Principles of Orthopedic Oncology: Biopsy, Limb Salvage, and Reconstruction
Key Takeaway
Orthopedic oncology demands a rigorous, multidisciplinary approach to maximize patient survival while optimizing limb function. This comprehensive guide details the critical principles of musculoskeletal tumor management, including biopsy techniques, neoadjuvant protocols, and advanced limb salvage reconstructions. From giant cell tumor curettage to complex periacetabular resections and expandable endoprostheses, mastering these evidence-based surgical strategies is essential for orthopedic residents, fellows, and practicing consultants managing primary sarcomas and metastatic bone disease.
Introduction to Modern Orthopedic Oncology
The surgical management of musculoskeletal tumors has undergone a paradigm shift over the last four decades. Historically, radical amputation was the standard of care for primary bone sarcomas. Today, advancements in neoadjuvant chemotherapy, high-resolution cross-sectional imaging (MRI, PET-CT), and modular endoprosthetic design have made limb salvage surgery the gold standard for over 85% of patients. The foundational literature—spanning Enneking’s staging systems to modern cooperative group trials (e.g., Rizzoli, COSS)—dictates a strict adherence to oncologic principles to prevent local recurrence while maximizing biomechanical function.
This masterclass synthesizes the critical tenets of orthopedic oncology, providing a comprehensive, step-by-step guide to biopsy principles, limb salvage biomechanics, pelvic resections, and the management of both benign aggressive lesions and metastatic bone disease.
Principles of Musculoskeletal Biopsy
The biopsy is the final, and arguably the most critical, staging procedure in orthopedic oncology. A poorly planned biopsy can contaminate neurovascular bundles, compromise limb salvage, and necessitate an otherwise avoidable amputation.
Modalities: FNA, Core Needle, and Open Biopsy
- Fine-Needle Aspiration (FNA): Highly specific for metastatic carcinomas and local recurrences but often yields insufficient architectural detail for primary bone sarcomas.
- Core Needle Biopsy (CNB): The preferred initial modality. Using a Jamshidi or Tru-Cut needle under fluoroscopic, ultrasound, or CT guidance provides a robust tissue cylinder, allowing for immunohistochemistry and molecular cytogenetics (e.g., identifying the t(11;22) translocation in Ewing's sarcoma).
- Open Incisional Biopsy: Reserved for cases where CNB is non-diagnostic. It provides maximum tissue yield but carries the highest risk of hematoma and local contamination.
Surgical Technique and Strict Oncologic Rules
When performing a biopsy, the surgeon must assume the lesion is highly malignant. The biopsy tract must be considered contaminated and must be excised en bloc during the definitive resection.
- Incision: Always use a strictly longitudinal incision. Transverse incisions contaminate multiple extensor/flexor compartments and complicate future wide excision.
- Approach: Traverse the minimum number of anatomic compartments. Go directly through muscle belly rather than intermuscular planes to prevent hematoma tracking along fascial planes.
- Hemostasis: Meticulous hemostasis is mandatory. A post-biopsy hematoma is considered contaminated with tumor cells and expands the required resection volume. Use a tourniquet without exsanguination (elevate only) and deflate prior to closure to ensure absolute hemostasis.
- Bone Window: If cortical bone must be breached, use a round burr or drill to create a circular window. Avoid square windows, which create stress risers and increase the risk of pathologic fracture. Plug the cortical defect with polymethylmethacrylate (PMMA) or bone wax to prevent tumor spillage.
Surgical Warning: Never place a drain after an open biopsy. If a drain is absolutely necessary, it must exit directly in line with the surgical incision so the drain tract can be excised en bloc with the tumor.
Surgical Margins and the Enneking Classification
The Enneking classification of surgical margins remains the cornerstone of musculoskeletal oncology. The margin achieved dictates the risk of local recurrence.
- Intralesional: The dissection passes directly through the tumor (e.g., curettage for Giant Cell Tumor). Leaves macroscopic disease.
- Marginal: The dissection passes through the reactive zone (pseudocapsule) surrounding the tumor. Leaves microscopic satellite lesions.
- Wide: The dissection passes entirely through normal tissue, removing the tumor, its pseudocapsule, and a cuff of normal tissue. This is the standard requirement for primary sarcomas (Osteosarcoma, Ewing's, Chondrosarcoma).
- Radical: The removal of the entire anatomic compartment containing the tumor (e.g., total femur resection).
