Chondrosarcoma: Comprehensive Diagnosis, Pathology, and Surgical Management
Key Takeaway
Chondrosarcoma is a malignant cartilage-forming tumor representing approximately 9% of primary bone malignancies. It predominantly affects the pelvis, proximal femur, and proximal humerus in adults aged 40 to 60. Differentiating low-grade chondrosarcoma from benign enchondroma relies heavily on clinical symptoms, specifically non-mechanical pain, and aggressive radiographic features like cortical erosion. Treatment ranges from extended intralesional curettage for low-grade lesions to wide en bloc resection for high-grade and dedifferentiated subtypes.
Introduction to Chondrosarcoma
Chondrosarcoma is a malignant, cartilage-matrix-producing neoplasm of the bone. It constitutes approximately 9% of all primary bone malignancies, making its incidence roughly half that of osteosarcoma. Unlike osteosarcoma, which predominantly affects the pediatric and adolescent populations, chondrosarcoma occurs over a broad age range. The epidemiological peak for primary chondrosarcoma is between 40 and 60 years of age, whereas secondary chondrosarcomas (arising from preexisting benign cartilaginous lesions) typically present earlier, peaking between 25 and 45 years of age.
While chondrosarcoma can manifest in any osseous location, the vast majority are found in the proximal appendicular skeleton and axial skeleton—most notably the pelvis, proximal femur, and proximal humerus. Although primary bone tumors of the hand are exceedingly rare, chondrosarcoma remains the most common malignancy of bone in this specific anatomical location. Similar to the demographic distribution of most bone tumors, there is a slight male predominance, with no significant racial predilection.
Clinical Pearl: The anatomical site of a cartilaginous lesion heavily influences its diagnostic probability. A lesion with identical histological features may be considered a benign enchondroma if located in the phalanges of the hand, but must be treated as a malignant chondrosarcoma if located in the pelvis or proximal femur.
Clinical Presentation and Evaluation
The clinical hallmark of primary chondrosarcoma is insidious, progressive, deep-seated pain. Because these tumors are frequently slow-growing, symptoms may be present for months or even years before the patient seeks formal medical evaluation. A palpable, firm mass may also be present, particularly in pelvic lesions where the tumor can grow to a substantial size before compressing adjacent neurovascular structures.
Pain in the absence of a pathological fracture is a critical clinical discriminator. It is the most reliable clinical symptom to help differentiate a benign, asymptomatic enchondroma from a low-grade chondrosarcoma.
Frequently, orthopedic oncologists receive referrals for patients with asymptomatic cartilaginous lesions discovered incidentally on radiographs or bone scans obtained for unrelated reasons (e.g., trauma, osteoarthritis). While an asymptomatic radiographic abnormality is highly characteristic of an enchondroma, a diagnosis of chondrosarcoma is exceedingly rare in a truly asymptomatic patient. However, if a chondrosarcoma occurs in the exact anatomical area of a previously treated "enchondroma," the original pathology specimen must be urgently reviewed by a specialized musculoskeletal pathologist to rule out an initial misdiagnosis.
Secondary Chondrosarcoma
Secondary chondrosarcomas arise from the malignant transformation of preexisting benign cartilage lesions. They occur most frequently in the setting of genetic syndromes such as multiple enchondromatosis (Ollier disease) and multiple hereditary exostoses (MHE).
- Ollier Disease: Characterized by multiple enchondromas, the incidence of malignant transformation to chondrosarcoma is approximately 25% by age 40.
- Maffucci Syndrome: Characterized by multiple enchondromas associated with soft-tissue hemangiomas. The incidence of malignant transformation is significantly higher than in Ollier disease, approaching 100% over a patient's lifetime in some cohorts.
- Multiple Hereditary Exostoses (MHE): The lifetime incidence of secondary chondrosarcoma arising from an osteochondroma in MHE is estimated at 5%.
- Solitary Osteochondroma: The risk of malignant transformation is approximately 1%.

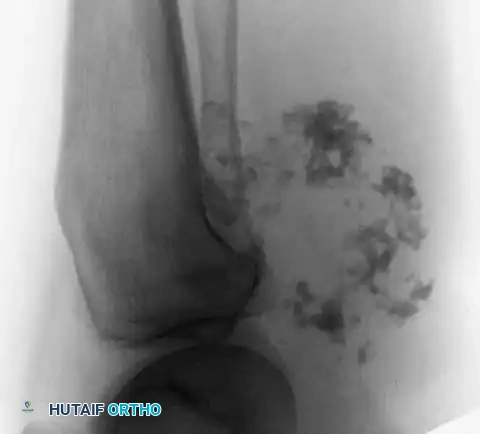
Fig. 22-6 A and B: Anteroposterior and lateral radiographs demonstrating a secondary chondrosarcoma arising from a preexisting osteochondroma in a 67-year-old woman. Note the massive, irregular cartilaginous cap.
