Anterior Cervical Spine Surgery: Comprehensive Guide to Deep Cervical Fascia & Surgical Anatomy
Key Takeaway
Understanding the deep cervical fascia is paramount in anterior cervical spine surgery (ACDF, ACCF) to minimize injury and optimize outcomes. It comprises the investing, pretracheal, and prevertebral layers. Each layer dictates specific surgical corridors and relationships with vital neurovascular structures. Mastery of this intricate anatomy ensures precise, safe decompression and stabilization.
Mastering the Deep Cervical Fascia for Anterior Cervical Spine Surgery
Introduction & Epidemiology
The anterior cervical approach to the spine represents a foundational pillar in the surgical management of cervical spine pathology. Since its popularization in the 1950s and 60s by Cloward, Smith, and Robinson, anterior cervical discectomy and fusion (ACDF) and anterior cervical corpectomy and fusion (ACCF) have become standard interventions for a myriad of conditions. These procedures effectively address nerve root and spinal cord compression caused by degenerative disc disease, osteophyte formation, disc herniation, trauma, infection, and neoplastic processes. The prevalence of cervical spondylotic myelopathy (CSM) is estimated at 1.6 cases per 100,000 annually, while cervical radiculopathy affects approximately 83 per 100,000, underscoring the significant clinical burden necessitating surgical expertise.
The anterior approach offers direct access to the ventral pathology, facilitating comprehensive decompression and robust stabilization. However, the cervical region is anatomically complex, densely packed with vital neurovascular structures, visceral organs, and a layered fascial system. A meticulous understanding of these anatomical relationships, particularly the deep cervical fascia, is paramount for minimizing iatrogenic injury, optimizing surgical outcomes, and ensuring patient safety. Mastery of this approach transcends mere technical skill; it demands a profound appreciation for the three-dimensional anatomy and the surgical implications of each fascial plane.
Surgical Anatomy & Biomechanics
The anterior cervical spine is approached through a carefully orchestrated dissection that respects the intricate layering of the neck's soft tissues. The foundational understanding of this pathway begins with the three principal fascial layers of the neck, which compartmentalize structures and dictate surgical corridors.
Investing Layer of Deep Cervical Fascia
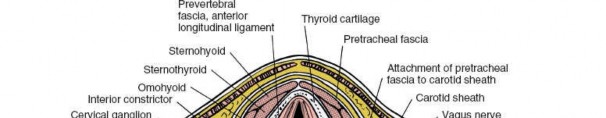
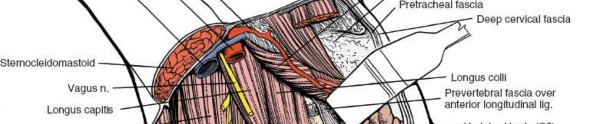
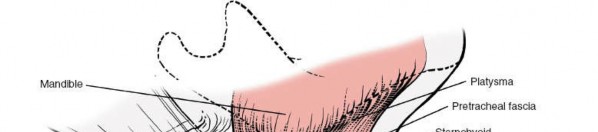
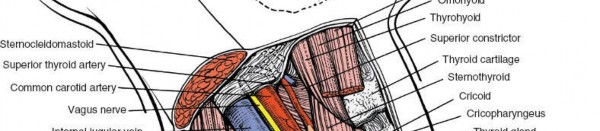
The most superficial of the deep cervical fascial layers is the investing layer . This robust fibrous sheath encircles the entire neck, akin to a collar. It originates dorsally from the ligamentum nuchae and the spinous process of C7, enveloping the trapezius and sternocleidomastoid (SCM) muscles before fusing anteriorly. Laterally, it splits to encapsulate both the SCM and trapezius, forming a protective sleeve around these large muscles. Superiorly, it attaches to the external occipital protuberance, superior nuchal line, mastoid process, zygomatic arch, and the body of the mandible. Inferiorly, it extends to the manubrium, clavicles, and acromion processes.
The clinical significance of the investing fascia lies in its role as the initial deep barrier encountered after skin and platysma. Its incision along the anterior border of the SCM is a critical step, enabling the muscle to be retracted laterally, thus exposing the underlying strap muscles. Structures superficial to the investing fascia are limited primarily to the platysma muscle, a thin, broad sheet of muscle representing a remnant of the panniculus carnosus, and the external jugular vein (EJV). The EJV, if encountered and deemed obstructive, can generally be safely ligated and divided without significant sequelae due to extensive collateral venous drainage. The greater auricular nerve may also course superficially to the SCM.

