Comprehensive Orthopaedic Management of Cerebral Palsy: Principles, Nonoperative Modalities, and Surgical Strategies
Key Takeaway
The orthopaedic management of cerebral palsy requires a multidisciplinary, patient-specific approach. While the primary central nervous system lesion is static, secondary musculoskeletal deformities are progressive, particularly during rapid growth phases. Treatment encompasses nonoperative modalities like targeted physical therapy, orthotics, and neuromodulators (intrathecal baclofen, botulinum toxin), alongside simultaneous multilevel orthopaedic surgery (SEMLS) to correct contractures, balance muscle forces, and optimize biomechanics while minimizing hospitalizations and psychosocial disruption.
Introduction and Core Principles of Treatment
The orthopaedic management of cerebral palsy (CP) is highly complex due to the profound heterogeneity of the condition. Clinical presentations range from mild hemiplegia with independent ambulation to severe spastic quadriplegia requiring total dependent care. Consequently, generalized treatment algorithms are insufficient; an individualized, patient-centric approach is mandatory. Optimal outcomes are consistently achieved within a multidisciplinary framework encompassing orthopaedic surgery, general pediatrics, neurology, physical and occupational therapy, orthotics, nutrition, and social work.
The foundation of orthopaedic intervention in cerebral palsy rests upon four immutable principles:
- Static Encephalopathy, Progressive Arthropathy: Although the primary central nervous system (CNS) insult is, by definition, nonprogressive, the secondary musculoskeletal deformities—driven by spasticity, abnormal muscle forces, and subsequent contractures—are relentlessly progressive.
- Symptomatic, Not Curative, Intervention: Current orthopaedic and pharmacological treatments address only the secondary musculoskeletal manifestations (the "end-organ" effects) rather than the primary neurological deficit. This reality necessitates careful management of family expectations.
- The Impact of Skeletal Growth: Deformities typically exacerbate during periods of rapid skeletal growth. The longitudinal growth of bone outpaces the elongation of spastic, fibrotic muscle, leading to relative muscle shortening and worsening contractures. When biomechanically feasible, delaying definitive surgical reconstruction until after significant growth spurts can substantially decrease the risk of recurrence.
- Psychosocial Preservation: Both operative and nonoperative treatments must be strategically timed to minimize disruption to the patient’s socialization, psychological development, and education.
Clinical Pearl: The modern standard of care strongly favors a combined approach. Nonoperative modalities are utilized to manage tone and delay contracture progression in the young child, bridging the gap until definitive operative intervention can be performed at an optimal skeletal age.
Nonoperative Management Strategies
Nonoperative modalities—encompassing pharmacotherapy, chemodenervation, orthotics, and physical therapy—are deployed as primary treatments in early childhood and as critical adjuncts to surgical intervention later in life.
Pharmacologic Interventions for Spasticity
Systemic medications are frequently utilized to manage generalized spasticity, though their efficacy is often limited by a narrow therapeutic index and significant side effect profiles. The three most common agents are diazepam, baclofen, and dantrolene.
- Diazepam: A benzodiazepine that acts centrally by potentiating the activity of γ-aminobutyric acid (GABA), a primary inhibitory neurotransmitter.
- Baclofen: A GABA-B receptor agonist that mimics the action of GABA centrally and peripherally, inhibiting both monosynaptic extensor and polysynaptic flexor reflexes.
- Dantrolene: Unlike centrally acting agents, dantrolene acts directly at the level of skeletal muscle by inhibiting calcium ion release from the sarcoplasmic reticulum via the ryanodine receptor. It exhibits an affinity for fast-twitch muscle fibers, selectively decreasing abnormal stretch reflexes.
Systemic Side Effects: Because diazepam and baclofen increase global inhibitory neurotransmitter activity, they frequently cause sedation, cognitive blunting, and balance disturbances. These effects can be profoundly detrimental to a child's ambulation, communication, and educational progress. Dantrolene is used less frequently due to the risk of profound generalized weakness and potential hepatotoxicity with long-term administration.
Intrathecal Baclofen (ITB) Therapy
Due to the dose-limiting systemic side effects of oral medications, targeted drug delivery systems have revolutionized spasticity management. Baclofen penetrates the blood-brain barrier poorly and possesses a short half-life (3 to 4 hours), requiring massive oral doses to achieve therapeutic cerebrospinal fluid (CSF) concentrations.
