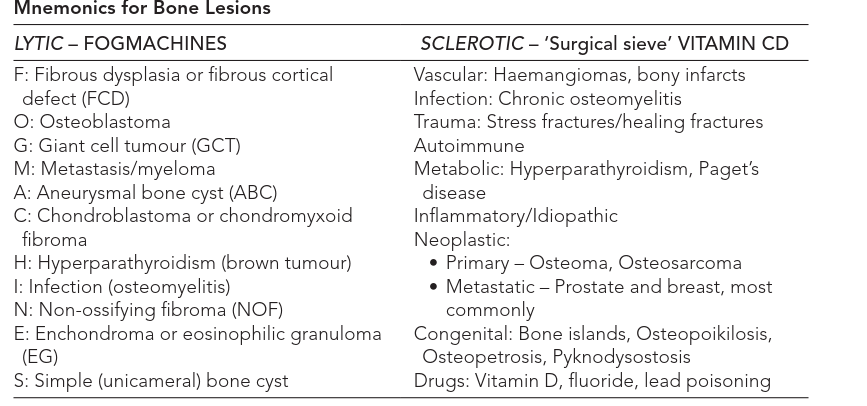
Pathological Fractures: Etiology, Diagnosis, Biomechanics & Mirels' Score
Key Takeaway
Pathological fractures occur through bone weakened by disease, not normal trauma. Common causes include metastatic bone disease (breast, prostate, lung cancer), primary bone tumors, and metabolic conditions. Diagnosis involves plain radiographs, CT, MRI, and systemic workup. The Mirels' Scoring System is used to assess fracture risk and guide prophylactic surgical stabilization.
Introduction & Epidemiology
Pathological fractures represent a critical challenge in orthopedic surgery, defined as a fracture occurring through bone that has been weakened by an underlying disease process, rather than solely by acute mechanical trauma that would typically be insufficient to fracture healthy bone. This distinction is crucial as it necessitates not only the management of the fracture itself but also the accurate diagnosis and treatment of the underlying osseous pathology. Unlike osteoporotic fragility fractures, which result from systemic skeletal demineralization, pathological fractures are due to focal or generalized structural compromise by a specific disease entity.
The epidemiology of pathological fractures is largely dictated by the prevalence of their causative conditions. In the adult population, metastatic bone disease is overwhelmingly the most common etiology, with primary tumors of the breast, prostate, lung, kidney, and thyroid accounting for a significant majority. Approximately 15-30% of cancer patients will develop skeletal metastases, and a substantial proportion of these will experience a pathological fracture during their disease course. Common sites include the proximal femur, humerus, spine, and pelvis, reflecting areas of high mechanical stress and rich vascularity conducive to tumor cell lodging. In younger patients, primary bone tumors (e.g., osteosarcoma, Ewing sarcoma, chondrosarcoma), benign bone lesions (e.g., aneurysmal bone cysts, fibrous dysplasia, unicameral bone cysts), and metabolic bone diseases (e.g., Paget's disease, osteomalacia, osteogenesis imperfecta) are more frequently implicated. Less common causes include osteomyelitis, stress shielding post-arthroplasty, and radiation-induced osteonecrosis.
The clinical presentation often involves localized pain, preceding the actual fracture event, particularly with impending fractures. This pre-fracture pain can be insidious and progressive, worsening with activity. An acute pathological fracture typically presents with sudden, severe pain, loss of function, and deformity, similar to a traumatic fracture, but often after minimal or no discernible trauma. The immediate goals of management are pain relief, restoration of function, local tumor control where applicable, and improvement in the patient's quality of life. Understanding the specific pathology and its systemic implications is paramount for optimal treatment planning and prognosis.
Radiographic evaluation is fundamental in diagnosing pathological fractures and identifying their underlying cause. Plain radiographs are the initial imaging modality, often revealing characteristic features such as lytic (bone-destroying), blastic (bone-forming), or mixed lesions. Cortical disruption, periosteal reaction, and the presence of a soft tissue mass are critical radiographic clues.

Advanced imaging modalities, including computed tomography (CT) and magnetic resonance imaging (MRI), provide superior detail regarding lesion morphology, cortical integrity, marrow involvement, and local soft tissue extension. A systemic workup, often including PET-CT or bone scintigraphy, is essential to determine the extent of disease, particularly in metastatic cases.
Surgical Anatomy & Biomechanics
Successful management of pathological fractures necessitates a thorough understanding of relevant surgical anatomy and the biomechanical implications of disease-compromised bone. The anatomical site of a pathological fracture significantly influences both its propensity to occur and the optimal surgical strategy for stabilization. Load-bearing bones, such as the femur and tibia, are inherently more susceptible to fracture when compromised by disease compared to less stressed bones.
Bone architecture, comprising dense cortical bone and porous cancellous bone, dictates its mechanical properties. Pathological lesions can disrupt either or both components. Lytic lesions, common in myeloma, renal cell carcinoma, and thyroid carcinoma, result in cortical thinning and endosteal scalloping, significantly reducing torsional and bending strength. Blastic lesions, characteristic of prostate and breast metastases, involve increased bone density but often with disorganized, weaker bone matrix, also leading to reduced mechanical integrity. Mixed lesions present a combination of these features.