Pretracheal Fascia
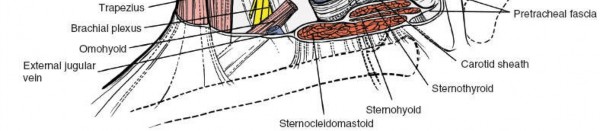
Deep to the investing layer lies the pretracheal fascia . This intermediate fascial layer extends from the hyoid bone superiorly, investing the infrahyoid "strap" muscles (sternohyoid, sternothyroid, omohyoid, and thyrohyoid) and descending into the thorax, where it blends with the fibrous pericardium. The pretracheal fascia's primary function is to provide a smooth gliding surface for these muscles during swallowing and neck movements, and to enclose the thyroid gland, trachea, and esophagus.
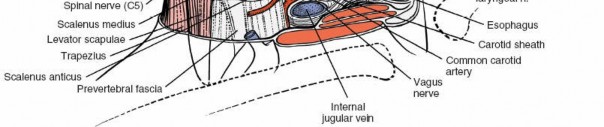
A key relationship of the pretracheal fascia is with the carotid sheath. While technically a distinct fascial tube, the pretracheal fascia is continuous laterally with the carotid sheath, which encases the common carotid artery, internal jugular vein (IJV), and the vagus nerve (CN X). This intimate relationship necessitates careful anterior-posterior retraction to avoid undue tension or injury to these vital neurovascular structures. The anterior jugular veins also run superficially to the strap muscles within or immediately deep to the investing fascia, and often require ligation.
Prevertebral Fascia
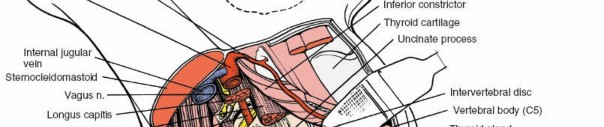
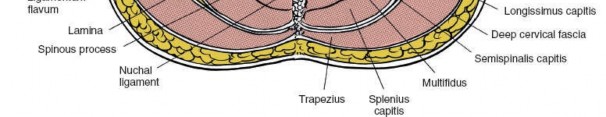
The deepest and surgically most critical of the cervical fascial layers for anterior spine access is the prevertebral fascia . This robust layer originates from the anterior longitudinal ligament and covers the prevertebral muscles (longus colli and longus capitis) and the scalene muscles. It extends from the base of the skull inferiorly to the T3 vertebral level, where it merges with the endothoracic fascia. The prevertebral fascia forms the posterior boundary of the retropharyngeal space, which is a key anatomical corridor for safe blunt dissection to the spine.
The prevertebral fascia is composed of two distinct layers:
1.
Alar fascia:
This anterior layer covers the longus colli muscles and extends laterally to form the floor of the retropharyngeal space.
2.
Prevertebral layer (true prevertebral fascia):
This posterior layer directly covers the vertebral bodies and the longus colli muscles, providing a robust, yet identifiable, plane for subperiosteal dissection.
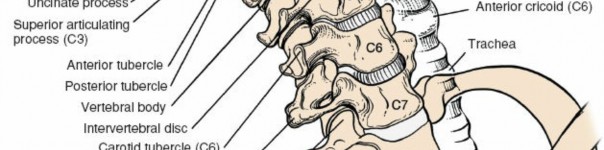
Deep to the prevertebral fascia, anterior to the vertebral bodies, are the longus colli muscles . These paired muscles originate from the anterior tubercles of the transverse processes and anterior surfaces of vertebral bodies, inserting on more superior vertebral bodies and transverse processes. Their medial borders are critical landmarks for identifying the midline of the cervical spine. The sympathetic chain typically lies on the prevertebral fascia, medial to the longus colli muscles at the level of the C6 transverse process, making it vulnerable to injury with excessively lateral dissection. The phrenic nerve (C3-C5) descends across the anterior surface of the anterior scalene muscle, beneath the prevertebral fascia, again emphasizing the need for medial, precise dissection.
