Transverse Deficiencies: Arrest of Development & Surgical Management
Key Takeaway
Transverse deficiencies represent a failure of formation in the upper extremity, characterized by the complete absence of parts distal to a specific level. Affecting 6.8 per 10,000 live births, these congenital amputations are predominantly unilateral. Management relies heavily on age-appropriate prosthetic fitting, though surgical interventions, such as the excision of nonfunctional digital nubbins or the Krukenberg reconstruction, are indicated to optimize bimanual function, cosmesis, and tactile sensibility.
FAILURE OF FORMATION (ARREST OF DEVELOPMENT)
INTRODUCTION TO TRANSVERSE DEFICIENCIES
Transverse deficiencies represent a profound failure of formation (arrest of development) within the upper extremity. These deformities are characterized by the complete absence of skeletal and soft tissue elements distal to a specific point on the limb, producing an amputation-like stump. The classification of these anomalies is strictly anatomical, named according to the exact level at which the remaining stump terminates (e.g., transradial, transhumeral, or midcarpal).
Epidemiologically, Wynne-Davies and Lamb reported the incidence of transverse deficiencies to be approximately 6.8 per 10,000 live births. The vast majority of these presentations (98%) are unilateral, and there is no established sex predilection. The most frequently encountered anatomical level for a transverse deficiency is the upper third of the forearm (proximal transradial). The etiology is generally considered to be a sporadic vascular insult or a spontaneous genetic mutation during the critical window of embryogenesis, rather than a predictable hereditary pattern.
EMBRYOLOGY AND PATHOGENESIS
Understanding the pathogenesis of transverse deficiencies requires a thorough grasp of upper limb embryology. Limb development occurs between the fourth and eighth weeks of gestation, driven by complex molecular signaling centers.
The limb bud is composed of a mesenchymal core derived from the lateral plate mesoderm, covered by a layer of surface ectoderm. The critical signaling centers include:
* Apical Ectodermal Ridge (AER): Extending from the anterior to the posterior along the dorsal/ventral boundary of the growing limb bud, the AER is responsible for proximal-to-distal growth via Fibroblast Growth Factor (FGF) signaling. An early arrest or vascular disruption of the AER results in a transverse deficiency.
* Progress Zone: Located immediately proximal to the AER, this is an area of highly proliferating, undifferentiated mesodermal cells. As cells leave the progress zone, they begin to differentiate into specific skeletal elements.
* Zone of Polarizing Activity (ZPA): Situated within the posterior mesoderm, the ZPA is the primary signaling center for the radioulnar (anterior-posterior) axis, mediated by Sonic Hedgehog (SHH) protein expression.
These signaling centers are intricately interconnected. Limb patterning and growth depend entirely on their coordinated function. A failure in this cross-talk, particularly involving the AER, leads to the abrupt cessation of distal limb formation.

CLINICAL PRESENTATION
The clinical presentation of a transverse deficiency is an amputation-like stump that is typically well-padded with soft tissue. In many cases, rudimentary, nonfunctional soft-tissue appendages known as "digital nubbins" are present at the distal end of the stump.
These nubbins rarely contain functional skeletal elements or tendinous attachments, though they may possess rudimentary fingernails. Despite their lack of prehensile function, the remaining stump and wrist motion (if preserved) allow the affected limb to be used effectively as an assisting hand in bimanual tasks.
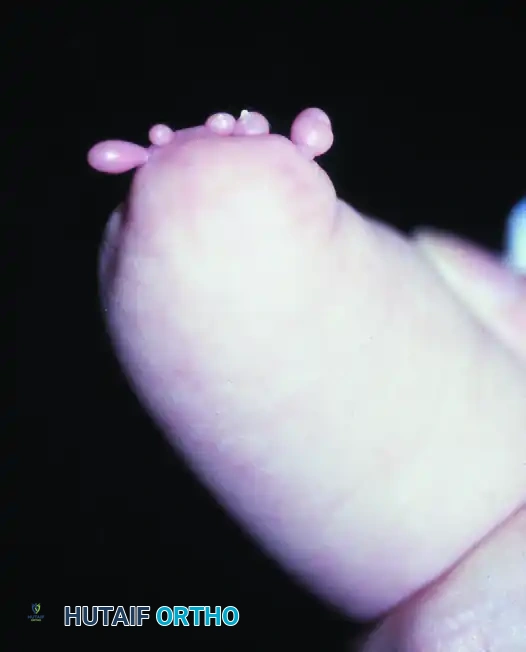
Clinical Pearl: Always evaluate the mobility and skeletal support of digital nubbins radiographically before considering excision. While most are flail and nonfunctional, any nubbin with underlying skeletal continuity and voluntary motor control should be preserved to maximize the child's tactile and prehensile capabilities.
PRINCIPLES OF PROSTHETIC MANAGEMENT
The cornerstone of managing transverse deficiencies is age-appropriate prosthetic fitting, designed to complement the child's natural neurodevelopmental milestones. Normal prehensile development dictates that gross grasp develops first, followed by thumb-to-finger pinch by 12 to 18 months of age. The ability to grasp an object precedes the ability to voluntarily release it. By 24 months, a child typically develops coordinated shoulder positioning, grasp, and release. Prosthetic introduction must align with these stages.
