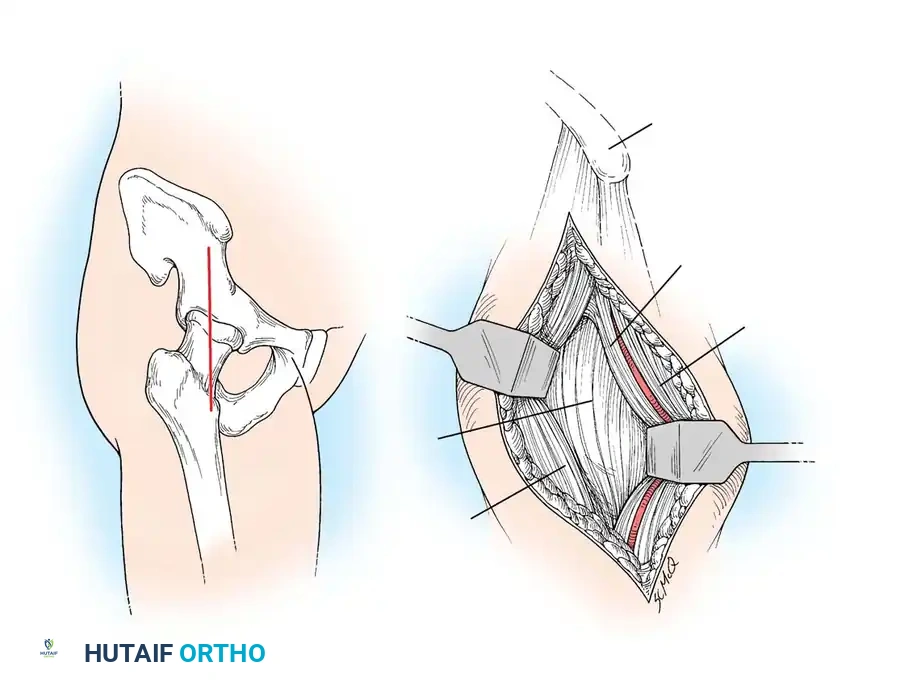
Unraveling Orthopaedic Infections and Osteomyelitis: History & Treatment
Key Takeaway
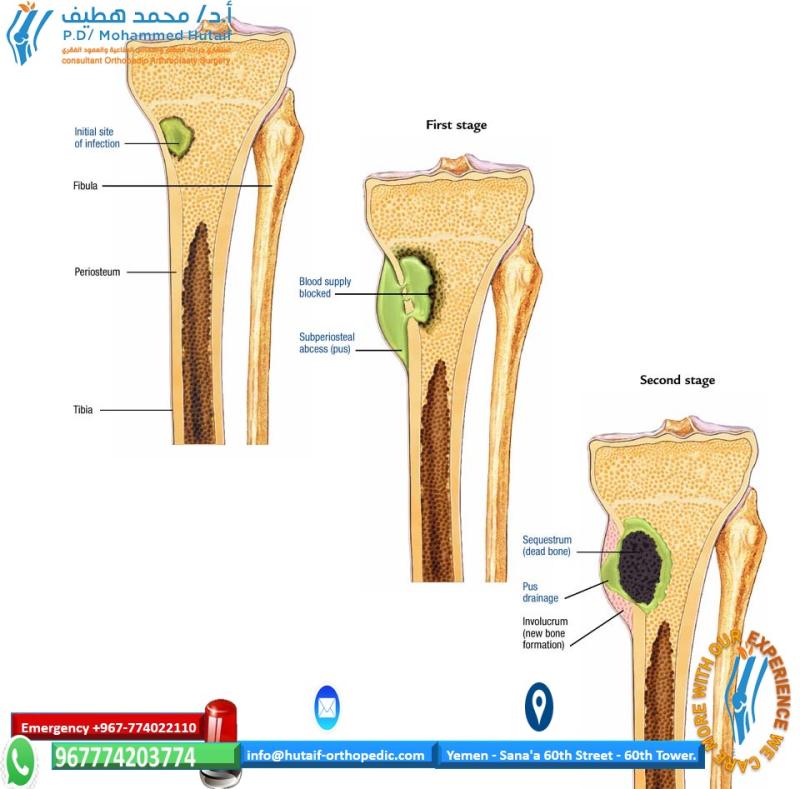
For anyone wondering about Unraveling Orthopaedic Infections and Osteomyelitis: History & Treatment, Infections, first described in ancient Sumerian carvings, were initially treated with remedies like honey before scientific methods emerged. The term osteomyelitis, coined in the mid-1800s, describes bone infections where a sequestrum (dead bone fragment) forms. The body's inflammatory response creates an involucrum, an enveloping sheath, to isolate the sequestrum, marking the natural history of infections and osteomyelitis.
INTRODUCTION
The first descriptions of infections date back to the
early Sumerian carvings, when the tenets of treatment were irrigation,
immobilization, and bandaging.82 In
these early times, the practice of infection and wound care was
essentially an art and there was very little science applied to it.
Treatment included the use of honey, wine, and donkey feces, and there
were a number of philosophies regarding the value of purulence.
Dominant personalities had a significant influence over medical
practice and the value of purulence persisted because of the writings
of Galen of Pergamum (120-201 A.D.). It was not until the latter third
of the second millennium that the concept of the value of purulence
would be challenged.82
In the past three centuries, the treatment of infection
has involved the use of local ointments or salves and the maintenance
of an open wound that permitted purulence to exit the body. Some
important terms were adopted into medical parlance. A sequestrum was defined as “a fragment of dead bone separated from the body.” The word sequestrum is derived from the Latin words sequester meaning “depositary” and sequestrate meaning “to give up for safe keeping.” The word sequestrum is used to describe a detached piece of bone lying within a cavity formed by necrosis. The term involucrum derives from the Latin word involucrum
meaning “enveloping sheath or envelope.” This term describes the
effects of the body’s inflammatory response when trying to envelope and
isolate the sequestrum from the host. The natural history of
osteomyelitis was seen as the process of isolation of the infective
material followed by a slow attempted resorption of the material by the
immune system. However, the term osteomyelitis was not coined until the mid-1800s, when it was adopted by Nelaton.98
In his book The Story of Orthopaedics, Mercer Rang describes the three pivotal discoveries that allowed orthopaedic surgery to be successful:98
anesthesia, antisepsis, and radiography. The first two were important
in all surgical specialties. Anesthesia made surgery tolerable, but
there was still considerable morbidity secondary to infection. It was
not until the mid-1800s that progress with antisepsis permitted
infection control and more effective surgical intervention. As a result
of this, infection issues
became
an integral part of medicine and were studied in a more formal basis.
However, descriptions of the first sequestrectomies of the tibia had
been illustrated as early as 1593 by Scultetus.98
Prior to anesthesia, most operative procedures were
performed using forced immobility and inebriation. Operating rooms were
created because procedures undertaken in the wards horrified patients
who witnessed them and the screams of agony did nothing to encourage
other patients to seek surgical treatment. Thus, the patients were
isolated from the rest of the ward. In the same era, many modern drugs
were developed, including morphine, heroin, nitrous oxide, and ether.
Ether was in fact serendipitously identified as an anesthetic agent
during one of the drug parties that were common at this time. However,
it was first used for anesthesia in Massachusetts General Hospital in
1846 by William T. G. Morton, and its use quickly caught on around the
world. This increased the incentive to undertake surgical procedures.
The ensuing increase in the number of surgical procedures, together
with the lack of antisepsis, meant that the morbidity and mortality of
surgery also increased.98 Pasteur
and Lister are most commonly credited as being the forerunners of
antisepsis, but the most notable achievement in demonstrating the
efficacy of bacterial transmission is the work of Semmelweiss, who, in
1848, demonstrated that hand washing between obstetric deliveries
reduced maternal mortality from 18% to about 1%. Lister read Pasteur’s
work on fermentation and likened tissue putrefaction to the same
process. He subsequently developed carbolic acid, which reduced
mortality from amputation from 43% in an untreated cohort of patients
to 15% in a treated cohort. Despite this significant discovery, his
findings were resisted for decades. Even when his concepts were
adopted, the remaining pieces of the puzzle required for successful
aseptic surgery did not come together for another 100 years.
The initial use of antibiotics was just as serendipitous
as the use of anesthesia and antisepsis. Some antibacterial treatments
were introduced, but it was not until the discovery of penicillin by
Alexander Fleming in 1928 that the proven usefulness of antibiotics
became understood. Even Fleming did not vigorously pursue his
discovery. However, when Florey and Chain read Fleming’s initial
report, they pursued and found the true impact of penicillin, which was
effective against streptococci. Since then, many antibiotics have been
developed, but the number of resistant bacteria has also increased.
