Axial Skeleton Fractures: Epidemiology, Surgical Anatomy, and Biomechanics of Spinal Trauma
Key Takeaway
Axial skeleton fractures, often from high-energy trauma, present significant neurological risk. Key insights cover their epidemiology (e.g., MVAs, falls, multifocal injuries), detailed surgical anatomy of vertebrae, spinal cord, and ligaments, plus core biomechanical principles like Denis's three-column theory. This knowledge is vital for accurate diagnosis and effective management.
Axial Skeleton Fractures: Critical Insights for Spine Injuries
Introduction & Epidemiology
Spinal column fractures represent a significant public health burden, demanding precise diagnosis and management to mitigate neurological sequelae and chronic pain. While injuries to the appendicular skeleton are statistically more frequent, vertebral column fractures, accounting for approximately 6% of all fractures, carry a disproportionately high risk for devastating neurological impairment. There are approximately 12,000 new spinal cord injuries (SCI) requiring treatment annually in the United States, with motor vehicle accidents (MVAs) contributing to nearly 50% of these traumatic SCIs. Falls, acts of violence, and sports injuries comprise other prevalent etiologies.
A critical aspect of vertebral trauma is the potential for multifocal injury; 15% to 20% of vertebral fractures can occur at multiple noncontiguous levels, necessitating thorough pan-spinal imaging. The cervical spine is particularly vulnerable, with 2% to 6% of trauma patients sustaining a cervical spine fracture. This vulnerability is attributed to its inherent mobility and exposure. Demographic analysis consistently demonstrates a male predominance, with a male-to-female ratio of 4:1 for vertebral fractures, likely reflecting higher rates of high-energy trauma in this population.
The societal and individual economic impact of SCI is profound. Beyond the acute morbidity and mortality (overall mortality during initial hospitalization for SCI patients is approximately 17%), the lifetime direct medical cost for a person injured at age 25 can range from an estimated $1.5 million to $4.6 million, contingent upon the severity and completeness of the neurological deficit. These figures underscore the imperative for optimized acute management and effective long-term rehabilitation strategies.
Surgical Anatomy & Biomechanics
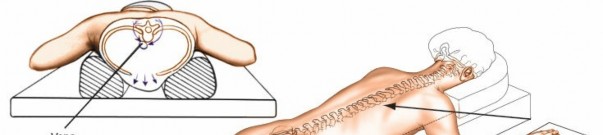
A comprehensive understanding of spinal anatomy and biomechanics is fundamental to the evaluation and management of axial skeleton fractures. The vertebral column comprises 33 vertebrae (7 cervical, 12 thoracic, 5 lumbar, 5 fused sacral, 4 fused coccygeal) separated by intervertebral discs, forming a flexible yet robust structure designed to protect the spinal cord and support the trunk.
Vertebral Anatomy:
*
Vertebral Body:
The primary weight-bearing structure, composed of cancellous bone encased in cortical bone.
*
Pedicles:
Stout bony projections extending posteriorly from the vertebral body, forming the lateral walls of the vertebral foramen. They serve as ideal conduits for instrumentation.
*
Laminae:
Broad, flat plates extending posteromedially from the pedicles, uniting in the midline to form the spinous process. They complete the posterior aspect of the vertebral foramen.
*
Articular Processes:
Superior and inferior facets projecting from the junction of the pedicles and laminae, forming synovial facet joints crucial for segmental stability and motion.
*
Transverse Processes:
Lateral projections from the junction of the pedicles and laminae, serving as attachment points for muscles and ligaments.
*
Spinous Process:
A posterior projection from the laminae, providing muscle attachment and aiding in palpation.
Spinal Cord and Neural Elements:
The spinal cord, a continuation of the brainstem, extends from the foramen magnum to the conus medullaris.
* The
conus medullaris
represents the caudal termination of the spinal cord, containing the sacral and coccygeal myelomeres. It typically lies dorsal to the L1 vertebral body and L1-L2 intervertebral disc in adults. Pathological variations in this level exist.
* The
cauda equina
(Latin for "horse's tail") comprises the motor and sensory nerve roots of the lumbosacral myelomeres that descend distally from the conus medullaris within the vertebral canal before exiting at their respective intervertebral foramina. These roots are more resilient to compression than the spinal cord itself due to their greater vascularity and peripheral nerve characteristics.