Midcarpal Amputations
Prosthetic treatment for a child with a midcarpal amputation remains highly controversial. Although the carpal bones do not ossify and cannot be seen radiographically until approximately 6 to 8 months of age, their cartilaginous presence improves the overall prognosis, as minimal shortening of the forearm is expected. Delay in carpal bone maturation is rarely encountered in these patients.
The long, below-elbow stump is exceptionally useful for stabilizing objects and assisting in bimanual functions. Because terminal sensibility is preserved and highly valuable, the benefits of a prosthesis are debatable. If a prosthesis is pursued, options include:
1. An open-ended volar plate secured to the forearm, permitting a simple grip between the stump and the plate.
2. An open-ended volar plate equipped with a terminal hook.
3. An artificial hand driven by preserved radiocarpal motion.
While the artificial hand provides a superior cosmetic effect, it sacrifices terminal sensibility. Regardless of the chosen device, therapist-supervised training sessions are mandatory. Coordinated follow-up among the patient, family, occupational therapist, orthotist, and orthopedic surgeon is essential to prevent prosthesis rejection, which is particularly common among adolescent boys.
Complete Arm Amputations
For the rare child presenting with a complete arm amputation (transhumeral or higher), especially in bilateral cases, conventional body-powered prostheses that include an articulated elbow are unlikely to provide significant functional benefit early on.
* Initial Fitting: A rigid elbow is utilized initially.
* 18 Months: When the passive mitten is exchanged for an actively opened split hook, the rigid elbow is replaced by a friction elbow.
* 3 Years: Dual-terminal devices and active elbow controls may be introduced. In bilateral above-elbow amputations, only the dominant (or preferred) side is fitted with a dual-control, articulated prosthesis to reduce cognitive and mechanical overload.
Upper Third Forearm Amputations
For the most common presentation—an amputation at the upper third of the forearm—the prosthetic protocol is well-standardized:
* 3 to 6 Months ("Sit to Fit"): A passive plastic mitten prosthesis is introduced when the child achieves independent sitting balance. This provides stability during sitting and assists the child in pulling to a standing position.
* 12 to 18 Months: The passive mitten is replaced with an actively opened, plastisol-covered split hook. Alternatively, a Child Amputee Prosthetic Program (CAPP) terminal device may be substituted. Training with this functional device begins immediately.
* Preschool (2 to 4 Years): A myoelectric prosthesis may be introduced. Early fitting of a passive or myoelectric prosthesis encourages bilateral limb use and cortical integration of the device.


SURGICAL MANAGEMENT: INDICATIONS AND PHILOSOPHY
There are strictly limited indications for surgical intervention in children with transverse deficiencies of the upper extremity. The primary goal of treatment is functional independence, which is usually achieved non-operatively. However, surgery is indicated in specific scenarios to improve prosthetic fit, enhance cosmesis, or construct a prehensile limb in severe bilateral cases.
Excision of Digital Nubbins
Amputation of nonfunctional digital remnants is frequently performed for psychological and cosmetic benefits. Complete amputation of all digits often gives the hand the bizarre appearance of a "little paw" with small nubbins attached.
As famously stated by Littler and emphasized by Flatt, it is often wise to alter the "stigma of congenitalism" and make the deformity appear acquired rather than congenital. A simple elliptical excision of the nubbins is appropriate.
Surgical Warning: When excising digital nubbins, meticulous dissection is required. Even rudimentary digits may contain small neurovascular bundles. Failure to identify and deeply ligate these nerves can result in painful terminal neuromas, which will severely compromise future prosthetic wear.
ADVANCED SURGICAL RECONSTRUCTION: THE KRUKENBERG PROCEDURE
In cases of bilateral transverse deficiencies (bilateral below-elbow amputations), particularly in patients who are blind or visually impaired, standard prostheses are often inadequate because they deprive the patient of essential tactile feedback. In such profound cases, the Krukenberg Reconstruction is a highly specialized, limb-salvaging procedure that converts the forearm stump into a sensate, pincer-like grasping mechanism.
Biomechanics of the Krukenberg Pincer
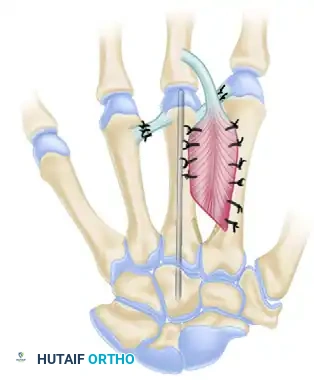
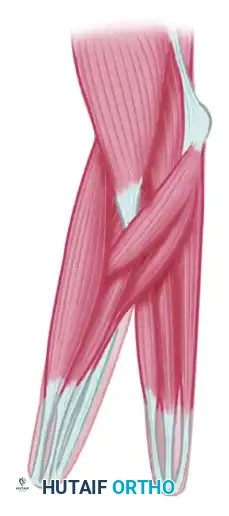
The Krukenberg procedure involves the longitudinal separation of the radius and ulna, along with their respective muscle groups, to create two independent "prongs" (a radial ray and an ulnar ray).