Hand washing, gloves, hats, enclosed rooms, aseptic techniques, and
early antibiotics all slightly decreased the incidence of surgical
infection. However, the operating theaters in the early 1900s still
admitted observers who coughed, did not use masks, and wore street
clothes. It was not until the mid-20th century that surgeons began to
integrate all the controllable aspects of patient exposure to
infectious agents by attempting to standardize the contributive effects
of the environment, patient, surgeon, wound, antisepsis, antibiotics,
and surgical techniques. It is likely, though, that many of the answers
to the problem of infection remain undiscovered, and it seems likely
that at the moment we do not fully understand the complex symbiosis
between bacteria and humans.
This chapter will concentrate on the description,
etiology, diagnosis, and management of orthopaedic infections but will
have a specific focus on posttraumatic conditions. Historically, the
treatment of orthopaedic infection was either ablative, when an
amputation was performed, or temporizing with treatment of a chronic
wound or sinus. There was little chance of limb salvage as we know it
today, and infections that were not adequately treated would
occasionally become systemic and fatal.
Certainly the high mortality of open gunshot wounds to
the femur in the American Civil War and World War I were largely due to
sepsis. In every war, the science of surgery and medicine advances, and
this is particularly true for trauma surgery and extremity injuries,
which still account for approximately 65% of all war-related injuries.83 Thus, many advances in infection treatment and extremity injuries have ironically come about as a result of war.
To treat orthopaedic infection, one must first
understand the basics of the interdependence of humans and bacteria.
Bacteria are a necessary part of our existence and normal flora live in
abundance on our bodies. It is worth considering that an individual’s
skin can contain up to 180 different types of bacteria at any given
time.45 There are up to 10
colony-forming units (CFUs) of bacteria in the mouth and perineum.
Nearly 95% of bacteria found in the hands exist under the fingernails.
The average human is composed of 100 trillion cells, but it is thought
that we harbor over a 1000 trillion bacteria in or on our bodies. Our
blood is constantly infiltrated with bacteria from breaks in the skin,
translocation across mucous membranes, and other roots. However, nearly
all of these bacteria are quickly and efficiently eradicated by our
host defense mechanisms. It is the disruption of our own homeostasis
that provides an opportunity for either external contaminant or
opportunistic host bacteria to become pathogenic and cause infection.
While colonization necessarily precedes infection, the presence of
bacteria by itself does not constitute infection. This is highlighted
by the findings of one study of hardware removal in which 50% of
cultures were positive in patients with no signs of symptoms or
infection.80 Thus, there is an
important distinction between colonization and infection. Understanding
the factors that have changed the local or systemic environment with
resultant bacterial infection is the key to effective prophylaxis,
treatment, and improved outcomes in orthopaedic surgery.
CLASSIFICATION
Historically, osteomyelitis was classified as either
acute or chronic depending on the duration of symptoms. Kelly
documented a classification system based on the etiology of the
osteomyelitis.61 There were four
types with type I being hematogenous osteomyelitis. Type II was
osteomyelitis associated with fracture union, while type III was
osteomyelitis without fracture union and type IV was postoperative or
posttraumatic osteomyelitis without a fracture. Weiland et al.123
in 1984 suggested another classification scheme based on the nature of
the bony involvement. In this classification system, there were three
types, with type I being characterized by open exposed bone without
evidence of osseous infection but with evidence of soft tissue
infection. In type II fractures, there was circumferential cortical and
endosteal infection, and in type III fractures, the cortical and
endosteal infection was associated with a segmental defect.
In 1989, May et al.71
proposed another classification scheme for osteomyelitis focusing on
the tibia. This system was based on the nature of the bone following
soft tissue and bony débridement. They proposed that there were five
different categories.
Type
I posttraumatic tibial osteomyelitis was defined as being present when
the intact tibia and fibula were able to withstand functional loads and
no reconstruction was required. In type II osteomyelitis, the intact
tibia was unable to withstand functional loading and required bone
grafting. In type III osteomyelitis, there was an intact fibula but a
tibial defect that measured no more than 6 cm. The tibial defect
required cancellous bone grafting, tibiofibular synostosis, or
distraction histogenesis. Type IV osteomyelitis was characterized by an
intact fibula but with a defect of more than 6 cm in length, which
required distraction osteogenesis, tibiofibular synostosis, or a
vascularized bone graft. Type V osteomyelitis was characterized by a
tibial defect of more than 6 cm without an intact fibula, which often
required amputation.
The Waldvogel classification121
categorized osteomyelitis into three primary etiologies—hematogenous,
contiguous (from an adjacent root such as an open fracture or a seeded
implant), or chronic, this being a longstanding osteomyelitis with
mature host reaction.
These various classification systems were predicated on
the beliefs and treatment options of the times, and they have all
become less relevant with current diagnostic and treatment modalities.
However, each classification represented an important effort to
categorize the pathophysiology of bone infection to facilitate the
choice of an effective treatment.
The currently accepted classification remains the Cierny-Mader classification,21 which not only describes the pathology in the bone but, more importantly, also classifies the host or patient (Tables 24-1 and 24-2).
The usefulness of the Cierny-Mader system is its applicability to
clinical practice and the wealth of experience and data gleaned from a
single surgeon’s practice with meticulous records. The hallmark of
Cierny’s approach is the use of oncologic principles for treatment. In
fact, osteomyelitis behaves very similarly to a benign bone tumor in
that it is rarely lethal but has a tendency to return without complete
eradiation. Interestingly, the outcome data reported by Cierny et al.21
indicate that once appropriate surgical treatment is undertaken, the
host may be the most important variable affecting treatment and outcome.
A novel aspect of the Cierny-Mader classification is its
analysis of the physiologic state of the patient or host. The host is
classified by the number of systemic and local comorbidities. An A host
has a healthy physiology and limb with little systemic or local
compromise. The B host is further divided into one with local
compromise (B local), systemic compromise (B systemic), or both (B
systemic/local systemic compromise, which includes any
immunocompromised condition, poor nutrition, diabetes, old age,
multiple trauma, chronic hypoxia, vascular disease, malignancy, or
organ failure such as renal insufficiency or liver failure). Local
compromise includes conditions such as previous surgery or trauma,
cellulitis, radiation fibrosis, scarring from burns or trauma, local
manifestations of vascular disease, lymphoedema, or zone-of-injury
issues. We believe that a new variable of compromise can be identified
in the trauma patient where systemic compromise is due to multiple
organ damage and the consequent systemic response to trauma and local
compromise is defined by the zone-of-injury effects on local tissues.
The C host is a patient in whom the morbidity of
treatment is greater than the morbidity of disease because of multiple
and severe comorbid conditions that cannot be treated safely. In these
patients, the risks of curative treatment such as extensive surgery, as
might be used with free flaps, or prolonged reconstruction with bone
transport would be greater than that caused by the infective condition
itself. Type C hosts are often better treated with limited nonablative
surgery and suppression or, if appropriate, by an amputation.
In the Cierny-Mader classification21,
the bone lesion is classified by the extent of involvement and
stability. Type I is a medullary or endosteal infection without
penetration through cortex. This is the type of infection that occurs
after intramedullary nailing. Type II is a superficial osteomyelitis
that involves only the outer cortex and is frequently contiguous with a
pressure ulcer or adjacent abscess. Type III is permeative in that
there is involvement of both cortical and medullary bone but,
importantly, there is no loss of axial stability of the bone. Type IV
also involves cortical and medullary bone but in a segmental fashion
such that axial stability is lost. Types III and IV would be typical
infections related to open fractures. In type IV lesions, the segmental
resection that is required necessitates reconstruction of the bone,
whereas in type III lesions, additional stabilization may not be
required (Tables 24-1 and 24-2).
The pairing of the four types of osteomyelitis with the
three host classes allows for the development of practical treatment
strategies. Cierny et al.21 proposed
a detailed treatment regimen defining optimal treatment modalities for
each stage. They achieved an overall clinical 2-year success rate of
91% for all states. As one would expect, when their results were broken
down by class of host and type of lesion. Class A hosts fared the best.