The recurrent laryngeal nerves (RLNs) are also of paramount importance. The right RLN typically loops around the subclavian artery, ascending more obliquely. The left RLN loops around the aortic arch, ascending more vertically in the tracheoesophageal groove. Both nerves course medial to the carotid sheath and posterior to the thyroid gland and trachea, making them susceptible to injury during medial retraction of the trachea and esophagus. The superior laryngeal nerve, which innervates the cricothyroid muscle, is less commonly injured but can contribute to dysphonia if affected.
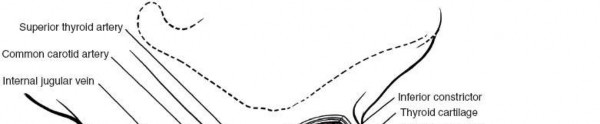
Vascular Anatomy: Beyond the carotid artery and IJV within the carotid sheath, knowledge of the superior and inferior thyroid arteries is important. These vessels may be encountered during dissection around the thyroid gland, if it requires mobilization for adequate exposure. The vertebral arteries ascend through the foramina transversaria of C6-C1, making them vulnerable during extensive lateral decompression or corpectomy, particularly at the C7-T1 level or with anatomical variations.
Biomechanics: The anterior column of the cervical spine bears approximately 80% of the axial load. Procedures like ACDF aim to restore disc height, decompress neural elements, and promote fusion, thereby stabilizing the motion segment and preventing further progression of deformity or neurological deficit. The choice of graft (autograft, allograft, PEEK cage) and instrumentation (plate and screws) directly impacts the biomechanical stability and fusion rates, with anterior plating significantly increasing construct rigidity and reducing graft subsidence.
Indications & Contraindications
The decision for anterior cervical spine surgery is multifactorial, balancing the severity of neurological deficit, extent of anatomical pathology, patient comorbidities, and response to conservative management.
Indications
-
Cervical Radiculopathy:
- Failure of conservative management (physical therapy, NSAIDs, epidural steroids) for 6-12 weeks.
- Progressive neurological deficit (e.g., motor weakness, profound sensory loss).
- Unremitting pain refractory to non-operative treatment.
- Imaging (MRI) demonstrating nerve root compression (disc herniation, foraminal stenosis).
-
Cervical Myelopathy:
- Diagnosis based on clinical signs (gait disturbance, spasticity, hyperreflexia, hand clumsiness) and symptoms (paresthesias, motor deficits).
- Imaging (MRI) demonstrating spinal cord compression with T2 signal change (myelomalacia) or significant canal stenosis.
- Progressive myelopathy is a strong indication for surgical intervention to prevent further deterioration.
- Even stable or mild myelopathy may warrant surgery to mitigate future progression, given the poor natural history without intervention.
-
Cervical Trauma:
- Unstable cervical spine fractures or fracture-dislocations.
- Traumatic disc herniations causing neurological compromise.
- Ligamentous injuries leading to instability.
- Often combined with posterior fixation for severe instability.
-
Spinal Deformity:
- Fixed cervical kyphosis or sagittal imbalance requiring anterior column support and correction.
-
Spinal Tumor:
- Primary or metastatic tumors causing spinal cord compression or instability.
- Need for tumor resection and reconstruction.
-
Spinal Infection (Osteomyelitis/Discitis):
- Abscess formation with neurological compromise.
- Progressive infection refractory to antibiotics.
- Debridement and stabilization.
-
Pseudarthrosis/Nonunion:
- Symptomatic failure of previous cervical fusion requiring revision.
Contraindications
-
Absolute Contraindications:
- Active systemic infection or local wound infection.
- Severe, uncorrectable coagulopathy.
- Critical medical comorbidities precluding safe anesthesia and surgery.
- Severe diffuse idiopathic skeletal hyperostosis (DISH) with ossified anterior longitudinal ligament (OALL) that obscures the surgical approach and increases risk of esophageal injury.
-
Relative Contraindications:
- Extensive scarring from previous anterior cervical surgery, particularly in the presence of dysphagia or dysphonia.
- Severe tracheal or esophageal pathology.
- Severe osteopenia, potentially compromising implant fixation and fusion rates.
- Vertebral artery anomaly or critical proximity to the surgical field, identified on pre-operative imaging.