* The spinal cord occupies approximately 35% of the canal at the level of the atlas (C1) and about 50% of the canal in the lower cervical spine and thoracolumbar segments. The remaining canal volume is occupied by epidural fat, cerebrospinal fluid (CSF), and the dura mater, offering a degree of protective cushioning. This canal occupancy percentage is critical; smaller percentages (e.g., C1) may imply less reserve, but also often less direct cord impingement unless there is significant displacement.
Ligamentous Structures:
The robust ligamentous complex provides significant stability to the spinal column.
*
Anterior Longitudinal Ligament (ALL):
Strongest spinal ligament, resisting hyperextension.
*
Posterior Longitudinal Ligament (PLL):
Within the vertebral canal, resists hyperflexion. Thinner and weaker than the ALL, particularly in the lumbar region.
*
Ligamentum Flavum:
Elastic ligament connecting adjacent laminae, preserving the upright posture and helping to limit hyperflexion.
*
Interspinous and Supraspinous Ligaments:
Connect adjacent spinous processes, resisting hyperflexion.
*
Capsular Ligaments:
Surround the facet joints, providing segmental stability.
Biomechanics and Stability:
The three-column theory by Denis is foundational for understanding spinal stability.
1.
Anterior Column:
Consists of the ALL, the anterior two-thirds of the vertebral body, and the anterior two-thirds of the annulus fibrosus.
2.
Middle Column:
Consists of the PLL, the posterior one-third of the vertebral body, and the posterior one-third of the annulus fibrosus.
3.
Posterior Column:
Comprises the posterior bony elements (pedicles, laminae, spinous processes) and the associated ligamentous structures (ligamentum flavum, interspinous, supraspinous, and facet joint capsules).
A fracture is considered unstable if two or more columns are disrupted, particularly if the middle column is involved. This framework guides the assessment of fracture patterns and helps determine the need for surgical intervention. The AO Spine Classification System further refines this understanding, categorizing fractures based on morphology (Type A: compression, Type B: distraction, Type C: translational/rotational), neurological status, and specific modifiers, providing a more granular and prognostic tool for treatment algorithms.
Indications & Contraindications
The decision to pursue operative versus non-operative management for axial skeleton fractures is multifactorial, requiring careful consideration of fracture morphology, neurological status, spinal stability, patient comorbidities, and surgeon expertise. The overarching goal is to optimize neurological recovery, restore spinal alignment, and achieve long-term stability and pain control.
Indications for Operative Management:
Operative intervention is typically indicated in situations where conservative management is unlikely to achieve a stable, aligned spine, or where there is ongoing neurological compromise.
-
Neurological Deficit:
- Progressive neurological deficit: Any worsening of motor, sensory, or bowel/bladder function.
- Incomplete spinal cord injury (SCI): Including central cord syndrome, Brown-Séquard syndrome, anterior cord syndrome, or cauda equina syndrome with demonstrable cord/root compression.
- Persistent spinal cord or nerve root compression: Demonstrated radiographically despite attempted closed reduction or external immobilization.
-
Spinal Instability:
- Fracture patterns involving significant disruption of two or more columns (e.g., Denis Type B or C burst fractures, AO Spine Type B or C fractures).
- Ligamentous disruption leading to gross instability (e.g., facet dislocations, complete disruption of the posterior ligamentous complex).
- Translation of >3.5 mm or angulation >11° in the cervical spine.
- Severe kyphosis (>20-30° depending on spinal region) or sagittal plane deformity, especially if progressive.
- Irreducible dislocations.
-
Specific Fracture Types:
- Unstable odontoid fractures (Type II/III with significant displacement or nonunion risk).
- Unstable hangman's fractures (Type II/IIa/III).
- Traumatic spondylolisthesis with neurological deficit or instability.
- Chance fractures (AO Type B3) due to their inherent instability and often associated visceral injuries.
- Burst fractures with >50% canal compromise, even in neurologically intact patients, if progressive deformity or delayed neurological deficit is a concern.
- Fracture-dislocations at any level.
- Failed Non-Operative Treatment: Progressive pain, deformity, or neurological decline despite appropriate conservative measures.
Indications for Non-Operative Management:
Non-operative management is considered for stable fractures in neurologically intact patients, where the risk of secondary neurological compromise or progressive deformity is low.
-
Stable Fractures:
- Isolated transverse process fractures or spinous process fractures.
- Stable wedge compression fractures (e.g., AO Spine Type A0, A1, A2, or stable A3) with minimal kyphosis (<20-30°) and no posterior column involvement.
- Certain stable odontoid fractures (Type I, minimally displaced Type II) or Type I hangman's fractures.
- Minor stable ligamentous injuries without significant displacement.
- Neurologically Intact Patients: Patients with no signs of spinal cord or nerve root compromise.
