Free Vascularized Fibular Grafts: Solving Complex Bone Defects
Key Takeaway
Looking for accurate information on Free Vascularized Fibular Grafts: Solving Complex Bone Defects? A free vascularized fibular graft is a viable solution for reconstructing large intercalary bone defects following tumor resection. This durable biologic reconstruction exploits normal fracture healing and benefits from an independent blood supply, allowing graft incorporation even in compromised soft tissue. It can hypertrophy under pressure, ensuring excellent long-term durability and minimal complications, making it superior to traditional methods.
Introduction and Epidemiology
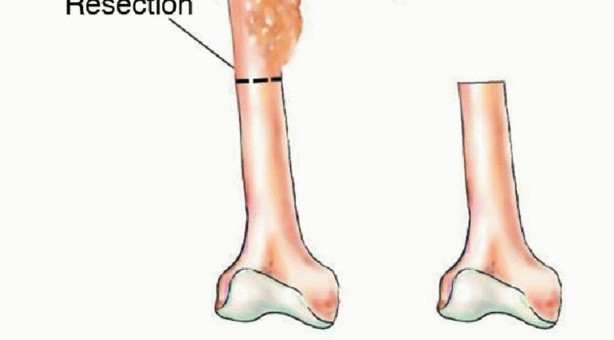
The management of massive intercalary bone defects—frequently resulting from the wide en bloc resection of primary bone sarcomas, high-energy polytrauma, or radical debridement of chronic osteomyelitis—presents one of the most formidable challenges in reconstructive orthopedic surgery. Historically, the treatment algorithm for defects exceeding critical dimensions was limited to definitive amputation. With the advent of limb-salvage surgery, reconstructive paradigms shifted toward the utilization of massive structural allografts, custom endoprostheses, and allograft-prosthetic composites (APCs). While these modalities reliably confer immediate structural stability and allow for early mobilization, their long-term survivorship is substantially compromised by high rates of aseptic loosening, progressive allograft resorption, late structural fracture, and deep periprosthetic infection.

Distraction osteogenesis, predicated on the Ilizarov principles of tension-stress, offers a true biologic reconstruction via the generation of vital, autogenous bone. However, its application is pragmatically constrained to small- and medium-sized defects. The bone transport process is exceedingly protracted, demanding up to two months for a single centimeter of consolidation, and is frequently complicated by pin-tract infections, axial deviations, and docking site nonunions. Furthermore, the requisite patient compliance is immense, and the technique is generally contraindicated in the oncologic setting, where adjuvant radiotherapy and cytotoxic chemotherapy severely compromise the biologic potential required for successful regenerate consolidation.
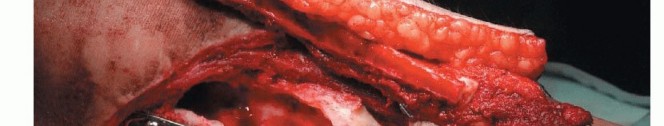
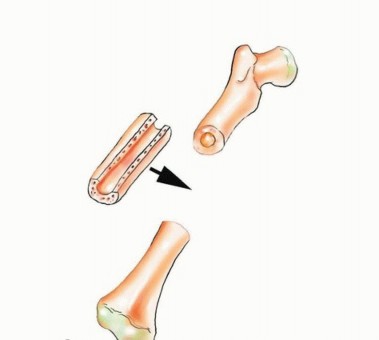
To circumvent the limitations of non-vascularized structural grafts—which rely on the slow and often incomplete process of creeping substitution—the free vascularized fibular graft (FVFG) emerged as a paradigm-shifting biologic solution. First described by Taylor et al. in 1975, the transfer of the fibula with its intact vascular pedicle allows for the transplantation of living osteocytes and osteoblasts. Because the microvascular anastomosis immediately restores endosteal and periosteal perfusion, the graft bypasses the need for creeping substitution and heals at the docking sites via primary fracture healing mechanisms.
