Deep Posterior Thigh Liposarcoma: An Orthopedic Oncology Case Study & Diagnostic Approach
Key Takeaway
Diagnosing deep posterior thigh liposarcoma (ALT) involves patient history of an enlarging painless mass and clinical exam of a firm, deep tumor. MRI is critical, revealing a fatty mass with thick enhancing septations and non-lipomatous nodules, vital for distinguishing it from benign lipomas and guiding treatment.
Patient Presentation & History
We present the case of Mr. J.A., a 62-year-old male, generally fit and active, who presented to our clinic with a chief complaint of a slowly enlarging, painless mass in his right posterior thigh. He first noticed the mass approximately 18 months prior to presentation, describing it as a small, deep-seated lump. Over the last 6 months, he reported a more noticeable increase in size, accompanied by a subtle feeling of pressure but no significant pain, paraesthesia, or motor weakness. He denied any history of trauma to the area.
His past medical history was significant for well-controlled hypertension and hyperlipidemia, managed with an ACE inhibitor and a statin, respectively. He had no history of prior malignancies or significant surgical interventions. His family history was non-contributory for sarcomas or other genetic conditions predisposing to malignancy. He was a non-smoker and consumed alcohol occasionally. Mr. J.A. remained functionally independent with an ECOG performance status of 0. His primary concern was the increasing size of the mass and a growing apprehension regarding its etiology.
Clinical Examination
Upon initial inspection, a prominent bulge was noted in the posteromedial aspect of the right mid-thigh. The overlying skin appeared normal, without erythema, discoloration, ulceration, or dilated superficial veins. There was no evidence of skin tethering. The mass appeared ovoid, with its long axis oriented longitudinally along the thigh.
Palpation revealed a deep-seated, firm-to-rubbery mass, approximately 10 x 8 cm in size, situated within the muscle compartment. The mass felt subtly lobulated and largely non-tender to deep palpation. It exhibited limited mobility in the transverse plane but was more mobile longitudinally, suggesting its origin within or intimately associated with muscular or fascial planes rather than being superficially encapsulated. There was no palpable warmth or pulsatility. Significant findings included:
- Size: Estimated 10 cm in greatest dimension.
- Consistency: Firm, rubbery, slightly heterogeneous.
- Mobility: Poorly mobile relative to deeper structures, suggesting deep fascial or intramuscular involvement.
- Tenderness: Mild, non-specific discomfort on deep palpation, not sharp.
- Neurovascular Assessment: Distal pulses (femoral, popliteal, dorsalis pedis, posterior tibial) were all 2+ and symmetrical. Capillary refill was brisk (<2 seconds). Sensation to light touch, pinprick, and proprioception was intact throughout the right lower extremity, corresponding to L2-S1 dermatomes. Motor strength was full (5/5) across all major muscle groups of the hip, knee, and ankle, including hip extension, knee flexion, and ankle plantarflexion, which could potentially be affected by a posterior thigh mass. No pathological reflexes were elicited.
- Range of Motion (ROM): Full active and passive range of motion of the right hip and knee joints was observed. There was no pain elicited with terminal flexion or extension of the knee or hip.
- Regional Lymph Nodes: No palpable inguinal lymphadenopathy.
Given the deep location, firmness, and progressive enlargement, a suspicious soft tissue mass requiring further investigation was immediately flagged.
Imaging & Diagnostics
Initial Radiographs
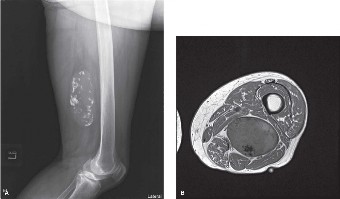
Plain radiographs of the right femur and knee (AP and lateral views) were performed. These films demonstrated a large, relatively radiolucent soft tissue mass in the posterior thigh, consistent with a fatty lesion. Importantly, there was no overt evidence of periosteal reaction, cortical erosion, or direct bony involvement. No calcifications within the mass were identified. While suggestive of a benign fatty tumor, the size and deep location mandated further, more advanced imaging.
Ultrasound
An initial focused ultrasound confirmed a large, hyperechoic mass with internal septations, consistent with a fatty tumor. It delineated the superficial margins but could not fully characterize the depth or relationship to neurovascular bundles due to its large size and deep location. Ultrasound-guided core needle biopsy was considered but deferred pending full MRI characterization.