- Minimal Deformity: Absence of significant kyphosis or scoliotic deformity that could progress or lead to pain.
- Patient Comorbidities: In patients with severe comorbidities precluding surgical intervention, non-operative management may be the only viable option, even for otherwise unstable fractures, with a focus on symptom management and supportive care.
Contraindications for Operative Management:
Absolute contraindications are rare and typically relate to the patient's physiological status, rather than the spinal injury itself.
- Severe Medical Comorbidities: Uncontrolled cardiac, pulmonary, renal, or metabolic conditions that render the patient an unacceptably high anesthetic or surgical risk.
- Active Systemic Infection or Sepsis: Surgery should be delayed until the infection is controlled.
- Patient Refusal: An informed patient's refusal of surgery.
- Complete Neurological Injury (Frankel A): While previously considered a contraindication, contemporary evidence supports early decompression and stabilization in these patients to facilitate nursing care, prevent pain, and potentially improve trunk balance, even if lower limb function is not expected to recover. However, the urgency and extent of intervention may be modified.
Table: Operative vs. Non-Operative Indications
| Feature | Operative Indications | Non-Operative Indications |
|---|---|---|
| Neurological Status | Progressive deficit, Incomplete SCI (any type), Cauda Equina Syndrome, Persistent compression | Neurologically intact |
| Spinal Stability | Unstable (≥2 column disruption, significant ligamentous disruption), Irreducible dislocation | Stable (isolated anterior column injury, intact PLL/posterior elements) |
| Deformity | Significant kyphosis (>20-30°), progressive angular deformity, >3.5mm translation | Mild kyphosis (<20-30°), no translation |
| Fracture Type (Examples) | Burst (severe), Fracture-dislocation, Unstable Odontoid (Type II/III), Unstable Hangman's (Type II/IIa/III), Chance, Type B/C AO Spine | Stable compression (Type A0-A3 AO Spine), Isolated process fractures, Stable Odontoid (Type I), Stable Hangman's (Type I) |
| Canal Compromise | Significant (>50% with neurologic deficit, or concern for delayed deficit) | Minimal (<50% and neurologically intact) |
| Treatment Response | Failure of non-operative treatment | Response to non-operative treatment |
| Overall Goal | Decompression, stabilization, fusion | Pain control, protection, natural healing |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning and appropriate patient positioning are paramount for optimizing surgical outcomes and minimizing complications in axial skeleton fracture management.
Pre-Operative Planning:
-
Comprehensive Patient Evaluation:
- Detailed History & Physical Exam: Ascertain injury mechanism, associated injuries (head trauma, chest/abdominal injury, long bone fractures), and pre-existing medical comorbidities. A thorough neurological examination, including ASIA Impairment Scale (AIS) assessment for SCI, is critical and should be serially documented.
- Review of Systems: Cardiopulmonary status, renal function, endocrine disorders (e.g., diabetes, osteoporosis), and medication history (anticoagulants, steroids).
-
Diagnostic Imaging:
- Plain Radiographs: AP, lateral, and open-mouth odontoid views (cervical spine); AP and lateral views (thoracolumbar spine). Essential for initial assessment of alignment, gross instability, and fracture morphology. Dynamic flexion-extension views may be considered for suspected ligamentous instability in stable, neurologically intact patients but should be performed with extreme caution.
- Computed Tomography (CT) Scan: The gold standard for bony detail. Provides high-resolution axial, sagittal, and coronal reconstructions, identifying fracture lines, comminution, canal compromise, and facet involvement. 3D reconstructions are invaluable for understanding complex fracture patterns and planning instrumentation trajectories.
- Magnetic Resonance Imaging (MRI): Essential for evaluating soft tissue structures, including the spinal cord, nerve roots, ligaments (ALL, PLL, ligamentum flavum), intervertebral discs, and presence of epidural hematoma or edema. It is indispensable for assessing the extent of SCI, identifying disc herniation, and determining the integrity of the posterior ligamentous complex.
-
Surgical Approach Selection:
- The choice of approach (anterior, posterior, or combined) depends on the fracture location, pathology (compression, distraction, translation), need for decompression (direct vs. indirect), and the goal of stabilization (short vs. long segment).
- Anterior: Often preferred for direct decompression of the ventral spinal cord and reconstruction of the anterior column (e.g., cervical corpectomy, anterior thoracolumbar reconstruction).
- Posterior: Provides excellent access for posterior element decompression (laminectomy, facetectomy), reduction of kyphosis or translation, and posterior instrumentation (pedicle screws, lateral mass screws). It is the workhorse for most thoracolumbar fracture stabilizations.