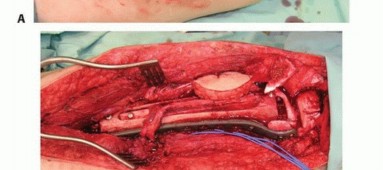
Epidemiologically, the demand for FVFG reconstruction is driven primarily by orthopedic oncology and severe orthopedic trauma. In the oncologic population, primary bone tumors such as osteosarcoma and Ewing sarcoma frequently necessitate intercalary resections of the diaphyseal femur, tibia, or humerus. In the trauma population, Gustilo-Anderson Type IIIB and IIIC open fractures often present with massive segmental bone loss that exceeds the 6-centimeter critical defect threshold, rendering non-vascularized autografting futile. Additionally, chronic recalcitrant osteomyelitis requiring radical diaphyseal excision, as well as pediatric congenital pseudarthrosis of the tibia (CPT), represent significant indications for this highly specialized microsurgical intervention.
Surgical Anatomy and Biomechanics
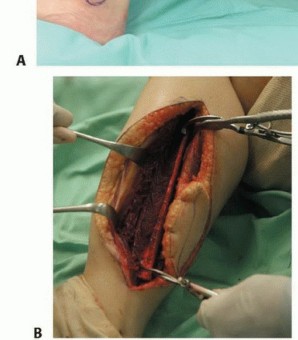
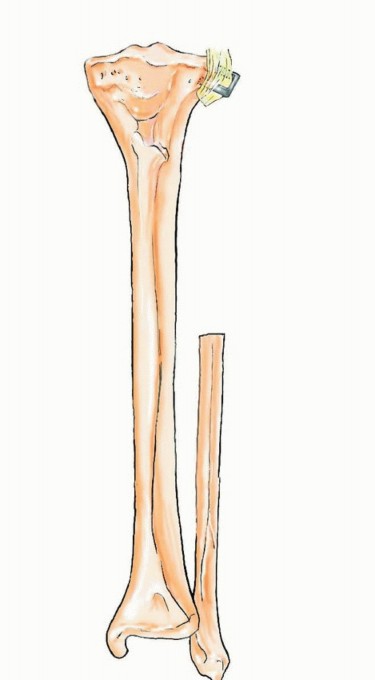
A profound understanding of the osteology, vascular topography, and regional biomechanics of the leg is an absolute prerequisite for the safe harvest and successful transfer of the free vascularized fibula. The fibula is a long, tubular bone that serves primarily as a site for muscular attachment rather than axial load-bearing, transmitting approximately 10 to 15 percent of the body's weight during normal ambulation. This mechanical redundancy permits the harvest of up to 25 centimeters of its diaphyseal length without significantly compromising the structural integrity of the lower extremity, provided the proximal and distal tibiofibular articulations are preserved.
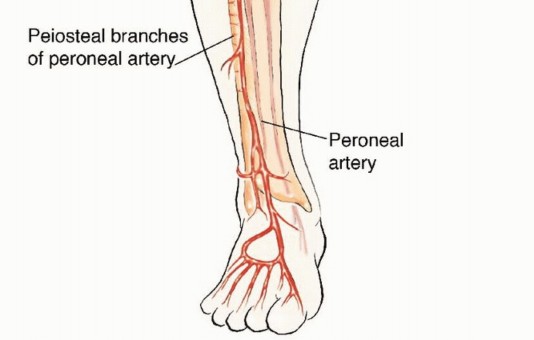
Vascular Anatomy of the Fibula
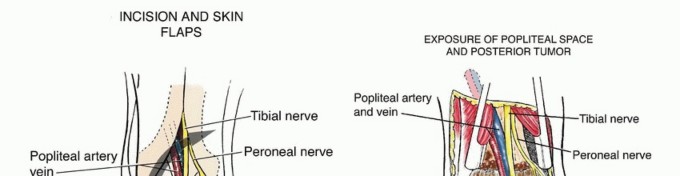
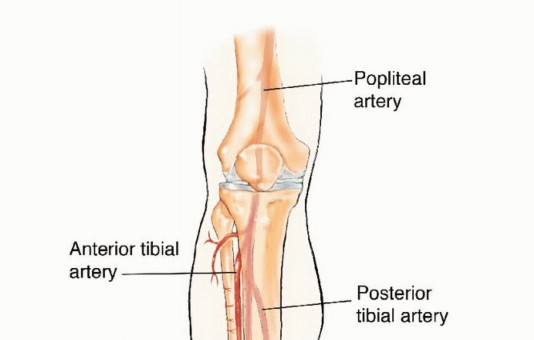
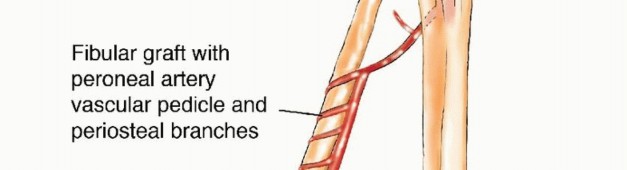
The vascular supply to the fibula is dual in nature, comprising an endosteal (medullary) system and a periosteal system, both of which are primarily derived from the peroneal artery. The peroneal artery typically originates from the tibioperoneal trunk, approximately 2 to 3 centimeters distal to the bifurcation of the popliteal artery. It descends through the deep posterior compartment of the leg, traveling intimately along the posterior aspect of the interosseous membrane, protected by the flexor hallucis longus (FHL) and the tibialis posterior muscles.