The size and location of the lesion within the bone are critical biomechanical factors. Cortical involvement exceeding 50% of the bone's circumference or diameter, particularly in weight-bearing areas, is a strong predictor of impending fracture. Lesions in regions of high stress concentration, such as the subtrochanteric femur, intertrochanteric region, and metadiaphyseal junctions of long bones, are at inherently higher risk. The concept of a "stress riser," where a localized defect concentrates mechanical forces, is paramount in pathological fractures.
The
Mirels' Scoring System
is a widely adopted clinical tool that quantitatively assesses the risk of impending pathological fracture in long bones, guiding prophylactic intervention. This system assigns points (1-3) based on four radiographic and clinical criteria:
*
Site:
Upper limb (1), lower limb (2), peritrochanteric (3).
*
Pain:
Mild (1), moderate (2), functional (3).
*
Lesion Type:
Blastic (1), mixed (2), lytic (3).
*
Size:
<1/3 (1), 1/3-2/3 (2), >2/3 (3) of cortical diameter.
A total score of 7 or higher is generally accepted as an indication for prophylactic surgical stabilization, as the risk of fracture significantly increases. While Mirels' score offers valuable guidance, clinical judgment, patient comorbidities, life expectancy, and specific tumor biology also play crucial roles in decision-making.
Biomechanical principles of internal fixation in pathological bone differ significantly from those in healthy bone. Compromised bone quality often leads to poor screw purchase, increased risk of pullout, and implant subsidence. Therefore, fixation strategies often require:
*
Longer implants:
Spanning a greater length of the bone, often from joint to joint or extending well beyond the lesion.
*
Augmentation:
Polymethylmethacrylate (PMMA) bone cement is frequently used, particularly with intramedullary nails or around screw tips, to enhance implant stability and fill lytic defects. This creates a composite construct with improved load-bearing capacity and facilitates immediate weight-bearing.
*
Enhanced construct stiffness:
Dual plating, locked plating systems, or large-diameter intramedullary nails are often preferred.
*
Bypass principle:
The fixation device should extend at least two cortical diameters (or preferably one full bone diameter) beyond the lesion proximally and distally to ensure adequate purchase in healthy bone.
Understanding these biomechanical challenges is crucial for selecting the appropriate implant, surgical technique, and ultimately achieving durable fixation that can withstand physiological loads, facilitate rehabilitation, and reduce the likelihood of mechanical failure.
Indications & Contraindications
The decision-making process for managing pathological fractures, whether actual or impending, is complex and requires a multidisciplinary approach involving orthopedic surgeons, oncologists, radiation oncologists, and palliative care specialists. Indications for surgical intervention are generally aggressive, prioritizing pain relief, functional restoration, local tumor control, and prevention of further morbidity.
Indications for Operative Management:
- Actual Pathological Fracture: Any complete fracture through diseased bone warrants surgical stabilization to alleviate pain, restore alignment, allow mobility, and prevent further damage. The urgency depends on factors like fracture location (e.g., weight-bearing bones), neurological compromise (e.g., spinal fractures), and patient's general condition.
-
Impending Pathological Fracture:
Prophylactic fixation is indicated to prevent fracture in high-risk lesions, especially in weight-bearing bones, to avoid the morbidity, pain, and more complex surgery associated with an acute fracture.
- Mirels' Score ≥ 7: As discussed, this is a strong indicator for prophylactic stabilization.
- Significant Cortical Involvement: Lesions involving >50% of the cortical circumference, particularly in high-stress areas (e.g., subtrochanteric femur).
- Lesion size: Large lesions (>2.5 cm or >50% diameter) within the diaphysis of long bones.
- Persistent pain: Intractable pain despite radiation therapy or systemic treatment, especially with activity.
- Progressive lytic destruction: Documented increase in lesion size or lysis despite systemic therapy.
- Spinal Instability/Neurological Compromise: Pathological vertebral fractures leading to spinal instability or evidence of spinal cord or nerve root compression necessitate surgical decompression and stabilization.
- Diagnostic Biopsy: In cases where the underlying pathology is unknown, an open biopsy may be performed concurrently with stabilization, especially if percutaneous methods are inconclusive or if a large tissue sample is required for diagnosis and molecular profiling.
- Local Tumor Control: Resection and reconstruction may be indicated for certain primary bone tumors or isolated metastases, especially if they are radioresistant or require surgical margins. This often involves wide en bloc resection with reconstructive options such as endoprostheses, allografts, or custom implants.
- Pain Management: Even in the absence of an impending or actual fracture, severe, localized pain from an active lytic lesion resistant to other therapies may be an indication for stabilization, often combined with cement augmentation (e.g., vertebroplasty, kyphoplasty, or intramedullary nailing with cement).
Contraindications for Operative Management:
- Limited Life Expectancy (<6-8 weeks): For patients with rapidly progressing systemic disease and a very short anticipated survival, the morbidity and recovery period associated with major surgery may outweigh the benefits. Palliative care with non-operative measures (radiation, pain medication, limited weight-bearing) is often more appropriate.