- Combined: Utilized for severe, highly unstable fractures or complex deformities requiring both ventral and dorsal stabilization and decompression.
-
Implant Selection:
- Determine the appropriate implants (e.g., pedicle screws, lateral mass screws, hooks, rods, plates, interbody cages, bone graft options).
- Consider construct length, stiffness, and fixation points. For unstable fractures, instrumentation should typically span at least two levels above and two levels below the injured segment.
-
Intraoperative Neuromonitoring (IONM):
- Somatosensory Evoked Potentials (SSEPs) and Motor Evoked Potentials (MEPs) are highly recommended, particularly for surgeries involving potential spinal cord compromise. Electromyography (EMG) is useful for nerve root monitoring. A baseline must be established pre-incision.
-
Blood Management:
- Anticipate potential blood loss. Type and cross-match blood, consider antifibrinolytic agents (e.g., tranexamic acid), and cell salvage.
-
Surgical Team & Equipment:
- Ensure all necessary instruments, C-arm fluoroscopy, microscope (if applicable), and specialty equipment are available and functioning. Briefing the surgical team is crucial.
Patient Positioning:
Correct patient positioning is critical to optimize surgical exposure, maintain spinal alignment, facilitate intraoperative imaging, and prevent iatrogenic injury.
-
General Principles:
- Maintain spinal precautions throughout transfer and positioning, often involving log-rolling with assistants to prevent inadvertent movement of unstable segments.
- Adequate padding of all pressure points (heels, knees, elbows, shoulders, face) to prevent nerve palsies and skin breakdown.
- Ensure proper ventilation and access for anesthesiology.
- Consider the implications of positioning on hemodynamics.
-
Cervical Spine:
- Anterior Approach (ACDF/Corpectomy): Supine position. Head often secured in a donut or gel pad, or with gentle traction (e.g., Gardner-Wells tongs if appropriate and stable) to extend the neck, facilitating access. Shoulders are pulled caudally with tape to improve fluoroscopic visualization of C7-T1.
- Posterior Approach (PCF): Prone position. Head secured in a Mayfield skull clamp or similar three-point fixation device. The torso is elevated to allow for natural lordosis and minimize venous congestion around the spine. Ensure the eyes are free from pressure.
-
Thoracic and Lumbar Spine:
- Posterior Approach (Pedicle Screw Fixation): Prone position. Typically positioned on a specialized spinal frame (e.g., Jackson table, Wilson frame) to allow the abdomen to hang freely, minimizing intra-abdominal pressure and epidural venous bleeding. Chest rolls or supports under the sternum and iliac crests are also used for less specialized tables. This also maintains lumbar lordosis. The arms are gently abducted and flexed.
- Anterior Thoracolumbar Approach (Thoracotomy/Retroperitoneal): Lateral decubitus position for access to the lateral and anterior aspects of the vertebral body. The ipsilateral arm is often suspended. Bean bags or specialized frames help maintain position.
Detailed Surgical Approach / Technique
Surgical intervention for axial skeleton fractures typically involves a combination of decompression, reduction, and stabilization with fusion. The specific technique varies significantly based on the spinal segment, fracture morphology, and neurological status.
General Principles of Spinal Trauma Surgery:
- Decompression: Relieving pressure on neural elements (spinal cord or nerve roots). This can be direct (e.g., laminectomy, corpectomy, fragmentectomy) or indirect (e.g., ligamentotaxis, distraction across disc spaces).
- Reduction: Restoring anatomical alignment of the fractured vertebrae. This may involve distraction, compression, translation, or cantilever maneuvers using instrumentation.
- Stabilization & Fusion: Providing immediate stability with instrumentation (screws, rods, plates) and promoting long-term biological fusion (arthrodesis) with bone graft.
Exemplary Surgical Approaches:
1. Posterior Thoracolumbar Pedicle Screw Fixation
This is the most common approach for unstable thoracolumbar fractures (e.g., burst fractures, fracture-dislocations).
- Positioning: Prone on a spinal frame (e.g., Jackson table) to allow free abdomen.
- Incision: Midline skin incision centered over the fractured segment, extending typically 2 levels above and 2 levels below the planned fusion.
- Muscle Dissection: Subperiosteal dissection of paraspinal musculature from the spinous processes, laminae, and facet capsules, extending laterally to the tips of the transverse processes. This exposes the posterior bony elements. Maintain musculature as much as possible, as extensive dissection can contribute to post-operative pain and atrophy.
