Background
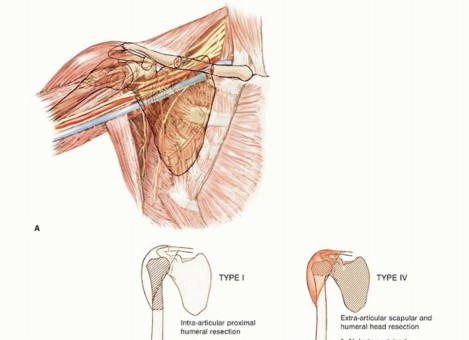
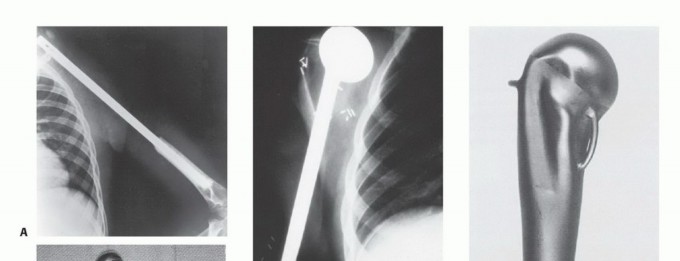
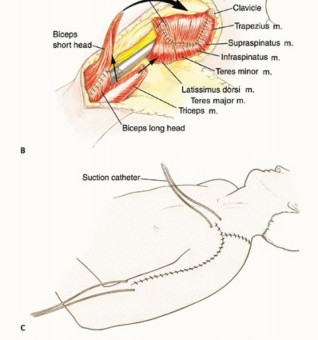
The proximal humerus is a common site for both primary osteosarcomas and chondrosarcomas and is the second most common site of metastatic disease involving long bones. Metastatic tumors occasionally involve the shoulder girdle and often are treated using the same resection and reconstruction techniques (FIG 1A).
Limb-sparing resection of the proximal humerus is challenging. Despite their complexity, these resections can be performed in about 95% of patients with high- or low-grade sarcomas. Amputations rarely are required. Endoprosthetic reconstruction is the most common technique for reconstructing large proximal humeral defects. It is used following both intra-articular (type I) and extra-articular (type V) resections.
This type of reconstruction is combined with local muscle transfers to create shoulder stability; cover the prosthesis; and provide a functional elbow, wrist, and hand (FIG 1B). The surgical and anatomic considerations of limb-sparing procedures of the proximal humerus and the specific surgical techniques for type I and type V resection and reconstruction are described in this chapter. Total humeral replacement is described briefly.

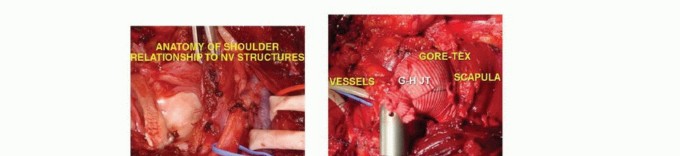
FIG 1A: Anatomy of the shoulder girdle.
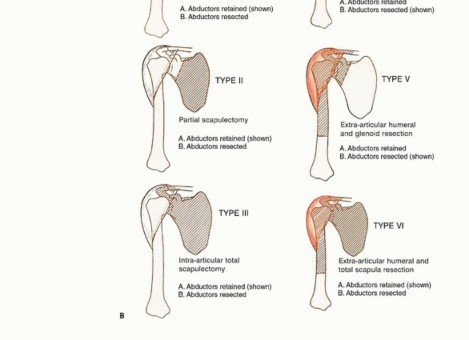
FIG 1B: Surgical classification of shoulder girdle resections. Types I-III are intra-articular, Types IV-VI are extra-articular.
The proximal humerus is one of the most common sites for high-grade malignant bone tumors in the adult, and it is the third most common site for osteosarcoma. Tumors in this location tend to have a significant extraosseous component. The proximal humerus also may be involved by metastatic cancer (especially renal cell carcinoma) and secondarily by soft tissue sarcomas, which require a resection similar to that used for primary bone sarcomas with extraosseous extension.
About 95% of patients with tumors of the shoulder girdle can be treated with limb-sparing resections. The Tikhoff-Linberg resection and its modifications are limb-sparing surgical options for bone and soft tissue tumors in and around the proximal humerus and shoulder girdle. Portions of the scapula, clavicle, and proximal humerus are resected in conjunction with all muscles inserting onto and originating from the involved bones.
