Glenohumeral Osteoarthritis: Etiology, Biomechanics, and Treatment Strategies
Key Takeaway
Glenohumeral osteoarthritis (GHOA) is a degenerative shoulder joint disease involving cartilage loss, pain, and stiffness. It arises from primary causes or secondary factors like trauma or rotator cuff arthropathy. Treatment involves initial non-surgical approaches, including rest and physical therapy, escalating to surgical interventions based on symptom severity, functional limitation, and specific anatomical considerations.
Shoulder Osteoarthritis Treatment: Surgical & Non-Surgical Paths
Introduction & Epidemiology
Glenohumeral osteoarthritis (GHOA) is a degenerative joint disease characterized by progressive loss of articular cartilage, subchondral bone remodeling, osteophyte formation, and capsular contracture, leading to pain, stiffness, and functional impairment. It is a significant cause of shoulder disability, with an increasing prevalence driven by an aging population and improved diagnostic capabilities.
Primary GHOA is idiopathic, though genetic predisposition and mechanical factors are implicated. Secondary GHOA can arise from a myriad of etiologies, including:
*
Post-traumatic arthritis:
Following fractures, dislocations, or labral injuries.
*
Rotator cuff arthropathy (Cuff Tear Arthropathy, CTA):
Chronic, massive, irreparable rotator cuff tears lead to superior migration of the humeral head, eccentric loading, and erosion of the glenoid cartilage. This distinct entity has specific treatment implications.
*
Inflammatory arthropathies:
Rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis.
*
Osteonecrosis:
Avascular necrosis of the humeral head.
*
Dysplasia or congenital abnormalities.
*
Prior surgery:
Such as capsulorrhaphy or instability procedures altering joint mechanics.
The prevalence of radiographic GHOA is estimated to be around 16% in adults over 65, with symptomatic GHOA affecting approximately 3% of this population. The economic and societal burden associated with GHOA is substantial, necessitating effective management strategies.
Surgical Anatomy & Biomechanics
A thorough understanding of the glenohumeral joint's complex anatomy and biomechanics is paramount for effective treatment of GHOA, particularly in surgical planning.
Relevant Anatomy
- Glenoid: The pyriform-shaped articular surface of the scapula, typically retroverted an average of 2-7 degrees relative to the scapular body, and superiorly tilted an average of 5 degrees. Glenoid bone loss, often posterior and superior in GHOA, significantly impacts prosthetic component positioning. The glenoid labrum enhances concavity and stability.
- Humeral Head: Approximately one-third of a sphere, retroverted relative to the humeral shaft (average 30-40 degrees). Its articulation with the glenoid forms a highly mobile but inherently unstable joint.
- Capsule and Ligaments: The glenohumeral capsule is lax, allowing extensive motion. The capsular ligaments (superior, middle, and inferior glenohumeral ligaments) provide static stability, with the inferior glenohumeral ligament (IGHL) complex being the primary stabilizer in abduction and external rotation.
-
Rotator Cuff Muscles:
The supraspinatus, infraspinatus, teres minor, and subscapularis comprise the dynamic stabilizers, creating a compressive force that centers the humeral head on the glenoid and facilitating humeral head rotation.
- Supraspinatus: Initiates abduction.
- Infraspinatus & Teres Minor: External rotators.
- Subscapularis: Internal rotator.
- Deltoid Muscle: The primary abductor of the glenohumeral joint, essential for overhead function, and a critical component for function in rTSA.
-
Neurovascular Structures:
- Axillary Nerve: Courses inferior to the humeral head, wraps around the surgical neck, and supplies the deltoid and teres minor. It is vulnerable during humeral head osteotomy, humeral component insertion, and inferior capsular release.
- Musculocutaneous Nerve: Pierces the coracobrachialis, supplying it and the biceps. It is a concern during coracoid approaches or extensive medial dissection.
- Brachial Plexus: Located medially in the axilla, susceptible to traction injuries, especially in the beach chair position or with vigorous retraction.
- Axillary Artery and Vein: Medial to the brachial plexus, critical to avoid injury during deep dissection.
- Cephalic Vein: Located in the deltopectoral groove, serves as a key landmark and often preserved during the deltopectoral approach.
Biomechanics
Normal glenohumeral biomechanics relies on the intricate balance between static (capsuloligamentous) and dynamic (rotator cuff) stabilizers. The rotator cuff muscles generate a "concavity compression" effect, centering the humeral head within the glenoid fossa. This force couple allows the large deltoid muscle to efficiently elevate the arm without superior humeral migration.
In GHOA, this delicate balance is disrupted:
*
Cartilage Loss:
Increases friction, leading to pain and reduced range of motion (ROM).
*
Osteophyte Formation:
Mechanically blocks motion, contributing to stiffness.
*
Capsular Contracture:
Further restricts motion.
*
Glenoid Erosion:
Posterior glenoid wear is common in primary GHOA, altering the effective glenoid version and shifting the center of rotation, which can lead to posterior humeral subluxation and increased stress on the posterior glenoid rim.
*
Rotator Cuff Dysfunction:
In CTA, the absence of a functional rotator cuff leads to superior migration of the humeral head, often impinging on the acromion, resulting in "femoralization" of the acromion and "acetabularization" of the proximal humerus. This shifts the center of rotation superiorly and medially, diminishing the deltoid's mechanical advantage.
Indications & Contraindications
Treatment for GHOA is fundamentally guided by patient symptoms, functional limitations, radiographic severity, and patient expectations. Initial management is invariably non-surgical, with surgical intervention reserved for refractory cases.
Non-Surgical Treatment
The initial approach for GHOA is conservative, aiming to alleviate pain and improve function.
*
Rest or activity modification:
Avoiding provocative activities and adjusting arm movements to minimize pain.
*
Physical therapy (PT):
A structured program focusing on maintaining or improving glenohumeral and scapulothoracic range of motion, stretching tight periarticular structures, and strengthening residual rotator cuff and periscapular musculature. It does not reverse cartilage damage but optimizes residual function.
*
Nonsteroidal anti-inflammatory drugs (NSAIDs):
Oral NSAIDs such as aspirin, ibuprofen, or naproxen, reduce inflammation and pain. Clinicians must be mindful of potential gastrointestinal irritation, bleeding risk, and cardiovascular side effects. Prescription strength NSAIDs or COX-2 selective inhibitors may be considered. These should be taken with food, and patient history of peptic ulcers, renal insufficiency, or concomitant anticoagulation therapy must be carefully evaluated.
*
Corticosteroid injections:
Intra-articular corticosteroid injections can provide significant, albeit often temporary, relief of inflammation and pain. The duration of efficacy is variable, and repeated injections carry risks including articular cartilage degradation and potential infection. Limiting injections to 3-4 per year is a common guideline.
*
Hyaluronic acid injections:
Viscosupplementation, while common in knee OA, has less robust evidence in the shoulder. Some patients may experience temporary pain relief.
*
Moist heat:
May provide temporary pain relief and muscle relaxation.
*
Icing:
Applying ice for 20-30 minutes two to three times daily can reduce inflammation and ease pain, particularly after activity.
*
Disease-modifying antirheumatic drugs (DMARDs):
For GHOA secondary to inflammatory conditions like rheumatoid arthritis, collaboration with a rheumatologist for systemic management with DMARDs (e.g., methotrexate) is essential.
*
Dietary supplements:
Glucosamine and chondroitin sulfate are marketed for joint health. Current scientific evidence from high-quality randomized controlled trials largely fails to support their efficacy in significantly relieving pain or slowing disease progression in osteoarthritis, including the shoulder. The U.S. Food and Drug Administration (FDA) does not regulate these as rigorously as pharmaceuticals.
Surgical Treatment
Surgical intervention is considered when non-surgical treatments fail to provide adequate relief from pain and functional limitations.
Indications for Operative Management:
- Persistent, disabling pain: Unresponsive to a comprehensive course (typically 3-6 months) of non-operative management.
- Significant functional impairment: Limiting activities of daily living (ADLs), sleep, and quality of life.
- Radiographic evidence of advanced GHOA: Samilson and Prieto classification (mild, moderate, severe) or Walch classification (A1, A2, B1, B2, C) for glenoid morphology.
- Patient readiness: Willingness to undergo surgery and comply with a rigorous post-operative rehabilitation program.
-
Specific conditions:
- Anatomic Total Shoulder Arthroplasty (TSA): Indicated for primary GHOA with an intact and functional rotator cuff.
- Reverse Total Shoulder Arthroplasty (rTSA): Primarily indicated for rotator cuff deficient arthropathy (CTA), but also for severe glenoid bone loss, failed prior TSA with rotator cuff deficiency, and specific complex fractures.
- Humeral Hemiarthroplasty (HHA): Primarily indicated in younger patients (<55-60 years) with GHOA, an intact rotator cuff, and significant glenoid cartilage loss where glenoid resurfacing is deferred due to concerns of future glenoid loosening. Its use has decreased significantly with improved TSA outcomes and glenoid components. It may also be used for certain humeral head fractures.
Contraindications for Operative Management:
- Active infection: Local or systemic.
- Neuropathic arthropathy (Charcot joint): Poor bone quality and high risk of instability.
- Paralysis of the deltoid and rotator cuff: An absolute contraindication for rTSA due to inability to achieve functional elevation. Relative contraindication for TSA.
- Inability to comply with post-operative rehabilitation protocol: Critical for successful outcomes.
- Severe medical comorbidities: Uncontrolled cardiovascular, pulmonary, or renal disease that significantly increases surgical risk.
- Insufficient bone stock: For implant fixation (relative, may necessitate specialized components or bone grafting).