- Poor General Medical Condition/High Anesthetic Risk: Patients with severe comorbidities that make them unfit for major surgery.
- Extensive, Unsalvageable Bone Destruction: If the bone destruction is so extensive that stable fixation is unlikely to be achieved or maintained, or if reconstruction would be futile.
- Rapidly Progressive Systemic Disease: If the systemic disease is progressing so aggressively that local bone intervention will not meaningfully impact the patient's overall prognosis or quality of life.
- Lesions Responsive to Non-Operative Treatment: Small, stable lesions, especially in non-weight-bearing bones, that respond well to radiation therapy or systemic anti-tumor agents may be managed non-operatively with careful monitoring.
- Uncontrolled Infection: Active systemic or local infection is a relative contraindication, as it significantly increases the risk of surgical site infection and hardware failure.
The decision to operate is always individualized, balancing the benefits of intervention against the risks and considering the patient's overall disease burden, functional goals, and personal wishes.
Summary of Operative vs. Non-Operative Indications
| Feature / Indication | Operative Management | Non-Operative Management |
|---|---|---|
| Pathological Fracture | Actual fracture (acute management) | Stable, non-displaced fracture (rarely) |
| Impending Fracture Risk | Mirels' score ≥ 7; >50% cortical destruction; >2.5 cm lesion | Mirels' score < 7; <50% cortical destruction; small lesions |
| Pain | Intractable pain despite systemic/radiation therapy | Mild to moderate pain controlled with analgesia/radiation |
| Neurological Status | Spinal cord compression, nerve root deficit | No neurological compromise |
| Lesion Type | Progressive lytic lesion | Stable blastic lesion, good response to systemic therapy |
| Functional Goal | Restoration of weight-bearing, mobility, QoL | Palliation, limited mobility |
| Life Expectancy | Anticipated survival >6-8 weeks | Anticipated survival <6-8 weeks |
| General Health | Medically fit for surgery | Unfit for major surgery; severe comorbidities |
| Biopsy Requirement | Concurrent open biopsy needed | Image-guided percutaneous biopsy sufficient (if needed) |
Pre-Operative Planning & Patient Positioning
Comprehensive pre-operative planning is critical for successful outcomes in pathological fracture management, given the complex nature of the underlying disease and the compromised bone stock. This phase ensures accurate diagnosis, appropriate staging, optimized patient condition, and precise surgical strategy.
Diagnostic Workup and Staging:
- History and Physical Examination: Detailed assessment of pain characteristics, functional limitations, neurological status, and systemic symptoms. Review of past medical history, particularly malignancy, and current systemic therapies.
-
Imaging:
- Plain Radiographs: Initial assessment, AP and lateral views, often revealing the characteristic lytic, blastic, or mixed appearance, cortical disruption, and periosteal reaction. These are essential for Mirels' scoring.
- Computed Tomography (CT): Provides superior detail regarding cortical integrity, endosteal scalloping, trabecular destruction, bone density, and local extent of the lesion, including soft tissue components. It is invaluable for surgical planning, especially for understanding the osseous defect and hardware placement.
- Magnetic Resonance Imaging (MRI): Crucial for evaluating marrow involvement, soft tissue extension, neurovascular bundle compromise, and differentiating between tumor, edema, and infection. It can detect skip lesions and is essential for spine pathology (cord compression).
- Bone Scintigraphy (Technetium-99m): A whole-body survey to identify other sites of skeletal metastasis. Highly sensitive but not specific.
- PET-CT (FDG-PET): Combines anatomical (CT) and metabolic (PET) information, offering high sensitivity and specificity for detecting metabolically active lesions, staging, and monitoring treatment response.
-
Biopsy:
- Image-Guided Core Needle Biopsy: Preferred method for diagnosis due to minimal invasiveness and lower risk of contamination. Performed by interventional radiology, ensuring the biopsy tract can be subsequently incorporated into a surgical incision if open surgery is needed.
- Open Biopsy: Reserved for cases where percutaneous biopsy is inconclusive or insufficient, or when a larger tissue sample is required for specialized analysis. The biopsy incision must be carefully planned to allow for future definitive surgery if necessary (e.g., limb salvage for a primary bone tumor).
- Timing: Biopsy generally precedes definitive stabilization unless the pathology is already known (e.g., known metastatic carcinoma with a typical radiographic appearance and a clear need for immediate stabilization).
- Systemic Staging: For metastatic disease, comprehensive staging often involves imaging of the chest, abdomen, and pelvis (CT or PET-CT) and brain (MRI) to identify other sites of disease. Tumor markers and blood work (e.g., CBC, metabolic panel, serum protein electrophoresis, PSA, SPEP, UPEP) are also integral.
Multidisciplinary Team Consultation:
Engaging a multidisciplinary team is essential.
*
Oncology/Hematology:
For systemic treatment strategies (chemotherapy, targeted therapy, immunotherapy, hormonal therapy).