Careful preoperative staging and selection of patients whose tumor does not encase the neurovascular bundle or invade the chest wall are required. A classification system for resection of tumors in this location is described in FIG 1B. The most common procedure for high-grade sarcomas of the proximal humerus, type VB, is described.
We do not recommend type I resection for high-grade tumors due to the increased risk of local recurrence. Optimal function is achieved with muscle transfers and skeletal reconstruction. A prosthesis is used to maintain length and stabilize the shoulder and distal humerus following resection.
A stable shoulder with normal function of the elbow, wrist, and hand should be achieved following most shoulder girdle resections and reconstructions performed using the techniques described.
Indications
Indications for limb-sparing procedures of the proximal humerus and shoulder girdle include:
- High-grade and some low-grade bone sarcomas.
- Some soft tissue sarcomas that secondarily invade bone.
- Solitary metastatic carcinomas to the proximal humerus.
- Multiple metastatic carcinoma to the proximal humerus with no option of stabilization and fixation (best treated by a wide excision / type I resection).
The decision to proceed with limb-sparing surgery is based on the location of the tumor and a thorough understanding of its natural history. Recently, we have treated patients with pathologic fractures with induction chemotherapy, immobilization, and limb-sparing surgery if there is a good clinical response and fracture healing.
Contraindications
**Absolute contraindications:** Include tumor involvement of the neurovascular bundle or extensive invasion of the adjacent chest wall (FIG 2), and extensive invasion of the muscles around the shoulder girdle.
**Relative contraindications:** Include chest wall extension, tumor contamination of the operative site from hematoma following a poorly performed biopsy or pathologic fracture, a previous infection, or lymph node involvement.
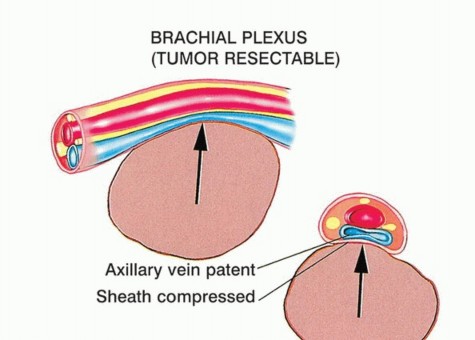
FIG 2A: Resectable tumor compressing but not invading the neurovascular bundle.
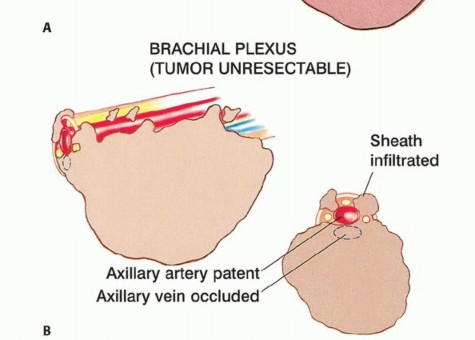
FIG 2B: Unresectable tumor infiltrating neurovascular structures and obliterating the axillary vein.
Unique Anatomic Considerations
Resection and reconstruction of the proximal humerus and shoulder girdle is a technically demanding procedure. The local anatomy of the tumor often determines the extent of the operation required. The surgeon should be experienced with all aspects of shoulder girdle anatomy and the unique considerations it may present.
Proximal Humerus
Malignant tumors often present with large soft tissue components (stage IIB) underneath the deltoid that extend medially and displace the subscapularis and coracobrachialis muscles. Pericapsular and rotator cuff involvement occur early and must be evaluated.
Glenohumeral Joint
The shoulder joint appears to be more prone to intra-articular or pericapsular involvement by high-grade bone sarcomas than are other joints. Four basic mechanisms exist for tumor spread: direct capsular extension, tumor extension along the long head of the biceps tendon, fracture hematoma from a pathologic fracture, and poorly planned biopsy. These mechanisms place patients undergoing intra-articular resections for high-grade sarcomas at greater risk for local recurrence than those undergoing extra-articular resections. Therefore, it often is necessary to perform an extra-articular resection for high-grade bone sarcomas of the proximal humerus or scapula.