Magnetic Resonance Imaging (MRI)
MRI of the right thigh was performed with and without intravenous gadolinium contrast. This was the critical imaging modality for local staging.
- Indications: To precisely define the size, extent, compartmental involvement, relationship to neurovascular structures, and characterize the internal architecture of the mass.
-
Findings:
- A large, multilobulated mass measuring approximately 10.5 x 8.0 x 7.2 cm was identified in the posteromedial compartment of the right mid-thigh.
- The mass was predominantly hyperintense on T1-weighted sequences and showed mild signal suppression on fat-saturated sequences, indicating a significant fatty component.
- Multiple, thick, enhancing septations and several focal, non-lipomatous nodular areas were observed within the mass. These nodules were isointense to muscle on T1, hyperintense on T2, and demonstrated heterogeneous post-contrast enhancement.
- The mass intimately abutted the semimembranosus and adductor magnus muscles, with evidence of mild displacement but no clear invasion of the muscle belly itself.
- The sciatic nerve and superficial femoral neurovascular bundle were displaced anteriorly and laterally by the mass but did not appear directly invaded.
- There was no direct cortical erosion or marrow invasion of the femur.
- Findings were highly suggestive of a well-differentiated liposarcoma (atypical lipomatous tumor - ALT).

- Interpretation: The presence of thick septations and non-lipomatous enhancing components within a predominantly fatty tumor in a deep location raised high suspicion for liposarcoma, differentiating it from a benign lipoma.
Computed Tomography (CT)
- CT Chest/Abdomen/Pelvis: A triple-phase CT scan of the chest, abdomen, and pelvis was performed for systemic staging. No evidence of pulmonary metastases, retroperitoneal adenopathy, or other distant metastatic disease was found. This ruled out systemic spread at the time of initial presentation.
- CT Thigh: Although MRI was primary for local staging, a CT of the thigh was also performed (primarily for surgical templating, though MRI provides superior soft tissue detail). It confirmed the low-attenuation fatty mass with internal septations and nodular components, consistent with MRI findings, further supporting the diagnosis of a liposarcoma. It helped confirm the absence of subtle calcifications or bony erosions not clearly visible on plain radiographs.
Biopsy
Given the imaging findings, a CT-guided core needle biopsy was performed by an interventional radiologist experienced in musculoskeletal oncology. The biopsy was planned longitudinally, directly over the palpable mass, in a manner that the biopsy tract could be entirely excised during definitive surgical resection. Multiple core samples were taken from both the fatty and the enhancing non-lipomatous areas.
- Histopathology: Microscopic examination revealed mature adipocytes with focal areas of atypical stromal cells exhibiting hyperchromatic, pleomorphic nuclei. Multiple thick fibrous septa were noted. Crucially, immunohistochemical staining showed amplification of MDM2 and CDK4 genes, which are characteristic genetic markers for well-differentiated liposarcoma/atypical lipomatous tumor (ALT). There was no evidence of a high-grade dedifferentiated component in the biopsied specimens.
- Diagnosis: Well-differentiated Liposarcoma (Atypical Lipomatous Tumor), consistent with a low-grade malignancy.
