Grand Rounds: Giant Cell Tumor of Bone – A Detailed Diagnostic Case Study
Key Takeaway
Giant Cell Tumor (GCT) diagnosis involves a comprehensive approach, typically starting with clinical presentation like pain and swelling. Imaging, including X-ray, CT, and MRI, reveals characteristic lytic, expansile lesions and assesses soft tissue extension. Definitive diagnosis relies on histopathological confirmation via biopsy, showing multinucleated osteoclast-like giant cells within mononuclear stromal cells.
Grand Rounds: Giant Cell Tumor: Understanding Its Aggression Through a Case Study
Patient Presentation & History
A 28-year-old active male presented to the orthopedic oncology clinic complaining of insidious onset, progressively worsening right knee pain over the preceding six months. The pain was dull, aching in nature, localized to the lateral aspect of the distal femur, and exacerbated by weight-bearing and strenuous activity. He reported nocturnal pain that occasionally disturbed sleep, partially relieved by over-the-counter NSAIDs. There was no history of acute trauma or specific mechanism of injury. He denied fevers, chills, night sweats, or unintentional weight loss. His past medical history was unremarkable, and he was not on any regular medications. He reported no known family history of bone tumors or endocrine disorders. Occupational history involved moderate physical activity.
Clinical Examination
On general examination, the patient was afebrile and hemodynamically stable. Local examination of the right knee revealed mild efflusion and a subtle fullness in the distal suprapatellar region laterally. There was no overlying skin erythema, warmth, or palpable mass. Tenderness was elicited on deep palpation over the lateral femoral condyle.
Range of motion (ROM) of the right knee was actively from 0° extension to 120° flexion, with terminal flexion causing pain. Passive ROM was similarly restricted by pain. Patellar tracking was normal. Ligamentous stability testing (anterior and posterior drawer, varus and valgus stress at 0° and 30° flexion) demonstrated no significant laxity. Meniscal tests were negative.
Neurological assessment revealed intact sensation to light touch and pinprick in the distribution of the femoral, sciatic, peroneal, and tibial nerves. Motor strength was 5/5 in all major muscle groups around the knee and ankle. Distal pulses (dorsalis pedis and posterior tibial) were bilaterally palpable and symmetric. Capillary refill was brisk in all digits. No significant lymphadenopathy was noted.
Imaging & Diagnostics
Initial diagnostic workup commenced with plain radiographs of the right knee.
Radiographs
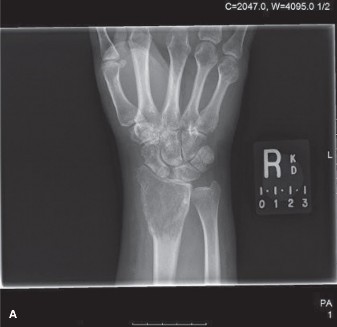
Anteroposterior and lateral radiographs of the right knee demonstrated an eccentric, purely lytic lesion in the metadiaphyseal region of the distal femur, extending into the subchondral bone of the lateral femoral condyle. The lesion was expansile, with a narrow zone of transition, and showed no significant sclerotic rim. Cortical thinning and a pathological stress fracture line were noted along the lateral cortex. There was no evidence of periosteal reaction or matrix mineralization. The epiphyseal plate was noted to be fused.
An example of typical radiographic findings in a similar case is shown:
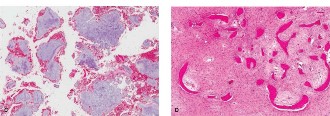
Figure 1: Initial AP radiograph demonstrating an expansile lytic lesion in the distal femur, extending to the subchondral bone, with cortical thinning.
Computed Tomography (CT)
A computed tomography scan of the right femur with intravenous contrast was subsequently obtained to further characterize the bony architecture, cortical integrity, and assess the true extent of the lesion. CT images confirmed a geographic, purely lytic lesion originating in the lateral femoral condyle, measuring approximately 4.5 x 3.0 x 3.5 cm. It showed significant cortical thinning and multiple areas of cortical breach, particularly along the lateral and posterior aspects, with associated subtle extraosseous soft tissue component. The subchondral bone plate was effaced, with tumor extending to within millimeters of the articular cartilage surface. No significant internal calcification or osteoid matrix was evident.
An example of relevant CT findings:

Figure 2: Coronal CT image showing the lytic, expansile nature of the tumor, cortical thinning, and subtle breach, indicating local aggression.