Neurovascular Bundle
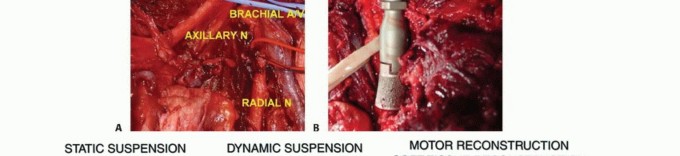
The subclavian artery and vein join the cords of the brachial plexus as they pass underneath the clavicle. Beyond this point, the nerves and vessels can be considered as one structure (ie, the neurovascular bundle). Large tumors involving the upper scapula, clavicle, and proximal humerus may displace the infraclavicular components of the plexus and axillary vessels.
Musculocutaneous and Axillary Nerves
The musculocutaneous and axillary nerves often are in close proximity or contact with tumors around the proximal humerus. The musculocutaneous nerve is the first nerve that leaves between the teres major and minor to innervate the deltoid muscle posteriorly. Tumors of the proximal humerus are likely to involve the axillary nerve as it passes adjacent to the inferior aspect of the humeral neck, just distal to the joint. Therefore, the axillary nerve and deltoid almost always are sacrificed during proximal humerus resections.
Radial Nerve
The radial nerve comes off the posterior cord of the plexus and continues anterior to the latissimus dorsi and teres major. Just distal to the teres major, the nerve courses into the posterior aspect of the arm to run between the medial and long head of the triceps. Although most sarcomas of the proximal humerus do not involve the radial nerve, it must be isolated and protected prior to resection.
Axillary and Brachial Arteries
The axillary artery is a continuation of the subclavian artery and is called the brachial artery after it passes the inferior border of the axilla. The axillary vessels are surrounded by the three cords of the brachial plexus. The axillary artery typically leaves the lateral cord just distal to the coracoid process, passes through the coracobrachialis, and runs between the brachialis and biceps.
Preservation of the musculocutaneous nerve and short head of the biceps muscle is important to ensure normal elbow function. The path of this nerve may vary extensively (within 2 to 8 cm of the coracoid) and should be identified before any resection is performed because the nerve can easily be injured.
The axillary nerve arises from the posterior cord and courses, along with the circumflex vessels, inferior to the distal border of the subscapularis. It then is tethered to the proximal humerus by the anterior and posterior circumflex vessels. Early ligation of the circumflex vessels is a key maneuver in resection of proximal humeral sarcomas because it allows the entire axillary artery and vein to fall away from the tumor mass.
Occasionally, anatomic variability in the location of the branches of the nerve may lead to difficulty in identification and exploration if the variation has not previously been recognized. A preoperative angiogram is helpful in determining vascular displacement and anatomic variability. Final determination of tumor resectability is made at surgery. Early exploration of the neurovascular structures is performed following division of the pectoralis major muscle. This approach does not jeopardize subsequent formation of an anterior flap in patients who require forequarter amputation.
Imaging and Other Staging Studies
Appropriate imaging studies are key to successful resections of tumors of the proximal humerus and shoulder girdle. The most useful imaging studies are plain radiography, computed tomography (CT) scans, magnetic resonance imaging (MRI), arteriography, and bone scan. Venography is only occasionally required.
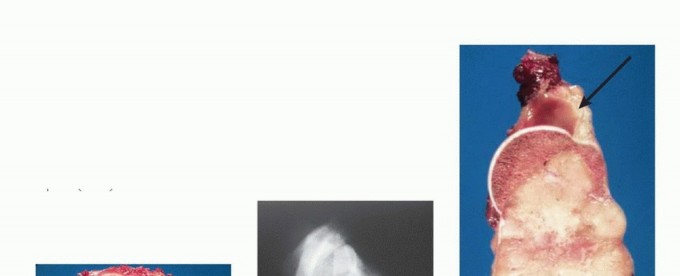
- Computed Tomography: CT is most useful for evaluating cortical bone changes and is considered complementary to MRI in evaluating the chest wall, clavicle, and axilla for tumor extension.
- Magnetic Resonance Imaging: MRI is useful to identify intraosseous tumor extent, which is necessary for determining the length of bone resection. It is the best imaging modality for evaluation of soft tissue tumor involvement, especially around the glenohumeral joint, suprascapular region, and chest wall.
