic HO: Excision after maturation (typically 6-12 months post-op), with continued HO prophylaxis. |
| Radial Nerve/PIN Injury (LCL) | <1% (PIN), higher for superficial radial nerve |
- Direct injury during lateral dissection.
- Compression from swelling or scar. - Traction. |
-
Superficial Radial Nerve:
Observation, sensory re-education.
- PIN: Observation for spontaneous recovery, neurolysis if no improvement after 3-6 months. Tendon transfers for persistent wrist drop. Careful identification and protection during initial exposure is paramount. |
| Graft Impingement/Pain | Rare |
- Graft placed too far anteriorly (UCL) or posteriorly (LCL).
- Bulky graft. |
- Local injection, physical therapy.
- Refractory: Surgical debridement of impinging tissue. |
| Donor Site Morbidity | Variable (e.g., palmaris longus <1%) |
- Nerve injury (e.g., superficial radial nerve, saphenous nerve for gracilis).
- Pain, hematoma, infection at harvest site. - Functional weakness (rare for palmaris/gracilis). |
- Careful dissection, precise surgical technique.
- Symptomatic treatment (NSAIDs, local care). - Neuropathy: Observation, neurolysis if persistent. |
General Principles of Management
- Early Recognition: Prompt identification of complications is critical.
- Conservative First (if applicable): Many mild complications (e.g., transient neuropathy, minor stiffness) can be managed non-operatively.
- Surgical Intervention: Reserved for persistent, severe, or functionally limiting complications.
- Prevention: Meticulous surgical technique, appropriate post-operative rehabilitation, and prophylactic measures (e.g., for HO) are the best strategies to mitigate complications.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is as critical as the surgical procedure itself for successful restoration of elbow stability and function. Protocols are typically phased, guided by the biology of graft healing and aimed at progressive stress application while protecting the repair/reconstruction. Specific timelines can vary based on surgeon preference, graft type (repair vs. reconstruction), and individual patient progress, but the principles remain consistent.
Phase I: Maximum Protection (Weeks 0-2/3)
- Goals: Protect the surgical repair/reconstruction, control pain and edema, initiate gentle active range of motion (AROM).
-
UCL:
- Immobilization: Posterior splint or hinged elbow brace at 90° flexion, forearm neutral, for 1 week. Then, transition to a hinged brace with motion limited to 30-100° flexion for the next 1-2 weeks. Avoid valgus stress.
- Motion: Gentle AROM (flexion/extension, pronation/supination) within protected range.
- Activities: Hand and wrist AROM, grip strengthening (submaximal isometrics), shoulder isometrics (scapular stabilization).
- Weight Bearing: Non-weight-bearing.
-
LCL (PLRI):
- Immobilization: Posterior splint or hinged elbow brace in 90° flexion, full forearm pronation (to tighten the LUCL), for 1-2 weeks. Then, brace with motion limited to 30-90° flexion, maintaining pronation. Avoid varus stress and supination.
- Motion: Gentle AROM (flexion/extension, pronation only) within protected range.
- Activities: Hand and wrist AROM, grip strengthening, shoulder isometrics.
- Weight Bearing: Non-weight-bearing.
- General: Cryotherapy, elevation, pain medication. Wound care.
Phase II: Moderate Protection & Progressive Motion (Weeks 3-6/8)
- Goals: Gradually increase range of motion, initiate light strengthening, protect the healing graft.
-
UCL:
- Brace: Progressively increase ROM in the hinged brace (e.g., 15-110° by week 4, 0-120° by week 6). May discontinue brace by week 6-8 if stable and good progress.
- Motion: Continue AROM, begin gentle passive range of motion (PROM) as tolerated, avoiding valgus stress.
- Strengthening: Begin light isometric elbow flexion/extension. Progress to light resistance with elastic bands for shoulder, scapula, wrist, and forearm.
-
LCL (PLRI):
- Brace: Progressively increase ROM in the hinged brace (e.g., 15-110° by week 4, 0-120° by week 6), maintaining forearm pronation for exercises involving elbow extension. May discontinue brace by week 6-8.
- Motion: Continue AROM/PROM within protected range, emphasizing full pronation.
- Strengthening: Similar to UCL, light isometrics and resistance for shoulder, scapula, wrist, and forearm. Avoid resisted supination.
- General: Continue edema control. Scar massage once wound is healed. Begin light cardiovascular exercise (e.g., stationary bike).
Phase III: Advanced Strengthening & Functional Progression (Weeks 6/8 - 12/16)
- Goals: Restore full pain-free ROM, progressively increase strength and endurance, prepare for sport-specific activities.
-
UCL:
- Motion: Achieve full, pain-free AROM.
- Strengthening: Progress to moderate resistance exercises for elbow, wrist, and shoulder (free weights, machines). Focus on eccentric control and kinetic chain integration.
- Proprioception: Begin dynamic stabilization exercises (e.g., plyoball throws against a wall).
- Sport-Specific: For throwing athletes, begin isolated throwing motion without a ball (dry throws).
-
LCL (PLRI):
- Motion: Achieve full, pain-free AROM.
- Strengthening: Progress similar to UCL, but continue to emphasize forearm pronation and avoid resisted supination until graft is well-healed (typically >12 weeks).
- Proprioception: Begin dynamic stabilization exercises.
- Activities: Gradual return to activities of daily living that require moderate force.
Phase IV: Return to Activity/Sport (Weeks 16 - 6-12+ months)
- Goals: Safely return to full sport-specific activities, optimize performance, prevent re-injury.
-
UCL:
- Interval Throwing Program (ITP): For throwing athletes, a highly structured, gradual ITP is initiated (typically at 4-6 months post-op). This progresses from short, light throws to increased distance, velocity, and pitch count over several months.