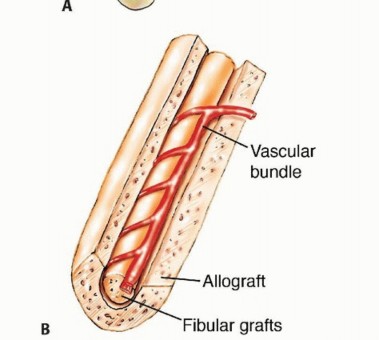
The principal nutrient artery to the fibula arises from the peroneal artery and enters the medullary canal through the nutrient foramen, which is generally located in the middle third of the diaphysis. This endosteal supply is critical for the survival of the diaphyseal osteocytes. Simultaneously, the peroneal artery gives off numerous segmental musculoperiosteal branches that supply the robust periosteal envelope. This dual perfusion allows the fibula to be osteotomized into multiple segments (e.g., a "folded" or double-barrel fibula) provided the periosteal sleeve and its segmental perforators remain intact.
Venous drainage is facilitated by the paired venae comitantes that accompany the peroneal artery. These veins are equipped with valves and eventually coalesce to drain into the popliteal vein. During microsurgical anastomosis, at least one, and preferably both, of these venae comitantes must be re-anastomosed to the recipient venous system to prevent catastrophic venous congestion of the graft.
Regional Neural Anatomy
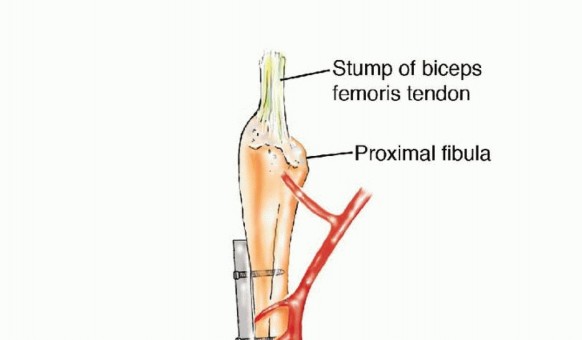
The common peroneal nerve (CPN) is the most critical neural structure at risk during proximal fibular dissection. The CPN courses distally from the popliteal fossa, wrapping around the fibular neck from posterior to anterior before dividing into the superficial and deep peroneal nerves within the substance of the peroneus longus muscle. To avoid iatrogenic denervation of the anterior and lateral compartments (resulting in foot drop), a minimum of 6 to 8 centimeters of the proximal fibula must be retained during harvest.
Biomechanical Principles of Graft Hypertrophy
The initial mechanical strength of a fibular graft is substantially inferior to that of the femur or tibia. The cortical diameter of the fibula is relatively small, making it susceptible to stress fractures when subjected to the massive axial and torsional loads of the lower extremity. However, because the FVFG is composed of living bone, it retains its intrinsic capacity to respond to mechanical stress in accordance with Wolff's Law.