*
Radiation Oncology:
For adjuvant or palliative radiation therapy, either pre- or post-operatively, to control local disease and reduce pain.
*
Palliative Care:
To optimize pain management, address quality of life issues, and assist in end-of-life planning.
*
Internal Medicine/Anesthesiology:
For pre-operative optimization of medical comorbidities, nutritional status, and comprehensive anesthetic risk assessment.
Pre-operative Optimization:
- Medical Comorbidities: Aggressive management of cardiac, pulmonary, renal, and endocrine conditions.
- Nutritional Status: Optimizing nutrition can improve wound healing and recovery.
- Pain Control: Adequate pre-operative analgesia.
- Blood Management: Type and cross-match blood products, especially for potentially hemorrhagic tumor resections. Consider anti-fibrinolytic agents like tranexamic acid.
- Anticipation of Adjuvant Therapies: Discuss the timing and sequencing of surgery relative to radiation and systemic therapies.
Implant Selection and Surgical Strategy:
- Implant Choice: Based on fracture location, bone quality, tumor type, patient's life expectancy, and expected functional demands. Options include intramedullary nails (often augmented with cement), locked plating systems, endoprosthetic replacement (e.g., hemiarthroplasty, total arthroplasty, tumor prostheses), or custom implants.
- Cement Augmentation: Plan for the use of PMMA cement, especially with IMN or screw fixation, to enhance stability and fill bone defects. Consider using cement impregnated with antibiotics if infection risk is high.
- Resection vs. Stabilization: Decide whether local tumor resection (e.g., curettage for benign, wide resection for primary malignant/solitary metastasis) or pure stabilization is the goal.
- Bone Grafting/Substitutes: Rarely indicated for pathological fractures due to tumor burden; PMMA is preferred for structural support.
- Radiation Beads: Consideration for local delivery of radiation (e.g., with PMMA) for radioresistant tumors.
Patient Positioning:
Positioning depends on the fracture site and chosen surgical approach.
*
General Principles:
* Adequate padding to prevent pressure injuries.
* Secure positioning to allow manipulation and fixation.
* Access for fluoroscopy (C-arm) is paramount.
* Preparation for potential extensive exposure and blood loss.
*
Specific Examples:
*
Proximal Femur:
Supine position on a fracture table or radiolucent table for intramedullary nailing; lateral decubitus for plating or arthroplasty approaches.
*
Diaphyseal Femur:
Supine on a fracture table for IMN.
*
Humerus:
Beach chair or supine position for proximal humerus; supine or lateral for diaphyseal.
*
Spine:
Prone position, often on a specialized spinal frame, for posterior stabilization.
Final plan should include the exact implant, approach, anticipated complications, and contingency plans.
Detailed Surgical Approach / Technique
Surgical management of pathological fractures aims to achieve immediate stability, alleviate pain, restore function, and, in selected cases, achieve local tumor control. The choice of surgical technique is highly individualized, dictated by the anatomical site, extent of bone destruction, tumor biology, patient's overall health, and anticipated life expectancy. While specific approaches vary, several overarching principles apply.
General Principles of Surgical Technique:
- Biopsy First (if unknown pathology): Unless the diagnosis is definitively established (e.g., known metastatic breast cancer with typical lytic lesion), a biopsy should precede definitive fixation. The biopsy tract must be planned carefully so it can be excised with the tumor or incorporated into the definitive surgical incision to minimize tumor seeding.
- Adequate Exposure: The surgical approach must provide sufficient exposure for tumor assessment, debridement, reduction, and secure fixation. Internervous planes are respected to minimize iatrogenic nerve injury.
-
Tumor Debridement/Resection:
- Benign Lesions (e.g., UBC, ABC): Aggressive curettage and burring of the cavity walls, followed by adjuvant treatment (e.g., phenol, cryotherapy) and bone cement filling or structural grafting if necessary.
- Metastatic Disease: Intralesional curettage of tumor mass to debulk and create space for cement augmentation, facilitating immediate weight-bearing and local control. Radiation beads can be added to the cement. Wide en bloc resection is generally reserved for isolated, radioresistant metastases in patients with good prognosis.
- Primary Malignant Tumors: Often requires wide en bloc resection to achieve oncologic margins, followed by complex reconstruction with endoprostheses, allografts, or custom implants. This is highly specialized limb-salvage surgery.
-
Stable Fixation:
This is paramount. Due to compromised bone quality, fixation must be inherently robust.
- Intramedullary Nailing (IMN): The preferred method for diaphyseal and many metaphyseal pathological fractures of long bones (femur, humerus, tibia) due to its load-sharing nature, minimally invasive approach, and ability to span the entire length of the bone. Reaming is often performed generously to allow for a thick cement mantle around the nail, enhancing stability and filling the tumor defect.
- Plating: Used for juxta-articular lesions, flat bones (pelvis), or when IMN is not feasible. Locked plating systems are favored for better screw purchase in osteopenic/compromised bone. Plates should be long, extending well beyond the lesion, and secured with multiple screws.