Differential Diagnosis
The presentation of a soft tissue mass necessitates a broad differential, particularly for deep-seated lesions in adults. For Mr. J.A., the imaging findings helped narrow the possibilities, but the core biopsy was ultimately diagnostic. Below is a detailed comparison of key differentials:
| Feature | Liposarcoma (Well-differentiated/Atypical Lipomatous Tumor) | Lipoma | Undifferentiated Pleomorphic Sarcoma (UPS/MFH) | Myxoid Liposarcoma | Schwannoma |
|---|---|---|---|---|---|
| Prevalence | Most common adult soft tissue sarcoma (20% of all STS); Well-differentiated is the most common subtype of liposarcoma. | Most common benign soft tissue tumor. | Common high-grade sarcoma, often a diagnosis of exclusion. | ~30% of all liposarcomas. | Common benign nerve sheath tumor. |
| Age Group | Predominantly 50-70 years. | Any age, most common in 40-60 years. | Typically 50-70 years. | Younger demographic (30-50 years). | Any age, typically 20-50 years. |
| Location | Deep soft tissues: retroperitoneum (most common), thigh, shoulder, popliteal fossa. Intracompartmental or intermuscular. | Subcutaneous (most common), less commonly deep (intramuscular, intermuscular). | Deep soft tissues: thigh, retroperitoneum, trunk, shoulder. | Deep soft tissues: thigh, buttock, popliteal fossa, retroperitoneum. | Typically associated with major nerves (e.g., sciatic, brachial plexus). |
| Growth Rate | Slow, insidious over months to years. Can accelerate if dedifferentiation occurs. | Very slow, stable for years. | Rapid, often noticed over weeks to months. | Slow, can accelerate. | Slow. |
| Pain | Usually painless until large enough to compress adjacent structures or nerves. | Painless, unless traumatized or if rapidly growing in a confined space. | Often painful, especially with rapid growth or necrosis. | Often painless; can cause pressure symptoms or nerve irritation. | Can be painful, often with neurological symptoms (paresthesia, weakness) if nerve is involved or compressed. |
| Mobility | Often fixed to deep structures dueperitumoral inflammation or infiltration. | Mobile, easily separable from surrounding tissues, encapsulated. | Fixed, infiltrative, often with indistinct margins. | Less mobile, often infiltrative, can appear pseudocapsulated on imaging. | Mobile perpendicular to the nerve, but fixed longitudinally (Tinell's sign). |
| MRI Features | Predominantly fat signal (T1 hyperintense, fat-saturated suppressed). Thick, irregular septations (>2mm), nodular non-fatty components with enhancement. | Uniform fat signal, thin or no septations (<2mm), no internal enhancement. | Heterogeneous signal (T1 variable, T2 hyperintense), marked heterogeneous enhancement, areas of necrosis, hemorrhage. | High water/mucinous content (T2 markedly hyperintense, T1 hypointense). Curvilinear "spiderweb" enhancement, nodular fatty components. | T1 isointense to muscle, T2 markedly hyperintense. "Target sign" (peripheral high signal, central low signal) and "split fat sign" (fat plane surrounding nerve). Cystic degeneration is common. Strong, homogeneous enhancement. |
| CT Features | Low attenuation (fat density), thick septations, enhancing non-lipomatous areas. | Uniformly low attenuation (fat density), usually well-circumscribed. | Heterogeneous attenuation, often with areas of higher density, necrosis, calcifications (rare but possible). | Variable attenuation, often low density due to myxoid component, can have internal calcifications. | Iso- or slightly hypo-attenuating on non-contrast CT. Homogeneous enhancement post-contrast. |
| Histology | Mature adipocytes with atypical stromal cells, hyperchromatic nuclei. MDM2/CDK4 amplification (FISH). | Mature adipocytes with no atypia or mitotic activity. | Pleomorphic spindle and polygonal cells, high mitotic activity, often with necrosis and hemorrhage. No specific line of differentiation. | Stellate cells in myxoid matrix, plexiform vascular pattern, occasional lipoblasts. FUS-DDIT3 or EWSR1-DDIT3 fusion. | Spindle cells (Schwann cells) in fascicular patterns (Antoni A) and myxoid areas (Antoni B). Verocay bodies. S100 positive. |
| Metastatic Potential | Low, but local recurrence is high (up to 50%). Risk of dedifferentiation. | None. | High (lung, bone). | Intermediate (lung, bone, other soft tissues - unusual for other sarcomas). | None. |
| Management | Wide local excision. Adjuvant RT considered for positive/close margins or large lesions. Long-term surveillance. | Simple marginal excision. | Wide local excision with adjuvant RT/chemotherapy. Staging scans essential. | Wide local excision. Adjuvant RT is highly effective (myxoid liposarcoma is very radiosensitive). Systemic therapy for advanced disease. | Enucleation or marginal excision. Neurolysis if needed. |
Surgical Decision Making & Classification
Mr. J.A.'s multidisciplinary tumor board discussion (Orthopedic Oncology, Medical Oncology, Radiation Oncology, Pathology, Radiology) concluded that the mass was a well-differentiated liposarcoma (atypical lipomatous tumor - ALT). The primary goal was complete surgical resection with clear margins to prevent local recurrence, given the negligible metastatic potential of this specific subtype.
Indications for Operative Intervention:
- Curative Intent: Surgical excision is the cornerstone of treatment for localized liposarcomas.
- Local Disease Control: The primary risk with well-differentiated liposarcoma/ALT is local recurrence, which can be morbid and increase the risk of dedifferentiation to a higher-grade sarcoma.