- Uncontrolled diabetes, active smoking, or other factors known to impair fusion.
| Indication Type | Operative Indications | Non-Operative Management (Trial Before Surgery) |
|---|---|---|
| Radiculopathy | Unremitting pain > 6-12 weeks refractory to conservative Rx | NSAIDs, physical therapy, cervical collar, epidural steroid injections, oral steroids |
| Progressive motor weakness or neurological deficit | ||
| Radiographic evidence of nerve root compression | ||
| Myelopathy | Clinical signs of myelopathy (spasticity, gait disturbance, hyperreflexia) | Close observation for mild, non-progressive cases, physical therapy for symptoms |
| Radiographic evidence of spinal cord compression | ||
| Progressive neurological deterioration | ||
| Trauma | Unstable fracture/dislocation | Stable, non-displaced fractures without neurological deficit |
| Traumatic disc herniation with neurological deficit | ||
| Deformity | Progressive kyphosis or sagittal imbalance | Minor postural correction, physical therapy |
| Tumor | Spinal cord compression, instability, uncontrolled pain | Adjuvant chemotherapy/radiation for radiosensitive tumors, watchful waiting for benign lesions |
| Infection | Abscess, neurological deficit, uncontrolled systemic infection | Targeted antibiotic therapy for early, uncomplicated infections |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is the cornerstone of a safe and successful anterior cervical spine operation.
Pre-Operative Planning
-
Imaging Review:
- Plain Radiographs: Anteroposterior, lateral, flexion/extension views to assess alignment, instability, disc space height, and osteophyte formation.
- MRI: Essential for evaluating neural element compression (disc herniation, osteophytes, ligamentum flavum hypertrophy), spinal cord signal changes (myelomalacia), and soft tissue pathology.
- CT Scan: Provides detailed bony anatomy, vertebral artery course (especially if corpectomy is planned or anatomical variations are suspected), fusion assessment, and for identifying uncinate hypertrophy. CT angiography (CTA) or magnetic resonance angiography (MRA) may be necessary for complex cases or high-risk vertebral artery anomalies.
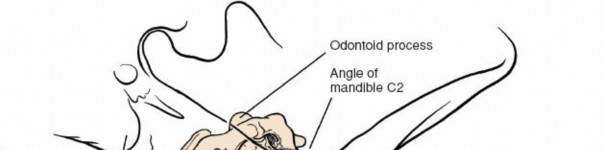
- Level Identification: Accurately identifying the target level pre-operatively and intra-operatively is paramount. Count vertebrae from C2 (dens) or C7 (vertebra prominens).
- Surgical Strategy: Determine the number of levels, type of decompression (discectomy vs. corpectomy), graft material, and fixation construct (plate design, screw type).
- Patient Optimization: Address comorbidities (e.g., diabetes, smoking cessation), optimize nutritional status, and manage anticoagulation.
- Informed Consent: Detailed discussion of risks, benefits, alternatives, and potential complications.
Patient Positioning
- Anesthesia Induction: Administer general endotracheal anesthesia. Neuromonitoring (SSEP, MEP) may be indicated.
- Supine Position: Patient is placed supine on the operating table.
-
Head and Neck Positioning:
- The head is typically placed in a neutral position or very slight extension using a donut or foam headrest. Excessive extension can stretch the spinal cord, especially in myelopathic patients, and complicate exposure by moving key anatomical landmarks.
- Slight rotation (e.g., to the right for a left-sided approach) can assist in retracting the SCM and neurovascular bundle, but must be minimal to avoid kinking the carotid artery or vertebral artery.
- Shoulder Roll: A bolster or rolled blanket is placed transversely under the shoulders to allow for gentle extension of the neck and to drop the shoulders inferiorly, thereby increasing access to the lower cervical segments (C6-T1).
- Shoulder Retraction: Broad pieces of tape are applied to the shoulders and pulled caudally towards the feet, secured to the operating table, further depressing the shoulders. This is crucial for optimal fluoroscopic visualization of the cervicothoracic junction.
- Surgical Site Preparation: The neck and upper chest are prepped and draped in a sterile fashion.
- Fluoroscopy Setup: The C-arm is positioned to allow for immediate anteroposterior and lateral radiographic confirmation of the surgical level throughout the procedure. Ensure the C-arm can move freely without obstruction.