Magnetic Resonance Imaging (MRI)
Magnetic resonance imaging of the right knee and distal femur was performed to evaluate soft tissue extension, marrow involvement, and neurovascular relationships. MRI sequences (T1-weighted, T2-weighted with fat suppression, and post-contrast T1-weighted with fat suppression) revealed a heterogeneous mass with intermediate signal intensity on T1-weighted images and mixed high signal intensity on T2-weighted images, consistent with a solid tumor component interspersed with cystic/hemorrhagic foci. Significant perilesional edema was noted. There was clear evidence of tumor extension into the articular surface and contiguous soft tissues, notably impacting the lateral collateral ligament origin and adjacent musculature. The neurovascular bundle was in close proximity but not overtly invaded. Fluid-fluid levels, often seen in Aneurysmal Bone Cysts, were also intermittently noted, raising the possibility of secondary ABC formation within the GCT.
An example of MRI findings:
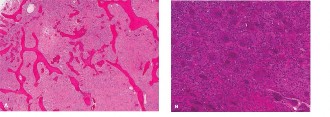
Figure 3: Sagittal T2-weighted MRI image demonstrating the heterogeneous nature of the lesion, high signal intensity, and clear extension towards the articular cartilage.
Biopsy
Given the imaging findings, an open incisional biopsy was performed. Histopathological examination revealed characteristic features of Giant Cell Tumor of Bone: numerous uniformly distributed multinucleated osteoclast-like giant cells within a proliferation of mononuclear stromal cells. Mitotic activity was present but atypical mitoses were absent. No osteoid formation, chondroid matrix, or overt features of malignancy were identified.
Differential Diagnosis
The presentation of a lytic lesion in the metaphysis/epiphysis of a long bone in a young adult necessitates a thorough differential diagnosis. For this case, considering the patient's age and imaging characteristics, the primary differentials include:
| Feature | Giant Cell Tumor (GCT) | Chondroblastoma | Aneurysmal Bone Cyst (ABC) | Telangiectatic Osteosarcoma |
|---|---|---|---|---|
| Typical Age | 20-40 years | 10-25 years | <20 years (primary), any age (secondary) | Any age, peak 10-30 years |
| Location | Epiphysis/Metaphysis (subchondral), often around knee | Epiphysis (ossification center), often around knee/shoulder | Metaphysis, less common in epiphysis (can be secondary to GCT) | Metaphysis (proximal humerus, distal femur, proximal tibia) |
| Radiographs | Purely lytic, eccentric, expansile, narrow zone of transition, no sclerotic rim, often subchondral, rarely periosteal reaction | Lytic, geographic, sclerotic margin often present, fine punctate calcifications (chondroid matrix) | Expansile, lytic, bubbly/multiloculated, thin sclerotic rim, cortical ballooning | Highly lytic, expansile, destructive, often with cortical breach and soft tissue mass, periosteal reaction (Codman's triangle/sunburst), often mimics ABC |
| CT | Cortical thinning/breach, purely lytic, no matrix | Cortical remodeling, calcifications common, often well-defined | Fluid-fluid levels characteristic (blood/serum), expansile, cortical destruction | Significant cortical destruction, soft tissue mass, potential mineralization of osteoid/chondroid matrix, fluid-fluid levels possible |
| MRI | Heterogeneous, intermediate T1, high T2, variable enhancement, sometimes fluid-fluid levels (if secondary ABC) | Low to intermediate T1, high T2 with surrounding edema, strong enhancement, calcification seen as low signal | Prominent fluid-fluid levels (diagnostic), internal septations, peripheral enhancement | Heterogeneous, infiltrative, T1 low, T2 high, strong enhancement, large soft tissue component, often hemorrhagic areas, fluid-fluid levels |
| Histology | Numerous uniform multinucleated osteoclast-like giant cells, monomorphic stromal cells. | Polyhedral chondroblast-like cells, giant cells, "chicken-wire" calcification. | Blood-filled cystic spaces separated by fibrous septa with spindle cells, giant cells, and osteoid. No endothelial lining. | Malignant spindle cells, pleomorphism, abundant telangiectatic spaces filled with blood, atypical giant cells, osteoid/chondroid matrix |
| Aggressiveness | Locally aggressive, high recurrence rate, rare metastasis (lungs) | Benign, low recurrence | Benign, locally aggressive, high recurrence rate, can be primary or secondary | Highly malignant, high metastatic potential |
Surgical Decision Making & Classification
Given the diagnosis of Giant Cell Tumor of Bone (GCT), a locally aggressive benign tumor with a high propensity for local recurrence and potential for malignant transformation or pulmonary metastasis (in rare cases), surgical intervention was deemed essential. The tumor's involvement of the subchondral bone and cortical breach, classified as Enneking Stage 2 (active) or 3 (aggressive, local recurrence) depending on interpretation of intra-operative findings and the full extent of soft tissue involvement, necessitated a comprehensive approach. Non-operative management with observation or intralesional ablation alone without formal curettage was not appropriate due to the size, active growth, and potential for pathological fracture or joint destruction.
The primary goals of surgery were:
1.
Complete intralesional tumor removal:
Aggressive curettage to minimize recurrence.
2.