Summary Table: Operative vs. Non-Operative Indications
| Feature | Non-Operative Treatment Indications | Operative Treatment Indications |
|---|---|---|
| Pain | Mild to moderate; intermittent; manageable | Severe, persistent, disabling; refractory to conservative care |
| Function | Minimal to moderate limitations; ADLs largely intact | Significant limitations in ADLs; poor quality of life |
| Radiographs | Early to moderate GHOA (mild joint space narrowing) | Advanced GHOA (severe joint space narrowing, osteophytes, subchondral sclerosis/cysts, glenoid erosion) |
| Rotator Cuff | Intact or minor tears | Intact (for TSA/HHA); Deficient/Irreparable (for rTSA) |
| Patient Age/Activity | All ages; lower demand | All ages; appropriate for activity level (e.g., rTSA for low-demand CTA) |
| Comorbidities | All | Medically fit for surgery |
| Failed Conservative Care | N/A | >3-6 months of failed non-operative management |
| Infection | N/A | Absence of active local or systemic infection |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is crucial to optimize outcomes and minimize complications in shoulder arthroplasty.
Pre-Operative Planning
-
Clinical Assessment:
- History: Detailed pain characterization, functional limitations, prior treatments, and patient expectations. Assess for inflammatory conditions or systemic comorbidities.
- Physical Examination: Evaluate ROM (active and passive), strength of rotator cuff and deltoid, stability, presence of crepitus, and neurovascular status.
-
Imaging:
- Standard Radiographs: True AP (Grashey view), scapular Y lateral, and axillary views are essential. These assess joint space narrowing, osteophyte formation, subchondral sclerosis, cyst formation, and glenoid morphology. The axillary view is critical for assessing glenoid retroversion and posterior erosion.
- CT Scan: Considered mandatory for most shoulder arthroplasty cases. Provides detailed assessment of glenoid bone stock, version, and inclination, humeral head retroversion, and the presence of cysts or defects. 3D reconstruction aids in identifying specific patterns of glenoid wear (e.g., Walch classification A1, A2, B1, B2, C glenoids) and allows for accurate templating.
- MRI: Primarily used to evaluate rotator cuff integrity, particularly important for distinguishing between primary GHOA (intact cuff, TSA candidate) and CTA (cuff deficient, rTSA candidate). Also assesses soft tissue pathology such as bursitis or tendinopathy.
- Templating: Utilize templating software (digital or manual with transparent overlays) to determine appropriate implant size, particularly for humeral head diameter, glenoid component size, and orientation. For rTSA, glenosphere size, offset, and baseplate position are critical to avoid scapular notching and optimize deltoid tension.
- Anesthesia Consultation: Discuss anesthetic options (general anesthesia, interscalene nerve block for post-operative pain control). Assess medical fitness for surgery.
- Blood Management: While transfusion rates are low, consider pre-operative type and screen, especially in revision cases.
- Antibiotic Prophylaxis: Administer pre-operative antibiotics (e.g., cefazolin) within 60 minutes of incision.
Patient Positioning
Two primary positions are utilized for shoulder arthroplasty: the beach chair position and the lateral decubitus position. The choice often reflects surgeon preference, but each has distinct advantages and disadvantages.
Beach Chair Position (Semi-Fowler)
- Advantages: Excellent surgical field visualization, easier conversion to open if arthroscopy is combined, ability to easily adjust arm position for exposure, better airway control compared to lateral.
- Disadvantages: Risk of cerebral hypoperfusion (especially if blood pressure is not maintained), potential for brachial plexus traction injury, neck and spinal positioning challenges.
-
Setup:
- Patient is supine on the operating table. The torso is elevated 30-70 degrees.
- Head is secured in a headrest (e.g., "donut" or gel headrest), positioned neutrally or slightly flexed, and carefully taped to prevent excessive lateral rotation, which can strain the brachial plexus.
- Legs are bent at the hips and knees, with sequential compression devices applied for DVT prophylaxis.
- The surgical shoulder is positioned at the edge of the table to allow for maximum maneuverability.
- The ipsilateral arm is draped free and placed on a sterile arm support or table, allowing full manipulation during surgery (abduction, adduction, rotation).
- The contralateral arm is tucked to the side or rested on an arm board, ensuring proper padding to prevent nerve compression.
- All pressure points are padded.
Lateral Decubitus Position
- Advantages: Less concern for cerebral hypoperfusion, potentially more stable position for prolonged cases, good for exposure of posterior structures if needed.
- Disadvantages: More challenging for arm manipulation, potential for nerve compression (brachial plexus, ulnar nerve) from prolonged positioning or inadequate padding, less optimal for deltopectoral approach for some surgeons, difficulty with C-arm access if required.
-
Setup:
- Patient is placed on their non-operative side.
- A beanbag or specialized positioning device is used to stabilize the torso.
- An axillary roll is placed in the contralateral axilla to protect the brachial plexus and axillary vessels from compression.
- The head is neutrally aligned with the spine and supported.
- The dependent arm is padded and positioned appropriately.
- The surgical arm is suspended in balanced traction, often from an overhead boom, allowing precise control of abduction, flexion, and rotation. Ensure traction is not excessive.
- All pressure points (knees, ankles, iliac crest) are meticulously padded.
Regardless of position, ensure adequate padding, confirm all bony prominences are clear of pressure, and monitor neurovascular status.
Detailed Surgical Approach / Technique
The primary surgical options for advanced GHOA are anatomic Total Shoulder Arthroplasty (TSA) and Reverse Total Shoulder Arthroplasty (rTSA). Humeral hemiarthroplasty (HHA) is used less commonly. The deltopectoral approach is the standard for both TSA and rTSA.
Deltopectoral Approach
- Incision: A curvilinear skin incision is made from the posterior acromial angle, extending anteriorly and inferiorly along the deltopectoral groove for approximately 10-15 cm.
- Deltopectoral Interval: Identify the cephalic vein. This marks the internervous plane between the deltoid (axillary nerve, C5-C6) laterally and the pectoralis major (medial and lateral pectoral nerves, C5-T1) medially. The cephalic vein is typically retracted medially with the pectoralis major to protect it, though some surgeons prefer lateral retraction or ligation if necessary.
-
Deep Dissection:
- Incise the clavipectoral fascia medial to the cephalic vein.
- Identify the coracoid process and its attached muscles (coracobrachialis, short head of biceps). The subscapularis muscle is deep and lateral to these structures.
- Release the upper 1-2 cm of the pectoralis major tendon from the humerus if additional exposure is required. This is generally avoided unless absolutely necessary to prevent weakening.
- Retract the conjoined tendon medially, protecting the musculocutaneous nerve which enters its deep surface 2-8 cm distal to the coracoid tip.
- Perform an anterior capsulotomy. Identify the subscapularis tendon, which spans from the lesser tuberosity to the glenoid labrum.
-
Subscapularis Management:
The subscapularis tendon must be detached from the lesser tuberosity to expose the glenohumeral joint.
- Tenotomy: Incise the subscapularis tendon approximately 1 cm medial to its insertion on the lesser tuberosity, preserving a cuff of tendon for later repair. Tag the tendon with sutures (e.g., #2 FiberWire) for identification and retraction. This is the most common method.
- Lesser Tuberosity Osteotomy: A small osteotomy (approximately 2 cm x 1 cm) of the lesser tuberosity with the attached subscapularis can be performed. This technique may provide a more robust repair, particularly in revision cases, but requires re-attaching the bone block with sutures or screws. This is less common in primary arthroplasty.
- Following subscapularis release, perform a wide anterior and inferior capsular release to achieve adequate external rotation for humeral head dislocation and glenoid exposure. The axillary nerve is vulnerable inferiorly.
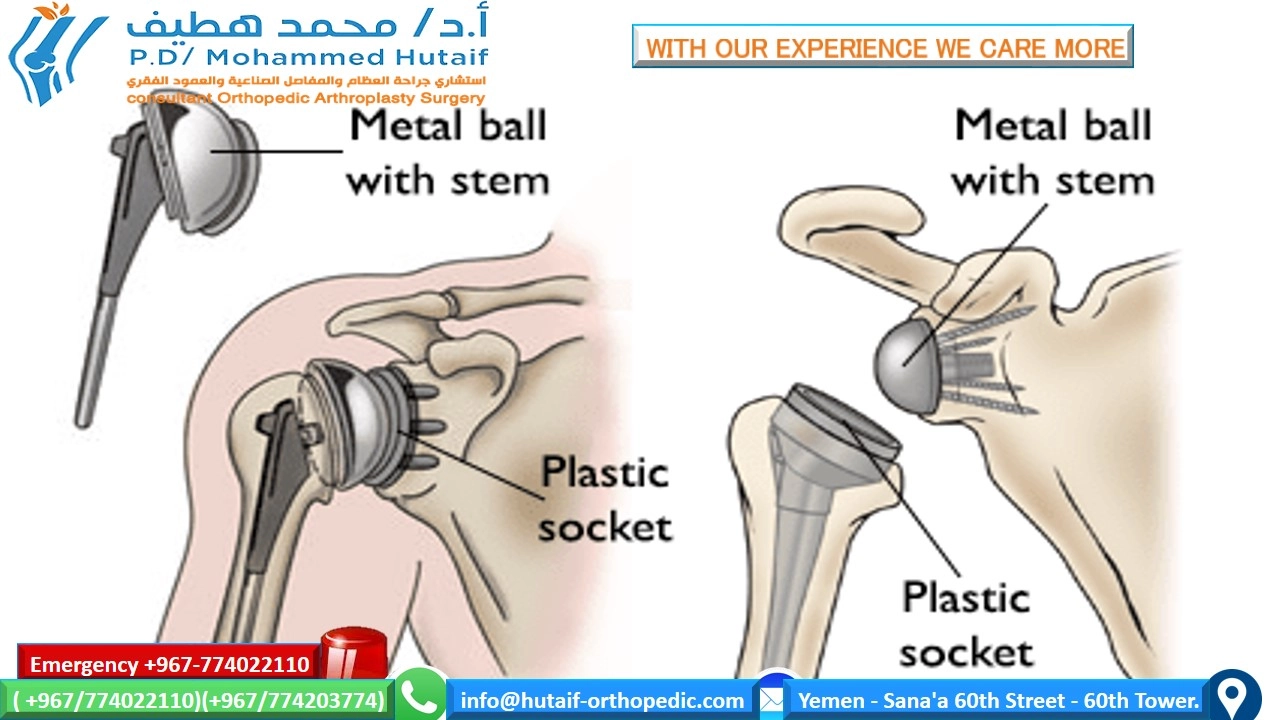
Anatomic Total Shoulder Arthroplasty (TSA)
TSA is indicated for GHOA with an intact rotator cuff.
