Comprehensive Management of Infantile Idiopathic Scoliosis: Biomechanics, Prognostication, and Surgical Techniques
Key Takeaway
Infantile idiopathic scoliosis presents before age three and carries a significant risk of cardiopulmonary compromise if progressive. Management hinges on differentiating resolving from progressive curves using Mehta’s rib-vertebral angle difference (RVAD). While 70% to 90% of curves resolve spontaneously, progressive deformities require aggressive early intervention. Treatment algorithms progress from serial elongation-derotation-flexion (EDF) casting and orthotics to growth-sparing surgical interventions, such as magnetically controlled growing rods, to maximize thoracic volume and preserve spinal growth.
SCOLIOSIS CLASSIFICATION AND CARDIOPULMONARY DYNAMICS
The Scoliosis Research Society (SRS) traditionally recommends that idiopathic scoliosis be classified chronologically according to the age of the patient at the time of initial diagnosis. Under this paradigm, infantile idiopathic scoliosis (IIS) occurs from birth to 3 years of age; juvenile idiopathic scoliosis presents between the ages of 4 and 10 years; and adolescent idiopathic scoliosis (AIS) manifests between 10 years of age and skeletal maturity. This chronological definition remains clinically vital because profound differences exist between these subtypes regarding natural history, curve progression, and systemic implications.
Modern orthopedic consensus, championed by Conner, Dickson, Davies, and Reid, strongly advocates for a broader physiological distinction between early-onset scoliosis (EOS) (presenting before age 5) and late-onset scoliosis. This paradigm shift is rooted in the devastating effects of early spinal deformity on cardiopulmonary development. During early childhood, the pulmonary system undergoes rapid and critical maturation. Not only do the lungs expand volumetrically, but alveolar arborization and arterial multiplication occur exponentially. The number of alveoli in the pulmonary tree increases approximately 10-fold between infancy and 4 years of age, with complete alveolar development not achieved until age 8.
A progressive scoliotic deformity in this age group severely limits the thoracic space available for lung growth, leading to Thoracic Insufficiency Syndrome (TIS). Children who develop significant, unyielding scoliosis before the age of 5 years are at an exceptionally high risk of developing disabling dyspnea, cor pulmonale, and premature cardiorespiratory failure.
Clinical Pearl: Early-onset scoliosis is not merely a cosmetic or structural orthopedic issue; it is a potentially lethal cardiopulmonary disease. The primary goal of treatment in the infantile and juvenile populations is the preservation of thoracic volume to allow for maximal alveolar development.
INFANTILE IDIOPATHIC SCOLIOSIS: EPIDEMIOLOGY AND ETIOLOGY
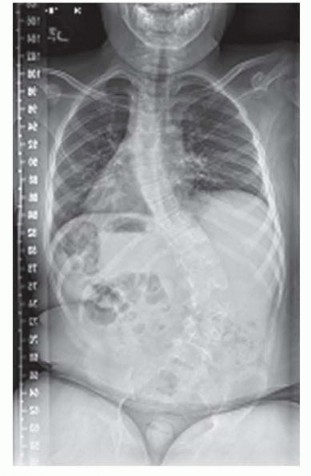
Infantile idiopathic scoliosis is defined as a structural, lateral curvature of the spine occurring in patients younger than 3 years of age without an identifiable congenital, syndromic, or neuromuscular etiology. James, who first coined the term, observed distinct epidemiological patterns: these curves present before 3 years of age, exhibit a higher prevalence in males than in females (in stark contrast to adolescent idiopathic scoliosis), and are predominantly thoracic with a left-sided convexity.
Wynne-Davies expanded on the phenotypic profile of these patients, noting a strong association with plagiocephaly. In a cohort of 97 children who developed curves in the first 6 months of life, plagiocephaly was nearly ubiquitous, with the flattened side of the infant's head consistently corresponding to the convex side of the spinal curve. Furthermore, her epidemiological studies revealed a higher incidence of concurrent anomalies, including mental retardation (13%), inguinal hernias in boys with progressive curves (7.4%), congenital dysplasia of the hip (CDH) (3.5%), and congenital heart defects (2.5%). These findings suggest a multifactorial etiology, likely involving a genetic predisposition that is either "triggered" or mitigated by external intrauterine or extrauterine mechanical factors.
The Postural Molding Hypothesis and Biomechanics
McMaster and Macnicol proposed a compelling biomechanical theory suggesting that infantile idiopathic scoliosis may be largely preventable, implicating infant positioning as a primary causative factor. Observational studies by Brown et al. and Mau demonstrated that during the first few months of life, infants exhibit a natural, asymmetrical tendency to turn their heads toward the right side when placed in a supine position.