Detailed Surgical Approach / Technique
The anterior cervical approach is a staged dissection, meticulously navigating through fascial planes to reach the prevertebral space.
1. Incision and Initial Dissection
-
Skin Incision:
- Transverse (Horizontal) Incision: Typically preferred for cosmetic reasons, placed within a skin crease (Langer's lines). The ideal location depends on the target level(s). For a single C5-C6 discectomy, an incision at this level is appropriate. For multilevel procedures or access to C3/C4 or C7/T1, a longer incision or a slightly oblique variant may be required.
- Longitudinal (Vertical) Incision: Less common, generally reserved for revision surgeries with extensive scarring or complex multilevel pathology, allowing for broader exposure. It is typically made along the anterior border of the SCM.
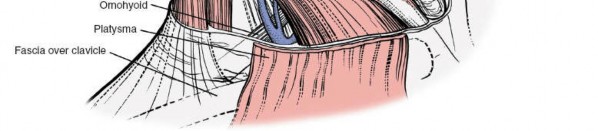
- Platysma: After incising the skin, the platysma muscle is encountered. It is incised transversely in line with the skin incision. Meticulous hemostasis is maintained.
- Subplatysmal Dissection: Subcutaneous flaps are raised superiorly and inferiorly, wide enough to expose the anterior border of the sternocleidomastoid muscle laterally and the midline strap muscles medially. This provides ample working space and protects the skin edges during retraction.


2. Incision of the Investing Layer of Deep Cervical Fascia
- The anterior border of the sternocleidomastoid muscle is identified.
- The investing layer of deep cervical fascia is incised longitudinally along the anterior border of the SCM. This allows the SCM to be retracted laterally, along with the underlying carotid sheath.
- The anterior jugular veins, if present, typically lie within or immediately deep to the investing fascia and often require ligation and division for adequate exposure.
3. Dissection of the Pretracheal Fascia and Strap Muscles
- Deep to the investing fascia, the infrahyoid "strap" muscles are identified: sternohyoid and sternothyroid are medial, omohyoid is lateral.
- The pretracheal fascia, which invests these strap muscles, is incised longitudinally in the avascular plane between the sternohyoid and sternothyroid muscles medially, and the omohyoid (and SCM) laterally. This internervous plane minimizes muscle injury.
- The strap muscles are then mobilized and gently retracted medially, exposing the trachea, esophagus, and thyroid gland.



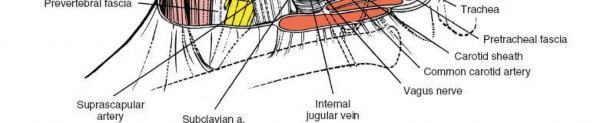
4. Carotid Sheath Identification and Retraction
- The carotid sheath, containing the common carotid artery, internal jugular vein, and vagus nerve, is palpated and identified.
- Using blunt dissection, the carotid sheath is carefully separated from the medial structures (trachea, esophagus) and gently retracted laterally, often with the SCM. Crucially, the retraction should be applied to the carotid sheath itself, not directly to the carotid artery, to minimize vagal nerve compression and protect the IJV.
- The entire neurovascular bundle is kept intact within the sheath and retracted as a single unit.


5. Entering the Retropharyngeal Space and Exposing the Prevertebral Fascia
- With the trachea and esophagus retracted medially and the carotid sheath laterally, the retropharyngeal space is entered bluntly. This avascular space provides a safe corridor to the anterior aspect of the vertebral column.
- The prevertebral fascia, covering the longus colli muscles and vertebral bodies, is now in view. The sympathetic chain typically lies on the prevertebral fascia, medial to the longus colli, requiring careful avoidance. The recurrent laryngeal nerve usually lies in the tracheoesophageal groove and is protected by medial retraction.
- Self-retaining retractors (e.g., Cloward, Caspar) are carefully placed to maintain the exposure. The blades should be placed under the prevertebral fascia to avoid direct pressure on the neurovascular bundle or esophagus/trachea.
6. Incision of the Prevertebral Fascia and Subperiosteal Dissection
- The prevertebral fascia is incised longitudinally in the midline of the vertebral bodies.