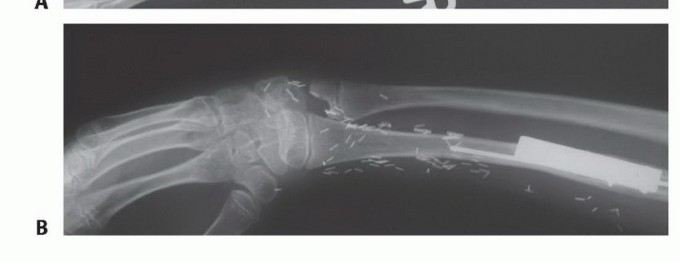
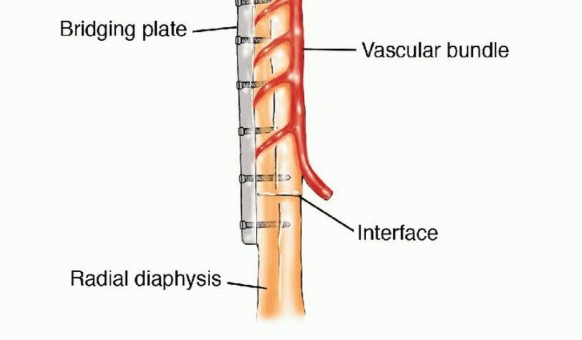
Over a period of 12 to 24 months, controlled, progressive weight-bearing stimulates profound cortical hypertrophy. The fibula can increase in cross-sectional area by up to 300 percent, eventually approximating the dimensions of the resected tibial or femoral diaphysis. To mitigate the risk of stress fracture during this vulnerable hypertrophic phase, surgeons frequently employ the Capanna technique—placing the vascularized fibula within the medullary canal of a massive structural allograft. The allograft provides immediate mechanical stability, while the vascularized fibula provides the biologic engine for long-term union and prevents late allograft collapse.
Indications and Contraindications
The decision to proceed with a free vascularized fibular graft must be carefully deliberated, weighing the magnitude of the defect, the biologic quality of the recipient bed, and the patient's physiologic capacity to tolerate a prolonged microsurgical procedure.
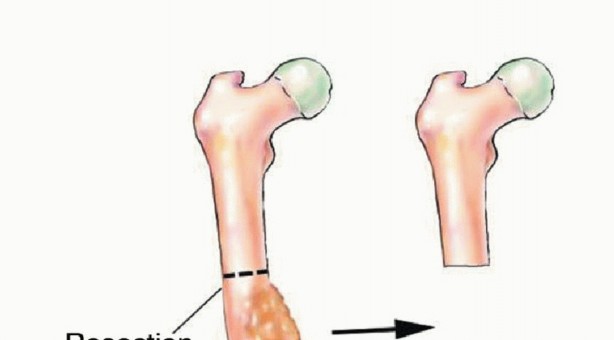
The classic indication for a FVFG is a diaphyseal bone defect exceeding 6 centimeters. For defects smaller than 6 centimeters, non-vascularized autogenous cancellous bone grafting (e.g., from the iliac crest) combined with rigid internal fixation or induced membrane techniques (Masquelet technique) is generally sufficient. However, when the defect exceeds this critical threshold, the central core of a non-vascularized graft undergoes ischemic necrosis, leading to a high probability of nonunion, resorption, and structural failure.
In the setting of osteonecrosis of the femoral head (AVN), particularly in young, active patients with pre-collapse or early post-collapse disease (Ficat Stage II or III), a free vascularized fibular graft can be utilized to decompress the necrotic subchondral bone and provide vital structural support to the articular cartilage, thereby delaying or preventing the need for total hip arthroplasty.
Contraindications to FVFG harvest are primarily vascular. Peripheral arterial disease (PAD) that compromises the inflow to the lower extremity is a strict contraindication. Furthermore, anatomical variants in which the peroneal artery provides the dominant or sole blood supply to the foot (arteria peronea magna) absolutely preclude fibular harvest, as sacrificing the vessel would result in critical limb ischemia and potential amputation of the donor foot.