Surgical Warning: The size of the cartilaginous cap of an osteochondroma is a critical metric. Evaluated via MRI or CT, a cartilaginous cap thicker than 2 cm in a skeletally mature patient is highly suspicious for secondary chondrosarcoma and warrants immediate biopsy or wide resection.
Other benign conditions occasionally associated with secondary chondrosarcoma include synovial chondromatosis, chondromyxoid fibroma, periosteal chondroma, chondroblastoma, fibrous dysplasia, and previous radiation therapy.
Radiographic and Advanced Imaging
The radiographic appearance of chondrosarcoma is frequently diagnostic. Similar to an enchondroma, it presents as a lesion arising within the medullary cavity featuring irregular matrix calcification. The calcification pattern is classically described as "punctate," "popcorn," "rings and arcs," or "comma-shaped."
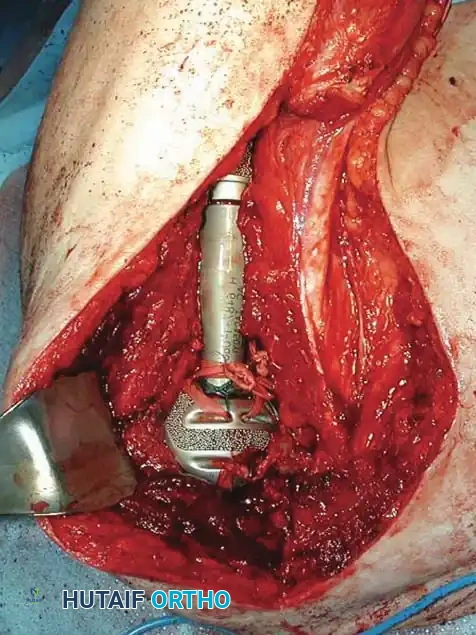
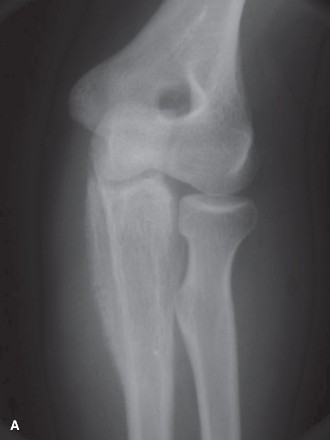
Fig. 22-7 A: Anteroposterior view of the proximal humerus in a 47-year-old man with conventional chondrosarcoma. Note the central calcification and extensive cortical erosion.
Compared to benign enchondromas, chondrosarcomas exhibit a much more aggressive radiographic phenotype. Key malignant features include:
1. Deep Endosteal Scalloping: Erosion of more than two-thirds of the cortical thickness.
2. Cortical Destruction: Frank breakthrough of the cortex.
3. Periosteal Reaction: Often multi-layered or demonstrating a Codman's triangle in high-grade lesions.
4. Soft-Tissue Mass: Indicates extraosseous extension.
Computed Tomography (CT) is invaluable for delineating subtle endosteal erosions and matrix mineralization patterns. Magnetic Resonance Imaging (MRI) is mandatory for assessing the intraosseous extent (skip lesions are rare but possible), soft-tissue extension, and proximity to major neurovascular bundles. On MRI, the high water content of hyaline cartilage results in a characteristic lobulated, hyperintense signal on T2-weighted images and peripheral rim enhancement post-gadolinium.
Histopathology and Subtypes
Histologically, conventional chondrosarcomas are composed of malignant chondrocytes producing an abundant cartilaginous matrix.
Pathology Pitfall: If malignant osteoid is present—even in microscopic amounts—the diagnosis defaults to chondroblastic osteosarcoma. This distinction is paramount, as osteosarcoma requires aggressive neoadjuvant and adjuvant chemotherapy, whereas conventional chondrosarcoma is notoriously chemoresistant.