- Bone Scintigraphy: Bone scintigraphy is used to determine the intraosseous tumor extent and to detect metastases.
- Angiography: Angiography is useful for evaluation of tumor vascularity and tumor response to neoadjuvant chemotherapy. It also is essential for determining the relation of the brachial vessels to the tumor or the presence of anatomic anomalies. A brachial venogram also may be necessary if there is evidence of distal venous obstruction suggesting a tumor thrombus. It is also relevant in cases where the decision to amputate is conflictive. Repeat staging studies are typically performed following surgical resection to determine patient response to chemotherapy.
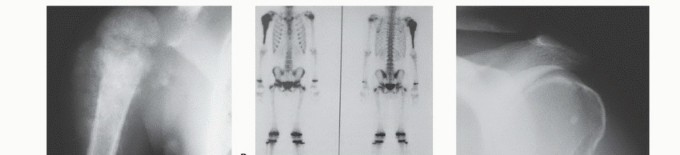
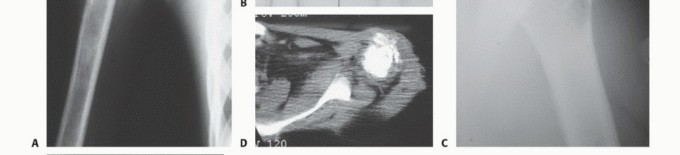
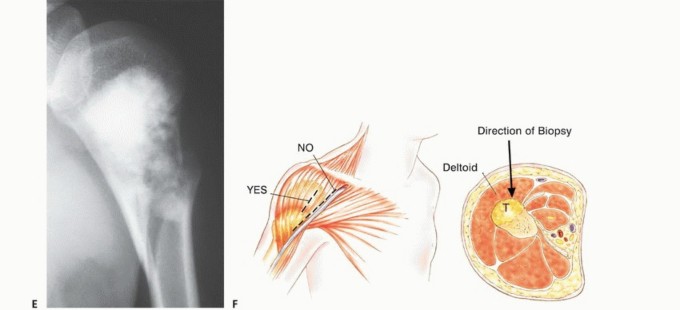
FIG 3 A-E: Imaging studies showing osteosarcoma presentation, CT/MRI evaluation, and clinical responses.
Biopsy
Needle or incisional biopsies of tumors of the proximal humerus should be performed through the anterior one-third of the deltoid muscle, not through the deltopectoral interval. A biopsy through the anterior one-third of the deltoid results in a limited hematoma that is confined by the deltoid muscle. This portion of the muscle and any biopsy hematoma are easily removed at the definitive resection.
A biopsy taken through the deltopectoral interval will contaminate the major pectoralis muscle, which is necessary for reconstruction; increase the risk of hematoma spread along the axillary vessels to the chest wall; and make a local resection difficult, if not impossible. If an open biopsy is required, a short longitudinal incision should be made just lateral to the deltopectoral interval. The dissection should be directly into the deltoid muscle and proximal humerus. The bone should be exposed lateral to the long head of the biceps. No flaps should be developed, and the glenohumeral joint should not be entered.
Surgical Techniques
1. Resection Techniques
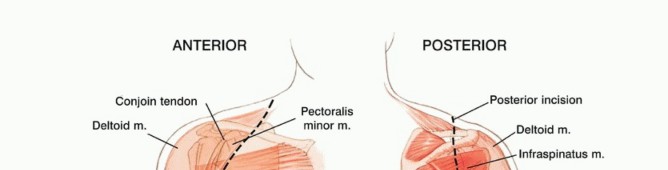
It is important to be extremely familiar with shoulder girdle anatomy and axillary and vascular structures. A utilitarian incision is used (TECH FIG 1 A-D). The anterior component is an extended deltopectoral incision that exposes the pectoralis major muscle, which is then released and retracted toward the chest wall. This exposes the axillary contents and permits exploration and safe dissection of the vascular structures and infraclavicular plexus.
An extra-articular resection is performed. Thus, the axillary nerve is identified and transected. The musculocutaneous nerve is identified and preserved. The radial nerve, which crosses the humerus posteriorly at the level of the deltoid insertion, is preserved. Approximately one-half to two-thirds of the humerus is resected.