In class A hosts, success rates of 98% were achieved even with type IV
osteomyelitis. The compromised class B host success rates were far
lower, ranging from 79% to 92% depending on anatomic type. In his
series Cierny found that the host class seemed to be more important
than the type of infection. A cumulative success rate of greater than
90% was achieved with most of the failures being in B hosts. C hosts
were recommended for amputation or suppressive treatment.22
The lessons that stem from their findings are that it is important not
just to treat the disease but also the host and that the patient’s
physiologic condition should be optimized. Thus, a B systemic-local
host who has had a previous open fracture but also smokes and has
uncontrolled diabetes, renal insufficiency, and malnutrition should
have all of these problems treated together with the bone disease.
Improving host status would appear to be a fruitful endeavor when one
| considers Cierny et al.’s21,22 findings.
TABLE 24-1 Cierny-Mader Classification of Bone
--- | Type I—Medullary | Osseous location | Involvement |
| --- | --- | --- |
| Infection is limited to the medullary canal. Typically seen after intramedullary nailing. | |
Type II—Superficial
Infection is limited to the exterior of the bone and does not penetrate the cortex. Typically seen from pressure ulcers. | |
Type III—Permeative/Stable
Infection
penetrates cortex but bone is axially stable and generally will not
require supplemental stabilization. Typically seen after internal
fixation with plates. | |
Type IV—Permeative/Unstable
Infection is
throughout the bone in segmental fashion and results in axial
instability. Typically seen in extensive infections or after aggressive
débridement of type III infections that results in loss of axial
stability. | |
It should be noted that Cierny et al.’s21,22
results used outcome criteria that were commonly used at that time.
Current outcome studies focus more on subjective patient-based
assessments than on surgeon-based assessments. We do not have much data
on the functional outcomes in the scenarios described by Cierny and
colleagues, and it is possible that some of the patients whom they
salvaged would have fared better with prosthetic replacement and vice
versa. The findings of the LEAP study for acute limb salvage have
raised new questions about the true nature of outcome and success.65
PATHOGENESIS
Before a discussion of diagnosis and treatment, it is
vital to understand the mechanisms by which infections occur. Most
infections encountered in orthopaedics are related to biofilm-forming
bacteria.
Much of our understanding of biofilm bacteria has come from the Centre
for Biofilm Engineering in Bozeman, Montana. Biofilm bacteria are also
important in the oil, food processing, naval, paper manufacturing, and
water processing industries.
Biofilm bacteria exist in one of two states—the planktonic state or the stationary state (Fig. 24-1).
Planktonic state bacteria are free floating in the extracellular matrix
and are isolated and relatively small in quantity. In this state, the
body host defenses can easily eradicate the organism through the usual
immunologic mechanisms. It is rare for planktonic bacteria to survive
long in the extracellular matrix despite numerous and repeated
occurrences of entry. However, if the bacterial load is large and
sustained, they can overwhelm the host defenses and escape the effects
of antibiotics. They can then invade tissue and blood, leading to
septicemia and death. Planktonic bacteria are also metabolically active
and reproductive. This is an important consideration for antibiotic
treatments that work by either interfering with cell wall or protein
synthesis or with reproduction.
If planktonic bacteria encounter a suitable inert surface such
as dead or necrotic tissue, foreign bodies, or any avascular body part
by either direct contamination, contiguous spreading, or hematogenous
seeding, they can attach and begin the process of colonization.
Juxtaposition of the bacteria with a surface or biomaterial is
accomplished by Van der Waals forces, which allow bacteria to develop
irreversible cross-links with the surface (adhesion-receptor
interaction).27
Adhesion is based on time-dependent specific protein adhesion-receptor
interactions, as well as carbohydrate polymer synthesis in addition to
| charge and physical forces.61 Following adhesion to a surface, bacteria begin to create a
TABLE 24-2 Cierny-Mader Classification of the Host
--- |
Host Class
|
Description
|
| --- | --- |
| A host | Healthy physiology and limb |
| B host: systemic | Diabetes, |
stable multiple organ disease, nicotine use, substance abuse,
immunologic deficiency, malnutrition, malignancy, old age, vascular
disease
Trauma context:
Multiple injuries
B host: local | Previous trauma, burns, previous surgery, vascular disease, cellulitis, scarring, previous radiation treatment, lymphedema
Trauma context:
Zone of injury
B host: systemic/ local | Combinations of systemic and local conditions
C host | Multiple
uncorrectable comorbidities. Unable to tolerate extent of surgical
reconstruction required. Treatment of the disease is worse than the
disease itself
 FIGURE 24-1
Illustration of bacterial attachment to a surface followed by
colonization and detachment. (Redrawn with permission after P. Dirckx,
MSU Center for Biofilm Engineering, Bozeman, MT.)
mucopolysaccharide layer called biofilm or slime. They then develop into colonies. These colonies exhibit remarkably resilient behavior. Figure 24-2
illustrates mature biofilm colonies where pillars of a mature biofilm
are visible distributed on top of a monolayer of surface-associated
cells. In addition to fixed cells, there are motile cells, which
maintain their association with the biofilm for long periods, swimming
between pillars of biofilm-associated bacteria.122
The interaction of the colonies and bacteria demonstrates complex
communication via proteins or markers that can alter bacterial behavior.
In the early stages of colonization, sessile bacteria
can be killed or neutralized by the host defenses. However, some of
these bacteria may escape destruction and potentially act as a nidus
for future infection. Transition from colonization to infection usually
requires other conditions to exist. This might occur if there was an
inoculum that was larger than threshold levels, impaired host immune
defense mechanisms, traumatized or necrotic tissues, foreign body, or
an acellular or inanimate surface such as dead bone, cartilage, or
biomaterials.
As previously discussed, the first step in the
transition from colonization to infection requires bacterial adhesion,
which will usually not occur on viable tissue surfaces. Thus, when
foreign material or dead tissue is found in the body, a “race for the
surface” begins. Host cells will attempt to incorporate nonliving
material or sequester nonviable tissue via encapsulation so that a
well-incorporated biomaterial implant that has such a tissueintegrated
neocapsule will be resistant to bacterial adhesion. Furthermore, the
same tissue integration can often isolate bacteria that have become
sessile on an implant surface by sequestering the bacteria from
necessary nutrients until host mechanisms can act.
However, if bacteria encounter the surface and develop
mature colonies, tissue integration by the host may be impaired and the
process of infection may proceed. Damaged bone, being relatively
acellular, acts as a suitable surface for bacterial adhesion and
colonization.69 Devitalized bone
devoid of normal periosteum presents a collagen matrix to which
bacteria can bind. Moreover, it has been suggested that bone
sialoprotein can act as a ligand for bacterial binding to bone.69
Biomaterials and other foreign bodies are usually inert and susceptible
to bacterial colonization because they are inanimate. Regardless of how
inert a metal is, it may still modulate molecular events on its
surfaces, these being receptor-ligand interactions, covalent bonding,
and thermodynamic interactions.44,50
The most important feature of any particular method is the interaction
between its outer surface atomic layer and the glycoproteins of
prokarytotic and eukaryotic cells. Stainless steel and cobalt-chromium
and titanium alloys are resistant to corrosion because of several
mechanisms including surface oxide passivates. These surface oxides
form a reactive interface with bacteria that can promote colony
formation. There is therefore a balance between implanting devices with
surface structures that lower corrosion rates but
might
increase the likelihood of surface binding by bacteria. Thus, a large
surface area and bacteria inoculum, combined with local tissue damage
and a compromised or insufficient host response, can collectively
create the necessary conditions for infection.
FIGURE 24-1
Illustration of bacterial attachment to a surface followed by
colonization and detachment. (Redrawn with permission after P. Dirckx,
MSU Center for Biofilm Engineering, Bozeman, MT.)
mucopolysaccharide layer called biofilm or slime. They then develop into colonies. These colonies exhibit remarkably resilient behavior. Figure 24-2
illustrates mature biofilm colonies where pillars of a mature biofilm
are visible distributed on top of a monolayer of surface-associated
cells. In addition to fixed cells, there are motile cells, which
maintain their association with the biofilm for long periods, swimming
between pillars of biofilm-associated bacteria.122
The interaction of the colonies and bacteria demonstrates complex
communication via proteins or markers that can alter bacterial behavior.
In the early stages of colonization, sessile bacteria
can be killed or neutralized by the host defenses. However, some of
these bacteria may escape destruction and potentially act as a nidus
for future infection. Transition from colonization to infection usually
requires other conditions to exist. This might occur if there was an
inoculum that was larger than threshold levels, impaired host immune
defense mechanisms, traumatized or necrotic tissues, foreign body, or
an acellular or inanimate surface such as dead bone, cartilage, or
biomaterials.