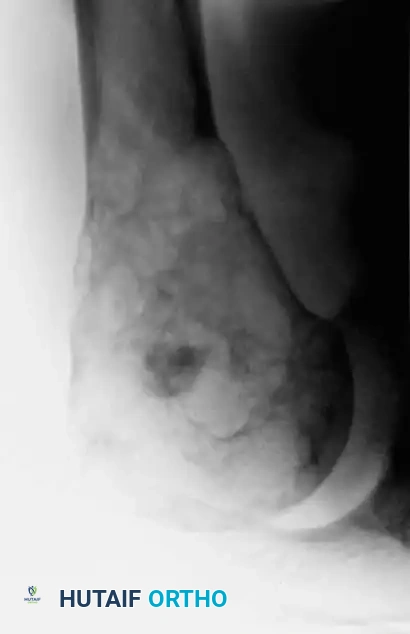
Fig. 22-7 E: Typical microscopic appearance of conventional chondrosarcoma, demonstrating hypercellularity, plump nuclei, and abundant cartilaginous matrix.
Differentiating a low-grade (Grade 1) chondrosarcoma from an enchondroma via a small core needle biopsy can be nearly impossible. Factors favoring a malignant diagnosis include:
* Hypercellularity
* Plump, pleomorphic nuclei
* More than occasional binucleate cells
* A permeative growth pattern
* Entrapment of host bony trabeculae
Less Common Histological Subtypes
Non-conventional subtypes account for less than 20% of all chondrosarcomas but carry distinct prognostic and therapeutic implications:
- Dedifferentiated Chondrosarcoma: A highly lethal variant where a high-grade sarcoma (most commonly osteosarcoma, fibrosarcoma, or undifferentiated pleomorphic sarcoma) arises directly adjacent to a typical low-grade chondrosarcoma. Radiographically, this appears as a bimorphic lesion: a heavily calcified cartilaginous tumor juxtaposed with a destructive, radiolucent, aggressive lytic area.
- Clear Cell Chondrosarcoma: A low-grade malignancy consisting of round cells with abundant clear cytoplasm and distinct borders. It has a strong predilection for the epiphysis (especially the proximal femur) and can be easily confused radiographically and histologically with chondroblastoma.
- Mesenchymal Chondrosarcoma: A high-grade, aggressive tumor consisting of sheets of small, round, blue cells interspersed with islands of benign-appearing cartilage. The cellular regions often exhibit a hemangiopericytomatous ("staghorn") vascular pattern.
Surgical Management
Because conventional chondrosarcoma is highly resistant to both chemotherapy and radiotherapy (due to its slow growth fraction, poor vascularity, and high expression of multidrug-resistance gene P-glycoprotein), surgical resection is the absolute mainstay of treatment.
Management of Low-Grade (Grade 1) Chondrosarcoma
The treatment of low-grade, intracompartmental chondrosarcoma (particularly in the appendicular skeleton) has evolved. Historically treated with wide resection, many centers now report excellent oncologic and functional outcomes using extended intralesional curettage.
Step-by-Step Extended Curettage:
1. Approach: A direct approach to the bone is made, ensuring the biopsy tract is excised in continuity with the cortical window.
2. Cortical Window: A large, oval cortical window is created to allow complete visualization of the medullary canal.
3. Gross Curettage: The macroscopic tumor is meticulously removed using progressively larger curettes.
4. High-Speed Burr: This is the most critical step. A high-speed burr is used to aggressively extend the curettage 2-3 mm into normal-appearing cancellous and cortical bone, eliminating microscopic disease in the bony interstices.
5. Adjuvant Therapy: The cavity is treated with chemical or physical adjuvants to induce further tumor necrosis. Options include:
* Phenol (followed by alcohol neutralization)
* Liquid Nitrogen (Cryotherapy)
* Argon Beam Coagulation
6. Reconstruction: The defect is packed with polymethylmethacrylate (PMMA) bone cement or bone graft. PMMA is often preferred as the heat of polymerization provides an additional thermal adjuvant effect, and it allows for immediate weight-bearing and easier radiographic surveillance for local recurrence.