1.
Humeral Head Osteotomy:
* Dislocate the humeral head anteriorly.
* Using an osteotomy guide, resect the humeral head at approximately 30-40 degrees of retroversion and 135-140 degrees of inclination (relative to the humeral shaft axis). The goal is to restore the natural humeral head geometry and joint line. Proper retroversion is critical to avoid instability and excessive internal rotation.
*

This image depicts a typical humeral component being prepared for insertion, emphasizing the importance of precise reaming and sizing to match the patient's anatomy.
2.
Humeral Canal Preparation:
* Progressively ream the humeral intramedullary canal to fit the chosen humeral stem. Start with a pilot hole, then use increasing reamer sizes.
* Trial humeral stem and head components are inserted to assess fit, stability, and range of motion.
3.
Glenoid Preparation:
* Retract the humeral shaft posteriorly to expose the glenoid. Use specialized glenoid retractors to protect the soft tissues (e.g., anterior deltoid, axillary nerve).
* Remove residual osteophytes from the glenoid rim.
* Assess glenoid bone loss and correct retroversion/inclination using reamers. Posterior eccentric reaming is often necessary to centralize the glenoid and address posterior wear (Walch B2 glenoids may require bone grafting or augmented glenoids).
* Drill pilot holes for the central peg/keel and peripheral pegs/screws of the glenoid component.
* For
cemented glenoids
, ensure a clean, dry surface. Apply cement (polymethylmethacrylate) to the glenoid component and press firmly onto the glenoid, removing excess cement.
* For
uncemented glenoids
, ensure precise fit and firm impaction. Screws provide initial stability.
4.
Component Implantation:
*
Glenoid Component:
Insert the final glenoid component. Ensure optimal version and inclination.
*
Humeral Component:
Insert the final humeral stem (cemented or uncemented, depending on bone quality and implant design). Attach the final humeral head component.
5.
Reduction and Stability:
* Reduce the humeral head onto the glenoid.
* Assess range of motion, stability in all planes (flexion, extension, abduction, adduction, internal and external rotation). Ensure appropriate soft tissue tension.
* Confirm no impingement or subluxation.
6.
Subscapularis Repair:
Reattach the subscapularis tendon using strong non-absorbable sutures (e.g., #2 FiberWire in a mattress or figure-of-eight fashion) to the lesser tuberosity footprint. A robust repair is paramount to prevent anterior instability and optimize internal rotation strength.
7.
Wound Closure:
Layered closure of the deltopectoral interval, subcutaneous tissue, and skin.
Reverse Total Shoulder Arthroplasty (rTSA)
rTSA is indicated for CTA, severe glenoid bone loss, and revision settings. The deltopectoral approach is identical for initial exposure.
1.
Humeral Head Osteotomy:
* The humeral head is resected, often at a slightly different angle than TSA (e.g., 155 degrees inclination), to accommodate the rTSA implant design. The goal is to remove the arthritic bone and prepare for the humeral tray.
2.
Glenoid Preparation:
* Similar exposure of the glenoid as in TSA.
* The primary goal is to place a stable
glenosphere baseplate
. Drill a central pilot hole, then ream the glenoid to a flat surface.
* Drill holes for the baseplate screws. The baseplate is often placed slightly inferiorly and with 5-10 degrees of inferior tilt to minimize scapular notching.
* Insert the
baseplate
and secure it with locking screws. Ensure bicortical fixation for maximal stability.
* Impact the
glenosphere
onto the baseplate. Glenosphere size and lateralization (via shims or lateralized baseplates) are chosen to optimize deltoid tension and impingement-free range of motion, balancing the risk of notching.
3.
Humeral Preparation:
* Ream the humeral intramedullary canal for the
humeral stem
.
* Insert the trial humeral stem and then the
humeral tray
with a polyethylene liner.
4.
Reduction and Stability:
* Reduce the humeral component onto the glenosphere.
* Assess range of motion, stability, and deltoid tension. The deltoid should be under appropriate tension to function effectively without being overtly tight, which could compromise nerve function or risk periprosthetic fracture.
* Ensure impingement-free motion.
5.
Subscapularis Repair:
In rTSA for CTA, the subscapularis is often deficient or irreparable. If a viable portion remains, a partial repair or imbrication can be performed, but it is not critical for stability as in TSA.
6.
Wound Closure:
Layered closure.
Humeral Hemiarthroplasty (HHA)
HHA involves replacing only the humeral head, leaving the glenoid intact. Its role is limited.
*
Technique:
Identical humeral head osteotomy and stem insertion as in TSA.
*
Glenoid:
The glenoid is left untouched or debrided of osteophytes.
*
Challenges:
Pain can persist due to continued glenoid arthritis and potential cartilage incongruity between the prosthetic head and native glenoid.
Arthroscopic Debridement
While not a definitive treatment for advanced GHOA, arthroscopic debridement, chondroplasty, and capsular release may be considered for early-stage OA with mechanical symptoms, loose bodies, or isolated areas of impingement. It typically offers temporary relief and does not alter disease progression.
Complications & Management
Despite advancements in surgical technique and implant design, shoulder arthroplasty carries inherent risks. A thorough understanding of potential complications and their management is essential.
| Complication | Incidence (%) | Management Strategy |
Description
|
Intervention / Indication (TSA/rTSA)
``
## Introduction & Epidemiology`
Glenohumeral osteoarthritis (GHOA) is a degenerative joint condition characterized by progressive damage to the articular cartilage, subchondral bone remodeling, osteophyte formation, and capsular thickening. This pathological cascade culminates in debilitating pain, restricted range of motion (ROM), and significant functional impairment of the shoulder, profoundly impacting a patient's quality of life. As the global population ages, the incidence and prevalence of GHOA are steadily rising, underscoring the critical need for effective diagnostic and therapeutic strategies.
GHOA can be broadly categorized into primary and secondary forms:
*
Primary GHOA:
Often idiopathic, its etiology is multifactorial, involving genetic predispositions, repetitive microtrauma, and age-related biochemical changes within the articular cartilage. It typically affects older individuals and presents with insidious onset.
*
Secondary GHOA:
Arises from identifiable causative factors, including:
*
Post-traumatic Arthritis:
Sequelae of prior shoulder injuries such as fractures (e.g., proximal humerus, glenoid), dislocations, or severe labral tears that disrupt joint congruity and mechanics.
*
Rotator Cuff Arthropathy (CTA):
A distinct and severe form of GHOA resulting from chronic, massive, and often irreparable rotator cuff tears. The superior migration of the humeral head due to lack of cuff compression leads to abnormal loading patterns, impingement against the acromion, and progressive erosion of both humeral and glenoid cartilage. This entity demands specialized surgical considerations.
*
Inflammatory Arthropathies:
Systemic autoimmune conditions such as rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and crystal-induced arthropathies (gout, pseudogout) can directly affect the glenohumeral joint, leading to synovitis, cartilage destruction, and bone erosion.
*
Osteonecrosis (Avascular Necrosis):
Compromised blood supply to the humeral head, often associated with corticosteroid use, alcohol abuse, sickle cell disease, or trauma, results in collapse of the subchondral bone and overlying cartilage.
*
Dysplasia or Congenital Abnormalities:
Anatomical deformities of the glenoid or humerus can lead to abnormal joint mechanics and accelerated degenerative changes.
*
Iatrogenic Causes:
Prior surgical interventions, particularly those altering capsular volume or glenoid mechanics (e.g., instability repairs, extensive open procedures), can predispose to premature GHOA.
Epidemiological studies indicate that radiographic evidence of GHOA affects approximately 16% of individuals over 65 years of age, with symptomatic GHOA impacting about 3% within this demographic. The economic burden, encompassing healthcare costs, productivity losses, and long-term care, highlights GHOA as a significant public health concern.
## Surgical Anatomy & Biomechanics
A comprehensive mastery of the intricate surgical anatomy and biomechanics of the glenohumeral joint is non-negotiable for any orthopedic surgeon managing GHOA. This foundational knowledge guides accurate diagnosis, precise surgical planning, and meticulous execution to optimize functional restoration and minimize complications.
Key Anatomical Considerations
- Glenoid: The articular surface of the scapula is a shallow, pyriform-shaped fossa, significantly smaller than the humeral head, facilitating extensive motion. It typically exhibits 2-7 degrees of retroversion relative to the scapular body and approximately 5 degrees of superior inclination. In GHOA, the glenoid is frequently affected by posterior erosion and eccentric wear patterns (e.g., Walch B2 glenoids with posterior biconcavity), necessitating careful assessment for prosthetic placement. The glenoid labrum, a fibrocartilaginous rim, deepens the socket and enhances stability.
