Kienböck Disease: Vascularized Bone Graft & Capitate Shortening Osteotomy Masterclass
Key Takeaway
This masterclass guides fellows through the intricate surgical management of Kienböck disease, combining vascularized bone grafting with capitate shortening osteotomy. We will meticulously detail preoperative planning, patient positioning, comprehensive dorsal wrist anatomy, and the precise, step-by-step intraoperative execution. Critical pearls, potential pitfalls, and comprehensive postoperative rehabilitation strategies are emphasized to optimize outcomes for this challenging condition.
Comprehensive Introduction and Patho-Epidemiology
Kienböck disease, or lunatomalacia, represents a profound and progressive avascular necrosis of the carpal lunate. First described by Robert Kienböck in 1910, this debilitating condition primarily afflicts young adults, typically between the ages of 20 and 40, leading to insidious wrist pain, diminished grip strength, and progressive loss of carpal kinematics. The etiology of Kienböck disease is widely considered multifactorial, driven by a complex interplay of anatomical predispositions, repetitive microtrauma, and precarious vascularity. The lunate, acting as the central keystone of the proximal carpal row, is subjected to immense compressive forces from the capitate distally and the radius proximally. When these mechanical demands exceed the biological capacity of the lunate's delicate intraosseous microcirculation, ischemic necrosis ensues, setting off a cascade of trabecular microfractures, subchondral collapse, and ultimately, severe radiocarpal and midcarpal arthrosis.
Understanding the vascular pathoanatomy is paramount to mastering the surgical management of this condition. Gelberman's seminal injection studies demonstrated that the lunate receives its blood supply through a variable network of volar and dorsal capsular vessels forming a Y-, I-, or X-shaped intraosseous anastomotic pattern. In approximately 20% of the population, the lunate relies on a single dominant volar or dorsal vessel, rendering it highly susceptible to ischemic insult following acute trauma or chronic repetitive loading. Furthermore, biomechanical factors such as ulnar variance play a critical role. While negative ulnar variance is classically associated with Kienböck disease due to increased radiolunate contact stresses, the condition frequently presents in patients with neutral or positive ulnar variance. In these instances, the "nutcracker" effect between the capitate and the lunate fossa remains a primary driver of mechanical overload, necessitating targeted surgical offloading.
The management of Kienböck disease is dictated by the Lichtman classification, which stages the disease based on radiographic and advanced imaging findings. Today’s masterclass focuses on the highly challenging Lichtman Stage IIIA, characterized by lunate sclerosis, cystic changes, and early coronal plane fragmentation, but crucially, without fixed scaphoid rotation or complete loss of carpal height. Most importantly, for the combined procedure we are discussing, the lunate must possess an intact cartilaginous shell. In these specific scenarios, simple mechanical unloading or isolated revascularization is often insufficient. Instead, a dual-modality approach is required: a vascularized bone graft (VBG) to biologically restore perfusion and osteogenesis to the necrotic lunate core, coupled simultaneously with a capitate shortening osteotomy to mechanically decompress the radiolunate joint and protect the delicate neo-vascularization.
This combined approach is designed to halt the inexorable progression of avascular necrosis, promote trabecular healing, and restore normal carpal mechanics before irreversible arthritic changes occur. The capitate shortening osteotomy is our preferred unloading procedure for patients presenting with neutral or positive ulnar variance, as radial shortening in these patients would induce an iatrogenic positive ulnar variance, leading to ulnocarpal impaction syndrome. By resecting a precise 2-millimeter wafer from the capitate waist, we effectively shorten the central column of the wrist, redistributing axial loads away from the fragile lunate and towards the more robust scaphoid and radioscaphoid articulation. This masterclass will exhaustively detail the anatomical, biomechanical, and technical nuances required to execute this complex, limb-salvaging reconstruction.
Detailed Surgical Anatomy and Biomechanics
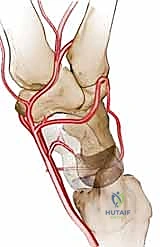
A profound, three-dimensional understanding of the dorsal wrist anatomy is the absolute foundation of a successful vascularized bone grafting procedure. The dorsal hand and wrist comprise a highly complex, layered anatomical region where precise navigation is required to harvest a viable vascularized pedicle while avoiding devastating neurovascular complications. The primary blood supply to the carpus and dorsal distal radius is derived from a rich, interconnected network of anastomotic arches, primarily fed by the radial and ulnar arteries, and supplemented by the anterior and posterior interosseous systems.