- Arthroplasty (Resection/Replacement): Indicated for extensive articular destruction, particularly in the proximal femur, humerus, or around the knee, especially in weight-bearing joints. Hemiarthroplasty or total arthroplasty (standard or tumor-specific endoprosthesis) allows for immediate function and pain relief by replacing the destroyed joint and adjacent bone.
- PMMA Cement Augmentation: Crucial for enhancing stability in nearly all types of fixation. For IMN, cement can be injected around the nail after insertion. For plating, cement can be injected into lytic defects prior to plate application or around screw tips. The heat generated by PMMA polymerization also provides a local ablative effect on tumor cells.
- Adjuvant Therapy Integration: Consideration of intraoperative radiation therapy or post-operative external beam radiation therapy.
Site-Specific Considerations (Illustrative Examples):
1. Femur
-
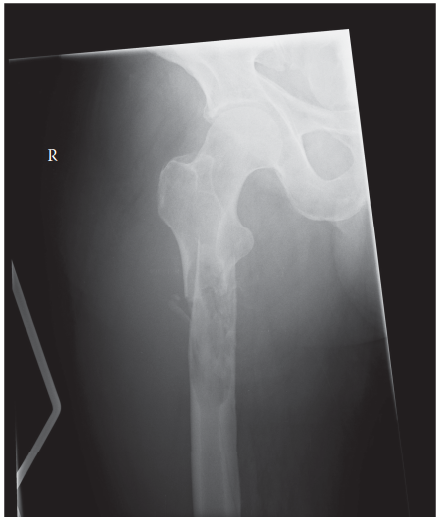
Proximal Femur (Femoral Neck, Intertrochanteric, Subtrochanteric):
- Approach: Lateral approach for IMN (trochanteric entry), standard hip approaches for arthroplasty.
-
Technique:
- Intramedullary Nailing: Cephalomedullary nails are often preferred. After initial reaming, an optional curettage of the tumor can be performed. The nail is inserted, followed by judicious injection of PMMA cement into the lytic defects around the nail and screws. The nail should span the entire length of the femur to provide comprehensive bypass.
- Arthroplasty: For extensive destruction of the femoral head/neck or poor bone quality not conducive to IMN, a hemiarthroplasty or total hip arthroplasty (THA) is performed, often with a long-stemmed cemented femoral component to bypass the lesion distally.
-
Reduction:
Due to compromised bone, reduction is often less rigid than in traumatic fractures; stable length and alignment are prioritized.

- Image Caption Context: This image could represent a successfully stabilized femoral shaft pathological fracture with an intramedullary nail and cement augmentation, allowing for immediate post-operative weight bearing.
-
Diaphyseal Femur:
- Approach: Lateral approach, typically through a small incision at the greater trochanter for nail insertion.
- Technique: Long intramedullary nailing spanning the entire femur, from the greater trochanter to the distal metaphysis. Over-reaming and cement augmentation are frequently employed.
-
Distal Femur:
- Approach: Lateral or medial parapatellar.
- Technique: Distal femoral locking plate, often with cement augmentation. If the knee joint is involved or extensively destroyed, distal femoral replacement (arthroplasty) may be required.
2. Humerus
-
Proximal Humerus:
- Approach: Deltopectoral approach.
- Technique: Short intramedullary nail (retrograde or antegrade) or locked plating system. For extensive destruction or impending fracture in patients with reasonable prognosis, a proximal humeral endoprosthesis or reverse shoulder arthroplasty may be considered.
-
Diaphyseal Humerus:
- Approach: Anterolateral or posterior, depending on lesion location.
- Technique: Long intramedullary nail (often antegrade from the greater tuberosity) spanning the entire humerus. Locked plating is an alternative, particularly if radial nerve integrity is a concern.
3. Spine
- Approach: Posterior for most stabilization and decompression. Anterior for direct tumor resection and reconstruction.
- Technique: Vertebroplasty or kyphoplasty for stable compression fractures. Posterior instrumentation (pedicle screws and rods) for unstable vertebral body fractures with or without laminectomy for decompression. Anterior corpectomy and reconstruction for severe anterior column destruction and/or spinal cord compression. This is a highly specialized area, often involving neurosurgeons.
Internervous Planes:
While the primary focus in pathological fractures is often lesion debridement and robust fixation, adherence to internervous planes remains crucial to minimize iatrogenic injury and preserve function. For example:
*
Deltopectoral approach (proximal humerus):
Utilizes the plane between the deltoid (axillary nerve) and pectoralis major (medial and lateral pectoral nerves).
*
Anterolateral approach (femur):
Often between the rectus femoris (femoral nerve) and vastus lateralis (femoral nerve) or more laterally between the vastus lateralis and tensor fascia lata (superior gluteal nerve).
*
Posterior approach (humerus):
Between the triceps medial (radial nerve) and lateral heads (radial nerve). Careful identification and protection of the radial nerve in the spiral groove are critical for diaphyseal humeral plating.
Thorough understanding of these planes and meticulous dissection are integral to the technical execution, even in complex oncologic resections.