An extra-articular resection is performed by exposing the glenohumeral joint both anteriorly and posteriorly. The scapula is osteotomized medial to the coracoid along with the distal portion of the clavicle. The resected specimen consists of the proximal one-half of the humerus, the glenohumeral joint, and the distal clavicle en bloc. A modular replacement proximal humeral prosthesis is used to reconstruct the skeletal defect.
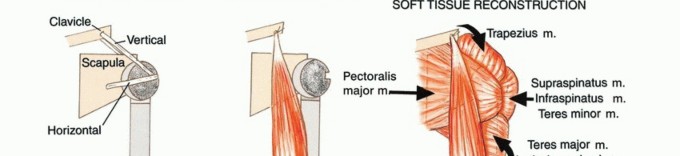
Attention must be paid to the reconstruction of the muscles for soft tissue coverage of the prosthesis. Static suspension is performed with Dacron tape, and the muscle reconstruction is performed with the pectoralis major muscle sutured to the remaining scapula. The remaining muscles are then tenodesed to the pectoralis major muscle. This technique permits immediate stability and restores motor power to the upper extremity. An epineural axillary sheath catheter is used to control postoperative pain. A 28-gauge chest tube is used for drainage through a Pleurovac. Postoperatively, the patient uses a sling for 2 weeks.
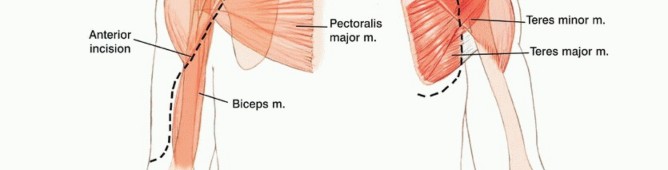
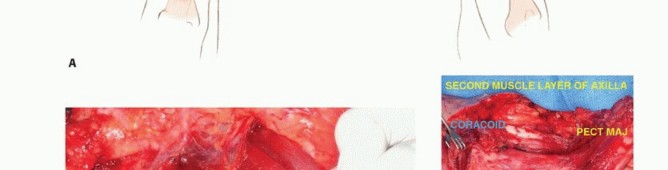

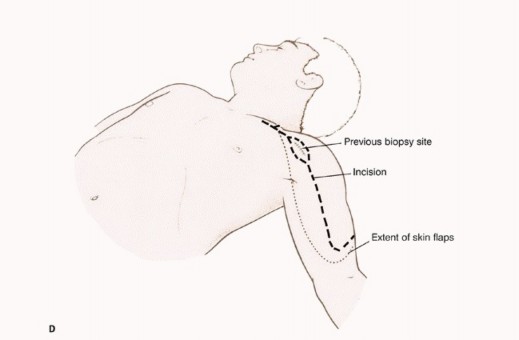
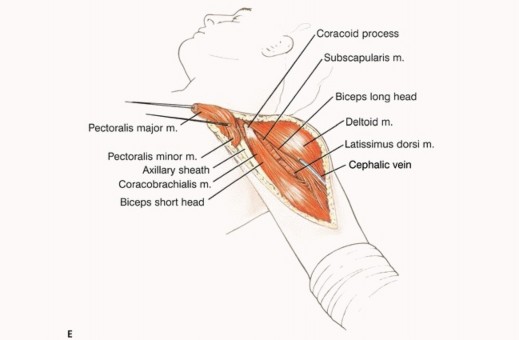
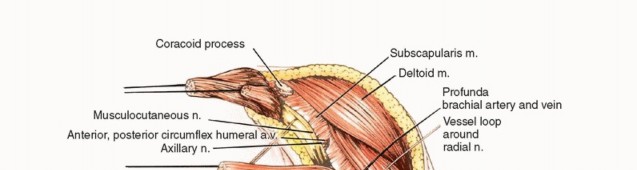
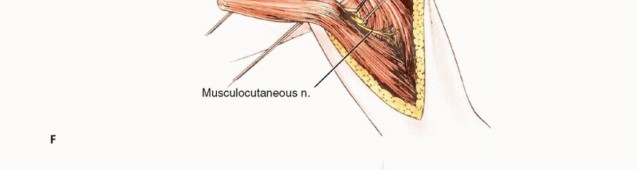
TECH FIG 1 A-G: Step-by-step utilitarian incision, deep muscular dissection, and exposure of the axillary space.