- Symptomatic Relief: Although currently asymptomatic, a growing mass of this size could eventually cause pain, functional impairment, or neurovascular compression.
- Definitive Diagnosis & Staging: While a biopsy provided initial diagnosis, the full pathological assessment of the resected specimen is crucial for complete staging and guiding any potential adjuvant therapy.
Classification (Enneking Staging System):
The Enneking Surgical Staging System for soft tissue sarcomas provides a framework for treatment planning and prognostication.
For Mr. J.A.'s case:
- Grade (G): Low-grade (G1) based on histopathology (well-differentiated liposarcoma).
- Site (T): Extracompartmental (T2) due to its deep location within the posterior thigh, potentially crossing fascial planes (MRI suggested abutment of multiple muscles).
- Metastasis (M): No regional or distant metastases (M0) based on clinical exam and staging CT scans.
Therefore, Mr. J.A.'s tumor was classified as Stage IA (G1, T2, M0) . This classification implies a low risk of systemic metastasis but a significant risk of local recurrence if not adequately excised.
Surgical Margins:
The target surgical margin for a low-grade extracompartmental sarcoma like this is a wide margin , defined as resection through a cuff of normal tissue at least 1 cm in all dimensions beyond the gross or microscopic limits of the tumor. For deep-seated tumors, this often necessitates the removal of adjacent muscle or fascia. The biopsy tract must also be excised en bloc with the main tumor specimen to prevent tumor cell seeding.
Neoadjuvant / Adjuvant Therapy:
- Neoadjuvant Radiation Therapy: Not routinely indicated for primary well-differentiated liposarcoma/ALT due to its low grade and excellent local control rates with adequate surgical margins. Its role is primarily for high-grade sarcomas or when very close margins are anticipated.
- Adjuvant Radiation Therapy: May be considered post-operatively if final pathological margins are found to be positive (R1) or very close (<1mm). In such cases, it aims to reduce local recurrence.
- Chemotherapy: Not effective for well-differentiated liposarcoma/ALT and was not considered.
The decision was made to proceed with wide local excision of the mass with a planned en bloc resection of the biopsy tract. The patient was counseled extensively on the risks and benefits, including the potential for neurovascular injury, significant soft tissue defect, and the need for possible reconstructive surgery.
Surgical Technique / Intervention
Pre-operative Planning:
Detailed pre-operative planning was paramount. MRI images were carefully reviewed to delineate tumor boundaries, its relationship to the sciatic nerve, profunda femoris artery/vein, and surrounding musculature. The biopsy tract was marked on the skin. A surgical incision was designed longitudinally in the posterior thigh, directly encompassing the biopsy site and allowing for a wide margin around the tumor, without crossing major anatomical planes unnecessarily.
Patient Positioning:
The patient was placed in the lateral decubitus position with the affected (right) leg uppermost, slightly flexed at the hip and knee. This provided optimal access to the posterior and posteromedial thigh. Adequate padding was used to protect pressure points.
Anesthesia and Preparation:
General anesthesia was administered. The limb was prepped and draped from the iliac crest to the foot in a sterile fashion, allowing for potential extension of the dissection or the harvesting of local/regional flaps if a large defect necessitated it.
Surgical Approach:
- Incision: A long, longitudinal skin incision was made in the posterior thigh, centered over the mass and incorporating the entire biopsy tract elliptically. The incision extended approximately 20 cm.
- Skin and Subcutaneous Dissection: Subcutaneous tissues were carefully incised. The skin flaps were raised full thickness to expose the underlying deep fascia, always maintaining a safe margin from the palpable tumor.
- Fascial Incision: The deep fascia of the thigh was incised longitudinally, well away from the apparent tumor margins, both proximally and distally, staying superficial to the mass initially.
- Identification of Neurovascular Structures: Meticulous dissection was performed to identify and protect the critical neurovascular structures. The sciatic nerve, lying deep and lateral to the tumor, was carefully identified and freed proximally and distally from its surrounding fascial attachments. The profunda femoris artery and vein were also identified anteriorly. These structures were found to be displaced by the tumor but were not adherent or invaded. Vessel loops were placed around the sciatic nerve for retraction and protection.
-
Tumor Resection (En Bloc Excision):
- The goal was a wide en bloc resection, ensuring at least 1-2 cm of healthy tissue circumferentially around the tumor and the biopsy tract.