Complications & Management
Despite meticulous surgical planning and execution, pathological fractures are associated with a higher rate of complications compared to traumatic fractures in healthy bone, primarily due to the compromised bone stock, systemic nature of the disease, and often aggressive adjuvant therapies. Anticipation and proactive management of these complications are crucial for optimizing patient outcomes.
Common Complications:
-
Mechanical Complications:
-
Hardware Failure:
(Incidence: 10-20%) This is the most common mechanical complication, including screw pullout, plate/nail breakage, or disengagement of components. It often results from progressive tumor lysis around the implant, inadequate initial fixation in compromised bone, or failure to bypass the entire diseased segment.
- Salvage Strategy: Revision surgery with a stronger construct (e.g., longer implant, larger diameter nail, additional plating), often incorporating extensive PMMA cement augmentation. Consideration of endoprosthetic replacement for unsalvageable bone loss.
-
Nonunion/Malunion:
(Incidence: 5-15%) Impaired bone healing due to poor biological environment (tumor, radiation, chemotherapy) and compromised blood supply. Malunion can result from inadequate initial reduction or loss of fixation.
- Salvage Strategy: For nonunion, revision surgery with enhanced fixation, cement augmentation, and consideration of adjunctive therapies (e.g., bone morphogenetic proteins are rarely used due to tumor risk, but local radiation may be re-evaluated). For symptomatic malunion, corrective osteotomy and revision fixation.
-
Aseptic Loosening/Dislocation (Arthroplasty):
(Incidence: 5-10%) For patients undergoing arthroplasty for pathological fractures, loosening of components or joint dislocation can occur due to poor bone quality, technical error, or soft tissue compromise.
- Salvage Strategy: Revision arthroplasty, often with long-stemmed cemented components or tumor prostheses. For dislocation, closed reduction may be attempted, but recurrent instability may require revision with constrained components.
-
Hardware Failure:
(Incidence: 10-20%) This is the most common mechanical complication, including screw pullout, plate/nail breakage, or disengagement of components. It often results from progressive tumor lysis around the implant, inadequate initial fixation in compromised bone, or failure to bypass the entire diseased segment.
-
Biological Complications:
-
Local Tumor Progression/Recurrence:
(Incidence: Variable, highly dependent on tumor type and margin) If tumor cells are left behind (e.g., after intralesional curettage or inadequate margins), the tumor can grow, leading to pain, further bone destruction, and potential hardware failure.
- Salvage Strategy: Aggressive re-resection if feasible, focused radiation therapy, systemic anti-tumor therapy, or chemotherapy.
-
Surgical Site Infection (SSI):
(Incidence: 2-5%) Higher risk in immunosuppressed cancer patients, those receiving chemotherapy, and with prolonged complex procedures involving cement.
- Salvage Strategy: Superficial infections may respond to antibiotics. Deep infections often require surgical debridement, pulsed lavage, appropriate antibiotic therapy, and potentially implant retention (if stable) or removal (if unstable or chronic). Two-stage revision is often necessary for implant-associated infections.
-
Hematoma Formation:
Can cause pain, swelling, and increase infection risk.
- Salvage Strategy: Observation, or surgical evacuation if large and symptomatic.
-
Tumor Seeding along Biopsy Tract:
Rare but serious complication if biopsy is performed carelessly or without excising the tract during definitive surgery.
- Salvage Strategy: Excision of the tract, radiation therapy.
-
Local Tumor Progression/Recurrence:
(Incidence: Variable, highly dependent on tumor type and margin) If tumor cells are left behind (e.g., after intralesional curettage or inadequate margins), the tumor can grow, leading to pain, further bone destruction, and potential hardware failure.
-
Systemic Complications:
-
Thromboembolic Disease (DVT/PE):
(Incidence: 5-10%) Increased risk in cancer patients due to hypercoagulable state and immobility.
- Salvage Strategy: Aggressive prophylaxis (mechanical and chemical), early mobilization. Treatment with anticoagulation for established DVT/PE.
-
Fat Embolism Syndrome:
Particularly with intramedullary reaming.
- Salvage Strategy: Supportive care, prophylaxis with careful reaming technique.
-
Cardiopulmonary Complications:
Due to general anesthesia and underlying patient comorbidities.
- Salvage Strategy: Pre-operative optimization, meticulous anesthetic management, post-operative monitoring.
-
Progressive Systemic Disease:
Ultimately, the patient's prognosis is tied to the underlying malignancy. Local surgical interventions do not address systemic progression.
- Salvage Strategy: Continued systemic oncologic management, palliative care.
-
Thromboembolic Disease (DVT/PE):
(Incidence: 5-10%) Increased risk in cancer patients due to hypercoagulable state and immobility.
Management Principles for Complications:
- Early Recognition: Vigilant post-operative monitoring for signs of complications.
- Multidisciplinary Approach: Collaboration with oncology, radiation oncology, infectious disease specialists, and palliative care.
- Individualized Treatment: Tailoring management strategies to the specific complication, patient's overall condition, and prognosis.
- Prophylaxis: DVT prophylaxis, pre-operative antibiotics, careful surgical technique, and meticulous soft tissue handling are crucial preventive measures.