- Level Identification: Crucial step using intraoperative fluoroscopy or navigation.
-
Pedicle Screw Insertion:
- Entry Point: Typically at the junction of the transverse process, superior articular process, and pars interarticularis. Specific entry points vary by vertebral level and surgeon preference.
- Awl/Drill: Create a pilot hole into the pedicle.
- Palpation: A pedicle probe is advanced, and all five walls of the pedicle tract are palpated to confirm containment within the cortical bone, minimizing the risk of canal or vascular violation.
- Tapping: The pedicle tract is tapped, if necessary, to create threads for the screw.
- Screw Insertion: Pedicle screws of appropriate diameter and length are inserted. Bicortical purchase into the anterior vertebral body cortex is often desired for maximal pullout strength, especially in osteoporotic bone, but care must be taken to avoid anterior vascular structures.
-
Decompression (if needed):
- For neural element compression (e.g., retropulsed bone fragments in burst fractures), indirect decompression via ligamentotaxis (distraction across the anterior column) may be sufficient.
- Direct decompression via laminectomy, facetectomy, or even costotransversectomy/transpedicular approaches to remove retropulsed fragments may be necessary, especially for severe canal compromise or persistent neurological deficits.
-
Reduction & Rod Placement:
- Contoured rods are placed into the screw heads.
- Reduction maneuvers (e.g., distraction across the injured segment, cantilever technique to restore kyphosis, sagittal correction) are performed using specialized tools (e.g., persuaders, compressors, distractors) to restore sagittal and coronal alignment.
- Final tightening of all set screws.
- Bone Grafting: Decortication of posterior elements (laminae, facets, transverse processes) is performed, and bone graft (autograft, allograft, or synthetic) is applied to promote arthrodesis.
- Closure: Layered closure of paraspinal fascia, subcutaneous tissue, and skin. Drain placement may be considered.
2. Anterior Cervical Discectomy and Fusion (ACDF)
Common for acute disc herniation with myelopathy/radiculopathy or stable cervical fractures involving the anterior column.
- Positioning: Supine, neck slightly extended, shoulders taped caudally.
- Incision: Transverse skin incision in a skin crease (e.g., C5/C6) or longitudinal incision along the anterior border of the sternocleidomastoid muscle (SCM).
-
Dissection:
- Subplatysmal dissection to the midline.
- Identification of the interval between the medial border of the SCM/carotid sheath (laterally) and the trachea/esophagus (medially).
- Retraction of these structures. Careful identification and preservation of the recurrent laryngeal nerve (right side is more variable).
- Identification of the prevertebral fascia, which is incised longitudinally.
- Level Identification: Fluoroscopy confirms the correct vertebral level.
- Discectomy: Incision of the annulus fibrosus, removal of the intervertebral disc and cartilaginous endplates.
- Decompression: Visualization and decompression of the spinal cord and nerve roots by removing osteophytes and posterior annulus.
- Graft/Cage Insertion: An appropriately sized interbody graft (autograft, allograft, PEEK cage) is impacted into the disc space.
- Plate Fixation: An anterior cervical plate is secured with bicortical screws into the vertebral bodies above and below the fused segment, providing immediate stability.
- Closure: Layered closure, ensuring hemostasis.
3. Posterior Cervical Fixation
Indicated for unstable posterior column injuries, ligamentous instability, or failed anterior approaches.
- Positioning: Prone, head in Mayfield skull clamp.
- Incision: Midline skin incision.
- Dissection: Subperiosteal dissection of paraspinal muscles from the posterior elements.
- Level Identification: Fluoroscopy confirms levels.
-
Instrumentation:
- Lateral Mass Screws (C3-C6): Entry points typically 2-3mm medial to the lateral mass midpoint, angled 30° laterally and 30° superiorly to avoid vertebral artery and nerve root.
- Pedicle Screws (C7, T1, C2): C7 and T1 pedicles are larger and often tolerate screws. C2 pedicle screws are also an option. Careful trajectory is critical due to proximity to the vertebral artery.
- Laminoforaminotomy/Laminectomy: Performed as needed for neural decompression.
- Rod Placement & Fusion: Rods are contoured and secured to the screws. Decortication of the posterior elements and application of bone graft promote fusion.
- Closure: Layered closure.
Complications & Management
Spinal surgery, particularly for trauma, carries inherent risks. A thorough understanding of potential complications, their incidence, and strategies for prevention and management is critical for patient safety and optimal outcomes.