* The Pronator Teres is the most critical muscle in this reconstruction; it serves as the primary motor for closing the pincer (bringing the radius toward the ulna).
* Opening the pincer is achieved passively by relaxation of the pronator teres, or actively by the supinator and extensor muscles.
* Tactile sensibility is preserved between the tips of the newly formed rays, allowing the patient to feel and manipulate objects without visual input.
Surgical Technique: Step-by-Step
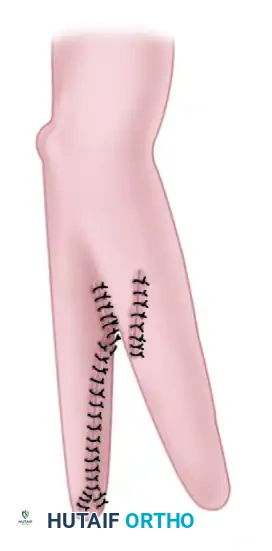
Step 1: Incision and Exposure
The procedure begins with precise incisions designed to allow the separation of the forearm while preserving adequate skin flaps for coverage.
* A longitudinal incision is made on the flexor (volar) surface of the forearm, slightly ulnar to the midline.
* A corresponding longitudinal incision is made on the dorsal surface.
Step 2: Muscle Separation
The deep fascia is incised, and the forearm musculature is meticulously separated into two distinct groups: a radial group and an ulnar group. The interosseous membrane is divided longitudinally from distal to proximal, taking extreme care to protect the interosseous vessels and the median and ulnar nerves.

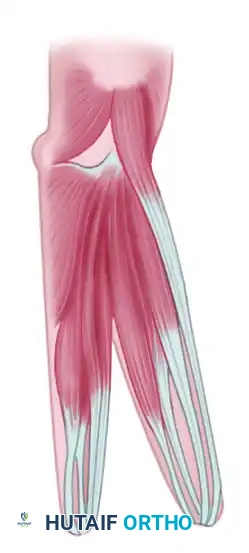
The muscles are divided as follows to ensure balanced biomechanics:
* Radial Group (Motorizing the Radius):
* Biceps brachii
* Supinator
* Brachioradialis
* Flexor carpi radialis (FCR)
* Radial half of the Flexor digitorum sublimis (FDS)
* Pronator teres (CRITICAL: Must remain attached to the radius)
* Radial half of the Extensor digitorum communis (EDC)
* Extensor carpi radialis brevis (ECRB)
* Extensor carpi radialis longus (ECRL)
- Ulnar Group (Motorizing the Ulna):
- Flexor carpi ulnaris (FCU)
- Ulnar half of the Flexor digitorum sublimis (FDS)
- Palmaris longus
- Brachialis
- Triceps brachii
- Ulnar half of the Extensor digitorum communis (EDC)
- Anconeus
- Extensor carpi ulnaris (ECU)
- Extensor digiti quinti proprius (EDQ)
Pitfall: Iatrogenic injury to the median nerve during the splitting of the flexor digitorum sublimis or the division of the interosseous membrane will result in a catastrophic loss of tactile sensibility on the opposing surfaces of the pincer, entirely defeating the purpose of the operation.
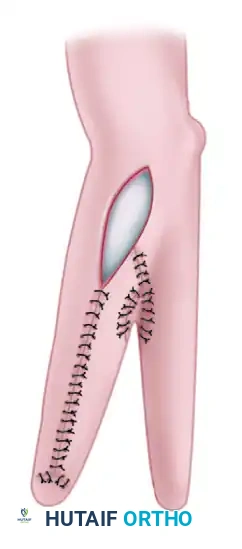
Step 3: Skin Closure and Grafting
Once the radius and ulna are fully mobilized and hemostasis is achieved, the skin flaps are wrapped around the newly created radial and ulnar rays.
* The flexor and dorsal skin flaps are closed over the opposing surfaces of the pincer to ensure that the grasping surfaces have normal, sensate skin.
* Because the surface area of the separated forearm exceeds the available local skin, a defect is inevitably left on the outer (non-opposing) aspects of the rays.
* An elliptical area on the flexor or dorsal surface will require coverage with a split-thickness skin graft harvested from the thigh or buttocks.

Postoperative Protocol
Postoperatively, the limb is immobilized in a bulky, non-compressive dressing with the pincer held in a widely abducted (open) position to prevent contracture of the pronator teres and to protect the skin grafts.
Once the grafts have fully taken (typically 2 to 3 weeks), aggressive occupational therapy is initiated. The patient is trained to use the pronator teres to actively close the pincer against the ulna. Sensory re-education is paramount, allowing the patient to integrate the new biomechanical construct into their daily activities. While cosmetically striking, the Krukenberg procedure provides unparalleled functional independence for the bilaterally amputated, visually impaired patient, representing a triumph of functional orthopedic reconstruction over aesthetic norms.
You Might Also Like