Table of Common Complications and Management Strategies
| Complication | Incidence (Approx.) | Salvage Strategy |
|---|---|---|
| Hardware Failure | 10-20% | Revision surgery with stronger/longer construct, PMMA cement augmentation, endoprosthetic replacement for extensive bone loss. |
| Nonunion / Malunion | 5-15% | Revision fixation, cement augmentation, possibly biologics (limited use), corrective osteotomy for malunion. Address underlying tumor progression. |
| Aseptic Loosening / Dislocation (Arthroplasty) | 5-10% | Revision arthroplasty (long-stem, cemented, constrained components), closed reduction for dislocation; open reduction/revision for recurrent dislocation. |
| Local Tumor Progression / Recurrence | Variable | Re-resection if oncologically feasible, focused radiation therapy, systemic anti-tumor therapies, palliative measures. |
| Surgical Site Infection | 2-5% | Debridement, irrigation, cultures, appropriate antibiotic therapy (often prolonged); implant retention vs. removal (depending on stability, chronicity). May require two-stage revision. |
| Thromboembolic Disease (DVT/PE) | 5-10% | Therapeutic anticoagulation, filter placement for PE contraindication. Aggressive prophylaxis (mechanical/chemical) and early mobilization are key for prevention. |
| Hematoma | 2-10% | Observation, aspiration, or surgical evacuation if symptomatic or at high risk for infection. |
| Nerve Palsy | <1-2% | Observation (if neuropraxia), nerve exploration and repair/grafting (if transection), palliative procedures (e.g., tendon transfer) for permanent deficit. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation following pathological fracture surgery is critical for achieving the primary goals of pain relief, functional restoration, and improving the patient's quality of life. The protocols are tailored to the individual patient, considering the stability of fixation, the extent of tumor resection, the underlying pathology, the patient's overall functional status, and life expectancy. The key advantage of stable surgical fixation is often the ability to initiate early mobilization and weight-bearing.
Immediate Post-Operative Phase (Days 0-7):
- Pain Management: Aggressive, multimodal analgesia is paramount. This includes patient-controlled analgesia (PCA), regional nerve blocks, oral opioids, NSAIDs (if not contraindicated), and adjuvant analgesics to facilitate early participation in therapy.
- DVT Prophylaxis: Given the hypercoagulable state associated with malignancy and surgical immobility, chemical (low molecular weight heparin or direct oral anticoagulants) and mechanical (intermittent pneumatic compression devices, anti-embolism stockings) prophylaxis are initiated promptly.
- Wound Care: Meticulous wound care to prevent infection. Regular dressing changes, monitoring for signs of hematoma or dehiscence.
-
Early Mobilization:
As soon as pain is controlled and hemodynamic stability achieved, emphasis is placed on out-of-bed activity.
- Weight-Bearing Status: This is determined by the stability of the surgical construct. For robustly fixed fractures, especially with cement augmentation or arthroplasty, immediate full weight-bearing (FWB) or weight-bearing as tolerated (WBAT) is often permissible. For less stable constructs, or if bone quality is severely compromised, protected weight-bearing (PWB) or touch-down weight-bearing (TDWB) may be prescribed, transitioning to FWB as tolerated.
- Range of Motion (ROM): Initiate gentle, active-assisted ROM exercises for adjacent joints. For example, knee flexion and extension for femoral fractures, shoulder pendulum exercises for humeral fractures.
Intermediate Rehabilitation Phase (Weeks 1-6):
-
Physical Therapy (PT):
- Ambulation Training: Progressive ambulation with appropriate assistive devices (walker, crutches) according to weight-bearing status. Focus on gait mechanics, balance, and endurance.
- Strengthening: Isotonic and isometric exercises for major muscle groups, gradually increasing resistance. Emphasis on core stability and strengthening muscles around the affected joint.
- Flexibility: Continued ROM exercises to prevent stiffness and contractures.
- Functional Mobility: Training for activities of daily living (ADLs) such as transfers, stair climbing, and adapting to assistive devices.
- Occupational Therapy (OT): Assessment and training for upper extremity function, fine motor skills, and adaptation of daily tasks to optimize independence. Provision of adaptive equipment if needed.
- Pain Management: Transition from hospital-based analgesia to oral pain regimens. Close monitoring for neuropathic pain or breakthrough pain.
- Monitoring for Complications: Continued vigilance for signs of infection, hardware failure, or disease progression.
Advanced Rehabilitation Phase (Beyond 6 Weeks):
- Progressive Loading: If bone healing is progressing and fixation remains stable, gradually increase weight-bearing and resistance in strengthening exercises.
- Endurance and Conditioning: Incorporate cardiovascular exercises to improve overall stamina.
- Return to Activity: Patients are encouraged to return to meaningful activities, within the limits of their underlying disease and functional capacity. High-impact activities are generally discouraged due to the risk of re-fracture or hardware failure.
- Collaboration with Oncology/Radiation Oncology: Rehabilitation plans must be coordinated with ongoing systemic cancer treatments (chemotherapy, radiation therapy), as these can impact strength, endurance, and healing.