Vascular Anatomy of the Dorsal Hand and Carpus
The dorsal carpal arch is the central vascular hub of the dorsal wrist, typically formed by the convergence of the dorsal carpal branch of the radial artery and the dorsal carpal branch of the ulnar artery. This arch lies transversely across the carpus, deep to the extensor tendons and directly upon the dorsal carpal ligaments, slightly distal to the radiocarpal joint line. Protecting this arch during capsulotomy is critical, as it serves as the origin for the dorsal metacarpal arteries (DMAs), which supply the metacarpals and digits, and provides retrograde flow to the distal carpal row.
The dorsal metacarpal arteries, specifically the second, third, and fourth DMAs, arise from the dorsal carpal arch and course distally over the dorsal interosseous muscles. The second dorsal metacarpal artery (2nd DMA) is a historically favored source for vascularized grafting due to its relatively consistent presence and adequate caliber. It travels distally within the fascial layer overlying the second dorsal interosseous muscle. However, utilizing the 2nd DMA requires a more distal dissection and carries a higher risk of injury to the dorsal sensory branches of the radial nerve. Furthermore, the venous drainage accompanying the 2nd DMA can be delicate and prone to congestion if the pedicle is kinked during rotation into the lunate defect.
In addition to the dorsal carpal arch, the deep palmar arch contributes significantly to the carpal blood supply via perforating branches that traverse the intermetacarpal spaces. While these volar structures are not directly visualized during a dorsal approach, understanding their contribution is essential, as they provide collateral flow that ensures the viability of the hand and carpus even when dorsal vessels are mobilized or sacrificed for grafting. The intricate balance of antegrade and retrograde flow within this anastomotic network allows for the remarkable versatility of pedicled flaps in hand surgery.
Vascular Anatomy of the Dorsal Distal Radius
For the revascularization of the lunate, the dorsal distal radius serves as an ideal donor site, providing structurally sound cancellous bone with a robust, predictable vascular pedicle. The blood supply to the dorsal metaphysis of the radius is predominantly derived from the posterior division of the anterior interosseous artery (pAIA), which pierces the interosseous membrane proximal to the distal radioulnar joint and arborizes over the dorsal wrist capsule and distal radius.
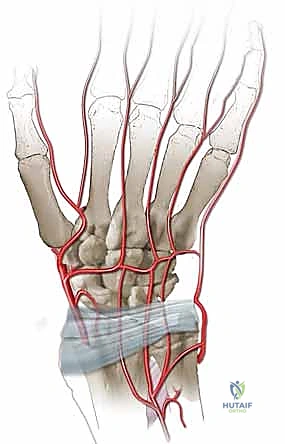
The pAIA gives rise to several critical branches, categorized by their relationship to the extensor retinacular compartments. The 1,2 and 2,3 intercompartmental supraretinacular arteries (ICSRA) course superficial to the extensor retinaculum. While the 1,2 ICSRA is famously used for scaphoid nonunions (the Zaidemberg flap), it is less ideal for the lunate due to its radial position and shorter pedicle length. The 4th and 5th extensor compartment arteries (ECAs), however, are located deep to the extensor retinaculum and are perfectly situated for lunate revascularization.
The 4th ECA travels along the radial floor of the fourth extensor compartment, closely associated with the posterior interosseous nerve (PIN). It provides numerous direct nutrient branches into the dorsal distal radius. The 5th ECA, typically the largest of these dorsal vessels, courses within the fifth compartment or the septum between the fourth and fifth compartments. Importantly, the 5th ECA anastomoses distally with the 4th ECA, the dorsal intercarpal arch, and the radiocarpal arch. This reliable distal anastomosis allows the surgeon to harvest a bone graft from the 4th ECA territory based on retrograde flow from the 5th ECA, providing an exceptionally long, robust pedicle that easily reaches the lunate without tension.