Indications Summary Table
| Clinical Parameter | Operative Indications for FVFG | Non-Operative / Alternative Management |
|---|---|---|
| Defect Size | Intercalary defects > 6 cm | Defects < 6 cm (Non-vascularized autograft) |
| Soft Tissue Bed | Hostile, irradiated, or extensively scarred bed | Well-vascularized bed supporting creeping substitution |
| Infection Status | Eradicated osteomyelitis requiring massive dead space management | Active, florid infection (requires staged debridement first) |
| Femoral Head AVN | Pre-collapse or early collapse (Ficat II/III) in young patients | Advanced collapse with secondary osteoarthritis (THA indicated) |
| Congenital Conditions | Congenital Pseudarthrosis of the Tibia (CPT) | Minimal deformity amenable to bracing (rare) |
| Vascular Anatomy | Normal three-vessel runoff to the foot | Arteria peronea magna or severe PAD (Absolute Contraindication) |
Pre Operative Planning and Patient Positioning
Meticulous preoperative planning is the cornerstone of successful microsurgical bone transfer. The evaluation begins with high-resolution orthogonal radiographs and full-length scanograms to precisely quantify the length of the bone defect and template the required length of the fibular graft. Advanced cross-sectional imaging, specifically Magnetic Resonance Imaging (MRI), is mandatory in oncologic cases to delineate the intramedullary extent of the tumor and plan the exact levels of the en bloc resection.
Vascular Imaging Protocols
The most critical preoperative study is vascular imaging of both the donor and recipient extremities. Computed Tomography Angiography (CTA) or Magnetic Resonance Angiography (MRA) must be obtained to confirm normal three-vessel runoff in the donor leg. The surgeon must definitively rule out the presence of an arteria peronea magna, a congenital variant where the anterior and posterior tibial arteries are hypoplastic or absent, and the peroneal artery serves as the primary arterial supply to the foot. In the recipient bed, angiography is utilized to identify suitable recipient arteries and veins, assess for post-traumatic or radiation-induced vascular stenosis, and plan the geometry of the microvascular anastomoses.
Multidisciplinary Coordination
Massive bone reconstruction is rarely a single-surgeon endeavor. It requires a highly coordinated, multidisciplinary approach involving orthopedic oncologists, orthopedic traumatologists, and microsurgeons (often plastic or specialized orthopedic hand/micro surgeons). A two-team approach is universally preferred to minimize operative time and tourniquet ischemia. Team A is responsible for the extirpation of the tumor, debridement of the infected bone, and preparation of the recipient vessels. Concurrently, Team B performs the fibular harvest.
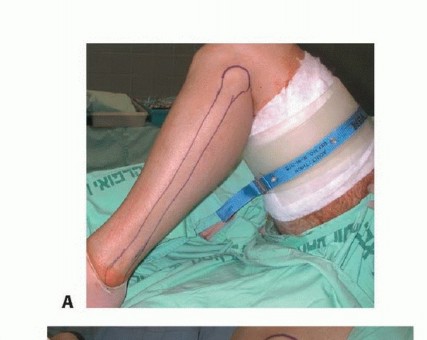
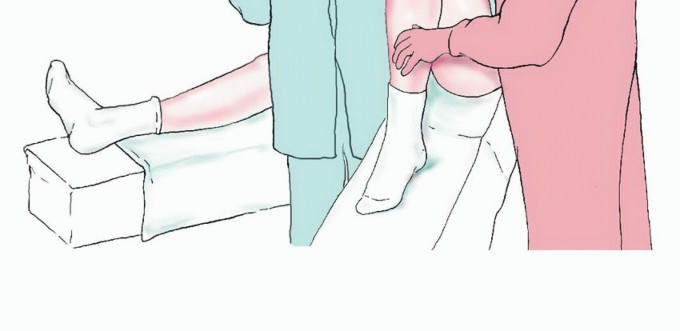
Patient Positioning and Setup
The patient is typically positioned supine on a radiolucent operative table to facilitate unhindered fluoroscopic imaging. A bump is placed under the ipsilateral hip of the donor leg to internally rotate the extremity, bringing the lateral aspect of the leg into optimal view. A sterile pneumatic tourniquet is applied high on the donor thigh. The entire lower extremity, from the iliac crest to the toes, is prepped and draped free in a sterile fashion. If a contralateral fibula is being harvested for an upper extremity defect, both the donor leg and the recipient arm are prepped simultaneously.