- Psychosocial Support: Addressing the psychological impact of cancer and surgery is crucial. Support groups, counseling, and psychological interventions can be beneficial.
Considerations for Specific Patient Populations:
- Limited Life Expectancy: For patients with very short prognoses, rehabilitation focuses on maximum functional independence for ADLs and comfort, rather than extensive long-term recovery.
- Primary Bone Tumors: Rehabilitation may be more aggressive, aiming for long-term functional recovery, particularly after limb salvage procedures. Close monitoring for tumor recurrence is essential.
- Spinal Fractures: Rehabilitation often includes brace management, spinal precautions, and specific core-strengthening exercises after appropriate healing.
The rehabilitation process for pathological fractures is often an ongoing journey, adapting to changes in the patient's condition and disease progression. Regular communication among the surgical team, rehabilitation specialists, and oncology team ensures a holistic and patient-centered approach.
Summary of Key Literature / Guidelines
The evidence base guiding the management of pathological fractures has evolved significantly, emphasizing a multidisciplinary, individualized approach. Several landmark papers and consensus guidelines form the bedrock of current practice.
-
Mirels' Criteria (1989): Perhaps the most seminal work in the field, Mirels' paper "Painless metastasizing lesion in a weight-bearing bone. Surgical decision-making based on a probability analysis" (Clin Orthop Relat Res. 1989 Dec;(249):256-64) introduced the scoring system for predicting impending pathological fractures. This quantitative tool, based on site, pain, lesion type, and size, provides an objective framework for decision-making regarding prophylactic stabilization, with a score of ≥7 generally indicating a high risk of fracture. While widely adopted, it's acknowledged that clinical judgment and consideration of tumor biology and patient prognosis are also vital. Subsequent studies have largely validated its utility, though some have proposed modifications or highlighted limitations for specific tumor types or sites.
-
National Comprehensive Cancer Network (NCCN) Guidelines: The NCCN publishes comprehensive, evidence-based guidelines for various cancer types, including specific sections on bone metastases. These guidelines provide detailed recommendations for diagnosis, staging, systemic therapy, local therapy (surgery, radiation), and supportive care for patients with skeletal involvement. They underscore the importance of early identification of impending fractures, multidisciplinary team discussions, and the integration of orthopedic surgery with systemic and radiation oncology treatments.
-
American Society of Clinical Oncology (ASCO) Guidelines: ASCO also produces clinical practice guidelines focusing on the management of bone metastases, including interventions for pain, prevention of skeletal-related events (SREs), and treatment of pathological fractures. These guidelines offer recommendations on systemic agents (e.g., bone-modifying agents like bisphosphonates or denosumab), radiation therapy, and surgical indications, often aligning with NCCN recommendations.
-
Orthopedic Oncology Society (OOS) / Musculoskeletal Tumor Society (MSTS) Recommendations: These societies, composed of orthopedic surgeons specializing in musculoskeletal oncology, provide expert consensus and publish research on the surgical management of bone tumors and metastases. Their work often details specific surgical techniques, implant selection, and outcomes for various anatomical sites and tumor types, emphasizing principles of stable fixation in compromised bone.
-
Literature on Fixation Techniques and Augmentation:
- Intramedullary Nailing vs. Plating: Numerous studies compare the efficacy and complications of IMN versus plating for long bone pathological fractures. IMN is generally favored for diaphyseal and many metaphyseal lesions due to its load-sharing characteristics, less soft tissue disruption, and ability to span the entire bone.
- Polymethylmethacrylate (PMMA) Cement Augmentation: Research consistently demonstrates the biomechanical benefits of PMMA cement augmentation in enhancing implant stability, filling lytic defects, and providing immediate load-bearing capabilities. Papers often highlight techniques for cement delivery (e.g., through IMN or directly into defects) and the local thermal cytotoxic effects on tumor cells.
- Endoprosthetic Reconstruction: For extensive bone destruction, particularly around joints, the literature supports endoprosthetic replacement (e.g., modular tumor prostheses, custom implants) as a durable option providing immediate stability and function, though with higher rates of surgical complexity and potential for mechanical failure or infection.
-
Palliative Care Integration: Growing literature emphasizes the critical role of palliative care specialists in managing pain, symptoms, and quality of life for patients with advanced cancer and pathological fractures. Discussions revolve around balancing aggressive surgical intervention with patient comfort and goals of care, especially in those with limited prognosis.
-
Advances in Imaging and Biopsy: The evolution of advanced imaging (CT, MRI, PET-CT) and image-guided biopsy techniques (core needle biopsy) has significantly improved diagnostic accuracy and reduced morbidity associated with tissue acquisition, allowing for more precise treatment planning.
In conclusion, the management of pathological fractures is a dynamic field that continues to integrate advances in surgical techniques, implant technology, systemic oncologic therapies, and a refined understanding of tumor biology. Adherence to established guidelines, a robust multidisciplinary approach, and continuous engagement with current literature are essential for providing optimal care to this complex patient population.
Clinical & Radiographic Imaging