- Humeral Head: Constituting roughly one-third of a sphere, the humeral head demonstrates an average of 30-40 degrees of retroversion relative to the epicondylar axis of the humerus. Its precise orientation and size are critical for restoring native joint kinematics.
-
Capsule and Glenohumeral Ligaments (GHLs):
The glenohumeral capsule is inherently lax, providing minimal static restraint unless reinforced by thickenings that form the GHLs.
- Superior GHL (SGHL): Limits inferior translation and external rotation in adduction.
- Middle GHL (MGHL): Important for anterior stability in mid-ranges of abduction.
- Inferior GHL (IGHL) Complex: The primary static stabilizer, composed of anterior and posterior bands and an intervening axillary pouch. It provides significant restraint against anterior and posterior translation, especially in abduction. Extensive capsular contracture is common in GHOA, restricting passive ROM.
-
Rotator Cuff Muscles:
The supraspinatus, infraspinatus, teres minor, and subscapularis are paramount for dynamic stability and smooth articulation.
- Supraspinatus: Initiates abduction and contributes to superior humeral head compression.
- Infraspinatus & Teres Minor: Primary external rotators and posterior compressors.
- Subscapularis: Primary internal rotator and anterior compressor. Its integrity and repair are critical for anterior stability and internal rotation function in anatomic shoulder arthroplasty.
- Deltoid Muscle: The powerful primary abductor of the glenohumeral joint, innervated by the axillary nerve. Its integrity is fundamental for functional outcomes, particularly in reverse total shoulder arthroplasty (rTSA).
-
Neurovascular Structures:
- Axillary Nerve: The most commonly injured nerve in shoulder surgery. It courses inferior to the humeral head, emerging from the quadrangular space, and wraps around the surgical neck of the humerus, supplying the deltoid and teres minor. It is at risk during inferior capsular release, humeral head osteotomy, and placement of glenoid retractors.
- Musculocutaneous Nerve: Pierces the coracobrachialis muscle 2-8 cm distal to the coracoid tip. It is vulnerable during dissection medial to the conjoined tendon and coracoid region.
- Brachial Plexus: Located medial to the coracoid process, superficial to the subscapularis, and inferomedial to the glenohumeral joint. Susceptible to traction injuries during vigorous retraction or prolonged positioning, especially in the beach chair.
- Cephalic Vein: A consistent landmark within the deltopectoral groove, providing a reliable internervous plane between the deltoid and pectoralis major.
- Axillary Artery and Vein: Positioned deep and medial to the brachial plexus, these major vessels are at risk with overly aggressive medial dissection or in revision settings with distorted anatomy.
Biomechanical Principles
Normal glenohumeral biomechanics relies on the harmonious interplay between static stabilizers (capsule, ligaments, glenoid labrum) and dynamic stabilizers (rotator cuff, deltoid).
*
Concavity Compression:
The rotator cuff muscles generate a compressive force that centers the humeral head within the glenoid fossa. This effectively increases the functional depth of the glenoid.
*
Force Couples:
The rotator cuff muscles act in concert to produce force couples that control humeral head translation. For instance, the superior cuff (supraspinatus) and inferior cuff (subscapularis, infraspinatus, teres minor) create a transverse force couple that prevents superior migration during deltoid contraction.
*
Center of Rotation (CoR):
In the native shoulder, the CoR is near the center of the humeral head. Maintaining this precise CoR is crucial for optimal deltoid leverage and overall joint function.
Biomechanical Alterations in GHOA
- Cartilage Degradation & Osteophytes: Increased friction, pain, and mechanical blocks to motion.
- Capsular Contracture: Progressive tightening of the capsule, particularly anteriorly and inferiorly, leads to significant loss of external rotation and elevation.
- Glenoid Erosion: Posterior glenoid wear, common in primary GHOA, shifts the effective CoR posteriorly. This leads to posterior humeral head subluxation, altered joint reaction forces, and increased shear stress on the remaining cartilage and soft tissues. This often necessitates eccentric reaming or augmented glenoid components in TSA.
- Rotator Cuff Dysfunction (CTA): In rotator cuff deficient shoulders, the superior force couple is lost. This results in unopposed deltoid contraction leading to superior migration of the humeral head, impinging against the acromion, and "acetabularization" of the proximal humerus and "femoralization" of the acromion. The CoR shifts superiorly and medially, diminishing the deltoid's mechanical advantage. This pathological state necessitates the unique biomechanical design of rTSA, which medializes and distalizes the CoR, restoring deltoid tension and its ability to elevate the arm.
Indications & Contraindications
The decision-making process for treating GHOA follows a staged approach, beginning with non-surgical interventions and progressing to surgical management when conservative measures fail. Patient-specific factors, including symptom severity, functional demands, radiographic findings, and overall health status, are paramount in guiding these decisions.
Non-Surgical Treatment Pathways
Initial management for GHOA is universally conservative, aiming to mitigate pain, preserve or restore joint mobility, and enhance functional capacity without invasive procedures. The core components include:
- Activity Modification: Advising patients to avoid repetitive overhead activities, heavy lifting, or positions that exacerbate pain. Education on proper body mechanics and ergonomic adjustments can be beneficial.
-
Physical Therapy (PT) and Home Exercise Programs:
Essential for maintaining and improving glenohumeral and scapulothoracic rhythm and range of motion. A structured program focuses on:
- Passive and Active-Assisted ROM exercises: To address capsular contracture and improve joint mobility.
- Stretching: Targeting internal rotators, posterior capsule, and pectoralis muscles.
- Strengthening: Emphasizing rotator cuff, deltoid, and periscapular stabilizers to improve dynamic stability and muscle balance.
- Modalities: Therapeutic ultrasound, electrical stimulation, or manual therapy may be used adjunctively.
-
Pharmacological Management:
- Nonsteroidal Anti-inflammatory Drugs (NSAIDs): Oral NSAIDs (e.g., ibuprofen, naproxen, celecoxib) are first-line agents to reduce pain and inflammation. Careful consideration of patient comorbidities (e.g., gastrointestinal history, renal function, cardiovascular risk, concomitant anticoagulation) is crucial. Gastroprotective agents or COX-2 selective NSAIDs may be indicated. Topical NSAIDs can also be used.
- Corticosteroid Injections: Intra-articular corticosteroid injections can provide potent, rapid, but often temporary relief from inflammation and pain. Efficacy duration is variable (weeks to months). Risks include infection (rare, <1%), cartilage damage with repeated injections, and hyperglycemia in diabetic patients. Limiting frequency (e.g., 3-4 injections per year) is generally advised.
- Hyaluronic Acid (HA) Injections: Viscosupplementation, while well-established for knee OA, has less compelling evidence for shoulder GHOA. Some patients may report transient pain relief, but its widespread use is not supported by robust, large-scale studies.
- Platelet-Rich Plasma (PRP) Injections: Emerging biological therapies, including PRP, are under investigation for GHOA. While anecdotal reports exist, strong evidence from randomized controlled trials supporting their efficacy in reducing pain or halting disease progression is currently limited and heterogeneous. These remain largely investigational.
- Disease-Modifying Antirheumatic Drugs (DMARDs): For GHOA secondary to systemic inflammatory arthropathies (e.g., rheumatoid arthritis), systemic medication management overseen by a rheumatologist is fundamental to control the underlying disease process and prevent further joint destruction.
-
Physical Modalities:
- Heat Therapy: Moist heat can alleviate muscle stiffness and pain.
- Cryotherapy: Ice application (20-30 minutes, 2-3 times/day) effectively reduces inflammation and localized pain, particularly post-activity.
- Dietary Supplements: Glucosamine and chondroitin sulfate are widely used, but meta-analyses and large clinical trials have largely demonstrated no significant clinical benefit over placebo in reducing pain or altering the course of osteoarthritis. The lack of stringent FDA regulation for these compounds necessitates cautious patient counseling regarding their limited scientific support.
Surgical Treatment Pathways
Surgical intervention is indicated when a patient experiences persistent, debilitating pain and functional impairment that is refractory to a thorough and appropriately administered course of non-operative management (typically 3-6 months). The choice of surgical procedure is highly individualized and depends on the specific etiology of the OA, the integrity of the rotator cuff, glenoid bone quality, patient age, activity level, and expectations.
Primary Surgical Options:
-
Anatomic Total Shoulder Arthroplasty (TSA):
- Indications: Best suited for primary GHOA with an intact and functional rotator cuff. The procedure replaces both the humeral head and glenoid articular surface, aiming to restore native joint kinematics.
- Patient Profile: Typically older, lower-demand patients with good bone quality.
-
Reverse Total Shoulder Arthroplasty (rTSA):
-
Indications:
The gold standard for
rotator cuff deficient arthropathy (CTA)
, where the rotator cuff is irreparably torn, leading to superior humeral head migration. Also indicated for:
- Severe glenoid bone loss that precludes anatomic glenoid component fixation.
- Failed TSA with irreparable rotator cuff failure.
- Specific complex proximal humeral fractures in elderly patients.
- Salvage for failed shoulder arthroplasty (TSA or HHA).
- Patient Profile: Often older, active individuals with CTA who rely on deltoid function for elevation. Requires an intact and functional deltoid muscle.