Management of Primary Bone Sarcomas
Osteosarcoma and Ewing's Sarcoma
High-grade central osteosarcoma and Ewing's sarcoma require a multidisciplinary approach. The standard protocol involves 10 to 12 weeks of neoadjuvant chemotherapy (e.g., MAP: Methotrexate, Doxorubicin, Cisplatin), followed by wide surgical resection, and subsequent adjuvant chemotherapy.
The degree of tumor necrosis observed in the resected specimen (Huvos grading) is the single most important prognostic factor for overall survival.
Limb Salvage vs. Amputation
Limb salvage is indicated when a wide margin can be achieved while preserving a functional limb. Absolute contraindications to limb salvage include:
1. Major neurovascular bundle encasement that cannot be bypassed or reconstructed.
2. Extensive soft tissue contamination from a poorly placed prior biopsy.
3. Infection in the tumor bed.
4. Immature skeletal age where expected leg length discrepancy exceeds 8-10 cm (though expandable prostheses and rotationplasty offer alternatives).
Rotationplasty (Van Nes)
For very young children with distal femoral osteosarcomas, rotationplasty is a highly durable, biologic reconstruction. The distal femur and knee are resected, and the tibia is rotated 180 degrees and fused to the proximal femur. The ankle joint functions as a knee joint, powering a custom below-knee prosthesis. This avoids the complications of multiple lengthening surgeries associated with expandable endoprostheses.
Reconstructive Strategies in Limb Salvage
Following wide resection, the surgeon must reconstruct the skeletal defect to restore biomechanical stability and joint kinematics.
1. Modular Endoprosthetic Reconstruction (Megaprostheses)
Megaprostheses are the workhorse of limb salvage, particularly for the distal femur and proximal tibia.
* Biomechanics: These implants rely on intramedullary stems (cemented or press-fit). Extramedullary porous-coated collars promote extracortical bone bridging, reducing stress shielding and aseptic loosening.
* Expandable Prostheses: In skeletally immature patients, non-invasive expandable prostheses (utilizing electromagnetic fields to lengthen the implant in the outpatient clinic) have revolutionized pediatric limb salvage, mitigating severe leg length discrepancies.
2. Allograft Reconstruction
Massive structural allografts (osteoarticular or intercalary) offer a biologic solution.
* Intercalary Allografts: Used for diaphyseal defects. They are fixed with rigid compression plating or intramedullary nails. Healing occurs via creeping substitution at the host-graft junction.
* Allograft-Prosthetic Composites (APC): Often used in the proximal femur or proximal tibia. The allograft provides bone stock for soft tissue reattachment (e.g., the patellar tendon or abductor mechanism), while the cemented prosthesis provides immediate articular stability.
Clinical Pearl: Soft tissue coverage is paramount in limb salvage. A medial gastrocnemius rotational flap is almost universally required following proximal tibia resections to cover the megaprosthesis and prevent catastrophic deep infection.
Pelvic Tumors and Internal Hemipelvectomy
Primary sarcomas of the pelvis (e.g., Chondrosarcoma, Ewing's) present formidable surgical challenges due to complex 3D anatomy and proximity to visceral and neurovascular structures. Enneking and Dunham classified pelvic resections into four types:
* Type I: Ilium resection.
* Type II: Periacetabular resection.
* Type III: Pubis and ischium resection.
* Type IV: Sacral resection (en bloc with the ilium).
Type II (Periacetabular) Resection and Reconstruction
A Type II resection is the most biomechanically devastating, as it removes the hip joint.
* Positioning: Floppy lateral decubitus, allowing access to both the anterior abdomen (ilioinguinal approach) and the posterior gluteal region.
* Approach: An extended utilitarian approach (e.g., modified Gibson combined with an ilioinguinal) is utilized. The femoral vessels and sciatic nerve must be meticulously mobilized and protected.
* Reconstruction Options:
1. Saddle Prosthesis: Articulates with the remaining ilium. Provides immediate stability but carries risks of superior migration and dislocation.
2. Custom Triflange Components: Patient-specific 3D-printed implants based on preoperative CT scans.
3. Massive Allograft: High rates of nonunion, infection, and fracture.
4. Flail Hip (Pseudarthrosis): No reconstruction is performed. The proximal femur migrates superiorly and scars into the gluteal musculature. While it results in a significant leg length discrepancy requiring a shoe lift, it has the lowest complication rate and allows for reasonable, pain-free ambulation with an assistive device.