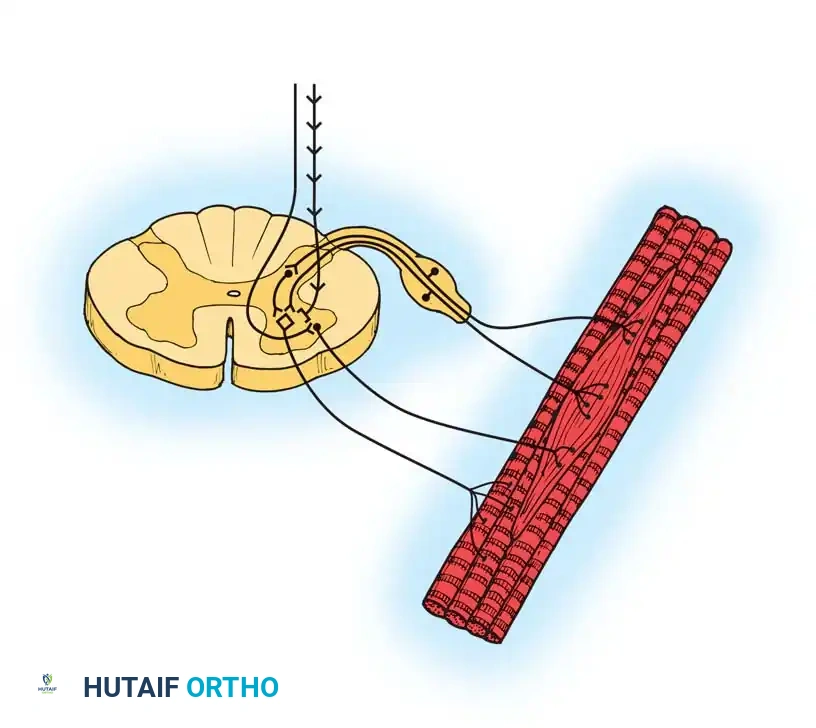
Intrathecal administration bypasses the blood-brain barrier, requiring approximately 1/30th of the oral dose to achieve superior spasticity reduction with minimal systemic sedation. Baclofen acts directly at the spinal cord level to slow abnormal spinal reflexes and decrease anterior horn cell motoneuron drive. Furthermore, it decreases substance P levels in the dorsal horn, effectively limiting nociception.

Surgical Implantation and Management:
The ITB system consists of a programmable pump implanted subcutaneously, typically in the lower abdominal wall, connected to a silastic catheter tunneled into the subarachnoid space of the spinal canal.
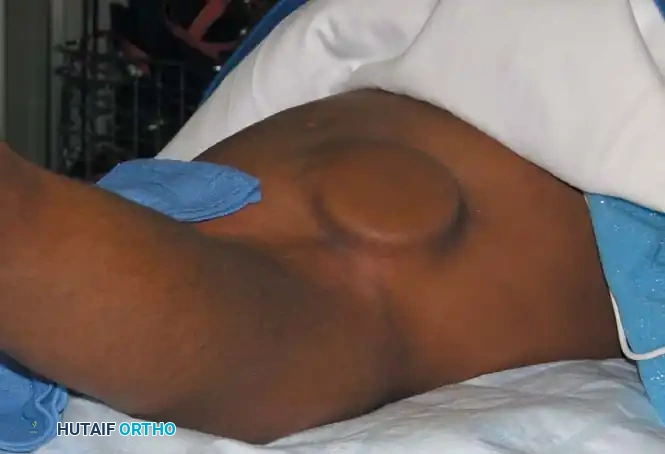
- Indications: ITB is indicated for patients with severe, generalized spasticity (often Gross Motor Function Classification System [GMFCS] levels IV and V) that significantly interferes with self-care, positioning, and quality of life, and who have failed or cannot tolerate oral medications.
- Maintenance: The pump reservoir requires percutaneous refilling every 2 to 6 months, depending on the programmed infusion rate and reservoir size.
- Complications: Approximately 10% to 20% of patients will require revision surgery. Complications include catheter kinking, disconnection, or migration; pump pocket infection; CSF leaks; and potentially life-threatening baclofen withdrawal or overdose. Overdosage presents as profound hypotonia, respiratory depression, and somnolence. There is also ongoing debate regarding the potential for ITB to accelerate the progression of paralytic scoliosis due to the sudden reduction in truncal muscle tone.
Chemodenervation: Botulinum Toxin Type A (BTX-A)
Botulinum toxin is a potent neurotoxin produced by Clostridium botulinum. Of its seven serotypes, Type A (BTX-A) is the most widely utilized in orthopaedics for targeted, reversible muscle weakening.
Mechanism of Action:
BTX-A is injected directly into the spastic muscle belly. It is internalized at the presynaptic motor endplate, where its light chain cleaves SNAP-25, a critical SNARE complex protein. This cleavage irreversibly blocks the vesicular fusion and subsequent release of acetylcholine into the synaptic cleft, resulting in a temporary, flaccid chemical denervation.