- A subperiosteal dissection is then performed using electrocautery or a periosteal elevator to detach the longus colli muscles from the anterior aspect of the vertebral bodies. These muscles are meticulously elevated and retracted laterally to expose the anterior longitudinal ligament and the disc spaces. Care must be taken to not dissect too laterally to avoid injury to the vertebral artery or sympathetic chain.
- Level Localization: Before any bony resection, an intraoperative lateral fluoroscopic image is obtained to confirm the correct surgical level. A needle can be placed into the disc space for definitive radiographic verification.

7. Decompression (Discectomy/Corpectomy)
-
Discectomy:
- An annular incision is made. The disc material is removed using rongeurs and curettes.
- The posterior longitudinal ligament (PLL) is meticulously identified. For radiculopathy, if the PLL is intact and not compressing the cord, it may be left intact. For myelopathy or significant central disc herniation, the PLL is typically resected to ensure complete decompression of the spinal cord and nerve roots.
- Osteophytes are carefully removed from the vertebral endplates and posterolateral margins to decompress nerve roots within the foramina.
- The superior and inferior endplates are prepared by removing cartilage using a high-speed burr or curettes, exposing bleeding subchondral bone to promote fusion.
-
Corpectomy:
- For multilevel compression or significant vertebral body pathology (e.g., tumor, trauma), a corpectomy is performed.
- After discectomy at the superior and inferior levels, a high-speed burr is used to remove the vertebral body (or bodies). Care is taken to protect the anterior and posterior dura and the nerve roots.
- The posterior cortex is meticulously thinned and removed to ensure complete spinal cord decompression.



8. Instrumentation and Fusion
-
Graft Insertion:
- A pre-selected interbody graft (e.g., allograft, autograft, PEEK cage) is impacted into the prepared disc space(s) or corpectomy defect. The graft should be appropriately sized to restore disc height and lordosis.
- For corpectomy, a structural cage (often expandable) or an autograft fibula/allograft strut is used.
-
Plate Application:
- An anterior cervical plate is contoured if necessary and applied to the anterior aspect of the vertebral bodies, spanning the fused segments.
- Screws are inserted into the adjacent vertebral bodies, bicortically or unicortically, ensuring appropriate trajectory to avoid posterior cortical breach or injury to the vertebral arteries.
- Dynamic plates allow for settling and load-sharing, while rigid plates provide immediate stability. The choice depends on surgeon preference and biomechanical considerations.






9. Closure
- Hemostasis: Thorough irrigation and careful hemostasis are achieved.
- Longus Colli Muscles: The longus colli muscles can be reapproximated in the midline, though this is not universally practiced.
- Prevertebral Fascia: The prevertebral fascia is closed, isolating the fusion construct from the visceral structures.
- Strap Muscles: The strap muscles (pretracheal fascia) are reapproximated loosely in layers.
- Investing Fascia: The investing fascia is closed.
- Platysma: The platysma muscle is closed meticulously to optimize cosmetic outcome.
- Skin: Skin edges are reapproximated with subcuticular sutures and sterile strips or tissue adhesive.
- Drainage: A small suction drain may be placed in the retropharyngeal space, particularly after multilevel procedures or in patients with bleeding diathesis, to prevent hematoma formation. The drain is typically removed within 24-48 hours.
Complications & Management
Despite its high success rate, anterior cervical spine surgery is not without potential complications, which range from transient to severe. A comprehensive understanding of these complications, their prevention, and management is crucial.