General Complications:
-
Infection:
- Incidence: Superficial infections 1-5%, deep infections 0.5-2%. Higher in open fractures, prolonged surgeries, or immunocompromised patients.
- Management: Prophylactic antibiotics, meticulous sterile technique. Superficial infections may respond to wound care and oral antibiotics. Deep infections often require surgical debridement, intravenous antibiotics, and potentially hardware removal (usually after fusion is complete, if possible).
-
Hemorrhage:
- Incidence: Variable, depends on approach and complexity. Can be significant in trauma.
- Management: Careful hemostasis, tranexamic acid, cell salvage, blood product transfusion. Pre-operative optimization of coagulation. Post-operative drains may be used.
-
Neurological Injury:
- Incidence: New or worsening deficit 0.5-5%. Can be due to direct surgical trauma, ischemia, distraction injury, or malposition of hardware.
- Management: Meticulous surgical technique, IONM. Immediate re-exploration and hardware removal/repositioning for new post-operative deficit. Steroids (though controversial, may be considered in acute injury).
-
Dural Tear/CSF Leak:
- Incidence: 5-15%, higher in revision surgery or complex decompressions.
- Management: Primary repair (suture), patch (muscle, fascia, dural substitute), fibrin sealant. Lumbar drain for 3-5 days may be used to reduce CSF pressure and aid healing. Post-operative strict bed rest.
-
Hardware Failure:
- Incidence: 1-10%, including screw pullout, rod fracture, or plate breakage. Higher with poor bone quality, pseudarthrosis, or inadequate construct.
- Management: Revision surgery, longer construct, additional fixation points, different screw types (e.g., expandable, cemented), osteotomy for sagittal balance correction, and robust bone grafting.
-
Pseudarthrosis (Non-Union):
- Incidence: 5-20%, higher in smokers, multi-level fusions, or patients with comorbidities affecting bone healing.
- Management: Non-operative (external bracing, electrical stimulation) for asymptomatic or mildly symptomatic non-unions. Symptomatic non-unions require revision surgery with re-grafting, stronger or extended instrumentation, and potentially biological adjuncts (e.g., BMP).
-
Adjacent Segment Disease (ASD):
- Incidence: 2-4% per year after fusion, manifesting as accelerated degeneration at levels adjacent to the fused segment.
- Management: Initial conservative management for symptomatic ASD. If symptoms persist or neurological compromise develops, further surgical intervention (decompression, fusion, or arthroplasty if indicated) may be required.
Specific Complications by Approach/Region:
-
Anterior Cervical:
- Dysphagia: 20-50% early post-op, usually transient. Persistent dysphagia <5%.
- Hoarseness/Recurrent Laryngeal Nerve Palsy: 1-5%, often transient.
- Vertebral Artery Injury: Rare, but catastrophic. Meticulous dissection and careful screw placement.
- Esophageal Perforation: Very rare, high morbidity.
-
Posterior Cervical:
- Vertebral Artery Injury: Risk with C1/C2 instrumentation or aberrant anatomy.
- C5 Palsy: Transient radiculopathy in 5-10% after posterior cervical decompression/fusion, likely due to cord shift or nerve root irritation.
-
Thoracolumbar:
- Great Vessel Injury (Aorta, Vena Cava): Rare with posterior pedicle screw fixation if proper technique is followed; higher risk with anterior approaches.
- Visceral Injury: Puncturing pleura, bowel, or ureter with anterior approaches.
- Pneumothorax/Hemothorax: Anterior transthoracic approaches.
- Retroperitoneal Hematoma: Anterior retroperitoneal approaches.
Table: Common Complications and Salvage Strategies
| Complication | Incidence | Salvage Strategies |
|---|---|---|
| New Neurological Deficit | 0.5-5% | Immediate re-exploration, hardware removal/repositioning, decompression, steroids (controversial). |
| Dural Tear / CSF Leak | 5-15% | Primary repair, patch (muscle, fascia, dural substitute), fibrin sealant, lumbar drain, strict bed rest. |
| Surgical Site Infection (Deep) | 0.5-2% | Surgical debridement, intravenous antibiotics, irrigation, consideration of hardware removal after fusion. |
| Hardware Failure | 1-10% | Revision surgery, longer construct, additional fixation points, osteotomy, robust bone grafting. Address underlying pseudarthrosis. |
| Pseudarthrosis (Non-Union) | 5-20% | Revision surgery with re-grafting (autograft preferred), extended/stronger instrumentation, biological adjuncts (e.g., BMP), electrical stimulation. |
| Dysphagia (Anterior Cervical) | 20-50% (transient) | Observation, soft diet, speech therapy. Consider corticosteroids for severe/prolonged cases. |
| Recurrent Laryngeal Nerve Palsy | 1-5% (transient) | Observation, speech therapy. Electromyography for persistent cases. |
| Vertebral Artery Injury | Rare, but severe | Direct surgical repair by vascular surgeon, embolization (if not amenable to repair), direct pressure, meticulous technique during drilling/screwing. |
| C5 Palsy (Posterior Cervical) | 5-10% (transient) | Observation, physical therapy. May indicate need for additional decompression or smaller laminoplasty opening to prevent cord shift in some cases. |
| Adjacent Segment Disease | 2-4% / year | Initial conservative management. Surgical intervention (decompression, fusion, or arthroplasty) for persistent symptoms or neurological compromise. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is an integral component of comprehensive spinal fracture management, aiming to restore function, prevent complications, and facilitate the patient's return to activities of daily living. Protocols are tailored to the individual patient, fracture stability, surgical construct, and neurological status.