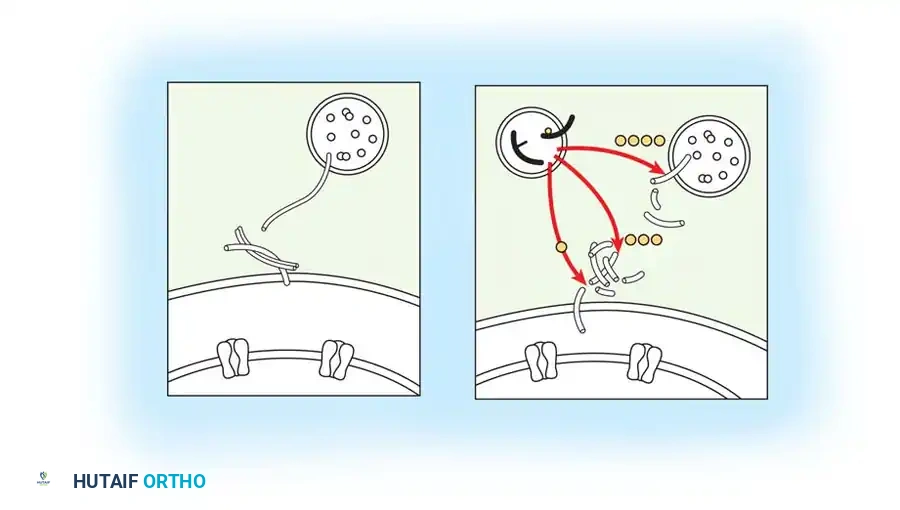
Clinical Application and Dosing:
* Diffusion: BTX-A diffuses 2 to 3 cm within the tissue, making it more forgiving than neurolytic agents like phenol or alcohol, which require precise motor point localization.
* Duration: The clinical effect begins 24 to 72 hours post-injection, peaks at 2 to 4 weeks, and lasts approximately 3 to 6 months as the nerve terminal sprouts new functional endplates.
* Dosing: While primate data suggests a maximum safe dose of 36 to 50 U/kg, clinical guidelines strongly recommend limiting total body doses to less than 15 to 20 U/kg per session to prevent systemic toxicity (respiratory depression).
* Indications: BTX-A is highly effective as an adjuvant to serial casting, bracing, and physical therapy. It is particularly beneficial in young children (ages 2-6) to manage dynamic contractures and delay the need for surgical lengthening until the child is older.
* Contraindications: Fixed myostatic contractures (where the muscle has physically shortened and fibrosed), concurrent use of aminoglycoside antibiotics (which potentiate the neuromuscular blockade), and known resistance/antibody formation. To minimize antibody formation, injections should be spaced at least 3 to 4 months apart.
Physical Therapy and Orthotic Management
Physical Therapy (PT):
PT is the cornerstone of CP management. The therapist is instrumental in spasticity management, contracture prevention, gait training, and caregiver education. For ambulatory patients (GMFCS I-III), goals focus on strengthening antagonists, balance training, and optimizing gait kinematics. For non-ambulatory patients (GMFCS IV-V), goals pivot toward sitting balance, transfer assistance, and facilitating perineal hygiene.
Surgical Warning: There is no robust evidence supporting the efficacy of lifelong intensive physical therapy. Over-prescription of PT can lead to physical burnout for the child and severe financial and emotional strain on the family. Therapy should be goal-directed and episodic (e.g., intensive bursts post-surgery or post-BTX-A injection).
Orthotics:
Bracing is utilized to prevent deformity progression, provide a stable base of support, and improve biomechanical alignment during gait.
* Ankle-Foot Orthoses (AFOs): The most common orthosis in CP. Solid AFOs block both plantarflexion and dorsiflexion, providing maximum stability. Hinged AFOs allow dorsiflexion while blocking plantarflexion, useful for children with adequate knee control but dynamic equinus.
* Ground-Reaction AFOs (GRAFOs): These are critical for managing "crouch gait" (excessive hip and knee flexion with ankle dorsiflexion). By utilizing a rigid anterior shell to block ankle dorsiflexion, the GRAFO forces the ground reaction force vector anterior to the knee joint during the stance phase. This creates a powerful extension moment at the knee, effectively coupling plantarflexion with knee extension and eliminating the crouch.
Operative Management: Principles and Preoperative Optimization
Operative intervention is indicated when fixed contractures, joint subluxation, or bony deformities cause pain, severely impair function, or interfere with activities of daily living and hygiene. Surgery is the only definitive treatment for fixed myostatic contractures.