As previously discussed, the first step in the
transition from colonization to infection requires bacterial adhesion,
which will usually not occur on viable tissue surfaces. Thus, when
foreign material or dead tissue is found in the body, a “race for the
surface” begins. Host cells will attempt to incorporate nonliving
material or sequester nonviable tissue via encapsulation so that a
well-incorporated biomaterial implant that has such a tissueintegrated
neocapsule will be resistant to bacterial adhesion. Furthermore, the
same tissue integration can often isolate bacteria that have become
sessile on an implant surface by sequestering the bacteria from
necessary nutrients until host mechanisms can act.
However, if bacteria encounter the surface and develop
mature colonies, tissue integration by the host may be impaired and the
process of infection may proceed. Damaged bone, being relatively
acellular, acts as a suitable surface for bacterial adhesion and
colonization.69 Devitalized bone
devoid of normal periosteum presents a collagen matrix to which
bacteria can bind. Moreover, it has been suggested that bone
sialoprotein can act as a ligand for bacterial binding to bone.69
Biomaterials and other foreign bodies are usually inert and susceptible
to bacterial colonization because they are inanimate. Regardless of how
inert a metal is, it may still modulate molecular events on its
surfaces, these being receptor-ligand interactions, covalent bonding,
and thermodynamic interactions.44,50
The most important feature of any particular method is the interaction
between its outer surface atomic layer and the glycoproteins of
prokarytotic and eukaryotic cells. Stainless steel and cobalt-chromium
and titanium alloys are resistant to corrosion because of several
mechanisms including surface oxide passivates. These surface oxides
form a reactive interface with bacteria that can promote colony
formation. There is therefore a balance between implanting devices with
surface structures that lower corrosion rates but
might
increase the likelihood of surface binding by bacteria. Thus, a large
surface area and bacteria inoculum, combined with local tissue damage
and a compromised or insufficient host response, can collectively
create the necessary conditions for infection.
---
FIGURE 24-2
Mature biofilm colonies showing potential intercolony communication.
(Redrawn with permission after P. Dirckx, MSU Center for Biofilm
Engineering, Bozeman, MT.)
Following bacterial adherence and colonization, the resistance to antibiotics appears to increase.84,86
This resistance is dependent on the type of surface to which the
organisms are attached. Organisms that adhere to hydrocarbon polymers
are extremely resistant to antibiotics. These same organisms, when
attached to metals, do not resist antibiotic therapy to the same
extent. Bacterial colonies can undergo phenotypic changes and appear to
hibernate. They can survive in a dormant state without causing
infection, and this can explain the recovery of bacteria from
asymptomatic hardware removal.80 So while colonization is a necessary antecedent for infection, colonization alone does not necessarily lead to infection.
Two characteristics of colonized bacteria may help
understand and explain this pseudo-resistance. Because the passage of
antibiotics through tissues is based on a diffusion gradient, colonized
bacteria are insulated with a natural barrier of glycocalyx, often
referred to as a slime, through which the circulating antibiotic must diffuse before arriving at the bacterial cell wall (Fig. 24-3).
The antibiotic molecules must then diffuse into the bacterial cell or
be transported by metabolically active bacterial cell membranes.
Because it is theorized that bacteria within biofilms have a decreased
metabolic rate and undergo phenotypic changes, active processes such as
cell membrane formation, which are targeted by antibiotics, would be
similarly decreased (Fig. 24-3).116
Consequently, antibiotic concentrations of 1500 times normal may be
required to penetrate both the biofilm and the bacterial cell wall.
Even then, most antimicrobials work via interference with cell wall
synthesis or cellular reproduction, and they therefore require
metabolically active bacteria to be effective. Thus, bacteria in the
biofilm may be dormant and appear to be pseudoresistant. The more
metabolically inactive the bacteria, the less bactericidal will be the
antibiotic therapy, which is why mature or chronic infections can
rarely be cured with antibiotics alone. Table 24-3
outlines the major antibiotic classes and their mechanisms of action,
all of which may be limited by the bacterial state in biofilm.
 ---
FIGURE 24-3
Biofilm creates a diffusion barrier that interferes with the ability of
antibiotics to reach bacterial organisms. Biofilm bacteria are
metabolically inactive and therefore not subject to the mechanism of
action of most antibiotics. They appear as “pseudoresistant.” (Redrawn
with permission after P. Dirckx, MSU Center for Biofilm Engineering,
Bozeman, MT.)
Once colonization occurs, body defenses continue to
identify bacteria as foreign. There may be chemotactic mechanisms that
keep immune cells active. The subsequent collection of inflammatory
cells brought in to wall off the bacteria via chemotaxis manifests as purulence,
which is a symptom of the host’s attempt to isolate and destroy the
infection. The acute inflammatory cells will also release a spectrum of
oxidative and enzymatic
products
in an attempt to penetrate the glycocalyx. These mediators and enzymes
are nonspecific and may be toxic to host tissue. Increased host tissue
damage can lead to more surface substrate for local bacteria, creating
a cycle of tissue damage, host response, and exacerbation of infection (Fig. 24-4).
The host tissues will eventually react to limit the spread of infection
macroscopically as well as microscopically. The clinical manifestation
of a sequestered infection is an abscess or involucrum. Alternatively,
if the infection grows and reaches the skin or an internal epithelial
surface, a sinus tract forms as a route to dispel detritus. While the
appearance of a sinus tract is a manifestation of a locally devastating
disease process and indicates severe underlying infection, it should be
remembered that it may also prevent the accumulation of internal
| fixation, which can lead to bacteremia and septicemia.
TABLE 24-3 Major Antibiotic Classes and Their Mechanism of Action
--- | Inhibition of cell wall synthesis/development | Penicillin, cephalosporins, vancomycin, bacitracin, chlorhexidine |
| --- | --- |
| Inhibition of protein synthesis | Chloramphenicol, macrolides, lincosamides, tetracyclines |
| Inhibition of RNA synthesis | Rifampin |
| Inhibition of DNA synthesis | Quinolones, macrolides |
| Inhibition of enzymatic/metabolic activity | Trimethoprim-sulfamethoxazole (blocks folic acid production) |
Source: www.sigmaaldrich.com/Area_of_Interest/Biochemicals/Antibiotic_Explorer/Mechanism_of_Action.html.
FIGURE 24-4
Autoinjury mechanism of host white cells in response to biofilm bacteria.
A.
Host white cell engulfs planktonic bacteria and then
B.
moves to engulf a bacterial colony that has developed but is unable to do so.
C.
Host white cell’s next response to engulfed bacteria is to release
oxidative enzymes, but those enzymes also cause damage to local host
cells.
D.
Unsuccessful eradication of
bacteria and colony growth attracts more host white cells, resulting in
increased damage to host tissue.
Eventually, an equilibrium may exist in the form of a chronic
infection, which is what many surgeons see in practice. There is
usually a history of intermittent symptoms and drainage that has
responded to some type of antibiotic regimen. What this probably
represents is the inhibition of colony expansion at the borders of the
infectious site. Clinically harmful manifestations of infection are
generally caused by the release of bacteria into the bloodstream that
are metabolically active and release toxins in addition to the release
of oxidative enzymes by the host cell. Although the bacteria remain
susceptible to the body’s host defenses and to antibiotics, their
numbers and continued release into the bloodstream represent a chronic
debilitating disease. Any acute stress on the host environment from
trauma, disease, or immunosuppression can allow the infection to
strengthen and spread. Thus, longstanding infections that were
tolerated by young healthy individuals may suddenly become limb or life
threatening as the individual’s age.
New developments stemming from the work of the Bozeman
group provide novel opportunities to treat bacterial infection of
orthopaedic implants. These include surface coatings, agents that
inhibit colonization or promote dissolution of colonies, small electric
fields, and low pH and acidic and negatively charged surfaces that are
resistant to biofilms. Surface properties of implants or local or
systemic drugs may help decrease the risk to infection, particularly in
the elderly population, who have decreased immune system activity.17
INFECTION AFTER FRACTURE
Infection after fracture is most likely to be associated
with open fractures or invasive surgical procedures. Few closed
fractures treated nonoperatively develop osteomyelitis. To improve the
diagnosis of posttraumatic bone infection, it is necessary to
understand the mechanisms of infection, particularly for open fractures.
---
FIGURE 24-3
Biofilm creates a diffusion barrier that interferes with the ability of
antibiotics to reach bacterial organisms. Biofilm bacteria are
metabolically inactive and therefore not subject to the mechanism of
action of most antibiotics. They appear as “pseudoresistant.” (Redrawn
with permission after P. Dirckx, MSU Center for Biofilm Engineering,
Bozeman, MT.)