- The tumor, along with a cuff of surrounding normal muscle (portions of semimembranosus and adductor magnus that were intimately associated), was carefully dissected.
- Sharp dissection was used throughout, minimizing blunt dissection near the tumor to avoid capsular rupture or tumor spillage.
- Feeding vessels to the mass were identified, ligated, and divided.
- The specimen was oriented with sutures and clips for the pathologist to accurately assess margins. Proximally, distally, medially, laterally, and deep margins were marked.
- The resected specimen measured approximately 13 x 10 x 9 cm, including the surrounding cuff of normal tissue and the biopsy tract.
Reconstruction:
Following tumor resection, a significant soft tissue defect remained in the posterior thigh.
- Muscle Closure: The remaining portions of the semimembranosus and adductor magnus muscles were mobilized and approximated loosely to fill the dead space, aiming to restore some anatomical contour and protect the exposed neurovascular structures. This also helped to reduce seroma formation.
- Fascial Closure: The deep fascia was reapproximated where possible.
- Subcutaneous and Skin Closure: A layered closure was performed. Subcutaneous tissues were closed with absorbable sutures. A large bore suction drain was placed within the defect. The skin was closed with non-absorbable staples.
Hemostasis:
Thorough hemostasis was achieved throughout the procedure using electrocautery and ligatures. Estimated blood loss was approximately 250 mL.
Intraoperative Pathology:
Frozen section analysis of the most suspicious deep and peripheral margins was performed and confirmed negative margins, reinforcing the success of the wide excision.
Post-Operative Protocol & Rehabilitation
Immediate Post-Operative Care:
- Pain Management: Multimodal analgesia including epidural, regional block (if performed pre-operatively), and oral/IV narcotics.
- Wound Care: Daily dressing changes, close monitoring for signs of infection, hematoma, or seroma. The suction drain was maintained until output was consistently less than 50 mL over 24 hours, typically 3-5 days.
- DVT Prophylaxis: Mechanical (SCDs) and chemical (LMWH) prophylaxis initiated post-operatively.
- Mobilization: Early mobilization was encouraged. The patient was allowed full weight-bearing on the affected leg as tolerated from Post-Op Day 1, with crutches for balance and comfort initially.
Pathology Report and Adjuvant Therapy:
The final pathology report confirmed a Well-differentiated Liposarcoma (Atypical Lipomatous Tumor) . All surgical margins were confirmed to be negative (R0 resection) , with the closest margin being >1 cm. Given the low-grade nature of the tumor and clear margins, no adjuvant radiation therapy or chemotherapy was indicated.
Rehabilitation Protocol:
-
Phase I (Weeks 0-2: Acute Recovery & Protection):
- Focus on pain control, wound healing, and minimizing edema.
- Gentle active and passive range of motion exercises for hip and knee within pain limits.
- Light isometric exercises for quadriceps, hamstrings, and gluteal muscles.
- Ambulation with crutches/walker for stability and confidence, progressing to partial weight-bearing as tolerated.
- Emphasis on deep breathing exercises to prevent pulmonary complications.
-
Phase II (Weeks 2-6: Early Strengthening & Functional Restoration):
- Once wound healing was satisfactory, progressive resistance exercises initiated.
- Focus on strengthening of the entire lower extremity, particularly the gluteal muscles and hamstrings to compensate for any muscle loss.
- Proprioceptive training (e.g., single-leg stance, balance exercises).
- Gradual reduction in assistive devices.
- Scar massage and desensitization initiated once wound closed.
-
Phase III (Weeks 6-12: Advanced Strengthening & Return to Activity):
- Continued progression of strengthening exercises, incorporating functional movements (squats, lunges, step-ups).
- Introduction of light cardiovascular activities (stationary cycling, swimming).
- Sport-specific or occupation-specific training as appropriate, depending on patient goals.
- Emphasis on regaining full range of motion and symmetrical strength.
-
Phase IV (Beyond 12 weeks: Long-Term Maintenance):
- Independent exercise program.
- Continued monitoring for local recurrence or functional deficits.
Mr. J.A. tolerated the surgery well and made an excellent recovery. He was discharged on Post-Op Day 5 with crutches and instructions for physiotherapy. By 6 weeks post-operatively, he was ambulating without aids and had regained near-full strength and range of motion, with minimal residual discomfort.