The Paradigm Shift: Single-Event Multilevel Surgery (SEMLS)
Historically, children with CP underwent sequential, isolated procedures as deformities arose—a phenomenon termed "birthday surgery" by Mercer Rang. This approach resulted in repeated hospitalizations, continuous cycles of pain and rehabilitation, and severe psychological trauma, often leading to "hospital burnout" before the child reached adolescence.
The modern gold standard is Single-Event Multilevel Surgery (SEMLS). This approach involves correcting all orthopaedic deformities—soft tissue and bony—in both lower extremities during a single anesthetic event. SEMLS optimizes the biomechanics of the entire kinetic chain simultaneously, requiring only one major rehabilitation period.
Preoperative Nutritional and Medical Optimization
Patients with CP, particularly those with severe involvement (GMFCS IV-V), frequently suffer from profound malnutrition, gastroesophageal reflux, and restrictive lung disease. Malnutrition exponentially increases the risk of catastrophic postoperative complications, including wound dehiscence, deep surgical site infections, and delayed union of osteotomies.
Clinical Pearl (Jevsevar and Karlin Criteria): Elective orthopaedic surgery should be delayed if the patient exhibits severe malnutrition. A serum albumin level of less than 3.5 g/dL (35 g/L) or a total lymphocyte count of less than 1,500 cells/mm³ (1.5 g/L) is associated with a statistically significant increase in postoperative infections. Preoperative optimization via gastrostomy tube feeding may be required.
Surgical Strategies and Biomechanics
Operative procedures in CP are broadly categorized into four domains: (1) correction of static deformity, (2) balancing muscle power, (3) joint stabilization, and (4) spasticity reduction.
1. Soft Tissue Procedures: Muscle-Tendon Lengthening
Spasticity causes a relative shortening of the musculotendinous unit, shifting the muscle's sarcomere length-tension curve and leading to abnormal joint loading. Surgical lengthening restores a more physiologic resting length, though it inherently weakens the muscle. The choice of lengthening technique is critical:
- Aponeurotic Recession (e.g., Strayer or Baumann procedure for the gastrocnemius): The fascial aponeurosis is transected, but the underlying muscle belly is left intact. This allows for controlled lengthening and preserves muscle continuity, significantly reducing the risk of overlengthening.
- Z-Plasty (e.g., Achilles tendon lengthening): The tendon is split longitudinally and transected at opposite ends. This allows for greater length but carries a high risk of overlengthening.
- Complete Tenotomy (e.g., Adductor longus or Psoas release): Complete transection of the tendon. Reserved for severe contractures or non-ambulatory patients where precise length-tension preservation is less critical.
Step-by-Step: Gastrocnemius Aponeurotic Recession (Strayer Procedure)
1. Positioning: The patient is positioned supine (if concurrent anterior procedures are planned) or prone. A thigh tourniquet is applied.
2. Incision: A 3-4 cm longitudinal incision is made over the posteromedial aspect of the mid-calf.
3. Dissection: The subcutaneous tissues are bluntly dissected. The sural nerve, located posterolaterally, must be identified and protected.
4. Exposure: The paratenon is incised, exposing the gastrocnemius aponeurosis where it merges with the underlying soleus fascia.
5. Release: A transverse incision is made through the gastrocnemius aponeurosis only. The underlying soleus muscle fibers must remain pristine.
6. Correction: The ankle is forcefully dorsiflexed with the knee extended. The aponeurosis will separate, yielding 1 to 2 cm of controlled lengthening.
7. Closure: The paratenon and skin are closed in layers.
Pitfall: Overlengthening the Achilles tendon or gastrocnemius in an ambulatory patient is a devastating complication. It destroys the plantarflexion-knee extension couple, leading to an iatrogenic, irreversible calcaneal crouch gait that is exceptionally difficult to salvage.
2. Bony Reconstruction: Osteotomies and Arthrodesis
Severe, long-standing contractures inevitably lead to bony deformity, such as increased femoral anteversion, tibial torsion, and hip subluxation. Soft tissue release alone is inadequate for these structural changes.