Adjuvant therapy application:
To further reduce recurrence rates, especially given the aggressive nature and subchondral involvement.
3.
Restoration of structural integrity:
Bone grafting or cementation to fill the defect and prevent collapse.
4.
Preservation of joint function:
Minimizing damage to articular cartilage and surrounding soft tissues.
Considering the patient's young age, the intra-articular and metadiaphyseal location, and the importance of preserving knee function, extended intralesional curettage with high-speed burring, adjuvant chemical cauterization (e.g., phenol), and cement augmentation was chosen as the optimal surgical strategy. Wide en bloc resection with subsequent prosthetic arthroplasty, while definitive for tumor control, was considered too morbid given the extent of involvement and would significantly compromise knee function unnecessarily at this stage. Denosumab, a RANKL inhibitor, was considered as a neoadjuvant or adjuvant therapy, but primary surgical management was prioritized due to the potential for rapid progression. The decision was made to proceed with surgery and consider Denosumab post-operatively in the event of high-risk features for recurrence or if recurrence occurred.
Surgical Technique / Intervention
The patient was positioned supine on a radiolucent operating table. General endotracheal anesthesia was administered. A high thigh tourniquet was applied to the right lower extremity. The limb was prepped and draped in a standard sterile fashion to the groin.
- Approach: A standard lateral parapatellar approach to the distal femur and knee joint was utilized. A longitudinal skin incision was made, centered over the lateral femoral condyle, extending from the vastus lateralis muscle proximally to just distal to the joint line. The fascia lata was incised, and the vastus lateralis was reflected anteriorly to expose the lateral femoral cortex. The common peroneal nerve was identified and protected.
- Cortical Window Creation: Intraoperative fluoroscopy was used to localize the precise extent of the lesion. A cortical window, approximately 3x2 cm, was carefully created using an osteotome and Kerrison rongeurs on the lateral aspect of the distal femur, avoiding entry into the articular surface. The window was designed to provide direct access to the entire tumor cavity.
-
Extended Intralesional Curettage:
Once the cortical window was created, the tumor tissue was identified. Grossly, the tumor was reddish-brown, soft, and friable. A curette was used to meticulously scrape out all visible tumor tissue from the bony cavity. This process was performed in a systematic manner, ensuring all walls of the cavity were debrided. Attention was paid to the subchondral bone, which was curetted aggressively but carefully to avoid breaching the articular cartilage.
Following initial curettage, a high-speed burr was used to further debride the tumor bed, creating a clean, sclerotic-like rim around the entire cavity. This mechanical adjuvant helps destroy microscopic tumor cells embedded in the bone trabeculae. -
Adjuvant Therapy:
After thorough burring and irrigation, the cavity was meticulously dried. Phenol (absolute alcohol, 90%) was applied to the entire tumor cavity for 30 seconds to one minute using gauze pledgets on a stick, followed by copious irrigation with sterile saline and hydrogen peroxide to neutralize the phenol. This chemical cauterization acts as an oncologic adjuvant to kill residual microscopic tumor cells. Care was taken to protect surrounding soft tissues during phenol application.
An example of the intraoperative appearance after thorough curettage and adjuvant application:
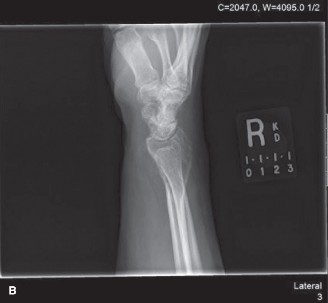
Figure 4: Intraoperative view after aggressive curettage and application of adjuvant, showing the clean bony cavity ready for reconstruction. - Cavity Reconstruction: The defect, measuring approximately 4.5 cm in its largest dimension, was then filled. Polymethylmethacrylate (PMMA) bone cement was mixed and impacted into the cavity under controlled pressure. The use of PMMA offers several advantages: it provides immediate structural support, reduces the risk of pathological fracture, and the exothermic reaction generated during polymerization may have an additional cytotoxic effect on residual tumor cells. A small amount of cancellous allograft chips were also mixed with the PMMA to promote osteoconduction at the margins. The cement was molded to fill the defect and restore the contour of the femoral condyle, with particular attention to supporting the subchondral bone.
- Closure: The cortical window fragment was not replaced. The vastus lateralis muscle was reapproximated, and the fascia lata closed. Subcutaneous tissues and skin were closed in layers. A sterile dressing was applied. The tourniquet was deflated, and hemostasis was secured.
Post-Operative Protocol & Rehabilitation
Immediate Post-Operative Period (Weeks 0-2)
- Weight Bearing: Non-weight-bearing on the operative limb, protected in a hinged knee brace locked in extension.
- Pain Management: Multimodal analgesia including NSAIDs, acetaminophen, and opioid analgesics as needed.