Once colonization occurs, body defenses continue to
identify bacteria as foreign. There may be chemotactic mechanisms that
keep immune cells active. The subsequent collection of inflammatory
cells brought in to wall off the bacteria via chemotaxis manifests as purulence,
which is a symptom of the host’s attempt to isolate and destroy the
infection. The acute inflammatory cells will also release a spectrum of
oxidative and enzymatic
products
in an attempt to penetrate the glycocalyx. These mediators and enzymes
are nonspecific and may be toxic to host tissue. Increased host tissue
damage can lead to more surface substrate for local bacteria, creating
a cycle of tissue damage, host response, and exacerbation of infection (Fig. 24-4).
The host tissues will eventually react to limit the spread of infection
macroscopically as well as microscopically. The clinical manifestation
of a sequestered infection is an abscess or involucrum. Alternatively,
if the infection grows and reaches the skin or an internal epithelial
surface, a sinus tract forms as a route to dispel detritus. While the
appearance of a sinus tract is a manifestation of a locally devastating
disease process and indicates severe underlying infection, it should be
remembered that it may also prevent the accumulation of internal
| fixation, which can lead to bacteremia and septicemia.
TABLE 24-3 Major Antibiotic Classes and Their Mechanism of Action
--- | Inhibition of cell wall synthesis/development | Penicillin, cephalosporins, vancomycin, bacitracin, chlorhexidine |
| --- | --- |
| Inhibition of protein synthesis | Chloramphenicol, macrolides, lincosamides, tetracyclines |
| Inhibition of RNA synthesis | Rifampin |
| Inhibition of DNA synthesis | Quinolones, macrolides |
| Inhibition of enzymatic/metabolic activity | Trimethoprim-sulfamethoxazole (blocks folic acid production) |
Source: www.sigmaaldrich.com/Area_of_Interest/Biochemicals/Antibiotic_Explorer/Mechanism_of_Action.html.
FIGURE 24-4
Autoinjury mechanism of host white cells in response to biofilm bacteria.
A.
Host white cell engulfs planktonic bacteria and then
B.
moves to engulf a bacterial colony that has developed but is unable to do so.
C.
Host white cell’s next response to engulfed bacteria is to release
oxidative enzymes, but those enzymes also cause damage to local host
cells.
D.
Unsuccessful eradication of
bacteria and colony growth attracts more host white cells, resulting in
increased damage to host tissue.
Eventually, an equilibrium may exist in the form of a chronic
infection, which is what many surgeons see in practice. There is
usually a history of intermittent symptoms and drainage that has
responded to some type of antibiotic regimen. What this probably
represents is the inhibition of colony expansion at the borders of the
infectious site. Clinically harmful manifestations of infection are
generally caused by the release of bacteria into the bloodstream that
are metabolically active and release toxins in addition to the release
of oxidative enzymes by the host cell. Although the bacteria remain
susceptible to the body’s host defenses and to antibiotics, their
numbers and continued release into the bloodstream represent a chronic
debilitating disease. Any acute stress on the host environment from
trauma, disease, or immunosuppression can allow the infection to
strengthen and spread. Thus, longstanding infections that were
tolerated by young healthy individuals may suddenly become limb or life
threatening as the individual’s age.
New developments stemming from the work of the Bozeman
group provide novel opportunities to treat bacterial infection of
orthopaedic implants. These include surface coatings, agents that
inhibit colonization or promote dissolution of colonies, small electric
fields, and low pH and acidic and negatively charged surfaces that are
resistant to biofilms. Surface properties of implants or local or
systemic drugs may help decrease the risk to infection, particularly in
the elderly population, who have decreased immune system activity.17
INFECTION AFTER FRACTURE
Infection after fracture is most likely to be associated
with open fractures or invasive surgical procedures. Few closed
fractures treated nonoperatively develop osteomyelitis. To improve the
diagnosis of posttraumatic bone infection, it is necessary to
understand the mechanisms of infection, particularly for open fractures.
 ---
FIGURE 24-5
Operative photographs of a severe open fracture.
A.
The appearance before surgical débridement.
B.
The appearance after surgical débridement. Note that after débridement,
the tissues and wound appear as if they were surgically created. While
it is unlikely that all bacteria have been removed, a thorough
exploration and débridement leaving behind only viable tissues will
minimize the risk of subsequent infection.
Approximately 60% to 70% of open fractures are
contaminated by bacteria, but a much small percentage develop
infection. The risk of infection correlates significantly with the
degree of soft tissue injury.117 If
one remembers that merely the presence of bacteria in an open wound is
not sufficient to cause infection, it is important to recognize that a
severely contaminated fracture can rarely be débrided to the point of
achieving a sterile or bacteria-free tissue bed. We believe that next
to removing the majority of bacteria from the contaminated tissue bed,
the second major goal of a wide and aggressive débridement is to leave
behind a viable tissue bed with minimal necrotic or inert surfaces for
the remaining bacteria to colonize. By minimizing the bacterial
contamination by eliminating adhesions and nutrition, the host gains an
opportunity to eradicate any remaining contaminants in the zone of
injury. Figure 24-5 demonstrates the concept
of open fracture débridement where a contaminated wound is débrided
until the remaining wound looks as if it is created surgically, with
residual tissue being healthy with little evidence of contamination. It
is important to remember that contamination can penetrate into tissue
planes or locations that are not obvious in the initial wound. The use
of pulsatile irrigation before surgical exploration and débridement may
in fact push the initial contaminants deeper into the tissues and
result in contaminants being left behind in a locally compromised
tissue bed. This will increase the likelihood of both acute and delayed
infection.
An important fact that is often unrecognized is that the
bacteria recovered from clinical infections are not necessarily the
bacteria found acutely in the contaminated tissue bed. Several studies
have found that routine cultures of open fractures are not useful
because the predominant organism recovered from acute cultures is
frequently not the organism recovered if and when an infection occurs.
Antibiotic treatment based on the acute culture, whether before or
after débridement, may be detrimental
because
the antibiotic that is chosen may not be specifically indicated and has
the potential to promote changes and overgrowth in the bacterial flora.
In the worst case scenario, routine antibiotic treatment based on
initial wound cultures may promote the development of resistant
bacterial strains.63,92,118
Many of the organisms responsible for eventual osteomyelitis are often hospital-acquired pathogens such as resistant Staphylococcus aureus or gram-negative bacilli, including Pseudomonas aeruginosa,51,67
which are not initially present in a traumatic wound. This does not
mean that other bacteria should not be considered and these may depend
on the environment. Clostridium perfringens must be considered if there is soil contamination and Pseudomonas, and Aeromonas hydrophila may be present following a freshwater injury. Vibrio and Erysipelothrix
may be present in saltwater injuries. One possible explanation for the
lack of correlation between acute cultures and the eventual infection
may be that the initial contaminants are of low virulence and easily
neutralized by a combination of débridement and antibiotics but that
the locally and, in polytrauma, the systemically, compromised tissue
bed is susceptible to the more aggressive nosocomial organisms.
ACUTE POSTTRAUMATIC OSTEOMYELITIS
Acute posttraumatic osteomyelitis is a bone infection
that results in traumatic injury that allows pathogenic organisms to
make contact with damaged bone and soft tissues, with a proliferation
and expression of infection.74 In a
patient with traumatic injuries, additional factors that contribute to
the subsequent development of osteomyelitis are the presence of
hypotension, inadequate débridement of the fracture site, malnutrition,
sustained intensive care unit hospitalization, alcoholism, and smoking.42,115
Trauma may lead to interference with the host response to infection.
Tissue injury or the presence of bacteria triggers activation of the
complement cascade that leads to local vasodilatation, tissue edema,
migration of polymorphonuclear leukocytes (PMNs) to the site of the
injury, and enhanced ability to phagocytes to ingest bacteria.56
Trauma has been reported to delay the inflammatory response to bacteria
as well as to depress cell-mediated immunity and to impair the function
of PMNs, including chemotaxis, superoxide production, and microbial
killing.56 The commonly used system of Cierny-Mader21
has been shown to have a close correlation with the general condition
of the patient rather than the specifics of bone involvement.