-
Indications:
The gold standard for
rotator cuff deficient arthropathy (CTA)
, where the rotator cuff is irreparably torn, leading to superior humeral head migration. Also indicated for:
-
Humeral Hemiarthroplasty (HHA):
-
Indications:
Historically used for GHOA, but its role has diminished significantly due to inferior long-term pain relief compared to TSA. Current indications are limited to:
- Younger patients (<55-60 years) with GHOA and an intact rotator cuff, where concerns regarding future glenoid component loosening or wear dictate deferring glenoid resurfacing. This remains a controversial indication.
- Acute, complex proximal humeral fractures in patients with good bone quality and potentially intact rotator cuff.
- Rarely, patients with significant glenoid bone loss or active infection where a glenoid component is contraindicated.
- Challenges: Persisting pain from continued glenoid arthritis and incongruity between the prosthetic humeral head and the native glenoid articular surface.
-
Indications:
Historically used for GHOA, but its role has diminished significantly due to inferior long-term pain relief compared to TSA. Current indications are limited to:
-
Shoulder Arthrodesis (Fusion):
- Indications: A salvage procedure for severe, intractable pain and instability in cases of failed arthroplasty (especially with infection), irreparable brachial plexus injury, massive irreparable rotator cuff and deltoid deficiency, or chronic instability.
- Consequences: Provides pain relief and stability but at the cost of complete loss of glenohumeral motion, necessitating compensatory scapulothoracic motion.
-
Arthroscopic Debridement and Capsular Release:
- Indications: Limited role in definitive treatment of advanced GHOA. May be considered for early-stage OA with mechanical symptoms (e.g., loose bodies, impingement from osteophytes) or for addressing isolated capsular contracture. Provides temporary relief but does not alter disease progression.
Contraindications to Surgical Management:
- Active Local or Systemic Infection: Absolute contraindication due to high risk of periprosthetic joint infection (PJI). Infection must be eradicated prior to arthroplasty.
- Neuropathic Arthropathy (Charcot Joint): Leads to severe bone destruction and instability, making prosthetic implantation highly prone to failure.
- Paralysis of the Deltoid Muscle: An absolute contraindication for rTSA, as functional elevation depends entirely on a healthy deltoid. Relative contraindication for TSA if the rotator cuff is intact but deltoid is deficient.
- Inability to Comply with Post-operative Rehabilitation: Successful outcomes are contingent upon patient adherence to structured rehabilitation protocols.
- Severe Medical Comorbidities: Uncontrolled cardiovascular, pulmonary, renal, or metabolic diseases that pose an unacceptably high anesthetic or surgical risk.
- Insufficient Bone Stock: Severe osteoporosis or glenoid/humeral bone loss may preclude stable implant fixation (relative contraindication, may require advanced techniques like bone grafting or custom implants).
- Unrealistic Patient Expectations: Important to counsel patients on realistic outcomes regarding pain relief, functional improvement, and potential complications.
Summary Table: Operative vs. Non-Operative Indications
| Feature | Non-Operative Treatment Indications | Operative Treatment Indications |
|---|---|---|
| Pain | Mild to moderate; intermittent; manageable with conservative measures | Severe, persistent, disabling pain; significantly impacting sleep and quality of life; refractory to >3-6 months of comprehensive non-operative care. |
| Functional Limitation | Minimal to moderate impact on activities of daily living (ADLs) | Severe limitations in ADLs (dressing, hygiene, reaching); inability to perform work or leisure activities. |
| Radiographic Severity | Early to moderate GHOA (mild joint space narrowing, early osteophytes) | Advanced GHOA (severe joint space narrowing, subchondral sclerosis/cysts, significant osteophyte formation, glenoid erosion, humeral head collapse/deformity). |
| Rotator Cuff Status | Intact or minor, non-symptomatic tears | TSA/HHA: Intact and functional rotator cuff. rTSA: Irreparable rotator cuff tear (Cuff Tear Arthropathy). |
| Patient Age/Demand | All ages; individuals with lower functional demands | All ages (adjusted for implant choice); active individuals seeking functional restoration. |
| Medical Fitness | All patients | Medically optimized and fit to undergo general anesthesia and surgery. |
| Compliance | Able to participate in therapy and activity modification | Able and willing to comply with a rigorous post-operative rehabilitation protocol. |
| Infection | No active infection | Absence of any active local or systemic infection. |
| Deltoid Status | Intact | TSA/HHA: Intact. rTSA: Intact and functional deltoid (absolute requirement). |
Pre-Operative Planning & Patient Positioning
Thorough and meticulous pre-operative planning, coupled with precise patient positioning, are fundamental to achieving optimal outcomes in shoulder arthroplasty, enhancing surgical efficiency, and minimizing potential complications.
Pre-Operative Planning
-
Comprehensive Clinical Assessment:
- History: Elicit detailed information regarding the onset, character, duration, and severity of pain, identifying exacerbating and alleviating factors. Quantify functional limitations (e.g., specific ADLs, work-related tasks, recreational activities) and assess the impact on sleep and overall quality of life. Document prior treatments (surgical and non-surgical) and their efficacy. Crucially, ascertain patient expectations regarding post-operative pain relief, functional recovery, and realistic limitations. Review all relevant medical history, including comorbidities (cardiovascular, pulmonary, renal, metabolic, neurological), allergies, and current medications (especially anticoagulants, immunosuppressants, or bisphosphonates).
-
Physical Examination:
Systematically evaluate the shoulder.
- Inspection: Observe for muscle atrophy (deltoid, rotator cuff), swelling, or skin changes.
- Palpation: Identify areas of tenderness.
- Range of Motion: Assess both active and passive ROM in all planes (flexion, extension, abduction, adduction, internal and external rotation). Note the presence of crepitus or painful arcs. Document the degree of capsular contracture.
- Strength: Dynamically assess rotator cuff (supraspinatus, infraspinatus, subscapularis) and deltoid strength. This is critical for surgical decision-making (TSA vs. rTSA).
- Stability: Evaluate for subtle instability.
- Neurovascular Status: Perform a detailed neurological examination of the ipsilateral upper extremity, assessing motor and sensory function (axillary, musculocutaneous, radial, ulnar, median nerves) and distal pulses.
-
Advanced Imaging:
-
Standard Radiographs:
Essential views include a true AP (Grashey view, 30-45 degrees oblique from the coronal plane), axillary lateral, and scapular Y lateral. These views are crucial for assessing:
- Joint space narrowing, subchondral sclerosis, and cyst formation.
- Osteophyte burden on the humeral head and glenoid.
- Humeral head superior migration (indicative of CTA).
- Glenoid version and inclination (best seen on axillary and true AP).
- Walch classification of glenoid morphology (A1, A2, B1, B2, C) guides glenoid component choice and potential need for augmentation.
-
Computed Tomography (CT) Scan:
Generally considered indispensable for shoulder arthroplasty. A high-quality CT scan with 1-2 mm cuts, often with 3D reconstruction, provides invaluable detailed information on:
- Precise quantification of glenoid bone loss, retroversion, and inclination. This allows for accurate templating and planning for corrective reaming or augmented glenoid components.
- Humeral head retroversion and the extent of humeral head deformity.
- Identification of large subchondral cysts or bone defects that may require grafting.
- Assessment of bone quality for implant fixation.
- Magnetic Resonance Imaging (MRI): Primarily indicated when rotator cuff integrity is uncertain, especially when distinguishing between primary GHOA (intact cuff, TSA candidate) and CTA (cuff deficient, rTSA candidate). It provides excellent soft tissue detail, evaluating tendon tears, muscle atrophy, and fatty infiltration. It can also detect avascular necrosis.
-
Standard Radiographs:
Essential views include a true AP (Grashey view, 30-45 degrees oblique from the coronal plane), axillary lateral, and scapular Y lateral. These views are crucial for assessing:
-
Surgical Templating:
- Digital templating software (preferred) or physical transparent overlays are used with calibrated radiographs and CT scans.
- Humeral Component: Determine appropriate humeral head size (diameter, articular depth), offset, and retroversion to match the native anatomy and restore joint line. Select stem type (cemented vs. uncemented) and length based on bone quality and implant design.
- Glenoid Component: Select appropriate glenoid component size, morphology (pegged vs. keeled, augmented designs for bone loss). Plan for correction of glenoid version/inclination.
- rTSA Specifics: Precisely plan glenosphere size, offset, and baseplate position (superior-inferior, anterior-posterior placement, inferior tilt) to optimize deltoid tension, avoid scapular notching, and achieve impingement-free motion.
- Anesthesia Consultation: Discuss anesthetic options (general anesthesia typically combined with an interscalene nerve block for peri-operative and post-operative pain management). Optimize patient medical status (e.g., blood pressure control, glycemic management, cardiac risk assessment).
- Blood Management: While elective shoulder arthroplasty typically has low blood loss, cross-matching or type and screen may be considered, especially for revision cases or patients with anticipated higher blood loss.
- Antibiotic Prophylaxis: Administer intravenous broad-spectrum antibiotics (e.g., cefazolin 2g for adults) within 60 minutes prior to skin incision, adhering to institutional guidelines.
Patient Positioning
The choice of patient position significantly impacts surgical exposure, operative efficiency, and patient safety. The two main positions are the beach chair (semi-Fowler) and lateral decubitus.
1. Beach Chair Position (Semi-Fowler)
-
Advantages:
- Excellent Visualization: Provides a familiar anatomical orientation and allows for superior visualization of the shoulder girdle.
- Arm Mobility: The ipsilateral arm is draped free, allowing full manipulation (flexion, extension, abduction, adduction, internal/external rotation) for optimal exposure of various aspects of the joint.
- Conversion to Arthroscopy: Facilitates easy transition to arthroscopy if indicated for diagnostic purposes or adjunct procedures.
- Airway Management: Easier for the anesthesiologist to manage the airway compared to lateral decubitus.