Neurovascular Risks and Osteological Landmarks
Navigating the dorsal wrist requires meticulous attention to adjacent neurovascular structures to prevent iatrogenic morbidity. The posterior interosseous nerve (PIN) is of paramount concern during the harvest of a 4th or 5th ECA-based graft. The PIN lies deep within the fourth extensor compartment, intimately associated with the 4th ECA and the dorsal capsule. While it is primarily a sensory nerve at this level, providing proprioception to the wrist capsule, injury or entrapment in scar tissue can lead to chronic, debilitating dorsal wrist pain. Careful, magnified dissection is required to separate the vascular pedicle from the nerve.
Superficially, the dorsal sensory branch of the radial nerve (DSBRN) and the dorsal cutaneous branch of the ulnar nerve (DCBUN) must be respected. The DSBRN emerges from beneath the brachioradialis in the distal third of the forearm and arborizes over the radial aspect of the dorsal wrist. Retraction injury or inadvertent transection during the initial skin incision or superficial dissection can result in painful neuromas or troublesome areas of anesthesia. Broad, even retraction and avoidance of sharp instruments near these superficial nerves are critical.
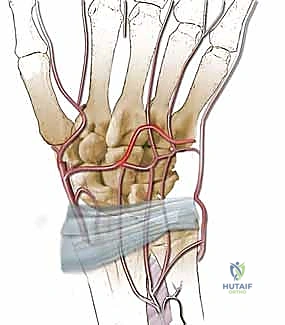
Osteologically, the surgeon must be intimately familiar with the topography of the carpus. Lister's tubercle serves as the primary landmark for identifying the third extensor compartment (extensor pollicis longus) and orienting the dissection toward the fourth and fifth compartments. The capitate, the largest carpal bone, is identified centrally. Its proximal pole (head) articulates with the lunate and scaphoid, while its waist—the narrowest portion—is the target for our shortening osteotomy. The lunate itself must be carefully exposed via a dorsal capsulotomy that preserves the critical scapholunate and lunotriquetral interosseous ligaments, which are essential for maintaining carpal stability following the procedure.
Exhaustive Indications and Contraindications
Patient selection is the single most critical determinant of success in the surgical management of Kienböck disease. The combined vascularized bone graft and capitate shortening osteotomy is a technically demanding, biologically intensive procedure that is only appropriate for a highly specific subset of patients. The primary indication is Lichtman Stage II or IIIA Kienböck disease in a patient with neutral or positive ulnar variance. In these stages, the lunate exhibits sclerosis and potential early fragmentation, but the overall carpal height is maintained, and crucially, the cartilaginous envelope of the lunate remains intact.
The integrity of the lunate cartilage is the absolute prerequisite for this procedure. If the subchondral bone has completely collapsed and the articular cartilage is severely fragmented or absent (Lichtman Stage IIIB or IV), a vascularized bone graft has no structural container to heal within, and the joint will inevitably progress to severe arthrosis regardless of revascularization. In such advanced stages, salvage procedures such as proximal row carpectomy (PRC) or limited intercarpal fusions are indicated. Furthermore, the patient's ulnar variance dictates the choice of unloading procedure. Capitate shortening is specifically indicated for neutral or positive ulnar variance; performing a radial shortening osteotomy in these patients would create an iatrogenic positive variance, leading to ulnocarpal impaction.
Age and systemic health also play vital roles in the decision-making process. The ideal candidate is a young, motivated patient (typically under 40-45 years of age) with robust healing potential. Older patients often have diminished vascular plasticity and a lower potential for successful graft incorporation and lunate remodeling. Smoking is an absolute contraindication for any vascularized bone grafting procedure, as nicotine-induced vasoconstriction and microvascular thrombosis will predictably lead to graft failure. Systemic conditions that impair microcirculation, such as poorly controlled diabetes mellitus, severe peripheral vascular disease, or autoimmune vasculitides, are also strong relative contraindications.
| Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Primary Indications | Lichtman Stage II or IIIA | Lunate architecture is compromised but not completely collapsed; carpal height is maintained. |
| Primary Indications | Intact Lunate Cartilage Shell | Provides a biomechanical container for the graft; prevents immediate progression to radiocarpal arthrosis. |
| Primary Indications | Neutral or Positive Ulnar Variance | Requires unloading via capitate shortening rather than radial shortening to avoid ulnocarpal impaction. |
| Absolute Contraindications | Lichtman Stage IIIB or IV | Fixed scaphoid rotation, severe lunate collapse, or established radiocarpal/midcarpal arthritis precludes joint preservation. |
| Absolute Contraindications | Active Tobacco Use | Nicotine causes severe microvascular vasoconstriction, leading to predictable failure of the pedicled bone graft. |
| Relative Contraindications | Age > 50-60 Years | Decreased biological healing capacity and diminished microvascular plasticity reduce the likelihood of lunate revascularization. |
| Relative Contraindications | Poorly Controlled Diabetes / PVD | Systemic microvascular disease compromises the viability of the delicate 4th/5th ECA or DMA pedicles. |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the cornerstone of executing this complex dual-intervention strategy. The surgeon must synthesize clinical findings with advanced imaging modalities to confirm the disease stage, assess the biological viability of the lunate, and precisely template the capitate osteotomy. A standard radiographic series, including high-quality posteroanterior (PA), true lateral, and oblique views of the wrist, is the starting point. These plain films allow for the calculation of ulnar variance, assessment of carpal height (carpal height ratio), and evaluation of the Lichtman stage based on lunate sclerosis and collapse.
Advanced Imaging Modalities
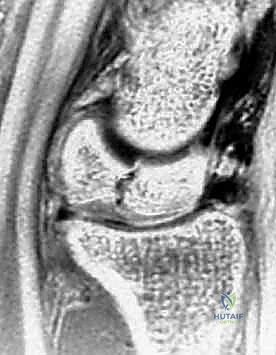
While plain radiographs are essential, Magnetic Resonance Imaging (MRI) is the definitive modality for staging Kienböck disease and determining candidacy for a vascularized bone graft. High-resolution MRI without and with intravenous gadolinium contrast is mandatory. T1-weighted coronal images are utilized to assess the extent of marrow replacement and ischemia; a homogeneous loss of the normal high-signal fatty marrow confirms avascular necrosis.
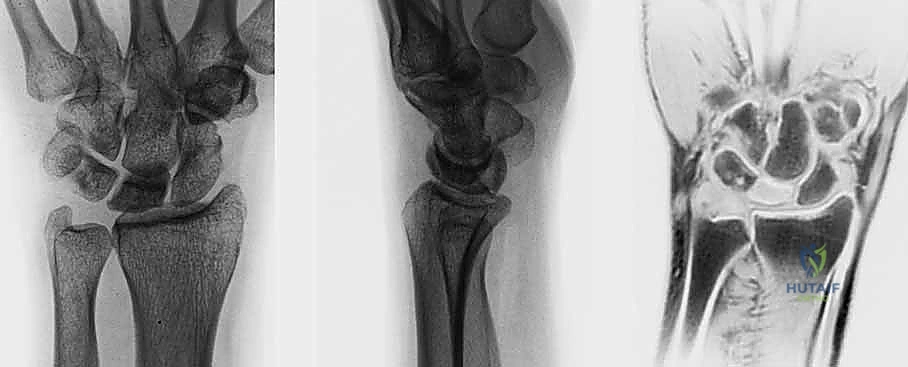
T2-weighted and Short Tau Inversion Recovery (STIR) sequences are critical for identifying occult fracture lines, bone marrow edema, and the presence of cysts within the lunate. Most importantly, high-resolution sagittal and coronal T2-weighted sequences must be rigorously scrutinized to evaluate the integrity of the lunate's articular cartilage envelope.
If the MRI reveals a complete disruption of the dorsal or volar cartilaginous shell, or severe fragmentation of the subchondral plate, the patient is downgraded from a joint-preserving procedure to a salvage operation. Gadolinium-enhanced sequences can occasionally demonstrate residual areas of perfusion within the lunate, which may influence the exact placement of the vascularized graft.
Templating the capitate shortening osteotomy is performed on the preoperative PA radiograph. The goal is to resect a precise 2.0-millimeter wafer of bone from the capitate waist. Resecting less than 1.5 mm may not provide sufficient mechanical offloading, while resecting more than 2.5 mm can lead to excessive laxity of the midcarpal ligaments and subsequent carpal instability. The osteotomy cuts must be planned perpendicular to the long axis of the capitate, ensuring that the proximal and distal fragments will appose flushly without angular deformity once compressed.