McMaster postulated that when an infant is laid supine and partially turned toward the right, gravity exerts a continuous, asymmetrical force on the highly compliant, cartilaginous immature thorax. This gravitational vector causes plastic deformation: the uppermost (left) hemithorax tends to fall backward, forcing the thoracic vertebrae to rotate posteriorly on their longitudinal axes.

Due to the inherent biomechanical coupling of the spine, axial rotation obligatorily induces lateral bending. This coupled motion produces a structural lateral curvature, manifesting as a left-sided thoracic scoliosis in infants who habitually lie on their right sides. Conversely, prone positioning prevents this asymmetrical molding. In the prone position, the infant cannot easily roll onto the side, which neutralizes the asymmetrical gravitational vectors and simultaneously promotes the early activation and maturation of the spinal extensor musculature. McMaster’s epidemiological observations in Edinburgh supported this, noting a near-total absence of IIS in infants who were strictly laid prone.
RADIOGRAPHIC EVALUATION AND PROGNOSTICATION
The natural history of infantile scoliosis follows one of two distinct paths: it may be progressive, increasing rapidly and relentlessly, or it may be resolving, spontaneously improving over a few years with or without intervention. Fortunately, the resolving type accounts for 70% to 90% of all IIS cases. The critical clinical challenge lies in differentiating a resolving curve from a malignant, progressive curve at the time of initial presentation, as early intervention is paramount for the latter.
James et al. noted that resolving curves generally present before 1 year of age, possess smaller initial Cobb angles, and lack compensatory structural curves. Lloyd-Roberts and Pilcher correlated resolving curves with plagiocephaly, suggesting an intrauterine molding etiology that corrects as the child gains mobility. Conversely, the development of secondary compensatory curves, or an initial Cobb angle exceeding 37 degrees, is highly predictive of relentless progression.
Mehta’s Rib-Vertebral Angle Difference (RVAD)
To provide a reliable, objective radiographic metric for prognostication, Min Mehta developed the Rib-Vertebral Angle Difference (RVAD). This measurement assesses the asymmetrical relationship between the apical vertebra and its articulating ribs, reflecting the degree of rotational deformity.
Technique for Measuring RVAD:
1. Identify the apical vertebra of the curve on a standard anteroposterior (AP) radiograph.
2. Draw a line perpendicular to the superior endplate of the apical vertebra.
3. Draw a second line bisecting the neck and head of the corresponding rib on the concave side. The angle formed by the intersection of these two lines is the Rib-Vertebral Angle (RVA).
4. Repeat this process for the corresponding rib on the convex side.
5. The RVAD is calculated by subtracting the convex RVA from the concave RVA.
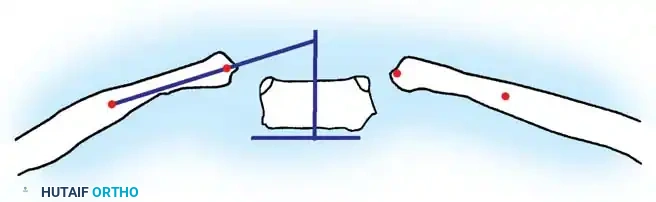
Interpretation of Mehta's Criteria:
* RVAD < 20 degrees: Indicates a high likelihood of spontaneous resolution. These patients require close clinical and radiographic observation (every 4 to 6 months) but generally do not require immediate active treatment.
* RVAD > 20 degrees: Strongly predictive of a progressive curve. If the curve is also clinically rigid, it must be treated as progressive until proven otherwise.
* Phase 1 vs. Phase 2 Rib: Mehta also described the relationship of the convex rib head to the apical vertebral body. In Phase 1, the rib head does not overlap the vertebral body. In Phase 2, the convex apical rib head overlaps the apical vertebral body on the AP radiograph. A Phase 2 rib is an ominous sign, indicating severe rotation and a highly progressive nature, mandating immediate intervention regardless of the exact RVAD value.
Surgical Warning: Failure to recognize a Phase 2 rib or an RVAD > 20 degrees can lead to catastrophic curve progression. These curves must be monitored aggressively to prevent severe thoracic deformity and subsequent restrictive pulmonary disease.
NON-OPERATIVE MANAGEMENT PROTOCOLS
The management of progressive infantile idiopathic scoliosis is a stepwise escalation from non-invasive techniques to surgical stabilization, always prioritizing the preservation of thoracic growth.
Serial Casting (Elongation-Derotation-Flexion)
For curves deemed progressive (RVAD > 20 degrees, Phase 2 rib, or documented clinical progression), serial casting is the gold standard of early management. The goal is not merely to halt progression, but to harness the rapid growth of the infant to achieve permanent curve correction.
- Technique: Casting is performed under general anesthesia to ensure complete muscle relaxation. The child is placed on a specialized casting frame (e.g., Mehta or Amica table). Traction is applied longitudinally (elongation), followed by derotation of the apical segments, and slight flexion to restore thoracic kyphosis. A well-molded plaster or fiberglass cast is applied over a protective stockinette and felt padding.