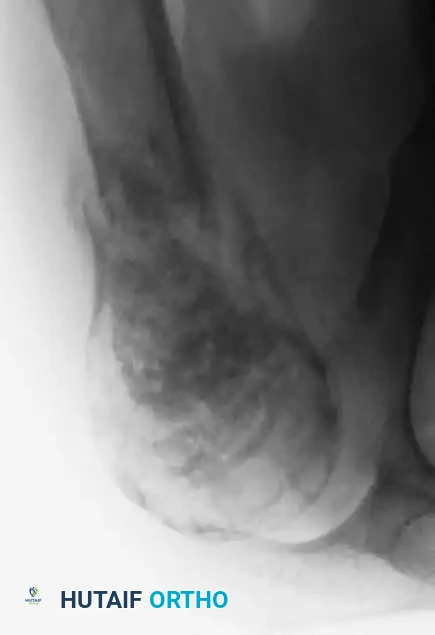
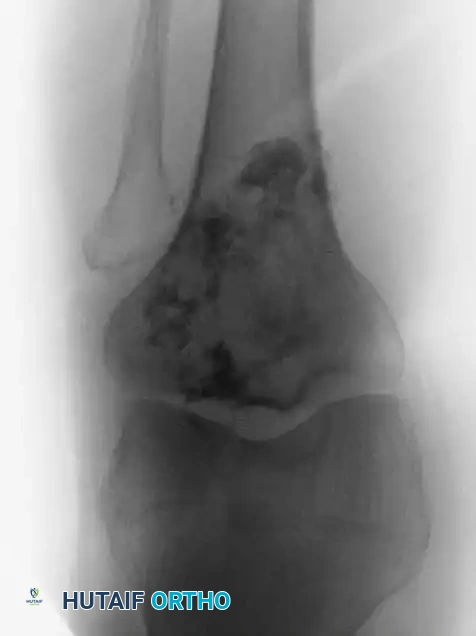
Fig. 22-7 B: A catastrophic failure of treatment. Malignant features were not recognized by the referring surgeon, who performed a simple curettage followed by iliac crest bone grafting without extended margins or adjuvants, leading to massive local recurrence.
Management of High-Grade and Dedifferentiated Chondrosarcoma
High-grade (Grade 2 and 3), dedifferentiated, and pelvic chondrosarcomas require wide en bloc resection with negative margins. Amputation is reserved for cases where neurovascular involvement precludes a limb-salvage procedure with negative margins.
Step-by-Step Wide Resection (Example: Proximal Humerus):
1. Positioning: Beach-chair position with the arm draped free.
2. Incision: An extended deltopectoral approach, incorporating the previous biopsy tract.
3. Neurovascular Dissection: The axillary nerve, radial nerve, and brachial plexus are identified and protected. The cephalic vein is mobilized.
4. Muscular Dissection: The deltoid is retracted laterally, and the pectoralis major medially. The conjoined tendon may be partially released for exposure. Rotator cuff tendons are tagged and transected at a safe oncologic margin from the tumor capsule.
5. Osteotomy: Based on preoperative MRI measurements, the humeral shaft is transected at least 3 cm distal to the most distal extent of the medullary tumor.
6. En Bloc Delivery: The proximal humerus is delivered en bloc, ensuring the tumor capsule is never breached.
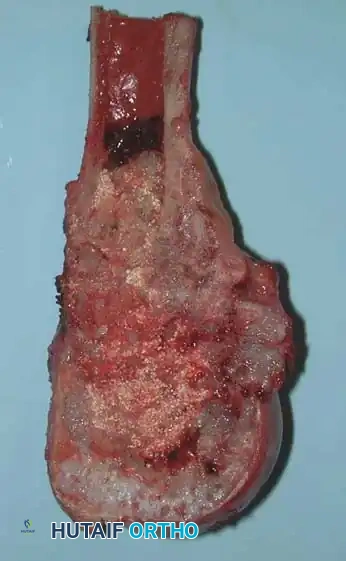
Fig. 22-7 C: Intraoperative photograph demonstrating the surgical bed after wide resection and subsequent endoprosthetic reconstruction of the proximal humerus.
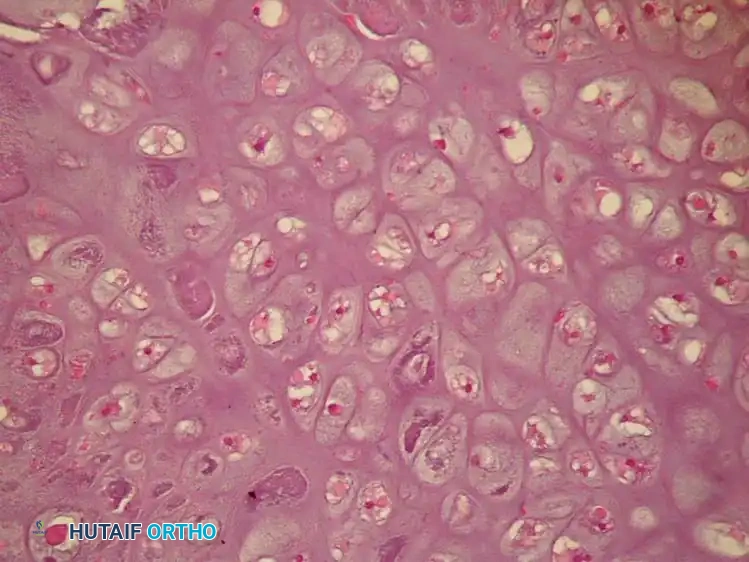
Fig. 22-7 D: The resected gross specimen. Note the large amount of residual, lobulated cartilaginous tumor invading the medullary canal and cortex, despite previous inadequate treatment with simple curettage.