Operative Suite Setup and Positioning
The operative setup must facilitate both microsurgical precision and unhindered fluoroscopic imaging. The patient is positioned supine on the operating table with the affected extremity extended onto a radiolucent hand table. The armboard must be completely free of metal components to allow for seamless, multi-planar C-arm fluoroscopy throughout the procedure. A proximal arm tourniquet is applied over generous padding.
Prior to inflation, the limb is elevated for gravity exsanguination for 3 to 5 minutes, rather than using a tightly wrapped Esmarch bandage. Gravity exsanguination is preferred because it leaves a small amount of venous blood within the microvascular network, making the tiny dorsal vessels (such as the 4th and 5th ECAs) slightly engorged and significantly easier to identify and dissect under loupe magnification. The tourniquet is then inflated to 250 mmHg.
The C-arm fluoroscope is draped sterilely and brought in from the head or the foot of the table, depending on the surgeon's preference, but it must be positioned such that true PA and lateral views of the wrist can be obtained instantly without moving the patient's arm. The surgical team must use high-quality loupe magnification (minimum 3.5x, preferably 4.5x) and a surgical headlamp to ensure adequate visualization of the delicate neurovascular structures during the pedicle harvest. Micro-instruments, including fine tenotomy scissors, micro-forceps, and a high-speed water-cooled burr or micro-sagittal saw, must be available on the sterile field.
Step-by-Step Surgical Approach and Fixation Technique
The execution of this combined procedure requires a systematic, unhurried approach. The capitate shortening osteotomy is typically performed first. This sequence is strategic: performing the mechanical unloading first ensures that the midcarpal joint is stabilized and the lunate is decompressed before the delicate vascularized bone graft is harvested and inset, thereby preventing inadvertent disruption of the pedicle during the manipulation required for the osteotomy.
Approach and Extensor Retinaculum Management
A dorsal longitudinal incision, approximately 6 to 8 centimeters in length, is made centered over the radiocarpal joint, extending from the distal radius to the base of the third metacarpal. This single incision provides excellent expansile access to both the capitate for the osteotomy and the dorsal distal radius for the graft harvest. Subcutaneous dissection is performed meticulously using fine scissors, taking great care to identify and protect the crossing branches of the dorsal sensory branch of the radial nerve (DSBRN) radially and the dorsal cutaneous branch of the ulnar nerve (DCBUN) ulnarly. The superficial veins should be preserved wherever possible to minimize postoperative edema.
The extensor retinaculum is exposed. To access the carpus and the distal radius, the retinaculum is typically opened over the third extensor compartment. The extensor pollicis longus (EPL) tendon is identified, mobilized, and retracted radially with a vessel loop. The extensor digitorum communis (EDC) tendons are retracted ulnarly. This exposes the floor of the fourth compartment and the dorsal wrist capsule. A ligament-sparing dorsal capsulotomy is then performed. We prefer a longitudinal or slightly oblique capsulotomy that explicitly avoids transecting the dorsal radiocarpal and dorsal intercarpal ligaments, preserving the essential stabilizers of the proximal carpal row. The capsular flaps are elevated to expose the lunate and the head and waist of the capitate.
Capitate Shortening Osteotomy Execution
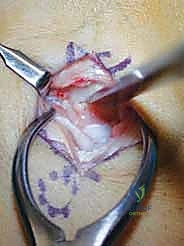
With the capitate exposed, the waist of the bone—its narrowest portion, located just distal to its articulation with the scaphoid and lunate—is clearly identified. Intraoperative fluoroscopy is utilized immediately to confirm the exact level of the planned osteotomy. A true lateral view is essential; the osteotomy should align with the level of the scaphotrapeziotrapezoidal (STT) joint to ensure appropriate mechanical shortening of the central column.

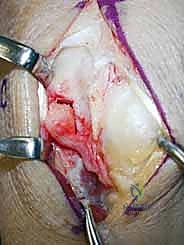
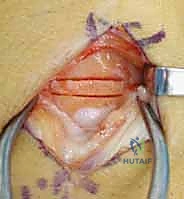
Using a fine, water-cooled micro-sagittal saw or a sharp osteotome, a precise 2.0-millimeter wafer of bone is marked and resected from the capitate waist. It is a critical technical pearl to complete the proximal osteotomy cut first. The proximal pole of the capitate is relatively small and can be difficult to control; making the proximal cut while the bone is still rigidly attached to the distal carpal row provides superior stability. Copious cold saline irrigation is maintained throughout the sawing process to prevent thermal necrosis of the capitate, which could lead to nonunion.