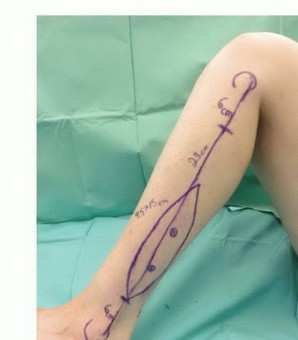
Detailed Surgical Approach and Technique
The surgical execution of a free vascularized fibular graft is technically demanding and can be broadly divided into four distinct phases: donor site dissection, recipient site preparation, skeletal fixation, and microvascular anastomosis.
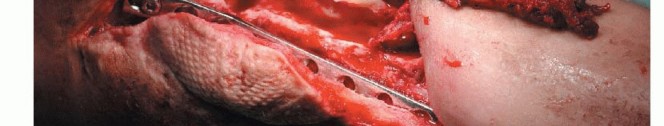
Donor Site Dissection
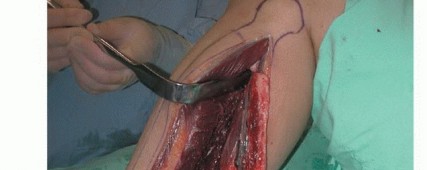
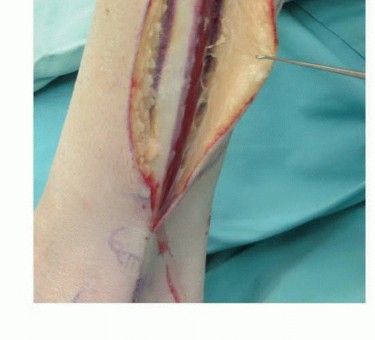
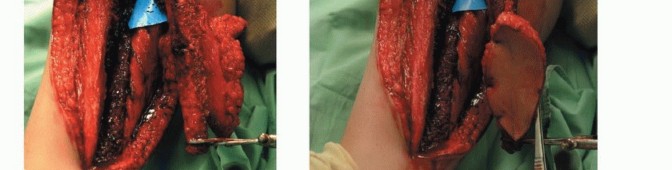
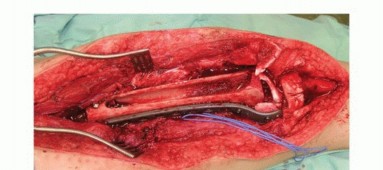
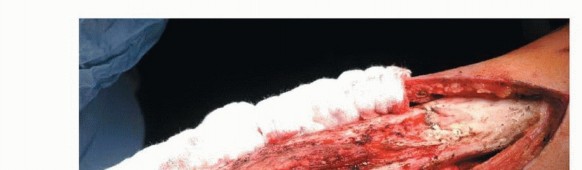
The harvest of the fibula is performed via a direct lateral approach. A longitudinal incision is made along the posterior border of the fibula, extending from the fibular head to the lateral malleolus. If a skin paddle is required to monitor the viability of the graft or to reconstruct an associated soft tissue defect (an osteocutaneous flap), the incision is designed to incorporate septocutaneous perforators traveling along the posterior intermuscular septum.

The deep fascia is incised, and the plane between the peroneus longus and brevis (anteriorly) and the soleus (posteriorly) is developed. The peroneal muscles are elevated off the lateral surface of the fibula via extraperiosteal dissection, taking extreme care to protect the superficial peroneal nerve. Proximally, the common peroneal nerve is identified and neurolysed to ensure its protection during the proximal osteotomy.