-
Disadvantages:
- Cerebral Hypoperfusion Risk: Reduced cerebral blood flow due to the elevated head, requiring careful blood pressure management to maintain adequate cerebral perfusion pressure.
- Brachial Plexus Traction: Susceptible to traction neuropraxia if the head is excessively flexed or rotated, or if shoulder retractors are used aggressively.
- Venous Air Embolism: A rare but potentially life-threatening complication, especially in cases of extensive venous exposure or open veins above the heart level.
-
Setup Protocol:
- Table Configuration: The patient is positioned supine, and the operating table is gradually articulated to a 30-70 degree semi-Fowler position. The knees are flexed to approximately 30 degrees, and the hips to 45 degrees, with feet flat on the table or supported, to reduce lumbar lordosis and prevent patient sliding.
- Torso Stabilization: The torso is secured to the table with a wide body strap (e.g., beanbag or Vac-Pac can also be used for additional stability).
- Head and Neck: The head is gently secured in a headrest (e.g., "donut" or gel headrest) in a neutral position, avoiding excessive flexion, extension, or rotation, which can strain the brachial plexus. Tape may be used across the forehead to the bed frame to prevent inadvertent movement. Ensure two fingerbreadths space between the chin and sternum.
- Surgical Arm: The ipsilateral arm is draped free into a sterile stockinette and placed on a stable arm table or bolster, allowing unhindered manipulation by the assistant.
- Contralateral Arm: The contralateral arm is padded and tucked securely to the patient's side, or placed on an arm board at less than 90 degrees abduction to avoid brachial plexus or ulnar nerve compression.
- Pressure Point Padding: Meticulous padding of all pressure points (sacrum, heels, elbows, ankles) is mandatory to prevent nerve damage or skin breakdown.
- Fluid Management: Maintain adequate hydration and avoid hypotension.
2. Lateral Decubitus Position
-
Advantages:
- Reduced Cerebral Hypoperfusion Risk: Head is at a similar level to the heart, minimizing concerns about cerebral blood flow.
- Stability: Often perceived as a more stable position for prolonged procedures.
- Specific Exposures: Can facilitate exposure of posterior structures if needed (though the deltopectoral approach is primary for arthroplasty).
-
Disadvantages:
- Arm Manipulation: May limit the ease and extent of arm manipulation compared to the beach chair, particularly for internal/external rotation and adduction.
- Nerve Compression: Increased risk of compression neuropraxia to the brachial plexus or ulnar nerve in the dependent arm if not properly padded with an axillary roll.
- Airway Management: More challenging for anesthesiologists to access the airway.
- C-Arm Access: Can be more difficult to position the fluoroscope for intraoperative imaging if required.
-
Setup Protocol:
- Patient Placement: Patient is placed on the non-operative side.
- Stabilization: A vacuum-pack beanbag or specialized surgical positioner is used to firmly stabilize the torso to prevent rolling.
- Axillary Roll: A padded axillary roll (not an IV bag) is strategically placed in the dependent axilla, just distal to the head of the humerus, to protect the neurovascular bundle from compression and elevate the chest.
- Head and Neck: The head is supported in a neutral position with padding, ensuring alignment with the spine.
- Lower Extremities: The dependent leg is flexed at the hip and knee, and the upper leg is extended, with a pillow between the knees and ankles.
- Surgical Arm: The ipsilateral arm is suspended in balanced traction (often from an overhead frame or boom) at approximately 30-70 degrees of abduction and 15-20 degrees of forward flexion, allowing for controlled manipulation of the arm during surgery. Ensure traction is not excessive.
- Dependent Arm: The dependent arm is well-padded and positioned on an arm board or tucked, ensuring no compression of the ulnar nerve at the elbow.
- Pressure Point Padding: All bony prominences (lateral malleolus, fibular head, greater trochanter, iliac crest) are meticulously padded.
Regardless of the chosen position, constant vigilance regarding patient alignment, padding, and neurovascular status is paramount throughout the surgical procedure.
Detailed Surgical Approach / Technique
The deltopectoral approach is the workhorse for both anatomic total shoulder arthroplasty (TSA) and reverse total shoulder arthroplasty (rTSA). Each procedure has distinct goals, necessitating variations in glenoid and humeral component preparation and implantation.
I. Deltopectoral Approach (Common for TSA, HHA, rTSA)
- Skin Incision: A curvilinear incision approximately 10-15 cm in length, originating from the posterior aspect of the acromion, extending inferiorly and medially along the natural skin lines of the deltopectoral groove. Precise placement minimizes scarring and optimizes exposure.
- Subcutaneous Dissection: Deepen the incision through the subcutaneous tissue to identify the deltopectoral groove.
-
Identify Deltopectoral Interval:
Locate the
cephalic vein
, which typically lies within this groove. This vein serves as a crucial landmark, as it defines the internervous plane between the deltoid muscle (innervated by the axillary nerve, C5-C6) laterally and the pectoralis major muscle (innervated by the medial and lateral pectoral nerves, C5-T1) medially.
- Vein Management: Most commonly, the cephalic vein is carefully preserved and retracted medially with the pectoralis major muscle. In some cases, lateral retraction with the deltoid, or selective ligation, may be necessary for adequate exposure or to prevent injury.
- Develop the Interval: Incise the clavipectoral fascia medial to the cephalic vein. This exposes the deeper structures.
- Coracoid Process and Conjoined Tendon: Identify the coracoid process, which serves as the origin for the conjoined tendon (coracobrachialis and short head of biceps brachii). These muscles are retracted medially. The musculocutaneous nerve typically enters the deep surface of the coracobrachialis approximately 2-8 cm distal to the coracoid tip and must be protected.
- Retraction: Retract the deltoid laterally and the pectoralis major/conjoined tendon medially. This exposes the subscapularis muscle and the anterior shoulder capsule.
II. Anatomic Total Shoulder Arthroplasty (TSA)
Indications: Primary GHOA with an intact and functional rotator cuff.
-
Subscapularis Management:
The subscapularis tendon must be released to gain access to the glenohumeral joint.
- Subscapularis Tenotomy (Most Common): Identify the superior and inferior borders of the subscapularis tendon. Perform a vertical tenotomy approximately 1-1.5 cm medial to its insertion on the lesser tuberosity. Preserve a cuff of tendon for later repair. Tag the superior and inferior edges of the tendon with strong non-absorbable sutures (e.g., #2 FiberWire) for retraction and subsequent repair.
- Lesser Tuberosity Osteotomy: Less common in primary TSA, this involves excising a small bone block of the lesser tuberosity with the attached subscapularis. This may offer a more robust repair, particularly in revision cases or when tendon quality is poor, but requires precise re-attachment.
-
Anterior Capsulotomy and Humeral Head Dislocation:
- Perform a thorough anterior and inferior capsular release to achieve adequate external rotation. The axillary nerve is vulnerable inferiorly.
- Dislocate the humeral head anteriorly, typically by externally rotating and extending the arm, leveraging it with a Hohmann retractor or specialized dislocator.
-
Humeral Head Osteotomy:
- Using an intramedullary or extramedullary cutting guide, perform the humeral head osteotomy. The goal is to accurately resect the humeral head to the correct retroversion (typically 30-40 degrees relative to the epicondylar axis), inclination (typically 135-140 degrees relative to the humeral shaft), and depth, restoring the native joint line. Precision here is critical for joint kinematics and stability.
-
This intraoperative image illustrates the preparation of the proximal humerus following osteotomy, demonstrating the exposed intramedullary canal ready for reaming and stem insertion. Accurate alignment and depth are crucial at this stage.
-
Humeral Canal Preparation:
- Progressively ream the humeral intramedullary canal to the appropriate size and depth for the chosen humeral stem. Ensure reaming follows the neutral axis of the humerus.
- Impact trial humeral stem and head components.
-
Glenoid Preparation:
- Retract the humerus posteriorly, exposing the glenoid fossa. Use dedicated glenoid retractors to protect the anterior deltoid and, critically, the axillary nerve inferiorly.
- Remove all peripheral osteophytes from the glenoid rim to allow for full exposure and proper seating of the glenoid component.
- Address glenoid bone loss and correct retroversion/inclination using reamers. For significant posterior wear (e.g., Walch B2 glenoids), eccentric reaming can be used to centralize the glenoid, potentially requiring augmented glenoid components or bone grafting for severe defects. The goal is to create a well-centered, stable platform for the glenoid component.
- Drill pilot holes for the central peg/keel and peripheral pegs/screws of the glenoid component.
- Cemented Glenoids: Common for good bone quality. Ensure the glenoid surface is clean and dry. Apply bone cement (polymethylmethacrylate) to the posterior surface of the glenoid component and press it firmly onto the prepared glenoid, ensuring complete coverage and appropriate seating. Remove all excess cement from the joint.
- Uncemented/Press-Fit Glenoids: Used in specific cases with excellent bone quality. Requires a precise fit and firm impaction for initial stability, often augmented with screws.
-
Definitive Component Implantation:
- Insert the chosen glenoid component, ensuring ideal version and inclination.
- Insert the final humeral stem (cemented or uncemented).
- Attach the final humeral head component to the stem.
-
Reduction and Stability Assessment:
- Carefully reduce the humeral head onto the glenoid.
- Perform a comprehensive stability assessment throughout the entire range of motion (flexion, extension, abduction, adduction, internal, and external rotation). Ensure the joint is stable, well-tracked, and impingement-free. Adjust soft tissue releases or component sizing if necessary.
- Subscapularis Repair: A robust repair of the subscapularis tendon is critical for anterior stability and function. Reattach the tendon using strong non-absorbable sutures (e.g., #2 FiberWire in a mattress or figure-of-eight fashion) to the lesser tuberosity footprint or drill holes in the bone.