Reconstruction Biomechanics:
Following wide resection, reconstruction of the proximal humerus or femur is typically achieved using a modular endoprosthesis. Endoprosthetic reconstruction allows for immediate stability and early mobilization. In the proximal humerus, a reverse total shoulder arthroplasty design is often utilized if the abductor mechanism (rotator cuff) has been resected, relying on the deltoid for elevation. In younger, high-demand patients, an osteoarticular allograft or allograft-prosthetic composite (APC) may be considered to allow for soft-tissue healing to the allograft bone, though this carries higher risks of nonunion and infection.
Postoperative Protocol and Surveillance
Because cartilage is relatively avascular, chondrosarcoma cells can survive in a dormant state for extended periods. Local recurrences have been documented decades after the primary surgical procedure. Furthermore, a significant percentage of local recurrences exhibit a higher histological grade than the original primary tumor (e.g., a Grade 1 recurring as a Grade 2).
Surveillance Protocol:
* Years 1-2: Physical exam, radiographs of the operative site, and non-contrast CT of the chest every 3 to 4 months.
* Years 3-5: Clinical and radiographic evaluation every 6 months.
* Years 5-10+: Annual evaluation.
Long-term, lifelong follow-up is imperative. Prompt identification of local recurrence or pulmonary metastasis is critical so that salvage surgical treatments can be initiated without delay.
📚 Medical References
- chondrosarcoma and extensive chondroma: report of two cases, Mayo Clin Proc 19:193, 1944.
- Gordon-Taylor G, Monro RS: Technique and management of “hindquarter” amputation, Br J Surg 39:536, 1952.
- Gordon-Taylor G, Wiles P, Patey DH, et al: The interinnominoabdominal operation: observations on a series of fi fty cases, J Bone Joint Surg 34B:14, 1952.
- Karakousis CP, Vezeridis MP: Variants of hemipelvectomy, Am J Surg 145:273, 1983.
- King D, Steelquist J: Transiliac amputation, J Bone Joint Surg 25:351, 1943.
- Lazzari JH, Rack FJ: Method of hemipelvectomy with abdominal exploration and temporary ligation of common iliac artery, Ann Surg 133:267, 1951.
- Littlewood H: Amputations at the shoulder and at the hip, BMJ 1:381, 1922.
- Luna-Perez P, Herrera L: Medial thigh myocutaneous fl ap for covering extended hemipelvectomy, Eur J Surg Oncol 21:623, 1995.
- Masterson EL, Davis AM, Wunder JS, et al: Hindquarter amputation for pelvic tumors, Clin Orthop Relat Res 350:187, 1998.
- Pack GT: Major exarticulations for malignant neoplasms of the extremities: interscapulothoracic amputation, hip-joint disarticulation, and interilio-abdominal amputation: a report of end results in 228 cases, J Bone Joint Surg 38A:249, 1956.
- Pack GT, Ehrlich HE: Exarticulation of the lower extremities for malignant tumors: hip joint disarticulation (with and without deep iliac dissection) and sacroiliac disarticulation (hemipelvectomy), Ann Surg 123:965, 1946; 124:1, 1946.
- Phelan JT, Nadler SH: A technique of hemipelvectomy, Surg Gynecol Obstet 119:311, 1964.
- Pinzur MS, Angelats J, Bittar T: Salvage of failed amputation about the hip in peripheral vascular disease by open wound care and nutritional support, Am J Orthop 8:561, 1998.
- Ross DA, Lohman RF, Kroll SS, et al: Soft tissue reconstruction following hemipelvectomy, Am J Surg 176:25, 1998.
- Sara T, Kour AK, De SD, et al: Wound cover in a hindquarter amputation with a free fl ap from the amputated limb, Clin Orthop Relat Res 304:248, 1994.
- Slocum DB: Atlas of amputations, St Louis, 1949, Mosby. Sorondo JP, Ferré RL: Amputación interilioabdominal, An Orthop Traumatol 1:143, 1948.
- Troup JB, Bickel WH: Malignant disease of the extremities treated by exarticulation: analysis of two hundred and sixtyfour consecutive cases with survival rates, J Bone Joint Surg 42A:1041, 1960.
You Might Also Like