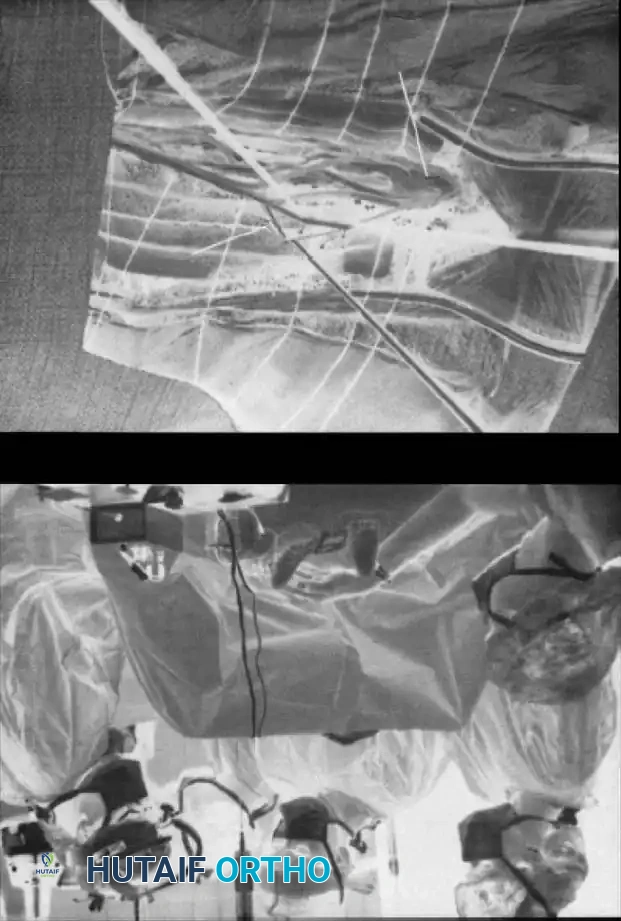
- Hip Reconstruction: Spasticity of the adductors and flexors leads to progressive posterolateral hip subluxation. Treatment requires a Varus Derotational Osteotomy (VDRO) of the proximal femur to redirect the femoral head into the acetabulum, often combined with a pelvic osteotomy (e.g., Dega or San Diego) to improve anterolateral acetabular coverage.
- Derotational Osteotomies: Excessive femoral anteversion (causing internal rotation gait) or external tibial torsion (causing lever-arm dysfunction) are corrected with diaphyseal or metaphyseal derotational osteotomies stabilized with rigid internal fixation (plates and screws or intramedullary nails).
- Foot and Ankle Arthrodesis: In older adolescents with rigid, painful planovalgus or equinovarus foot deformities, joint-sparing osteotomies may fail. Extra-articular subtalar arthrodesis (Grice-Green) or triple arthrodesis provides a stable, plantigrade, and braceable foot.
3. Neuromodulation and Spasticity Reduction (Neurectomy and SDR)
While ITB and BTX-A provide reversible spasticity management, surgical interventions can offer permanent tone reduction.
* Selective Dorsal Rhizotomy (SDR): A neurosurgical procedure involving laminectomy and the selective transection of abnormal sensory nerve rootlets (Ia afferent fibers) in the cauda equina. By interrupting the sensory limb of the reflex arc, SDR permanently reduces spasticity in the lower extremities. It is most effective in young (ages 4-8), ambulatory children with pure spastic diplegia and excellent underlying motor control.
* Peripheral Neurectomy: Selective division of motor nerve branches (e.g., the obturator nerve for severe adductor spasticity). This is less commonly performed today due to the advent of BTX-A, but remains a viable option in severe, non-ambulatory patients to assist with perineal hygiene and prevent hip dislocation.
Postoperative Protocols and Rehabilitation
The success of SEMLS is heavily dependent on rigorous postoperative care.
- Pain Management: Multilevel bony and soft tissue surgery is intensely painful. Continuous epidural analgesia for 48 to 72 hours is the gold standard. Intravenous diazepam is critical for managing severe postoperative muscle spasms, which can otherwise compromise surgical repairs.
- Immobilization: Depending on the procedures performed, patients are immobilized in short leg casts, long leg casts, or hip spica casts. Casts are typically maintained for 3 to 6 weeks to allow for tendon healing and early bony consolidation.
- Mobilization: Early mobilization is encouraged to prevent osteopenia and deconditioning. For soft tissue lengthenings, weight-bearing in casts is often permitted immediately. For major osteotomies, protected weight-bearing is maintained until radiographic union is evident.
- Orthotic Transition: Upon cast removal, patients are immediately transitioned into custom-molded AFOs to protect the lengthened tendons and maintain surgical correction during the vulnerable remodeling phase. Night splinting is often continued for 12 to 24 months postoperatively.
-
Intensive Rehabilitation: A structured, intensive physical therapy program begins immediately after cast removal, focusing on restoring range of motion, rebuilding strength in the newly lengthened muscles, and retraining the central nervous system to adapt to the altered biomechanical alignment. Maximum functional recovery following SEMLS may take 12 to 18 months.
You Might Also Like