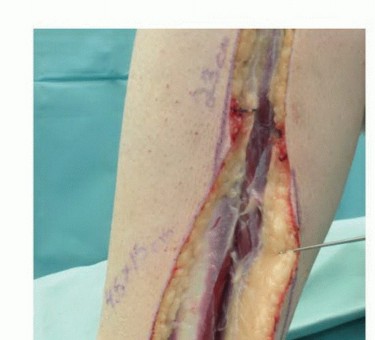
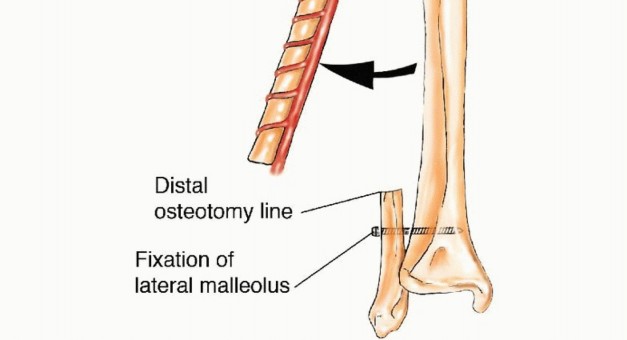
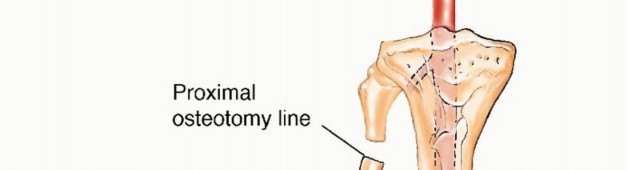
The anterior intermuscular septum is divided, and the anterior compartment musculature (extensor digitorum longus, extensor hallucis longus, tibialis anterior) is elevated. The interosseous membrane is then sharply divided. At this juncture, the planned osteotomy sites are marked. To preserve the stability of the ankle mortise and prevent proximal migration of the lateral malleolus, a minimum of 6 to 8 centimeters of the distal fibula must be retained. Similarly, 6 centimeters of the proximal fibula is retained to protect the common peroneal nerve and the collateral ligamentous structures of the knee.
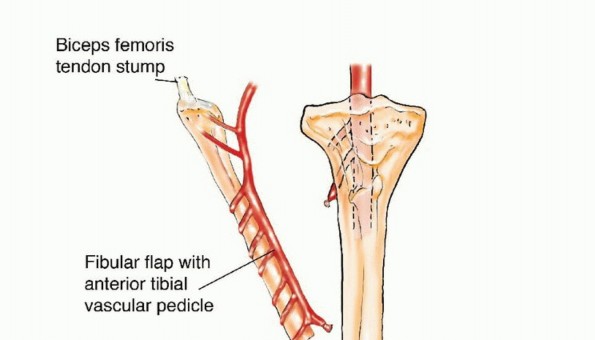
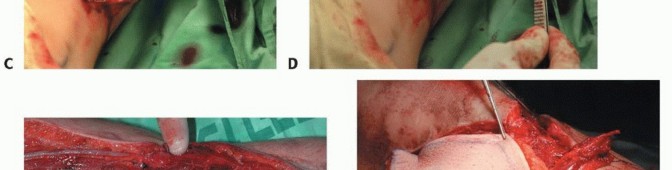
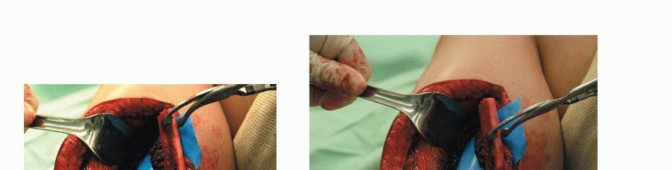
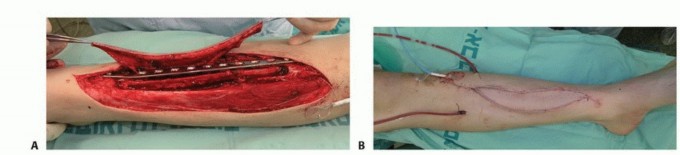
The osteotomies are performed using an oscillating saw or a Gigli saw under continuous saline irrigation to prevent thermal necrosis. Once the bone is divided, the distal peroneal artery and venae comitantes are identified, ligated, and divided. The fibula is then retracted laterally, allowing direct visualization of the posterior tibialis muscle. The muscle is divided, exposing the proximal peroneal pedicle. The pedicle is dissected proximally to its origin at the tibioperoneal trunk to maximize pedicle length. The tourniquet is deflated to confirm perfusion of the graft prior to pedicle division.