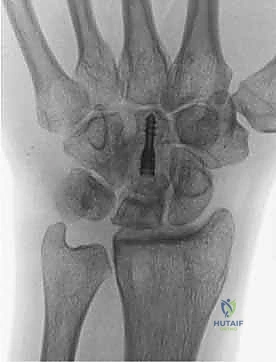
Once the 2.0-mm wafer is removed, the distal pole of the capitate is translated proximally to close the gap. The osteotomy is then provisionally stabilized with a smooth 0.045-inch Kirschner wire. Fluoroscopy is used to confirm excellent apposition of the osteotomy surfaces and appropriate shortening. Final fixation is typically achieved using one or two headless compression screws (e.g., 2.4mm or 3.0mm) placed from dorsal-distal to volar-proximal, ensuring the threads are fully buried beneath the articular cartilage of the capitate head.
Alternatively, multiple crossed K-wires can be used if screw fixation is not feasible, though screws provide superior biomechanical compression and allow for earlier mobilization.
Lunate Preparation and Vascularized Bone Graft Harvest
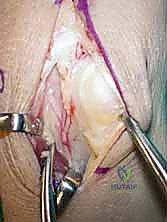
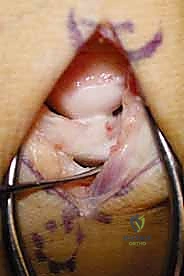
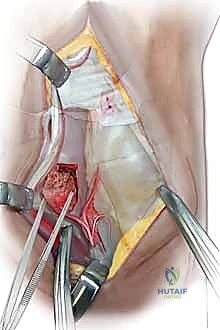
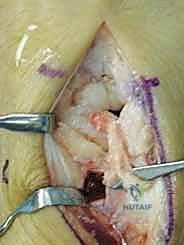
Attention is now turned to the necrotic lunate. Using a high-speed burr or fine curettes, a dorsal cortical window is created in the lunate. The necrotic, avascular cancellous bone is meticulously debrided. It is imperative to remove all sclerotic and non-viable bone to create a healthy, vascularized bed for the incoming graft. However, the surgeon must exercise extreme caution to preserve a 1- to 2-millimeter shell of subchondral bone and the overlying articular cartilage on the volar, proximal, and distal surfaces. Breaching this shell will compromise the structural integrity of the lunate and lead to rapid joint destruction.
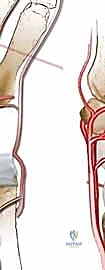
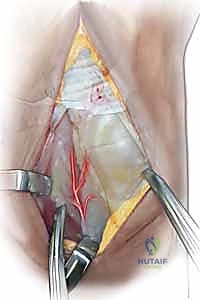
With the lunate prepared, the vascularized bone graft is harvested from the dorsal distal radius. We prefer the 4th/5th ECA pedicled graft. The 5th ECA is identified within the septum between the fourth and fifth extensor compartments. It is traced distally to its anastomosis with the 4th ECA. A bone block, typically measuring 10mm x 5mm x 5mm, is designed over the dorsal metaphysis of the radius, centered on the nutrient branches of the 4th ECA.
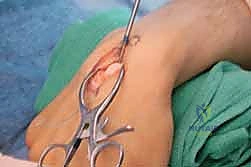
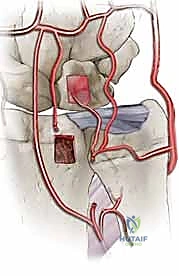
Using fine osteotomes, the cortical-cancellous bone block is carefully elevated. The pedicle is mobilized proximally, taking great care to separate it from the underlying posterior interosseous nerve (PIN). A cuff of periosteum and retinacular tissue is left attached to the bone block and pedicle to protect the delicate microscopic vessels. The tourniquet may be temporarily deflated at this stage to confirm pulsatile bleeding from the cancellous surface of the harvested bone block, ensuring graft viability.
Graft Inset and Final Fixation
The vascularized bone block is then gently rotated distally on its pedicle toward the lunate defect. The pedicle must not be twisted, kinked, or placed under any tension. If the lunate defect is larger than the vascularized
Clinical & Radiographic Imaging Archive