- Thromboprophylaxis: Chemical (e.g., LMWH) and mechanical (e.g., SCDs) thromboprophylaxis initiated.
-
Physical Therapy:
- Initiate active ankle pumps and isometric quadriceps/hamstring exercises.
- Gentle passive range of motion (PROM) for knee flexion (0-30 degrees) to prevent stiffness, initiated cautiously by a therapist.
Early Rehabilitation (Weeks 2-6)
- Weight Bearing: Gradual progression to touch-down weight-bearing (TDWB) with crutches, followed by partial weight-bearing (PWB) as tolerated, guided by pain and radiographic stability. The knee brace may be unlocked for controlled range of motion.
-
Physical Therapy:
- Increase knee ROM (aiming for 0-90 degrees by 6 weeks).
- Quadriceps and hamstring strengthening (closed kinetic chain exercises where appropriate).
- Proprioception and balance exercises.
- Scar management.
Intermediate Rehabilitation (Weeks 6-12)
- Weight Bearing: Progress to full weight-bearing (FWB) as tolerated, typically by 8-10 weeks, transitioning off crutches.
-
Physical Therapy:
- Continue to advance knee ROM and strengthening exercises.
- Initiate light functional activities (e.g., stationary cycling with low resistance).
- Focus on gait training and normalization of movement patterns.
Advanced Rehabilitation (Weeks 12+)
- Return to Activity: Gradual return to activities of daily living and work.
-
Physical Therapy:
- Progress to higher-level strengthening and conditioning.
- Sport-specific training, if applicable, with appropriate bracing for protection.
- Monitoring: Close radiographic and clinical follow-up is crucial for surveillance of local recurrence and pulmonary metastasis. Typically, radiographs are obtained at 3, 6, 12 months, and then annually for at least 5-10 years. Chest CT is recommended annually for the first 3-5 years to screen for pulmonary metastases, which, though rare, can occur.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- Location, Location, Location: GCT is classically an epiphyseal/meta-epiphyseal lesion in young adults (20-40 years old) after physeal closure, frequently found around the knee (distal femur, proximal tibia).
- Radiographic Features: Purely lytic, eccentric, expansile, narrow zone of transition, no sclerotic rim. Subchondral involvement is characteristic.
- Histology is Key: Definitive diagnosis requires biopsy showing numerous uniformly distributed multinucleated osteoclast-like giant cells and a monomorphic stromal cell population.
- Local Aggression: Despite being histologically benign, GCT is locally aggressive with a significant risk of recurrence (20-50% with intralesional curettage alone).
- Adjuvant Therapy: Essential for reducing recurrence rates. High-speed burr, phenol, cryotherapy, and argon beam coagulation are commonly used. PMMA cement offers structural support and an additional thermal cytotoxic effect.
- Denosumab: A RANKL inhibitor, can be used pre-operatively to devitalize tumor cells and facilitate intralesional surgery, or for recurrent/unresectable disease, and in cases of pulmonary metastasis. It can, however, make subsequent surgery more challenging due to increased bone sclerosis and can sometimes mask residual viable tumor.
- Pathological Fracture: Management of GCT with pathological fracture usually involves initial stabilization followed by definitive tumor surgery.
- Malignant Transformation: Rare but possible, often after multiple recurrences or radiation therapy.
- Pulmonary Metastasis: Occurs in 2-5% of cases, typically histologically benign but requires close follow-up with chest CT. These are often amenable to surgical resection.
Pitfalls
- Inadequate Curettage: The most common cause of local recurrence. Aggressive, thorough curettage of all tumor tissue, especially from corners and subchondral areas, is paramount.
- Failure to Use Adjuvants: Relying solely on curettage, particularly for aggressive lesions, significantly increases recurrence risk.
- Misdiagnosis: Confusion with other lytic lesions (e.g., ABC, chondroblastoma, fibrous dysplasia, brown tumor of hyperparathyroidism, or even telangiectatic osteosarcoma) can lead to inappropriate management. Always verify with biopsy.
- Articular Cartilage Breach: Aggressive curettage in the subchondral region risks damaging the articular cartilage, leading to premature osteoarthritis. Careful technique and fluoroscopic guidance are essential.
- Neurovascular Injury: Large lesions, especially around the knee, can abut neurovascular structures. Meticulous dissection and protection are critical.
- Post-operative Instability/Fracture: Large bone defects, especially after extensive curettage, may require robust reconstruction (cement, bone graft, internal fixation) to prevent collapse or fracture.
- Complacency in Follow-up: The risk of local recurrence and rare pulmonary metastasis necessitates long-term, diligent follow-up with clinical examination and imaging (plain radiographs, chest CT).
- Denosumab-induced Sclerosis: While beneficial, denosumab can make the tumor margins difficult to delineate intraoperatively due to reactive sclerosis, potentially increasing the risk of incomplete resection. Careful planning and imaging correlation are needed.