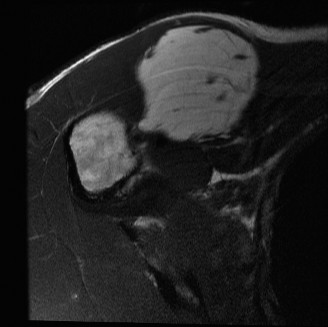
Benign Aggressive Lesions: Giant Cell Tumor of Bone (GCT)
Giant Cell Tumor is a locally aggressive, osteolytic lesion typically occurring in the epiphysis/metaphysis of long bones in young adults (Campanacci staging). While histologically benign, it can cause massive joint destruction and, rarely, metastasize to the lungs.
Surgical Technique: Extended Curettage
The goal is to eradicate the tumor while preserving the native joint.
1. Cortical Window: A large cortical window is created to allow complete visualization of the tumor cavity.
2. Intralesional Curettage: Aggressive removal of all gross tumor using hand curettes.
3. High-Speed Burr: A high-speed burr is used to break down the bony ridges within the cavity, extending the margin by 2-3 mm into normal cancellous bone.
4. Chemical/Thermal Adjuvants: The cavity is treated to kill microscopic residual cells. Options include:
* Phenol (89%): Causes protein coagulation. Must be neutralized with alcohol.
* Cryotherapy (Liquid Nitrogen): Induces cell necrosis via ice crystal formation. Carries a risk of pathologic fracture.
* Argon Beam Coagulation: Provides thermal necrosis to the cavity walls.
5. Reconstruction: The defect is typically packed with PMMA bone cement. Cement provides immediate structural stability, allows for early weight-bearing, and its exothermic reaction during curing provides an additional thermal adjuvant effect. Furthermore, the radiopaque cement allows for easy radiographic detection of local recurrence (which appears as a radiolucent halo around the cement mantle).
Management of Metastatic Bone Disease
The skeleton is the third most common site for metastatic disease (following lung and liver). Breast, prostate, lung, thyroid, and renal cell carcinomas frequently metastasize to bone.
Evaluation and Prognostication
The goal of surgery is palliative: to relieve pain, restore function, and prevent or treat pathologic fractures. The Mirels' Criteria is used to predict the risk of pathologic fracture based on four variables:
1. Site: Upper extremity (1), Lower extremity (2), Peritrochanteric (3).
2. Pain: Mild (1), Moderate (2), Functional/Severe (3).
3. Lesion Type: Blastic (1), Mixed (2), Lytic (3).
4. Size: <1/3 cortex (1), 1/3 to 2/3 cortex (2), >2/3 cortex (3).
A score of 9 or greater is a strong indication for prophylactic surgical fixation.
Surgical Principles for Metastatic Disease
- Renal Cell and Thyroid Carcinoma: These metastases are highly vascular. Preoperative transarterial embolization (within 24-48 hours of surgery) is mandatory to prevent catastrophic intraoperative hemorrhage.
- Diaphyseal Lesions: Best managed with locked intramedullary nailing. The entire bone should be bypassed to protect against future metastases.
- Periarticular Lesions: Extensive destruction of the femoral head or neck is best managed with prosthetic replacement (hemiarthroplasty or total hip arthroplasty) rather than internal fixation, as the irradiated, tumor-laden bone will not heal.
- Adjuvant Radiotherapy: Postoperative radiation is generally required to halt local tumor progression, typically initiated 2-3 weeks post-surgery once the wound has healed.
Postoperative Protocols and Rehabilitation
Rehabilitation in orthopedic oncology is highly individualized, depending on the reconstruction type and the patient's adjuvant therapy schedule.
- Wound Healing: Chemotherapy and radiation severely compromise wound healing. Sutures or staples should remain in place for a minimum of 3 to 4 weeks.
- Weight-Bearing: Cemented endoprostheses allow for immediate weight-bearing as tolerated. Allograft reconstructions require strict non-weight-bearing or partial weight-bearing until radiographic evidence of host-graft union is observed (often 6-9 months).
- Functional Evaluation: The Musculoskeletal Tumor Society (MSTS) scoring system, developed by Enneking, is utilized to objectively evaluate postoperative function, assessing parameters such as pain, range of motion, strength, joint stability, and emotional acceptance.
- Surveillance: Intensive surveillance is required to detect local recurrence and pulmonary metastases. For high-grade sarcomas, this involves physical examination, local imaging (MRI), and chest CT every 3 months for the first 2 years, every 6 months for years 3-5, and annually thereafter.
By adhering to these rigorous, evidence-based principles, the orthopedic oncologist can navigate the complex balance between complete tumor eradication and the preservation of a functional, durable extremity.
You Might Also Like