- Wound Closure: Layered closure of the deltopectoral interval, subcutaneous tissues, and skin.
III. Reverse Total Shoulder Arthroplasty (rTSA)
Indications: Rotator cuff deficient arthropathy (CTA), severe glenoid bone loss, failed TSA with rotator cuff deficiency, complex proximal humeral fractures in elderly. Requires a functional deltoid.
- Initial Approach & Humeral Head Dislocation: Same deltopectoral approach as TSA. Humeral head dislocated anteriorly.
-
Humeral Head Osteotomy:
- Perform the humeral head osteotomy according to the specific implant system's guidelines, often with a slightly greater inclination angle (e.g., 155 degrees) than TSA. The goal is to remove the arthritic head and prepare for the humeral tray, often removing less bone distally to preserve more metaphyseal bone.
-
Glenoid Preparation:
- Thoroughly expose the glenoid. Remove all glenoid osteophytes.
-
Glenosphere Baseplate Placement:
The primary step is to establish a stable foundation for the glenosphere.
- Drill a central guide wire into the central glenoid, ensuring proper orientation to match the scapular spine.
- Ream the glenoid to a flat surface that allows for optimal seating of the glenosphere baseplate.
- The baseplate is typically positioned inferiorly on the glenoid and with 5-10 degrees of inferior tilt to maximize glenoid bone contact and minimize the risk of scapular notching (impingement of the humeral polyethylene liner on the inferior scapular neck).
- Drill holes for the baseplate locking screws. Secure the baseplate with bicortical screws for maximum stability.
- Glenosphere Implantation: Impact the glenosphere onto the baseplate. Glenosphere size and lateralization (using shims or lateralized baseplates) are chosen to optimize deltoid tension, improve external rotation, and avoid impingement, balancing the risk of notching. The CoR is effectively moved medially and distally.
-
Humeral Preparation:
- Ream the humeral intramedullary canal for the chosen humeral stem. Ensure appropriate sizing and depth.
- Insert the trial humeral stem and then the humeral tray with a polyethylene liner. Humeral tray retroversion is typically neutral or slight retroversion (0-10 degrees).
-
Definitive Component Implantation:
- Insert the final glenosphere and secure the final humeral stem and tray/liner.
-
Reduction and Stability:
- Reduce the humeral component onto the glenosphere.
- Assess range of motion, stability, and deltoid tension. The deltoid should be under appropriate tension to achieve functional elevation without being overtly tight (risk of nerve injury, periprosthetic fracture) or too loose (poor function).
- Ensure impingement-free motion, particularly for internal and external rotation.
- Subscapularis Management: In rTSA for CTA, the subscapularis is often irreparable. If a viable portion remains, a partial repair or imbrication can be performed, but it is not critical for joint stability in the same way as in TSA, as the implant design provides inherent stability.
- Wound Closure: Layered closure.
IV. Humeral Hemiarthroplasty (HHA)
Indications:
Limited, as discussed previously (younger patients, specific fractures).
*
Technique:
The humeral approach, subscapularis management, humeral head osteotomy, and humeral stem implantation are identical to the TSA procedure.
*
Glenoid:
The glenoid is left untouched. Osteophytes are debrided to improve motion. The humeral head prosthesis articulates directly with the native glenoid cartilage.
*
Reduction and Subscapularis Repair:
As in TSA.
Complications & Management
Shoulder arthroplasty, while highly effective, is not without risks. An in-depth understanding of potential complications, their incidence, and evidence-based management strategies is vital for orthopedic surgeons.
| Complication | Incidence (%) | Management Strategy
Microbial Spore Contamination (PJI Risk)
|
|
PJI (Periprosthetic Joint Infection)
|
0.5-2%
|
Early (Acute, <3-4 weeks):
Debridement and implant retention (DAIR) with antibiotics.
Late (Chronic):
One-stage or two-stage revision arthroplasty with prolonged antibiotic regimen.
Unsalvageable:
Resection arthroplasty or shoulder arthrodesis. |
|
Instability / Dislocation
|
1-5%
(higher for rTSA, up to 10% in some series) |
Conservative:
Closed reduction, immobilization, and guided rehabilitation.
Recurrent:
Soft tissue repair/reconstruction, component revision (e.g., larger humeral head, different glenoid/glenosphere offset), conversion from TSA to rTSA for rotator cuff deficiency. |
|
Rotator Cuff Failure (TSA)
|
1-10%
(subscapularis tear most common) |
Partial/Symptomatic:
Conservative management, physical therapy.
Complete/Dysfunctional:
Revision subscapularis repair, conversion to rTSA if irreparable or combined with glenoid loosening. |
|
Nerve Injury
|
1-4%
(axillary nerve most common, brachial plexus) |
Mild Neuropraxia:
Observation, supportive care, nerve conduction studies/EMG. Most recover spontaneously.
Severe/Persistent:
Nerve exploration, neurolysis, or grafting (rarely). |
|
Glenoid Loosening (TSA)
|
5-15%
(aseptic) |
Symptomatic:
Revision glenoid component (with or without bone grafting), conversion to rTSA (especially for B2/C glenoids or cuff deficiency), HHA, or arthrodesis.
Asymptomatic:
Observation. |
|
Humeral Loosening (TSA/rTSA)
|
1-5%
(aseptic) |
Symptomatic:
Revision humeral stem. Consider conversion to rTSA if rotator cuff deficient. |
|
Periprosthetic Fracture
|
<1-5%
(intraoperative or postoperative) |
Intraoperative:
Fixation with cables, plates, or wires around the stem.
Postoperative:
ORIF with plate/screws, sometimes revision stem (longer stem). Depending on stability and location. |
|
Scapular Notching (rTSA)
|
10-70%
(radiographic, clinically symptomatic lower) |
Prevention:
Inferior glenosphere placement, inferior glenosphere tilt, lateralized glenosphere/baseplate.
Management (symptomatic):
Observation for mild, revision for severe cases (often with lateralized components or larger glenospheres). |
|
Acromial/Spine Fracture (rTSA)
|
1-10%
(due to increased deltoid tension) |
Non-Displaced:
Conservative management, immobilization.
Displaced/Symptomatic:
ORIF (challenging due to poor bone quality), potential for revision. |
|
Deltoid Dysfunction (rTSA)
|
<1-5%
|
Partial:
Rehabilitation, physical therapy.
Complete/Avulsion:
Surgical repair, but prognosis often guarded. May necessitate salvage procedures. |
|
Heterotopic Ossification
|
1-20%
(clinically significant lower) |
Prophylaxis:
NSAIDs (e.g., Indomethacin) post-op in high-risk patients.
Management (symptomatic):
Resection if mature and causing impingement/pain after 6-12 months. |
|
Persistent Pain
|
5-10%
| Rule out infection, loosening, instability, nerve injury, referred pain. Consider revision surgery if identifiable mechanical cause, otherwise pain management strategies. |
General Principles of Management:
- Early Diagnosis: Prompt recognition of complications is key to effective management.
- Thorough Work-up: Clinical examination, appropriate imaging (X-rays, CT, MRI, bone scan), laboratory tests (ESR, CRP, joint aspiration for PJI) are crucial.
- Multidisciplinary Approach: Involve infectious disease specialists for PJI, neurologists for nerve injuries, and rehabilitation specialists for all complications.
- Prevention: Meticulous surgical technique, appropriate patient selection, strict adherence to sterile protocols, and careful post-operative rehabilitation are the best preventative measures.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is a cornerstone of successful shoulder arthroplasty outcomes, inextricably linked to the surgical procedure. Protocols are meticulously structured into phases, with specific goals and precautions tailored to the type of arthroplasty performed and the integrity of repaired soft tissues (e.g., subscapularis in TSA). The overarching objectives are to protect the repair, restore range of motion, regain strength, and optimize functional return.
General Principles Across All Protocols:
- Pain Management: Crucial for patient participation; often includes multimodal analgesia.
- Protection of Surgical Repair: Emphasizing non-weight bearing, avoiding sudden movements, and respecting tissue healing timelines.
- Progressive Loading: Gradual increase in stress to tissues as healing progresses.
- Patient Education: Empowering the patient with knowledge about their recovery, precautions, and exercise execution.
- Communication: Regular communication between surgeon, therapist, and patient.
Phase 1: Immediate Post-Operative / Protection Phase (0-6 Weeks)
Goals:
* Control pain and swelling.
* Protect the healing tissues (subscapularis repair in TSA).
* Begin gentle, controlled passive range of motion (PROM).
* Prevent capsular adhesions and stiffness.
Precautions:
*
TSA:
No active internal rotation (AIROM), no lifting, no supporting body weight with the operative arm. Avoid excessive external rotation beyond limits set by surgeon (typically 20-30 degrees) to protect subscapularis repair. No extension past neutral.
*
rTSA:
No active internal/external rotation against resistance. Avoid shoulder adduction with internal rotation and extension, which can lead to dislocation. No lifting, no supporting body weight. Protect deltoid integrity if surgically repaired/reattached.
Activities:
*
Sling Immobilization:
Worn continuously, typically for 4-6 weeks, removed only for exercises and hygiene. For rTSA, some surgeons may opt for less strict immobilization depending on deltoid integrity.
*
Pendulum Exercises:
Gentle, gravity-assisted swings.
*
Passive Range of Motion (PROM):
Performed by a therapist or with the non-operative arm.
*
Forward Flexion:
Supine position, up to 90-120 degrees (surgeon dependent).