Early Post-Operative Phase (Days 0-7):
- Pain Management: Multimodal analgesia (opioids, NSAIDs, acetaminophen, muscle relaxants) to control acute pain, facilitate mobilization, and minimize chronic pain development.
- Wound Care: Daily assessment of incision for signs of infection (erythema, discharge, dehiscence). Dressing changes as per institutional protocol.
- Neurological Monitoring: Frequent neurological checks to detect any new or worsening deficits, particularly in the first 24-48 hours.
- Deep Vein Thrombosis (DVT) Prophylaxis: Pharmacological (low molecular weight heparin, fondaparinux) and mechanical (sequential compression devices) prophylaxis, especially critical in SCI patients.
-
Mobilization:
- Bracing: Depending on surgical stability and fracture characteristics, a rigid orthosis (e.g., TLSO for thoracolumbar, rigid cervical collar for cervical) may be prescribed for ambulation and out-of-bed activities. The duration and type of bracing are surgeon-dependent.
- Early Ambulation: As tolerated, usually within 24-48 hours post-op, to prevent deconditioning, reduce DVT risk, and improve bowel function.
- Physical Therapy (PT) & Occupational Therapy (OT): Initial focus on bed mobility, transfers, proper body mechanics, and instruction on brace donning/doffing. For SCI patients, early initiation of comprehensive rehabilitation is critical.
- Activity Restrictions: Strict limitations on lifting, twisting, and bending ("BLT" precautions) to protect the fusion and hardware.
Subacute Phase (Weeks 2-12):
- Progressive Mobilization: Increase duration and intensity of walking.
- Spinal Hygiene: Education on ergonomic principles, proper posture, and avoidance of provocative movements.
-
Targeted PT/OT:
- Core Strengthening: Focus on stabilizing the trunk musculature (abdominal obliques, transversus abdominis, multifidus) without stressing the fusion site.
- Posture Correction: Exercises to improve alignment and reduce compensatory strain.
- Range of Motion (ROM): Gentle, pain-free ROM exercises for adjacent, unfused segments, and peripheral joints (hips, shoulders) to prevent stiffness. Avoidance of direct motion through the fused segment.
- Functional Training: Gradual progression towards functional activities, incorporating spinal precautions.
- Brace Management: Gradual weaning from orthosis as clinical healing and radiographic evidence of fusion progress, typically between 6-12 weeks, depending on the case.
Chronic Phase (Months 3-12+):
- Advanced Strengthening: Progressive resistance exercises targeting global strength, endurance, and balance.
- Return to Activity: Gradual return to work, recreational activities, and low-impact sports. High-impact sports are typically restricted until complete fusion and clearance by the surgeon.
- Radiographic Follow-up: Serial radiographs at 3, 6, 12 months, and sometimes 2 years to assess fusion progression and hardware integrity. CT scans may be used to confirm fusion, especially if symptomatic non-union is suspected.
- Management of Residual Pain/Deficit: Address any persistent neuropathic pain, muscle weakness, or functional limitations with targeted therapies (e.g., nerve blocks, medication adjustments, assistive devices).
- Patient Education: Emphasis on lifelong spinal health, weight management, and smoking cessation (critical for fusion success).
For patients with SCI, rehabilitation is an intensive, multidisciplinary process that extends well beyond typical orthopedic protocols, focusing on maximizing functional independence, adapting to disability, and managing secondary complications (e.g., spasticity, neurogenic bladder/bowel, pressure sores). Early referral to a specialized SCI rehabilitation center is paramount.