CHRONIC OSTEOMYELITIS
This condition is often the result of an acute
osteomyelitis that is inadequately treated. General factors that may
predispose to chronic osteomyelitis include the degree of bone
necrosis, poor nutrition, the infecting organism, the age of the
patient, the presence of comorbidities, and drug abuse.26
The infecting organism generally varies with the cause of the chronic
osteomyelitis. Chronic osteomyelitis results from acute osteomyelitis
and is frequently caused by S. aureus,
although chronic osteomyelitis that occurs after a fracture can be
polymicrobial or gram negative. Intravenous drug users are commonly
found to have Pseudomonas as well as S. aureus
infections. Gram-negative organisms are now seen in up to 50% of all
cases of chronic osteomyelitis, and this may be due to variables such
as surgical intervention, chronic antibiotics, nosocomial causes, or
changes in the bacterial flora of the tissue bed.26
The fundamental problem in chronic osteomyelitis is a slow progressive
revascularization of bone that leaves protected pockets of necrotic
material to support bacterial growth that are relatively protected from
systemic antibiotic therapy. This collection of necrotic tissue, bone,
and bacteria is what becomes termed a sequestrum,
and the body’s attempt to wall off the offending material with reactive
inflammatory tissue, whether this is bone or soft tissue, is termed the
involucrum. The involucrum can be highly
vascular and may be viable and structural, and this should be taken
into consideration during surgical débridement.
FUNGAL OSTEOMYELITIS
Fungal osteoarticular infections are caused by two groups of fungi. The dimorphic fungi, which include Blastomyces dermatitidis, Ciccidioides sp., Histoplasma capsulatum, and Sporothrix schenckii, typically cause infections in healthy hosts in endemic regions, while Candida sp., Cryptococcus, and Aspergillus
cause infections in immunocompromised hosts. Infection is introduced by
direct trauma or injury but may be associated with a penetrating
foreign body or hematogenous spread.
Candida sp. is the most
common fungus seen in osteomyelitis. It affects both native and
prosthetic joints, vertebrae, and long bones. Risk factors include loss
of skin integrity, diabetes, malnutrition, immunosuppressive therapy,
intravenous drug use, hyperalimentation, the use of central venous
catheters, intra-articular steroid injections, and the use of
broad-spectrum antibiotics. A combined approach to therapy using
medical and surgical modalities is necessary for optimal results. Azole
antifungals and lipid preparations of Amphotericin B have expanded the
therapeutic options in fungal osteomyelitis as there is reduced
toxicity associated with long-term therapy.74
CLINICAL AND LABORATORY DIAGNOSTIC TESTS
A history of infection or intercurrent illness as well
as of remote surgery or trauma should raise the clinical suspicion of
osteomyelitis. Normal signs of inflammation may be absent and thus the
diagnosis of infection may be difficult. Patients may have a history of
infection at another site, such as the lungs, bladder, or skin in
conjunction with a history of trauma. They usually complain of pain in
the affected area and feel generally unwell. Moreover, reduced
activity, malaise, anorexia, fever, tachycardia, and listlessness may
be present. Local findings include swelling and warmth, occasional
erythema, tenderness to palpation, drainage, and restricted range of
motion in adjacent joints.
Aspects of the clinical history that should alert the
surgeon to look for infection include a history of open fracture,
severe soft tissue injury, a history of substance abuse and smoking,
inadequate previous treatment, or an immunocompromised state. These are
all factors that contribute to a B host. Factors affecting treatment
that need to be assessed include the time of onset of the infection,
the status of the soft tissues, the viability of the bone, the status
of fracture healing, implant stability, the condition of the host, and
the neurovascular examination (Fig. 24-6).
---
FIGURE 24-6
Typical appearance of a postoperative wound. The limb looks relatively
benign. This patient had an extensive type III infection and had been
treated with attempted débridement on several occasions before
referral. Poor nutrition and nicotine use together with her previous
multiple surgeries made her a B systemic/local host.
Routine blood cultures are of little help unless
patients show manifestations of systemic disease, but they may be
positive in up to 50% to 75% of cases where there is concomitant
bacteremia or septicemia.124 Blood
cultures that yield coagulasenegative staphylococci, a common
contaminant and pathogen, must be correlated with other clinical
findings before attribution of clinical significance. Blood results
that are suggestive of infection include an elevation of the white
blood cell (WBC) count and elevations in the C-reactive protein (CRP)
and erythrocyte sedimentation rate (ESR) levels. The ESR may be normal
in the first 48 hours but rises to levels about 100 mm/hr and may
remain elevated for several weeks. It is, however, a nonspecific marker.124
Combination of the ESR with the CRP improves specificity such that if
both are negative, the specificity is 90% to 95% for acute
osteomyelitis. In other words, a negative CRP and ESR makes
osteomyelitis unlikely. Their values are also age dependent, and there
is a steady increase in normal values with aging. In one recent study,
the ESR and CRP were found to be useful diagnostic tools for the
detection of an infected arthroplasty. While they had low sensitivities
and positive predictive values and therefore were of little value for
screening, they had high specificity and negative predictive value and
therefore were useful for treatment decisions.49
These studies and other diagnostic studies may not be as useful in
acute postoperative and chronic infections. In the acute setting, the
ESR and CRP are expected to be elevated due to local and systemic
inflammation from the surgical procedure. In chronic infections, the
host has had time to adapt to the offending condition and thus may not
mount the response required to trigger an elevation in these tests.
Once osteomyelitis treatment is initiated, the CRP and ESR are useful
in following the response to treatment. We use the ESR and CRP to
establish a baseline value before débridement and initiation of
antibiotic therapy and to monitor the subsequent response to treatment.
Radiographic Imaging
Radiologic findings in the initial presentation of acute
osteomyelitis are often normal. The most common radiographic signs of
bone infection are rarefaction, which represents diffuse
demineralization secondary to inflammatory hyperemia; soft tissue
swelling with obliteration of tissue planes; trabecular destruction;
lysis; cortical permeation; periosteal reaction; and involucrum
formation. Radiologically detectable demineralization may not be seen
for at least 10 days after the onset of acute osteomyelitis.124
When present, mineralization usually signifies trabecular bone
destruction. If the infection spreads to the cortex, usually within 3
to 6 weeks, a periosteal reaction may be seen on radiographs. One study
reported that in cases of proven osteomyelitis, 5% of radiographs were
abnormal initially, 33% were abnormal by 1 week, and 90% were abnormal
by 4 weeks.6 In trauma and fracture
treatment, the nature of callus formation and the obfuscation of bone
by hardware may make radiologic changes difficult to recognize in the
early or middle states of infection. Often it is not until there is a
clear sequestrum, sinus, or involucrum that parallels the clinical
findings that specific radiographic changes are recognized (Fig. 24-7).
Bone Scintigraphy
Scintigraphy has been widely used and remains a very
useful diagnostic tool. There are numerous types of scintigraphy, but
three scan types are commonly used to diagnose musculoskeletal
infection. These are the bone scan, which uses tagged red cells; the
leukocyte scan, which uses tagged white cells; and the bone marrow
scan, which investigates marrow cell activity. Recently, positron
emission tomography (PET) has shown promise and is undergoing increased
investigation and use.
Technetium-99m is the principal radioisotope used in most whole body red cell bone scans.28,32,43
Technetium is formed as a metastable intermediate during the decay of
molybdenum-99. It has a 6-hour half-life and is relatively inexpensive
and
readily available.28
After intravenous injection, there is a rapid distribution of this
agent throughout the extracellular fluid. Within several hours, more
than half the dose will accumulate in bone, while the remainder is
excreted in the urine. Technetium phosphates bind to both the organic
and inorganic matrix. However, the key characteristic that makes
technetium scanning useful is that there is preferential incorporation
into metabolically active bone. Bone images are usually acquired 2 to 4
hours following intravenous injection of the isotope. A triple-phase
bone scan is one that is useful for examining general inflammation and
related processes. Following the initial injection, dynamic images are
captured over the specified region. These are followed by static images
at later time points. The first phase represents the blood flow phase,
the second phase immediately postinjection represents the bone pooling
phase, and the third phase is a delayed image made at 3 hours when
there is decreased soft tissue activity. Classically, osteomyelitis
presents as a region of increased blood flow, and it should appear
“hot” in all phases with focal uptake in the third phase (Fig. 24-8).
Other processes such as healing fractures, loose prostheses, and
degenerative change do not appear hot in the early phase despite a hot
appearance in the delayed phase. Reported sensitivities of bone
scintigraphy for the detection of osteomyelitis vary considerably from
32% to 100%. Reported specificities have ranged from 0% to 100%.103,120
---
FIGURE 24-5
Operative photographs of a severe open fracture.
A.
The appearance before surgical débridement.
B.
The appearance after surgical débridement. Note that after débridement,
the tissues and wound appear as if they were surgically created. While
it is unlikely that all bacteria have been removed, a thorough
exploration and débridement leaving behind only viable tissues will
minimize the risk of subsequent infection.