*
External Rotation:
Supine position, up to 20-30 degrees for TSA (avoiding stress on subscapularis). For rTSA, external rotation may be more limited initially by prosthesis design and deltoid tension.
*
Internal Rotation:
Hand to abdomen.
*
Abduction:
Limited to 90 degrees or less initially.
*
Scapular Mobilization:
Gentle scapular glides and retraction exercises to maintain mobility of the scapulothoracic joint.
*
Elbow, Wrist, Hand AROM:
To prevent stiffness and maintain circulation.
*
Cryotherapy and Edema Control:
Regular icing and elevation.
Phase 2: Intermediate / Controlled Active Motion Phase (6-12 Weeks)
Goals:
* Gradually increase active range of motion (AROM).
* Initiate gentle strengthening of rotator cuff and periscapular muscles.
* Improve neuromuscular control.
Precautions:
* Continue to avoid heavy lifting and sudden movements.
*
TSA:
Continue to protect subscapularis; avoid resisted internal rotation.
*
rTSA:
Emphasize deltoid activation; avoid combined internal rotation, adduction, and extension past neutral to minimize dislocation risk.
Activities:
*
Sling Weaning:
Gradual discontinuation of the sling, as tolerated, particularly for rTSA where deltoid activation is encouraged earlier.
*
Active-Assisted Range of Motion (AAROM):
Progress to patient-assisted exercises using a pulley, cane, or the contralateral hand.
*
Active Range of Motion (AROM):
Initiate gravity-eliminated or supported AROM exercises as control improves.
*
Gentle Isometric Strengthening:
*
TSA:
Isometrics for external rotation (subscapularis protected), abduction, flexion, extension.
*
rTSA:
Isometrics for deltoid (flexion, abduction), and periscapular muscles.
*
Scapular Strengthening:
Retraction and stabilization exercises (e.g., prone scapular squeezes).
*
Proprioceptive Exercises:
Early balance and coordination drills.
Phase 3: Strengthening / Return to Function Phase (12 Weeks - 6+ Months)
Goals:
* Restore full functional ROM (as tolerated by implant and soft tissue status).
* Progressive strengthening of rotator cuff, deltoid, and periscapular muscles.
* Improve endurance and functional capacity.
* Return to light, modified activities.
Precautions:
* Avoid contact sports or activities involving forceful impact.
*
TSA:
Continue to protect subscapularis repair, avoiding heavy, resisted internal rotation for several months.
*
rTSA:
Avoid internal rotation combined with extension and adduction (dislocation risk). Long-term protection of deltoid.
Activities:
*
Progressive Resistive Exercises (PREs):
Using resistance bands, light weights, and bodyweight.
*
TSA:
Focus on external rotation, abduction, flexion, and internal rotation (gradually).
*
rTSA:
Emphasize deltoid strength (forward flexion and abduction) and periscapular muscles. Rotator cuff strengthening is less relevant if irreparable.
*
Functional Exercises:
Incorporate activities that mimic daily tasks, gradually increasing complexity and load.
*
Advanced Proprioceptive Training:
Balance boards, plyometric activities (if appropriate for patient goals and implant type).
*
Cardiovascular Conditioning:
Maintain overall fitness.
*
Return to Activity Counseling:
Gradual return to sports or physically demanding occupations, typically after 6-12 months, with modifications as necessary. High-impact or contact sports are generally discouraged.
Specific Considerations for rTSA Rehabilitation:
- Early Deltoid Activation: Due to the biomechanical shift of the CoR, the deltoid becomes the primary elevator. Early, controlled deltoid activation is crucial for functional recovery.
- Dislocation Precautions: Lifelong adherence to precautions to prevent dislocation (avoiding combined internal rotation, adduction, and extension, especially for anterior/inferior dislocations).
- Scapular Notching: Rehabilitation should avoid activities that excessively impinge the humeral component on the scapular neck.
- Acromial/Spine Fracture Risk: Increased deltoid tension can predispose to these fractures, requiring careful monitoring during strengthening.
The post-operative rehabilitation protocol must be individualized, dynamic, and responsive to the patient's progress, pain levels, and any complications. Close collaboration between the surgeon and physical therapist ensures adherence to protective guidelines while maximizing functional recovery.
Summary of Key Literature / Guidelines
The landscape of shoulder arthroplasty for GHOA has evolved significantly, driven by extensive research and technological advancements. Key literature and professional guidelines provide evidence-based recommendations for optimal patient selection, surgical technique, and post-operative management.
Outcomes and Survivorship:
-
Anatomic Total Shoulder Arthroplasty (TSA):
- Systematic reviews and national joint registries consistently report excellent long-term pain relief and functional improvement with TSA for primary GHOA with an intact rotator cuff.
- 10-year implant survivorship: Typically ranges from 90-95%.
- 20-year survivorship: Ranges from 70-85%.
- Primary modes of failure: Aseptic glenoid loosening (most common), rotator cuff failure (especially subscapularis), and infection. Patient-reported outcomes (PROMs) such as the Constant-Murley Score, ASES score, and SF-36 demonstrate significant and sustained improvements.
-
Reverse Total Shoulder Arthroplasty (rTSA):
- Revolutionized the treatment of cuff tear arthropathy (CTA) and complex proximal humeral fractures.
- Short-to-mid-term outcomes (5-10 years): Excellent pain relief and functional restoration (especially elevation and abduction), often surpassing the outcomes of HHA for CTA.
- 10-year implant survivorship: Varies from 85-95% in recent series.
- Primary modes of failure: Scapular notching (radiographic but not always symptomatic), glenoid baseplate loosening, infection, instability, and acromial/scapular spine fractures.
- Functional gains: While pain relief is consistent, achieving full external rotation can be challenging, and internal rotation is often limited.
Comparison of TSA vs. rTSA:
- For primary GHOA with an intact rotator cuff , TSA remains the gold standard, offering superior restoration of native kinematics and often better external and internal rotation.
- For CTA , rTSA provides more predictable pain relief and functional elevation compared to HHA or failed rotator cuff repairs. Conversion to rTSA for failed TSA (due to rotator cuff failure or glenoid loosening) is a recognized salvage strategy with good outcomes.
Humeral Hemiarthroplasty (HHA):
- Historically, HHA was favored for younger patients or those with severe glenoid bone loss. However, numerous studies have shown that HHA for GHOA provides less consistent and often inferior long-term pain relief compared to TSA due to persistent pain from glenoid arthrosis. Its role is now primarily limited to specific fracture patterns or situations where glenoid implantation is contraindicated.
Glenoid Component Design (TSA):
-
Cemented vs. Uncemented:
- Cemented polyethylene glenoids have a long track record with good survivorship.
- Uncemented (all-polyethylene, metal-backed) glenoids were initially problematic due to high rates of loosening. Modern press-fit, all-polyethylene, or hybrid (glenoid with central screw) designs have shown improved outcomes, but long-term data on uncemented components is still accruing.
- Pegged vs. Keeled: Pegged designs are generally favored as they provide more even stress distribution and greater initial stability compared to keeled designs, potentially reducing loosening rates.
- Augmented Glenoids: For Walch B2 and C glenoids with significant posterior bone loss, augmented glenoid components (wedged designs) or glenoid bone grafting (autografts or allografts) are utilized to restore glenoid version and provide a stable platform, preventing early loosening and posterior subluxation. Long-term results with augmented components are promising but require careful patient selection.
Patient-Specific Factors and Outcomes:
- Age: While advanced age is not a contraindication for arthroplasty, patient physiology and activity level are more important determinants.
- Comorbidities: Smoking, diabetes, obesity, and immunocompromised states are associated with higher complication rates (infection, wound healing issues).
- Pre-operative Functional Status: Patients with higher pre-operative function often achieve better post-operative outcomes.
- Patient Expectations: Realistic expectations are crucial for patient satisfaction.
Current Trends and Future Directions:
- Pre-operative Planning Software and 3D Templating: Increasingly used to virtually plan cases, assess bone loss, and optimize implant positioning, particularly for complex glenoid deformities.
- Navigation and Robotics: Emerging technologies aim to enhance precision in component placement, especially for glenoid reaming and version correction.
- Biologics: Continued research into adjunctive therapies like PRP, bone marrow aspirate concentrate (BMAC), and stem cells, though their definitive role in OA treatment or arthroplasty outcomes remains to be established.
- Conforming Hemiarthroplasty: Newer hemiarthroplasty designs featuring conforming articular surfaces aiming to improve glenoid contact and reduce pain, but long-term comparative data with TSA is limited.
- Short-Stem and Stemless Humeral Components: These designs are gaining traction, potentially offering bone preservation, easier revision, and quicker recovery, particularly for certain patient demographics and bone quality.
Key Guidelines:
- American Academy of Orthopaedic Surgeons (AAOS): While specific comprehensive clinical practice guidelines for shoulder arthroplasty are less prescriptive than for hip or knee, AAOS provides position statements and evidence-based reviews supporting TSA for end-stage GHOA and rTSA for CTA.
- Arthroscopy Association of North America (AANA) and American Shoulder and Elbow Surgeons (ASES): These societies contribute significantly to the literature and consensus recommendations on surgical techniques and post-operative care.
In conclusion, the management of shoulder osteoarthritis is a dynamic field that balances patient-centered care with evidence-based interventions. Non-surgical modalities remain the initial approach, but for advanced disease, shoulder arthroplasty offers predictable and durable solutions, with TSA being the benchmark for intact rotator cuff GHOA and rTSA transforming outcomes for rotator cuff deficient conditions. Continued research, technological innovation, and adherence to meticulous surgical principles and rehabilitation protocols will further refine treatment strategies and improve patient lives.
You Might Also Like