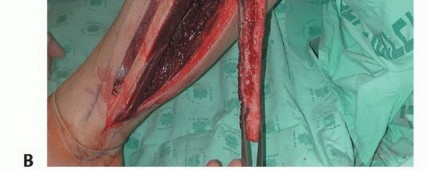
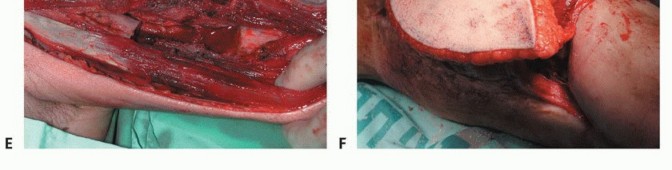
Recipient Site Preparation
While the harvest is underway, the recipient site team performs the necessary oncologic resection or radical debridement. The
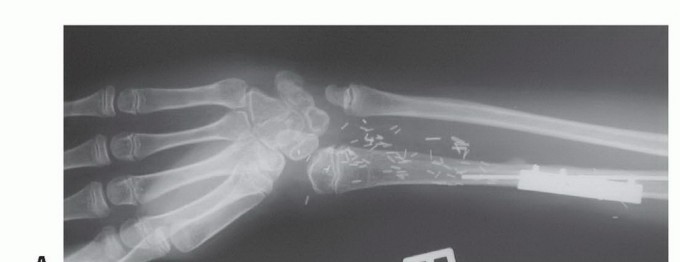
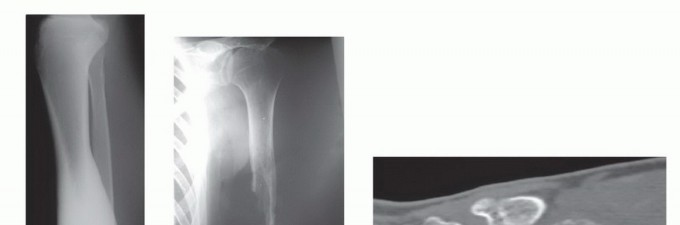
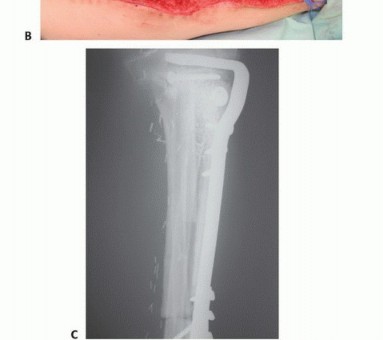
Clinical & Radiographic Imaging
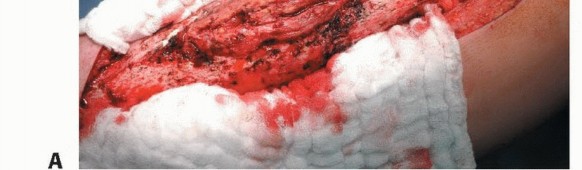
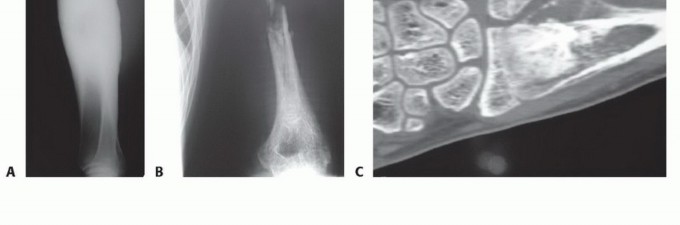

You Might Also Like