- Strength & Conditioning: Continue advanced strengthening, power, and endurance training.
- Return to Play: Typically 9-12 months post-op for pitchers, possibly sooner for other positions or sports. A functional assessment should demonstrate full strength, stability, and lack of symptoms.
-
LCL (PLRI):
- Sport-Specific Drills: Gradually integrate specific movements and drills required for their sport/activity.
- Strength & Conditioning: Continued emphasis on global upper body and core strength.
- Return to Play: Generally 6-9 months post-op, but can vary. Functional stability tests should be performed prior to full clearance.
Key Principles for All Phases:
*
Pain as a Guide:
Activities should not elicit significant pain.
*
Individualization:
Protocols should be adapted to the individual patient's progress, sport, and functional demands.
*
Therapist Communication:
Close collaboration between the surgeon, physical therapist, and athletic trainer is paramount.
*
Avoidance of Provocative Stress:
Strict adherence to avoiding valgus stress for UCL and varus/supination stress for LCL is critical until sufficient graft healing.
Summary of Key Literature / Guidelines
The landscape of elbow collateral ligament management has evolved significantly, underpinned by robust clinical research and refined surgical techniques. Numerous studies have shaped current best practices for both UCL and LCL injuries.
Ulnar Collateral Ligament (UCL)
- Outcomes of UCL Reconstruction: Jobe's seminal work in the 1980s revolutionized the treatment of UCL injuries in throwing athletes. Early studies reported return-to-play rates for professional baseball pitchers around 80%, with subsequent studies showing similar or improved outcomes (e.g., Conte et al., Ahmad et al. consistently report 80-90% return to prior or higher level of competition). Factors associated with successful return to sport include younger age, isolated UCL injury, and absence of revision surgery.
- Techniques: While the original Jobe technique used a figure-of-8 approach, the modified docking technique (e.g., developed by Altchek, Dines, and others) has become widely adopted due to its strong biomechanical properties, requiring fewer tunnels and minimizing stress risers. Internal bracing, utilizing a high-strength suture augmentation, is an emerging technique that may allow for accelerated rehabilitation, particularly for acute repairs or as an adjunct to reconstruction. Biomechanical studies have demonstrated comparable strength to traditional reconstructions.
- Ulnar Nerve Management: The role of routine ulnar nerve transposition remains controversial. While some advocate for transposition to prevent post-operative neuropathy, others report similar outcomes with in situ decompression, reserving transposition only for pre-existing neuropathy or gross subluxation. Current literature leans towards selective transposition.
- Graft Choice: Palmaris longus is the most common autograft, but gracilis and semitendinosus are excellent alternatives. Allografts are increasingly used, particularly in revision cases or when autograft is limited, with comparable mid-term outcomes.
- Guidelines: The American Academy of Orthopaedic Surgeons (AAOS) and American Orthopaedic Society for Sports Medicine (AOSSM) provide clinical practice guidelines and position statements emphasizing a structured rehabilitation program and shared decision-making with patients regarding surgical versus non-surgical management.
Lateral Collateral Ligament (LCL) / Posterolateral Rotatory Instability (PLRI)
- Diagnosis and Classification: The posterolateral rotatory instability (PLRI) test (pivot shift test) remains the cornerstone of clinical diagnosis, complemented by stress radiography and MRI. O'Driscoll's classification system for elbow instability provides a framework for understanding injury patterns.
- Outcomes of LCL Reconstruction: Surgical outcomes for symptomatic chronic PLRI are generally favorable, with most studies reporting good-to-excellent results and resolution of instability in 80-95% of patients. Recurrent instability, while uncommon, can occur due to inadequate tensioning or missed associated pathology.
- Anatomical Reconstruction: The anatomical reconstruction of the LUCL is the preferred method, aiming to recreate the precise origin and insertion of the native ligament. Various techniques exist, often using gracilis or palmaris longus autograft, typically fixed with interference screws or suture anchors in a single humeral tunnel and an ulnar tunnel on the supinator crest. Proper tensioning in pronation and slight flexion is critical to avoid overtightening and subsequent varus stiffness.
- Associated Pathology: Recognition and management of associated injuries, such as radial head fractures or coronoid fractures (e.g., Regan-Morrey type I or II), are crucial for successful outcomes in cases of complex posterolateral elbow dislocations.
- Iatrogenic PLRI: PLRI can occur following aggressive lateral epicondylar debridement for lateral epicondylitis. Prevention involves careful assessment of stability during epicondylar release and conservative debridement. Treatment often involves LUCL reconstruction.
- Rehabilitation: Post-operative rehabilitation for LCL reconstruction heavily emphasizes protecting against varus stress and supination, maintaining the forearm in pronation during the initial healing phase to prevent recurrent instability.
Future Directions
- Biologics: The role of platelet-rich plasma (PRP) and stem cell therapies, both as adjunctive treatments to surgical repair/reconstruction and as stand-alone treatments for partial tears, continues to be investigated. Current evidence for these approaches, especially as stand-alone treatments, remains equivocal and requires further high-quality research.
- Advanced Imaging: Dynamic ultrasound and advanced MRI sequences may further improve diagnostic accuracy, especially for subtle instability patterns.
- Robotics and Navigation: While not yet mainstream for collateral ligament surgery, the use of robotics and navigation tools holds potential for precise tunnel placement and tensioning in the future.
In conclusion, the successful management of elbow collateral ligament instability hinges on a meticulous understanding of anatomy, a nuanced approach to indications, precise surgical technique, and a rigorous, individualized post-operative rehabilitation protocol, all guided by the continually evolving body of scientific literature.
Clinical & Radiographic Imaging

You Might Also Like