Hemophilic Arthropathy: Understanding Joint Damage, Pathophysiology & Orthopedic Considerations
Key Takeaway
Hemophilic arthropathy is severe joint damage in hemophilia, driven by recurrent intra-articular bleeding. This leads to chronic synovitis, iron deposition, and synovial hypertrophy, which directly degrades articular cartilage and erodes subchondral bone, causing pain, contractures, and severe disability, particularly in knees, elbows, and ankles.
Hemophilic Arthropathy: Understanding & Managing Joint Damage
Introduction & Epidemiology
Hemophilic arthropathy represents the most debilitating complication of severe hemophilia A (factor VIII deficiency) and hemophilia B (factor IX deficiency), both X-linked recessive coagulopathies. While modern prophylactic factor replacement therapy has significantly reduced its incidence and severity, managing established arthropathy remains a formidable challenge for orthopedic surgeons. The core pathology stems from recurrent intra-articular hemorrhage (hemarthrosis), primarily affecting large synovial joints such as the knees, elbows, and ankles. Without adequate treatment, this progressive process invariably leads to chronic pain, synovial hypertrophy, cartilage destruction, subchondral bone erosion, joint contractures, and ultimately severe disability, often culminating in joint ankylosis. Early diagnosis and aggressive management, integrating both medical and surgical interventions, are paramount to preserving joint function and improving quality of life. Understanding the complex interplay between factor deficiency, bleeding events, and the inflammatory cascade within the joint is critical for effective management.
Surgical Anatomy & Biomechanics
The anatomical and biomechanical characteristics of affected joints dictate the clinical manifestations and surgical strategies for hemophilic arthropathy. The knee, elbow, and ankle are most commonly involved dueizing to their complex synovial lining, extensive range of motion, and susceptibility to trauma.
Knee Joint
The knee, a complex hinge joint, is frequently affected. Its large synovial membrane and extensive capsule make it prone to hemarthrosis. Recurrent bleeding leads to synovial hypertrophy, which, in turn, increases the vascularity and fragility of the synovium, creating a vicious cycle of bleeding. The articular cartilage, particularly on the femoral condyles and tibial plateau, is gradually eroded. The intercondylar notch may widen due to chronic inflammation and bone remodeling, and the patella can exhibit inferior squaring due to growth disturbances and chronic effusions. Biomechanically, loss of cartilage and subchondral bone leads to altered joint mechanics, malalignment, and progressive valgus or varus deformity, further exacerbating wear and pain.
Elbow Joint
The elbow, a trochleoginglimoid joint, is also a common site for hemophilic arthropathy. The radiocapitellar and ulnohumeral articulations are particularly vulnerable. Synovial hypertrophy within the olecranon fossa and coronoid fossa can impinge during flexion and extension, leading to contractures. The radial head may show degenerative changes and subluxation. Loss of the normal valgus carrying angle is common, often progressing to cubitus varus. The elbow's role in daily activities, especially lifting and reaching, means even minor functional deficits significantly impact quality of life.
Ankle Joint
The tibiotalar joint, a hinge joint, is frequently affected. The relatively small volume of the ankle joint predisposes it to rapid pressure increases with hemarthrosis, leading to cartilage damage. Talar flattening is a characteristic radiographic finding. Subtalar joint involvement, though less common, can also contribute to hindfoot pain and deformity. Progressive ankle dorsiflexion and plantarflexion limitations impair gait and balance.
Pathophysiology of Joint Damage
The seed content correctly identifies the core pathophysiology:
1.
Recurrent bleeds and chronic synovitis:
Intra-articular hemorrhage provides iron to the synovial lining.
2.
Synovial hypertrophy/hyperplasia:
Iron-laden macrophages (type A synovial cells) and fibroblasts proliferate, creating a thickened, friable, hypervascular synovium. This hypertrophic synovium is itself prone to bleeding and produces pro-inflammatory cytokines, proteases (e.g., collagenases, metalloproteinases), and angiogenesis factors.
3.
Iron deposition:
Hemosiderin deposition within the synovium is central to the inflammatory process. Iron acts as a catalyst for free radical formation, causing oxidative stress and cellular damage.
4.
Cartilage destruction:
The inflamed, hypertrophic synovium directly invades and degrades articular cartilage, leading to chondrocyte apoptosis and extracellular matrix breakdown. Subchondral bone erosion and cyst formation follow. This process differentiates it from typical osteoarthritis where cartilage degradation is primary.