- Molding: Precise molding is critical. Windows are cut out anteriorly over the abdomen to allow for diaphragmatic excursion and feeding, and posteriorly on the concave side to create a "relief space" into which the derotated spine and ribs can migrate during growth.
- Protocol: Casts are typically changed every 2 to 3 months to accommodate the infant's rapid somatic growth. This process continues until the curve resolves, plateaus, or the child grows large enough to transition to an orthosis.
Orthotic Management
A custom-fabricated Thoracic-Lumbar-Sacral Orthosis (TLSO) or Cervical-Thoracic-Lumbar-Sacral Orthosis (CTLSO) is utilized once the child has achieved sufficient size, or as a step-down therapy following successful serial casting.
* Indications: Flexible curves under 40 degrees, or maintenance of correction post-casting.
* Efficacy: Orthoses are highly effective at delaying progression during the early period of skeletal growth. Brace wear is mandated full-time (20-22 hours/day) until curve stability is maintained for at least 2 years, after which a gradual weaning protocol is initiated.
SURGICAL MANAGEMENT OF PROGRESSIVE INFANTILE SCOLIOSIS
When a curve is severe at presentation, or relentlessly progresses despite optimal serial casting and orthotic management, surgical stabilization becomes imperative. The surgical philosophy in the infantile and juvenile populations is fundamentally different from that in adolescents. The primary objective is to halt curve progression while simultaneously allowing for continued longitudinal growth of the spine and volumetric expansion of the thorax.
Growth-Sparing Techniques: Subcutaneous Instrumentation
For children under the age of 8 years, definitive spinal fusion is contraindicated due to the severe stunting of truncal height and the induction of iatrogenic thoracic insufficiency syndrome. Instead, growth-friendly constructs, such as traditional growing rods (TGR) or magnetically controlled growing rods (MCGR), are utilized.
Step-by-Step Surgical Approach for Growing Rods:
- Preoperative Planning & Positioning:
- The patient is positioned prone on a pediatric Jackson table or specialized gel rolls. All bony prominences are meticulously padded.
- Neuromonitoring (Somatosensory Evoked Potentials [SSEPs] and Motor Evoked Potentials [MEPs]) is mandatory.
- Incision and Exposure:
- Rather than a single long midline incision, two separate, short midline incisions are made: one over the proximal foundation (upper thoracic spine) and one over the distal foundation (lower lumbar spine or pelvis).
- The intervening spine is left entirely undisturbed to prevent spontaneous autofusion of the immature posterior elements.
- Foundation Creation:
- Proximal Foundation: Typically spans 2 to 3 levels. Fixation is achieved using pedicle screws or a combination of pedicle screws and transverse process/rib hooks to distribute pull-out forces.
- Distal Foundation: Spans 2 to 3 levels in the lower lumbar spine using pedicle screws. In cases of pelvic obliquity, fixation may be extended to the pelvis using iliac or S2-alar-iliac (S2AI) screws.
- Subfascial Tunneling:
- A specialized tunneling device is passed subfascially or submuscularly between the proximal and distal incisions. This creates a soft-tissue envelope for the rods while avoiding periosteal stripping of the apical vertebrae.
- Rod Contouring and Insertion:
- Dual rods are contoured to maintain physiological sagittal alignment (thoracic kyphosis and lumbar lordosis).
- The rods are passed through the subfascial tunnels and secured to the proximal and distal foundations.
- In traditional growing rods, a tandem connector is placed in the thoracolumbar region to link the proximal and distal rod segments, allowing for future surgical lengthenings. In MCGRs, the magnetic actuator is positioned in the mid-subfascial tunnel.
- Distraction and Closure:
- Gentle distraction is applied to achieve curve correction. Over-distraction must be avoided to prevent anchor pull-out or neurological injury.
- The wounds are closed in multiple layers to prevent hardware prominence and infection, a common complication in this malnourished, low-BMI population.
Postoperative Protocol for Growth Rods:
Traditional growing rods require repeated surgical lengthenings (typically every 6 months) under general anesthesia until the child reaches skeletal maturity. Magnetically controlled growing rods allow for non-invasive outpatient lengthenings using an external remote controller, significantly reducing the psychological and physiological burden of repeated surgeries.
Definitive Arthrodesis and the Crankshaft Phenomenon
If surgical stabilization is required in an older child (approaching 10-11 years of age), or if growth-sparing techniques have reached their maximum utility, a definitive spinal fusion is performed.
Pitfall: Performing an isolated posterior spinal fusion (PSF) in a skeletally immature patient (open triradiate cartilage, Risser 0) carries a high risk of the "Crankshaft Phenomenon."