| Complication | Incidence (%) | Etiology | Salvage/Management Strategies |
|---|---|---|---|
| Recurrent Laryngeal Nerve Injury (RLNI) | 0.5-12 | Direct nerve traction/compression by retractors, thermal injury, transection, hematoma. Left RLN more common than right. | Prevention: Meticulous surgical technique, proper retractor placement (under prevertebral fascia), minimizing retraction time/force, careful hemostasis. Management: Voice rest, speech therapy. Most resolve spontaneously. Rarely requires vocal cord injection/medialization. |
| Dysphagia | 10-70 (transient), 1-10 (persistent) | Esophageal edema/spasm, direct trauma, nerve injury (RLN, superior laryngeal), prevertebral soft tissue swelling, hardware prominence. | Prevention: Gentle tissue handling, minimize retraction time, appropriate plate/graft sizing. Management: Soft diet, anti-inflammatory medications, proton pump inhibitors (PPIs). Swallowing therapy for persistent cases. Revision surgery for hardware issues. |
| Esophageal Perforation | <0.1 | Direct surgical trauma (burr, drill, retractor, implant), thermal injury. Often missed intraoperatively. | Prevention: Careful identification of the esophagus, proper retractor placement, avoid excessive anterior drilling. Management: Immediate surgical repair (primary closure), broad-spectrum antibiotics, possible diversionary feeding (J-tube), NPO. May require sternocleidomastoid or other muscle flap. |
| Hematoma (Retropharyngeal) | 0.1-2 | Inadequate hemostasis, vessel injury. | Prevention: Meticulous hemostasis, drain placement. Management: Prompt recognition (stridor, swelling, dysphagia), emergent evacuation in OR, airway management (intubation, tracheostomy if severe). |
| Vascular Injury | <0.1 | Carotid artery/IJV (retraction, direct trauma), Vertebral artery (excessive lateral decompression, aberrant course). | Prevention: Careful dissection within fascial planes, proper retractor placement, pre-op imaging review. Management: Direct repair, vascular surgery consultation. Ligature for IJV. Vertebral artery injury may require packing, muscle patch, or vascular repair, with risk of stroke. |
| Cerebrospinal Fluid (CSF) Leak / Dural Tear | 0.1-1 | Direct laceration during decompression, especially with ossified PLL (OPLL) or re-operative surgery. | Prevention: Careful decompression, use of blunt instruments near dura, small high-speed burrs. Management: Primary repair (suture, fibrin glue, dural substitute), Valsalva maneuver to check for leak, lumbar drain for 3-5 days, bed rest, prophylactic antibiotics. |
| Infection | 0.1-1 | Wound infection, discitis, osteomyelitis. | Prevention: Strict sterile technique, prophylactic antibiotics. Management: Debridement, targeted antibiotics, hardware removal in chronic cases. |
| Hardware-Related Complications | 2-5 | Screw pullout/breakage, plate migration/breakage, graft subsidence, pseudarthrosis. | Prevention: Proper biomechanical principles (plate length, screw trajectory, graft preparation), patient compliance. Management: Revision surgery with removal of failed hardware, re-decompression, revision fusion (possibly posterior approach). |
| Adjacent Segment Disease (ASD) | 2-4% per year | Increased stress on adjacent levels after fusion. | Prevention: Maintain sagittal balance, meticulous endplate preparation. Management: Non-operative treatment initially, if refractory, ACDF or arthroplasty at the new symptomatic level. |
| Neurological Deficit (New/Worsened) | <1 | Spinal cord contusion, nerve root injury, retractor paralysis, ischemia. | Prevention: Intraoperative neuromonitoring, careful decompression, avoid excessive traction. Management: Steroids, careful observation, re-exploration if acute neurological decline. |
Post-Operative Rehabilitation Protocols
Post-operative management following anterior cervical spine surgery aims to optimize recovery, promote fusion, and prevent complications. Protocols vary based on the extent of surgery, stability of the construct, and surgeon preference.
Immediate Post-Operative Period (Day 0-7)
- Pain Management: Multimodal analgesia including opioids, NSAIDs (if not contraindicated), and muscle relaxants.
- Airway Management: Close monitoring for signs of airway compromise (stridor, dyspnea) due to retropharyngeal swelling or hematoma. Early ambulation is encouraged.
-
Cervical Collar:
- Soft Collar: Often prescribed for comfort and proprioceptive feedback, particularly for single-level fusions with stable plating. Worn for 2-4 weeks.
- Rigid Collar (Philadelphia or Miami J): May be indicated for multilevel fusions, corpectomies, unstable constructs, or revision surgeries. Worn for 6-12 weeks, with removal for hygiene.
-
Activity Restrictions:
- Avoid heavy lifting (>5-10 lbs), pushing, pulling.
- Avoid bending, twisting, or excessive extension of the neck.
- No driving while on opioid pain medication or if a rigid collar impairs head checks.
- Wound Care: Keep incision clean and dry. Staples or sutures typically removed at 10-14 days.
Early Rehabilitation Phase (Weeks 2-6)
- Collar Weaning: Gradual weaning from collar as per surgeon's protocol, if used.