2. Endoprosthetic Replacement of the Proximal Humerus
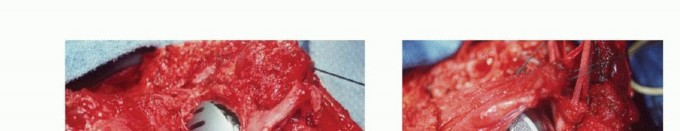
The Modular Replacement System (MRS; Stryker Orthopaedics, Mahwah, NJ), which is used for reconstruction of the shoulder girdle, is shown. Results of the MRS are predictable and successful, and the device is used for both intra- and extra-articular resections. Endoprosthetic reconstruction following tumor resection entails the following steps:
1. Fixation of the endoprosthesis in the remaining distal humerus.
2. Fixation and stabilization of the prosthetic humeral head to the scapula to provide a stable shoulder joint.
3. Soft tissue reconstruction to cover the prosthesis completely and optimize postoperative function.

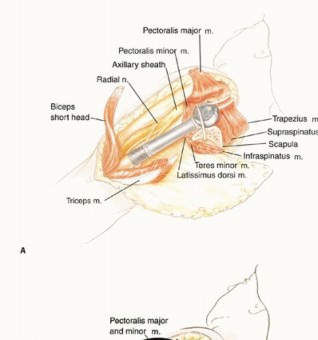
TECH FIG 2 & 3: Various methods for reconstruction of the proximal humerus and securing the prosthesis.
3. Dual Suspension Technique
A dual suspension (ie, static and dynamic) technique is used to create shoulder stability (TECH FIG 4). In the static reconstruction, drill holes are made in the distal portion of the osteotomized clavicle and through the remaining scapula at the level of the spine. The head of the prosthesis is secured to the remaining portion of the scapula with 3-mm Dacron tape so that the prosthesis is suspended mediolaterally to provide horizontal stability. It then is suspended, using more Dacron tape, in a craniocaudal direction from the end of the clavicle to provide vertical stability. Dynamic suspension is provided by transfer of the short head of the biceps muscle to the stump of the clavicle, which allows elbow flexion.
4. Soft Tissue Reconstruction
The remaining muscle groups are tenodesed to the pectoralis major and osteomized border of the scapula with Dacron tape. This mechanism offers dynamic support, assists in the suspension of the prosthesis, and provides soft tissue coverage. Soft tissue coverage is essential to cover the prosthesis and prevent skin problems and secondary infections.
5. Type I Resection
Intra-articular resection of the proximal humerus is indicated for low-grade sarcomas or high-grade sarcomas confined to the bone without extraosseous extension (stage IIA). The abductor mechanism and axillary nerve usually are preserved. This procedure is not recommended for high-grade sarcomas with soft tissue extension. The prosthesis is suspended from the glenoid with a Gore-Tex graft, which is reinforced by any remaining capsule. Anterior utilitarian shoulder incision is not required. The posterior component is not used. The axillary nerve is explored early and preserved. If there is tumor extension to the nerve, then the procedure is converted to a type V resection.
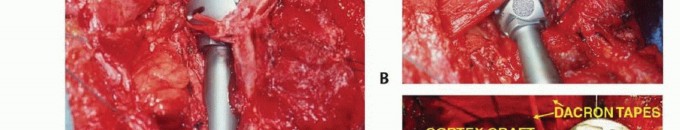

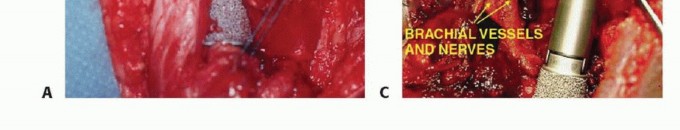
TECH FIG 4: Reconstruction showing static and horizontal tapes for immediate stability.
6. Total Humeral Resection and Prosthetic Reconstruction
Total humeral replacement is unusual but is indicated when the tumor involves a large portion of the diaphysis, such as in Ewing sarcoma, or when an extremely short segment of distal humerus remains following adequate tumor resection. The surgical technique is a combination of that used for proximal and distal humerus resections. Reconstruction provides stability of both shoulder and elbow joints.