The Crankshaft Phenomenon:
When a solid posterior fusion mass is created, the posterior elements are tethered. However, the anterior neurocentral synchondroses and vertebral body growth plates remain open and continue to grow. This continued anterior longitudinal growth, restricted by the posterior tether, forces the spine to buckle and rotate around the fusion mass, leading to a severe, progressive rotational deformity and recurrent clinical scoliosis despite a solid posterior arthrodesis.
Prevention and Surgical Strategy:
To prevent the crankshaft phenomenon in patients with significant remaining growth (typically girls < 10 years, boys < 12 years, or open triradiate cartilages), a combined anterior and posterior spinal fusion (ASF-PSF) is mandatory.
* The anterior approach (via thoracotomy or thoracoabdominal approach) involves complete discectomies and disruption of the anterior growth plates over the length of the structural curve, followed by anterior bone grafting.
* This is immediately followed by a standard posterior instrumentation and fusion. By arresting both anterior and posterior growth simultaneously, the spine is definitively stabilized without the risk of rotational buckling.
In modern practice, the advent of highly rigid, all-pedicle-screw posterior constructs has reduced the incidence of the crankshaft phenomenon, but combined anterior-posterior approaches remain the gold standard for large, rigid curves in highly immature patients requiring definitive fusion.
📚 Medical References
- infantile idiopathic scoliosis, J Bone Joint Surg 62A:863, 1980.
- Conner AN: Early onset scoliosis: a call for awareness, Br Med J 289:962, 1994.
- Davies G, Reid L: Effect of scoliosis on growth of alveoli, pulmonary arteries, and the right ventricle, Arch Dis Child 46:623, 1971.
- Dickson RA: The etiology of spinal deformities, Lancet 1:1151, 1988.
- Dickson RA, Lawton JO, Archer IA, et al: The pathogenesis of idiopathic scoliosis: biplanar spinal asymmetry, J Bone Joint Surg 66B:8, 1984.
- Dimeglio A: Growth of the spine before 5 years, J Pediatr Orthop 1B:102, 1993.
- Dubousset J, Herring JA, Shuffl ebarger H: The crankshaft phenomenon, J Pediatr Orthop 9:541, 1989.
- Evans SC, Edgar MA, Hall-Craggs MA, et al: MRI of “idiopathic” juvenile scoliosis, J Bone Joint Surg 78B:314, 1996.
- Figueiredo UM, James JIP: Juvenile idiopathic scoliosis, J Bone Joint Surg 63B:61, 1981.
- Gupta P, Lenke LG, Bridwell KH: Incidence of neural axis abnormalities in infantile and juvenile patients with spinal deformity. Is a magnetic resonance image screening necessary? Spine 23:206, 1998.
- Hefti FL, McMaster MJ: The effect of the adolescent growth spurt on early posterior spinal fusion in infantile and juvenile idiopathic scoliosis, J Bone Joint Surg 65B:247, 1983.
- James JIP: Infantile idiopathic scoliosis, Clin Orthop 21:106, 1961.
- James JIP: The management of infants with scoliosis, J Bone Joint Surg 57B:422, 1975.
- James JIP, Lloyd-Roberts GC, Pilcher MF: Infantile structural scoliosis, J Bone Joint Surg 41B:719, 1959.
- Kager AN, Marks M, Bastrom T, et al: Morbidity of iliac crest bone graft harvesting in adolescent deformity surgery, J Pediatr Orthop 26:132, 2006.
- Kahanovitz N, Levine DB, Lardone J: The part-time Milwaukee brace treatment of juvenile idiopathic scoliosis: long-term follow-up, Clin Orthop Relat Res 167:145, 1982.
- Koop SE: Infantile and juvenile idiopathic scoliosis, Orthop Clin North Am 19:331, 1988.
- Krismundsdottir F, Burwell RG, James JIP: The rib-vertebra angles on the convexity and concavity of the spinal curve in infantile idiopathic scoliosis, Clin Orthop Relat Res 201:205, 1985.
- Lewonowski K, King JD, Nelson MD: Routine use of magnetic resonance imaging in idiopathic scoliosis patients less than 11 years of age, Spine 17(suppl):510, 1992.
- Lloyd-Roberts GC, Pilcher MF: Structural idiopathic scoliosis in infancy, J Bone Joint Surg 47B:520, 1965.
- Lonstein JE, Carlson JM: The prediction of curve progression in untreated idiopathic scoliosis during growth, J Bone Joint Surg 66A:1061, 1984.
- Lowe TG, Peters JD: Anterior spinal fusion with Zielke instrumentation for idiopathic scoliosis. A frontal and sagittal curve analysis in 36 patients, Spine 18:423, 1993.
- Luhmann SJ, Lenke LG, Kim YJ, et al: Thoracic
You Might Also Like