Follow-up Protocol:
Long-term surveillance is crucial due to the risk of local recurrence and potential for dedifferentiation.
- Clinical Examination: Every 3 months for the first 2 years, then every 6 months for the next 3 years, then annually.
- Local Imaging: MRI of the right thigh at 6 months post-op, then annually for 5 years, and then as clinically indicated. This is to detect early signs of local recurrence.
- Distant Metastasis Surveillance: Chest CT scan annually for the first 5 years, then every 2-3 years, given the very low but not zero risk of metastasis or dedifferentiation with distant spread over time.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
- Any Deep-Seated Soft Tissue Mass in an Adult is Malignant Until Proven Otherwise: This principle is paramount in orthopedic oncology. Never assume benignity based on palpation or initial plain films.
- MRI is the Gold Standard for Local Staging: Provides unparalleled detail on tumor size, exact location, compartmental involvement, and relationship to neurovascular structures. Essential for surgical planning.
-
Biopsy Planning is Critical:
- Performed by the Definitive Surgeon (or with their input): The biopsy tract must be planned so it can be entirely excised en bloc with the definitive specimen. Longitudinal incisions are preferred.
- Core Needle Biopsy (CNB) is Preferred: Less tissue disruption, lower risk of seeding compared to incisional biopsy. Avoid FNA for diagnosis of sarcomas as it often provides insufficient tissue for grading and subtyping.
- Adequate Tissue Sampling: Multiple cores from different areas, especially enhancing/solid components, are necessary to capture potential heterogeneity or higher-grade foci.
- Multidisciplinary Team (MDT) Approach: Essential for optimal management of sarcomas. Orthopedic oncologist, radiation oncologist, medical oncologist, radiologist, and pathologist must collaborate.
- Wide Surgical Margins are the Cornerstone: For localized sarcoma, achieving R0 (negative) margins is the most critical prognostic factor for local recurrence. Enneking's principles of surgical staging guide the extent of resection.
- Biopsy Tract Excision: Always excise the entire biopsy tract en bloc with the main tumor to prevent tumor cell seeding and recurrence along the tract.
-
Myxoid Liposarcoma Specifics:
- Highly radiosensitive, making neoadjuvant/adjuvant RT very effective.
- Metastases are common to unusual sites: retroperitoneum, other soft tissues, and bone, in addition to lung.
-
Well-Differentiated Liposarcoma (ALT):
- Rarely metastasizes but has a high local recurrence rate if margins are inadequate.
- Risk of dedifferentiation (transformation into a higher-grade sarcoma) in recurrent lesions, which then behave aggressively. Long-term surveillance is crucial.
- MDM2/CDK4 amplification is a key diagnostic marker.
Pitfalls:
-
Inadequate Biopsy:
- Fine-Needle Aspiration (FNA): Often insufficient for accurate sarcoma diagnosis, grading, and subtyping, leading to misdiagnosis or delayed treatment.
- Poorly Planned Incisional Biopsy: Transverse incisions, incisions directly over neurovascular bundles, or tracts that cross multiple compartments can compromise subsequent definitive resection, necessitating wider or more complex resections, or even amputation.
- Misdiagnosing Liposarcoma as Lipoma: Based solely on imaging showing a fatty mass without careful assessment for thick septations, non-lipomatous nodules, or enhancement patterns, especially in deep-seated lesions. Biopsy is mandatory for any suspicious fatty mass.
- Failure to Perform Complete Staging: Omitting a Chest CT for pulmonary metastases (even for low-grade lesions, as they can dedifferentiate) is a significant error.
- Inadequate Surgical Margins: Leading to high rates of local recurrence, which is the most common mode of failure for many sarcomas.
- Not Involving a Specialist Orthopedic Oncologist: Sarcoma management is complex and requires specialized expertise for optimal outcomes.
- Ignoring the Biopsy Tract: Failure to excise the biopsy tract en bloc with the tumor can lead to isolated local recurrence along the tract.
- Underestimating Dedifferentiation: While well-differentiated liposarcoma is low-grade, the potential for dedifferentiation, particularly in recurrent lesions, necessitates aggressive treatment and surveillance as it then behaves like a high-grade sarcoma.
- Lack of Long-Term Follow-up: Sarcomas can recur or metastasize years after initial treatment. A structured, long-term surveillance protocol is essential.