Exposure and Extension of Type V Procedure: The surgical approach is similar to that used for a type V resection (ie, anterior utilitarian approach), but it requires additional distal exposure and identification and mobilization of the brachial artery and vein and the radial, ulnar, and median nerves. The incision and exposure are continued down the anteromedial aspect of the arm, across the antecubital fossa, and, if necessary, down the anterior aspect of the forearm. The brachial vessels, along with the median and ulnar nerves, are identified medially in the arm. The medial intermuscular septum is transected to allow further dissection and mobilization of the ulnar nerve so that it can be retracted medially with the brachial vessels and median nerve. The biceps is retracted medially with the neurovascular bundle. The radial nerve is identified where it passes around the humerus and into the interval between the brachialis and brachioradialis muscles and continues into the forearm.
The pronator teres and common flexor origins are transected medially, and the brachioradialis, extensor carpi radialis longus, and common extensor origins are released laterally to expose the distal humerus. A small cuff of muscle is left around the tumor as needed. The medial triceps muscle usually is resected with the tumor, but the lateral and long heads are retained. The triceps tendon is kept attached to the olecranon. The olecranon is not osteotomized. The elbow joint is opened anteriorly and the capsule released circumferentially. The humeroulnar and radiohumeral joints are then disarticulated.
Prosthetic Reconstruction, Muscle Reconstruction, and Postoperative Management: Reconstruction of the total humerus is similar to that of the proximal humerus. Distally, an ulnar endoprosthetic component with an intramedullary stem is cemented, with the olecranon left intact. Several articulating elbow devices are available. The reconstruction technique is similar to that used for the proximal humerus, with the addition of distal soft tissue and joint capsule reconstruction. The brachioradialis, pronator teres, and flexor carpi radialis muscles are sutured to the remaining biceps and triceps muscles to secure soft tissue around the flared distal portion of the humeral endoprosthesis. The remaining muscles are closed in layers in an attempt to cover the entire prosthesis. A posterior splint is used to protect the elbow reconstruction for 7 to 10 days. The surgical incision and wounds are examined on the fourth to fifth postoperative day.
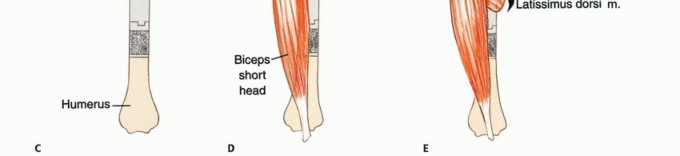
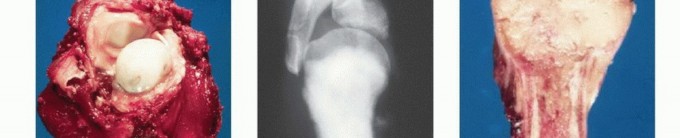
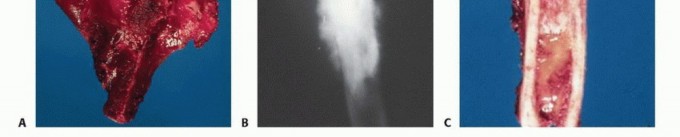
TECH FIG 5 & 6: Intraoperative photographs of intra-articular and extra-articular resections.
Pearls and Pitfalls
This chapter contains a complete description of the technique for a modified Tikhoff-Linberg procedure in patients with extirpation rare sarcomas of the proximal humerus. Modifications of the procedure also have been used for tumors at other anatomic sites. Proximal humeral lesions require resection of about two-thirds of the humerus.
The technique of resection and reconstruction requires a thorough knowledge of the regional anatomy and technique of musculoskeletal reconstruction. Essential aspects of the treatment plan should be emphasized:
- Biopsy: The initial biopsy should be performed through the anterior portion of the deltoid muscle for a lesion of the proximal humerus. The deltopectoral interval should not be used because biopsy here would contaminate the deltopectoral fascia, the subscapularis, and the pectoralis major muscles and would jeopardize the possibility of performing an adequate resection through uninvolved tissue planes.
- Incision: For the definitive resection, the initial incision extends along the medial aspect of the biceps muscle, divides the pectoralis major, and exposes the neurovascular structures, thereby enabling the surgeon to determine resectability early in the dissection. This incision does not jeopardize construction of an anterior skin flap in patients who will require forequarter amputation.