Summary of Key Literature / Guidelines
The management of axial skeleton fractures has evolved significantly with advances in imaging, surgical techniques, and a deepening understanding of spinal biomechanics. Several classification systems and evidence-based guidelines inform current practice.
Classification Systems:
- Denis Three-Column Theory (1983): As detailed previously, this foundational model categorizes the spine into anterior, middle, and posterior columns. Disruption of two or more columns, particularly the middle column, signifies instability and often indicates a need for surgical intervention. While superseded by more comprehensive systems for fracture morphology, its conceptual framework for stability remains highly relevant.
-
AO Spine Classification System:
This is the most widely adopted and robust classification system, replacing older systems for morphological characterization.
- Type A (Compression): Axial compression forces, affecting the anterior and/or middle columns without significant posterior ligamentous complex (PLC) injury. Further subdivided (A0-A4) based on involvement of endplates and posterior wall.
- Type B (Distraction): Tension forces causing failure of either the anterior (B1), posterior (B2), or combined anterior and posterior columns (B3) via distraction. These are inherently unstable due to PLC disruption.
- Type C (Translation/Rotation): Multiplanar forces leading to translational displacement of one vertebral body relative to another. These represent the most unstable injuries.
- The AO Spine system also incorporates neurological status (N0-N4), facet injury (F0-F4), and specific patient modifiers (M1-M4), creating a comprehensive tool for prognostication and treatment planning. This system is heavily endorsed by international spine societies for its reliability and validity.
Key Literature & Guidelines:
-
Timing of Decompression in Spinal Cord Injury (SCI):
- Historically debated, but a growing body of evidence, including the STASCIS trial (Surgical Timing in Acute Spinal Cord Injury Study) , supports early surgical decompression (within 24-72 hours) for acute traumatic SCI, particularly for incomplete injuries. While direct evidence of improved neurological outcome remains controversial for complete SCI, early decompression facilitates rehabilitation, reduces pain, and improves the overall hospital course.
- Guidelines from AO Spine and NASS generally advocate for early surgical stabilization and decompression in patients with acute SCI and demonstrable neural compression, provided the patient is medically stable.
-
Role of Methylprednisolone:
- The use of high-dose intravenous methylprednisolone for acute SCI remains highly controversial. Based on the NASCIS (National Acute Spinal Cord Injury Study) trials , there was a narrow therapeutic window for administration, but subsequent meta-analyses and systematic reviews have failed to consistently demonstrate significant clinical benefit and have highlighted potential adverse effects (e.g., gastrointestinal hemorrhage, sepsis).
- Most major spine societies (e.g., NASS, AOSNA, Congress of Neurological Surgeons) no longer routinely recommend high-dose methylprednisolone for acute SCI due to a lack of clear evidence of benefit outweighing risks.
-
Anterior vs. Posterior Approaches:
- The choice of approach often depends on the predominant pathology. Anterior approaches are superior for direct ventral decompression and reconstruction of the anterior column, particularly in the cervical spine. Posterior approaches offer robust stabilization for posterior and middle column injuries and are excellent for kyphosis correction.
- For thoracolumbar burst fractures with significant canal compromise, the optimal approach (anterior vs. posterior vs. combined) is still debated. Modern posterior-only instrumentation with indirect decompression (ligamentotaxis and distraction) has shown comparable outcomes to combined anterior/posterior approaches for many burst fractures, with reduced morbidity. However, anterior reconstruction may be preferred for specific pathologies or for severely comminuted anterior column defects requiring structural support.
-
Minimally Invasive Spine Surgery (MISS) for Trauma:
- Increasingly utilized for select stable or semi-stable fractures. MISS techniques (e.g., percutaneous pedicle screw fixation) offer advantages of reduced muscle dissection, blood loss, and shorter hospital stays.
- While effective for achieving stabilization, MISS may have limitations in achieving direct decompression or aggressive deformity correction in highly unstable or complex fracture patterns. The learning curve is significant.
-
Bone Graft Options:
- Autograft (iliac crest): Remains the gold standard due to its osteoconductive, osteoinductive, and osteogenic properties. However, donor site morbidity can be significant.
- Allograft and synthetic bone graft substitutes: Increasingly used, especially with improved biologics, reducing donor site complications. The choice is often dictated by the specific needs of the fusion and patient factors.
These insights, grounded in evidence and clinical experience, underscore the complexity and evolving nature of axial skeleton fracture management. Continued adherence to sound surgical principles, meticulous technique, and critical appraisal of emerging literature are essential for optimizing patient outcomes.
You Might Also Like