Approximately 60% to 70% of open fractures are
contaminated by bacteria, but a much small percentage develop
infection. The risk of infection correlates significantly with the
degree of soft tissue injury.117 If
one remembers that merely the presence of bacteria in an open wound is
not sufficient to cause infection, it is important to recognize that a
severely contaminated fracture can rarely be débrided to the point of
achieving a sterile or bacteria-free tissue bed. We believe that next
to removing the majority of bacteria from the contaminated tissue bed,
the second major goal of a wide and aggressive débridement is to leave
behind a viable tissue bed with minimal necrotic or inert surfaces for
the remaining bacteria to colonize. By minimizing the bacterial
contamination by eliminating adhesions and nutrition, the host gains an
opportunity to eradicate any remaining contaminants in the zone of
injury. Figure 24-5 demonstrates the concept
of open fracture débridement where a contaminated wound is débrided
until the remaining wound looks as if it is created surgically, with
residual tissue being healthy with little evidence of contamination. It
is important to remember that contamination can penetrate into tissue
planes or locations that are not obvious in the initial wound. The use
of pulsatile irrigation before surgical exploration and débridement may
in fact push the initial contaminants deeper into the tissues and
result in contaminants being left behind in a locally compromised
tissue bed. This will increase the likelihood of both acute and delayed
infection.
An important fact that is often unrecognized is that the
bacteria recovered from clinical infections are not necessarily the
bacteria found acutely in the contaminated tissue bed. Several studies
have found that routine cultures of open fractures are not useful
because the predominant organism recovered from acute cultures is
frequently not the organism recovered if and when an infection occurs.
Antibiotic treatment based on the acute culture, whether before or
after débridement, may be detrimental
because
the antibiotic that is chosen may not be specifically indicated and has
the potential to promote changes and overgrowth in the bacterial flora.
In the worst case scenario, routine antibiotic treatment based on
initial wound cultures may promote the development of resistant
bacterial strains.63,92,118
Many of the organisms responsible for eventual osteomyelitis are often hospital-acquired pathogens such as resistant Staphylococcus aureus or gram-negative bacilli, including Pseudomonas aeruginosa,51,67
which are not initially present in a traumatic wound. This does not
mean that other bacteria should not be considered and these may depend
on the environment. Clostridium perfringens must be considered if there is soil contamination and Pseudomonas, and Aeromonas hydrophila may be present following a freshwater injury. Vibrio and Erysipelothrix
may be present in saltwater injuries. One possible explanation for the
lack of correlation between acute cultures and the eventual infection
may be that the initial contaminants are of low virulence and easily
neutralized by a combination of débridement and antibiotics but that
the locally and, in polytrauma, the systemically, compromised tissue
bed is susceptible to the more aggressive nosocomial organisms.
ACUTE POSTTRAUMATIC OSTEOMYELITIS
Acute posttraumatic osteomyelitis is a bone infection
that results in traumatic injury that allows pathogenic organisms to
make contact with damaged bone and soft tissues, with a proliferation
and expression of infection.74 In a
patient with traumatic injuries, additional factors that contribute to
the subsequent development of osteomyelitis are the presence of
hypotension, inadequate débridement of the fracture site, malnutrition,
sustained intensive care unit hospitalization, alcoholism, and smoking.42,115
Trauma may lead to interference with the host response to infection.
Tissue injury or the presence of bacteria triggers activation of the
complement cascade that leads to local vasodilatation, tissue edema,
migration of polymorphonuclear leukocytes (PMNs) to the site of the
injury, and enhanced ability to phagocytes to ingest bacteria.56
Trauma has been reported to delay the inflammatory response to bacteria
as well as to depress cell-mediated immunity and to impair the function
of PMNs, including chemotaxis, superoxide production, and microbial
killing.56 The commonly used system of Cierny-Mader21
has been shown to have a close correlation with the general condition
of the patient rather than the specifics of bone involvement.
CHRONIC OSTEOMYELITIS
This condition is often the result of an acute
osteomyelitis that is inadequately treated. General factors that may
predispose to chronic osteomyelitis include the degree of bone
necrosis, poor nutrition, the infecting organism, the age of the
patient, the presence of comorbidities, and drug abuse.26
The infecting organism generally varies with the cause of the chronic
osteomyelitis. Chronic osteomyelitis results from acute osteomyelitis
and is frequently caused by S. aureus,
although chronic osteomyelitis that occurs after a fracture can be
polymicrobial or gram negative. Intravenous drug users are commonly
found to have Pseudomonas as well as S. aureus
infections. Gram-negative organisms are now seen in up to 50% of all
cases of chronic osteomyelitis, and this may be due to variables such
as surgical intervention, chronic antibiotics, nosocomial causes, or
changes in the bacterial flora of the tissue bed.26
The fundamental problem in chronic osteomyelitis is a slow progressive
revascularization of bone that leaves protected pockets of necrotic
material to support bacterial growth that are relatively protected from
systemic antibiotic therapy. This collection of necrotic tissue, bone,
and bacteria is what becomes termed a sequestrum,
and the body’s attempt to wall off the offending material with reactive
inflammatory tissue, whether this is bone or soft tissue, is termed the
involucrum. The involucrum can be highly
vascular and may be viable and structural, and this should be taken
into consideration during surgical débridement.
FUNGAL OSTEOMYELITIS
Fungal osteoarticular infections are caused by two groups of fungi. The dimorphic fungi, which include Blastomyces dermatitidis, Ciccidioides sp., Histoplasma capsulatum, and Sporothrix schenckii, typically cause infections in healthy hosts in endemic regions, while Candida sp., Cryptococcus, and Aspergillus
cause infections in immunocompromised hosts. Infection is introduced by
direct trauma or injury but may be associated with a penetrating
foreign body or hematogenous spread.
Candida sp. is the most
common fungus seen in osteomyelitis. It affects both native and
prosthetic joints, vertebrae, and long bones. Risk factors include loss
of skin integrity, diabetes, malnutrition, immunosuppressive therapy,
intravenous drug use, hyperalimentation, the use of central venous
catheters, intra-articular steroid injections, and the use of
broad-spectrum antibiotics. A combined approach to therapy using
medical and surgical modalities is necessary for optimal results. Azole
antifungals and lipid preparations of Amphotericin B have expanded the
therapeutic options in fungal osteomyelitis as there is reduced
toxicity associated with long-term therapy.74
CLINICAL AND LABORATORY DIAGNOSTIC TESTS
A history of infection or intercurrent illness as well
as of remote surgery or trauma should raise the clinical suspicion of
osteomyelitis. Normal signs of inflammation may be absent and thus the
diagnosis of infection may be difficult. Patients may have a history of
infection at another site, such as the lungs, bladder, or skin in
conjunction with a history of trauma. They usually complain of pain in
the affected area and feel generally unwell. Moreover, reduced
activity, malaise, anorexia, fever, tachycardia, and listlessness may
be present. Local findings include swelling and warmth, occasional
erythema, tenderness to palpation, drainage, and restricted range of
motion in adjacent joints.
Aspects of the clinical history that should alert the
surgeon to look for infection include a history of open fracture,
severe soft tissue injury, a history of substance abuse and smoking,
inadequate previous treatment, or an immunocompromised state. These are
all factors that contribute to a B host. Factors affecting treatment
that need to be assessed include the time of onset of the infection,
the status of the soft tissues, the viability of the bone, the status
of fracture healing, implant stability, the condition of the host, and
the neurovascular examination (Fig. 24-6).
---
FIGURE 24-6
Typical appearance of a postoperative wound. The limb looks relatively
benign. This patient had an extensive type III infection and had been
treated with attempted débridement on several occasions before
referral. Poor nutrition and nicotine use together with her previous
multiple surgeries made her a B systemic/local host.
Routine blood cultures are of little help unless
patients show manifestations of systemic disease, but they may be
positive in up to 50% to 75% of cases where there is concomitant
bacteremia or septicemia.124 Blood
cultures that yield coagulasenegative staphylococci, a common
contaminant and pathogen, must be correlated with other clinical
findings before attribution of clinical significance. Blood results
that are suggestive of infection include an elevation of the white
blood cell (WBC) count and elevations in the C-reactive protein (CRP)
and erythrocyte sedimentation rate (ESR) levels. The ESR may be normal
in the first 48 hours but rises to levels about 100 mm/hr and may
remain elevated for several weeks. It is, however, a nonspecific marker.124
Combination of the ESR with the CRP improves specificity such that if
both are negative, the specificity is 90% to 95% for acute
osteomyelitis. In other words, a negative CRP and ESR makes
osteomyelitis unlikely. Their values are also age dependent, and there
is a steady increase in normal values with aging. In one recent study,
the ESR and CRP were found to be useful diagnostic tools for the
detection of an infected arthroplasty. While they had low sensitivities
and positive predictive values and therefore were of little value for
screening, they had high specificity and negative predictive value and
therefore were useful for treatment decisions.49
These studies and other diagnostic studies may not be as useful in
acute postoperative and chronic infections. In the acute setting, the
ESR and CRP are expected to be elevated due to local and systemic
inflammation from the surgical procedure. In chronic infections, the
host has had time to adapt to the offending condition and thus may not
mount the response required to trigger an elevation in these tests.