- Resection: The length of bone resection is determined preoperatively from a bone scan and MRI. To avoid a positive margin at the site of humeral transection, the distal osteotomy is performed 3-5 cm distal to the area of abnormality on the scan.
- Alternative Methods: Alternatively, other surgeons use autografts (usually fibulas) or allografts as spacers in obtaining an arthrodesis. We do not recommend osteoarticular allografts or intra-articular resections for high-grade bone sarcomas; those techniques were developed during the 1960s and 1970s and are inferior to current standards. Superior results routinely are obtained with modular prosthetic replacements combined with reconstruction of the soft tissues (FIG 4).
- Reconstruction: Segmental reconstruction of the resultant humeral defect is necessary to create shoulder stability. We do not leave a flail extremity. Reconstruction is necessary to maintain length of the arm and to create a fulcrum for elbow flexion. We recommend a custom or modular prosthesis. The key to success is reconstruction of the stability of the joint and soft tissue coverage of the prosthesis.
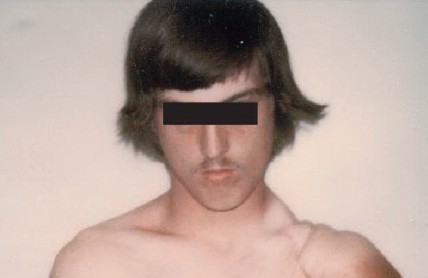
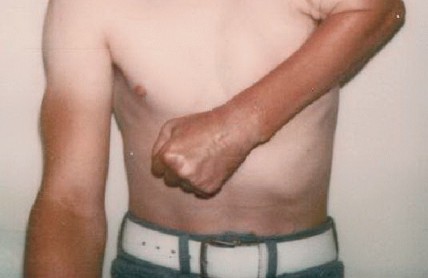
FIG 4: Historical results of early extra-articular resections showing marked shortening but functional preservation. (Courtesy of Ralph C. Marcove, MD.)
Outcomes
The proximal humerus resection, the prosthetic reconstruction and muscle plasty, is a very reliable procedure with good oncologic and functional results. Pain is well controlled in the majority of patients. The motor dexterity of the hand and the range of motion (ROM) of the elbow and wrist generally are preserved.
The Musculoskeletal Tumor Society upper extremity functional scores ranged from 24 to 27 (80% to 90%) with stable shoulders in the vast majority of the patients. Normally, all patients could do activities of daily life with the involved extremity. Some restrictions in activities can be seen, but most patients are capable of participating in some recreational activities. Most restrictions are seen in high-level athletes. Prosthetic survival is optimal with extremely low percentages of loosening and revisions. The rate of dislocation is minimal if static and dynamic reconstruction technique is followed.
Complications
Neurologic complications are infrequent and transient. Normally, 6 to 12 months after the surgery, all the nerve palsies are resolved. Neurapraxias due to late traction from the weight of the upper extremity occurs rarely. Loss of function is contingent on the extent of the muscle resection. Patients who present with extensive tumors around the shoulder girdle tend to have decreased ROM. Skin necrosis and superficial infections are uncommon. Dislocation of the shoulder is seen in less than 5% of the cases and no surgery is required.
Scientific References
- Cannon CP, Paraliticci GU, Lin PP, et al. Functional outcome following endoprosthetic reconstruction of the proximal humerus. J Shoulder Elbow Surg 2009;18(5):705-710.
- Malawer MM, Link M, Donaldson S. Sarcomas of bone. In: Devita VT, Hellman S, Rosenberg SA, eds. Cancer: Principles and Practice of Oncology , ed 3. Philadelphia: JB Lippincott, 1989.
- Malawer MM, Sugarbaker PH, Lambert MH, et al. The Tikhoff-Linberg procedure and its modifications. In: Sugarbaker PH, ed. Atlas of Sarcoma Surgery . Philadelphia: JB Lippincott, 1984.
- Wittig JC, Bickels J, Kellar-Graney KL, et al. Osteosarcoma of the proximal humerus: long-term results with limb-sparing surgery. Clin Orthop Relat Res 2002;(397):156-176.
Detailed Chapters & Topics
Dive deeper into specialized chapters regarding proximal-humerus-resection-with-endoprosthetic-replacement-intra-articular-and-extra-articular-resections