- Gentle Range of Motion (ROM): Commence gentle, active, pain-free cervical ROM exercises (flexion, extension, lateral bending, rotation). Avoid aggressive stretching.
- Isometrics: Begin isometric neck strengthening exercises under guidance, emphasizing activation of deep cervical flexors.
- Posture Education: Reinforce proper posture and ergonomic principles.
- Return to Light Activities: Gradual increase in ambulation and light daily activities.
Intermediate Rehabilitation Phase (Weeks 6-12)
- Progressive Strengthening: Advance cervical strengthening exercises. Focus on endurance and controlled movements.
- Scapular and Thoracic Mobility: Incorporate exercises to improve scapular stabilization and thoracic spine mobility, reducing compensatory strain on the cervical spine.
- Aerobic Conditioning: Encourage low-impact aerobic activities (walking, stationary cycling).
- Functional Training: Gradually reintroduce functional movements required for daily living and work.
Advanced Rehabilitation Phase (Months 3-6+)
- Return to Activity: Gradual return to more demanding activities, including work and recreational sports, provided fusion is confirmed radiographically and symptoms allow.
- Sports-Specific Training: For athletes, a structured program to rebuild strength, power, and agility specific to their sport.
- Radiographic Follow-up: Obtain follow-up X-rays (AP/Lateral with flexion/extension views) at 3, 6, and 12 months post-operatively to assess fusion status, hardware integrity, and sagittal alignment. CT scan may be used to confirm fusion, especially if symptoms persist or in cases of suspected pseudarthrosis.
Summary of Key Literature / Guidelines
The evolution of anterior cervical spine surgery has been driven by decades of rigorous research and clinical experience, shaping current guidelines and best practices.
- ACDF vs. Arthroplasty: Landmark studies such as the FDA investigational device exemption (IDE) trials have established the efficacy and safety of cervical disc arthroplasty (CDA) as an alternative to ACDF for single-level cervical radiculopathy or myelopathy without significant facet degeneration or instability. CDA aims to preserve motion and potentially reduce the risk of adjacent segment disease (ASD), though long-term data regarding ASD superiority over ACDF remains an area of ongoing research and debate.
- Graft Options: Meta-analyses and systematic reviews consistently demonstrate comparable fusion rates and clinical outcomes between autograft and allograft in ACDF. PEEK cages, often augmented with bone morphogenetic protein (BMP) or local autograft, have also shown high fusion rates and are widely utilized. The use of rhBMP-2 is controversial due to potential complications like dysphagia, seroma, and osteolysis, thus its use should be judicious and off-label in the cervical spine.
- Anterior Plating: The use of anterior cervical plates has significantly improved fusion rates, reduced graft subsidence, and minimized the need for external immobilization compared to non-instrumented fusions. Principles of proper plate length, screw trajectory, and unicortical vs. bicortical purchase are critical for construct stability.
- Management of Myelopathy: The AOSpine North America Cervical Spondylotic Myelopathy Study, among others, has reinforced that surgical decompression is superior to non-operative treatment for patients with moderate to severe CSM. Earlier intervention in progressive myelopathy generally leads to better neurological recovery.
- Recurrent Laryngeal Nerve Protection: Consensus is that meticulous surgical technique, minimizing retraction duration and pressure, and careful identification of anatomical planes are the primary means of preventing RLN injury. Intraoperative neuromonitoring of the RLN is technically challenging and its utility remains debated for routine ACDF.
- Dysphagia Mitigation: Strategies include minimizing retractor pressure, careful patient positioning, and avoiding oversized implants. Studies suggest that transient dysphagia is common, but chronic, severe dysphagia is less frequent and may warrant investigation for hardware prominence or esophageal injury.
- Evidence-Based Protocols: Current guidelines emphasize the importance of multimodal pain management, early mobilization, and tailored rehabilitation protocols to optimize patient recovery and facilitate return to function. Radiographic follow-up is essential to confirm fusion and detect complications.
Continued advancements in imaging, instrumentation, and surgical techniques underscore the dynamic nature of anterior cervical spine surgery. Adherence to sound anatomical principles, meticulous surgical execution, and a commitment to evidence-based practice remain paramount for achieving superior clinical outcomes in this technically demanding field.
Clinical & Radiographic Imaging



You Might Also Like