Once osteomyelitis treatment is initiated, the CRP and ESR are useful
in following the response to treatment. We use the ESR and CRP to
establish a baseline value before débridement and initiation of
antibiotic therapy and to monitor the subsequent response to treatment.
Radiographic Imaging
Radiologic findings in the initial presentation of acute
osteomyelitis are often normal. The most common radiographic signs of
bone infection are rarefaction, which represents diffuse
demineralization secondary to inflammatory hyperemia; soft tissue
swelling with obliteration of tissue planes; trabecular destruction;
lysis; cortical permeation; periosteal reaction; and involucrum
formation. Radiologically detectable demineralization may not be seen
for at least 10 days after the onset of acute osteomyelitis.124
When present, mineralization usually signifies trabecular bone
destruction. If the infection spreads to the cortex, usually within 3
to 6 weeks, a periosteal reaction may be seen on radiographs. One study
reported that in cases of proven osteomyelitis, 5% of radiographs were
abnormal initially, 33% were abnormal by 1 week, and 90% were abnormal
by 4 weeks.6 In trauma and fracture
treatment, the nature of callus formation and the obfuscation of bone
by hardware may make radiologic changes difficult to recognize in the
early or middle states of infection. Often it is not until there is a
clear sequestrum, sinus, or involucrum that parallels the clinical
findings that specific radiographic changes are recognized (Fig. 24-7).
Bone Scintigraphy
Scintigraphy has been widely used and remains a very
useful diagnostic tool. There are numerous types of scintigraphy, but
three scan types are commonly used to diagnose musculoskeletal
infection. These are the bone scan, which uses tagged red cells; the
leukocyte scan, which uses tagged white cells; and the bone marrow
scan, which investigates marrow cell activity. Recently, positron
emission tomography (PET) has shown promise and is undergoing increased
investigation and use.
Technetium-99m is the principal radioisotope used in most whole body red cell bone scans.28,32,43
Technetium is formed as a metastable intermediate during the decay of
molybdenum-99. It has a 6-hour half-life and is relatively inexpensive
and
readily available.28
After intravenous injection, there is a rapid distribution of this
agent throughout the extracellular fluid. Within several hours, more
than half the dose will accumulate in bone, while the remainder is
excreted in the urine. Technetium phosphates bind to both the organic
and inorganic matrix. However, the key characteristic that makes
technetium scanning useful is that there is preferential incorporation
into metabolically active bone. Bone images are usually acquired 2 to 4
hours following intravenous injection of the isotope. A triple-phase
bone scan is one that is useful for examining general inflammation and
related processes. Following the initial injection, dynamic images are
captured over the specified region. These are followed by static images
at later time points. The first phase represents the blood flow phase,
the second phase immediately postinjection represents the bone pooling
phase, and the third phase is a delayed image made at 3 hours when
there is decreased soft tissue activity. Classically, osteomyelitis
presents as a region of increased blood flow, and it should appear
“hot” in all phases with focal uptake in the third phase (Fig. 24-8).
Other processes such as healing fractures, loose prostheses, and
degenerative change do not appear hot in the early phase despite a hot
appearance in the delayed phase. Reported sensitivities of bone
scintigraphy for the detection of osteomyelitis vary considerably from
32% to 100%. Reported specificities have ranged from 0% to 100%.103,120
 ---
FIGURE 24-7
Radiograph of patient in Figure 24-6. The arrow points to periosteal reaction.
Gallium-67 citrate binds rapidly to serum proteins, particularly Transferrin.10,100
There is also uptake in the blood, especially by leukocytes. Gallium
has been used in conjunction with technetium-99 to increase the
specificity of the bone scanning.40,52
Several mechanisms have been postulated to explain the increased
activity at sites of inflammation. Enhanced blood flow and increased
capillary permeability cause enhanced delivery. Bacteria have high iron
requirements and thus take up gallium. Gallium is strongly bound to
bacterial siderophores and leukocyte lactoferrins. In regions of
inflammation, these proteins are available extracellularly and can bind
with gallium avidly. Chemotaxis also acts to localize gallium-labeled
WBCs at the sites of infection. In a typical study, gallium is injected
intravenously and delayed images are acquired at 48 to 72 hours. The
hallmark of osteomyelitis is the focal increased uptake of gallium.
Unfortunately, gallium’s nonspecific bone uptake can be problematic
because any processes causing reactive new bone formation will appear
hot. In patients with fractures or a prosthesis, osteomyelitis cannot
be easily diagnosed with gallium alone. Gallium images are usually
interpreted in conjunction with a technetium bone scan. Gallium
activity is interpreted as abnormal either if it is incongruous with
the bone scan activity or if there is a matching pattern with gallium
activity. Reported sensitivities and specificities for the diagnosis of
osteomyelitis range from 22% to 100% and 0% to 100%, respectively.2,52,76,103
Despite its lower-than-optimal diagnostic value, gallium still has some
advantages. It is easily administered and it is the agent of choice in
chronic soft tissue injection, although it is less effective in bone
infections. It has also proved useful in following the resolution of an
inflammatory process by showing a progressive decline in activity.
---
FIGURE 24-8
Red cell scan of patient in Figure 24-6 demonstrating increased activity in distal femur.
An indium-111 or 99mTc-hexamethylpropyleneamine osime
(99mTc-HMPAO) (Ceretec; GE Healthcare) -labeled leukocyte scan is the
most common scan used in conjunction with a standard bone scan. The
labeled leukocytes migrate to the region of active infection resulting
in a hot white cell scan over the area of active inflammation. The use
of a combined red cell and white cell scan significantly increases both
the sensitivity and specificity and now represents the g
---
FIGURE 24-7
Radiograph of patient in Figure 24-6. The arrow points to periosteal reaction.
Gallium-67 citrate binds rapidly to serum proteins, particularly Transferrin.10,100
There is also uptake in the blood, especially by leukocytes. Gallium
has been used in conjunction with technetium-99 to increase the
specificity of the bone scanning.40,52
Several mechanisms have been postulated to explain the increased
activity at sites of inflammation. Enhanced blood flow and increased
capillary permeability cause enhanced delivery. Bacteria have high iron
requirements and thus take up gallium. Gallium is strongly bound to
bacterial siderophores and leukocyte lactoferrins. In regions of
inflammation, these proteins are available extracellularly and can bind
with gallium avidly. Chemotaxis also acts to localize gallium-labeled
WBCs at the sites of infection. In a typical study, gallium is injected
intravenously and delayed images are acquired at 48 to 72 hours. The
hallmark of osteomyelitis is the focal increased uptake of gallium.
Unfortunately, gallium’s nonspecific bone uptake can be problematic
because any processes causing reactive new bone formation will appear
hot. In patients with fractures or a prosthesis, osteomyelitis cannot
be easily diagnosed with gallium alone. Gallium images are usually
interpreted in conjunction with a technetium bone scan. Gallium
activity is interpreted as abnormal either if it is incongruous with
the bone scan activity or if there is a matching pattern with gallium
activity. Reported sensitivities and specificities for the diagnosis of
osteomyelitis range from 22% to 100% and 0% to 100%, respectively.2,52,76,103
Despite its lower-than-optimal diagnostic value, gallium still has some
advantages. It is easily administered and it is the agent of choice in
chronic soft tissue injection, although it is less effective in bone
infections. It has also proved useful in following the resolution of an
inflammatory process by showing a progressive decline in activity.
---
FIGURE 24-8
Red cell scan of patient in Figure 24-6 demonstrating increased activity in distal femur.
An indium-111 or 99mTc-hexamethylpropyleneamine osime
(99mTc-HMPAO) (Ceretec; GE Healthcare) -labeled leukocyte scan is the
most common scan used in conjunction with a standard bone scan. The
labeled leukocytes migrate to the region of active infection resulting
in a hot white cell scan over the area of active inflammation. The use
of a combined red cell and white cell scan significantly increases both
the sensitivity and specificity and now represents the g
You Might Also Like