5.
Subchondral bone changes:
Chronic inflammation and mechanical stress lead to osteopenia, subchondral cyst formation, and eventual osteophyte formation and joint space narrowing.

This illustration depicts a cross-section of a joint, likely showing the progression from synovial inflammation to cartilage and subchondral bone destruction, characteristic of hemophilic arthropathy. The interplay between the hypervascular synovium and the articular cartilage is crucial to understanding the disease progression.
Indications & Contraindications
Management of hemophilic arthropathy follows a spectrum from non-operative prophylaxis to complex reconstructive surgery. Indications for intervention are guided by disease severity, joint function, pain levels, and patient's overall health and factor status.
Non-Operative Indications
Non-operative management forms the cornerstone of care, particularly in early stages, and is often continued post-operatively.
*
Prophylactic Factor Replacement:
This is the primary intervention to prevent bleeds and is indicated for all patients with severe hemophilia to prevent or delay arthropathy.
*
On-demand Factor Replacement:
For acute hemarthrosis, to stop bleeding and prevent further joint damage.
*
Physical Therapy & Rehabilitation:
To maintain range of motion (ROM), strengthen periarticular muscles, improve proprioception, and protect joints.
*
Pain Management:
Non-opioid analgesics, NSAIDs (with caution due to bleeding risk), neuromodulators, and topical agents.
*
Orthotics/Bracing:
To support unstable joints, correct deformities, or provide temporary immobilization after acute bleeds.
*
Radiosynovectomy (Medical Synovectomy):
Indicated for chronic synovitis and recurrent hemarthrosis refractory to factor replacement, particularly in early-stage arthropathy (Pettersson stage I or II) where cartilage is relatively preserved. This is a minimally invasive medical procedure often performed by interventional radiology with orthopedic input.
Operative Indications
Surgical intervention is considered when conservative measures fail, and progressive joint damage significantly impairs function and quality of life.
*
Synovectomy (Arthroscopic or Open):
* Indicated for chronic, hypertrophic synovitis causing recurrent hemarthrosis, pain, and early ROM limitations (Pettersson stage II or III) where conservative management including radiosynovectomy has failed.
* Goal is to remove the inflamed synovium, reduce bleeding, and preserve articular cartilage.
*
Joint Debridement / Arthrolysis:
For symptomatic osteophytes, loose bodies, or adhesions causing mechanical impingement and ROM limitation. Often performed concurrently with synovectomy.
*
Osteotomy (e.g., High Tibial Osteotomy):
For correctable axial malalignment (varus/valgus) in younger patients with unicompartmental disease, aiming to redistribute joint loads and delay arthroplasty.
*
Arthrodesis (Joint Fusion):
* Indicated for severely destroyed, painful, unstable joints where arthroplasty is not feasible or has failed, particularly in younger, high-demand patients or those with recurrent infection.
* Commonly performed in the ankle and less frequently in the knee or elbow.
*
Total Joint Arthroplasty (TJA) / Total Knee Arthroplasty (TKA), Total Elbow Arthroplasty (TEA), Total Ankle Arthroplasty (TAA):
* Indicated for end-stage arthropathy (Pettersson stage IV or V) with severe pain, loss of function, significant radiographic changes, and failed conservative and joint-preserving surgeries.
* Goals are pain relief, improved function, and correction of deformity.
Contraindications
- Active Infection: Absolute contraindication for elective arthroplasty or synovectomy. Must be aggressively treated and resolved prior to surgery.
- Uncontrolled Coagulopathy: Inadequate factor replacement or presence of inhibitors (neutralizing antibodies to factor VIII or IX) poses a significant risk of severe bleeding and is a relative contraindication. Surgery may proceed only after factor levels are optimized and inhibitor management (e.g., bypassing agents, immune tolerance induction) is established.
- Profound Systemic Comorbidities: Cardiopulmonary compromise, severe renal or hepatic disease that significantly increases surgical risk.
- Patient Unwillingness/Non-compliance: Especially regarding rigorous post-operative factor replacement and rehabilitation.
- Inadequate Bone Stock: May contraindicate arthroplasty in cases of severe osteolysis, requiring complex reconstruction or alternative approaches like arthrodesis.
Table of Operative vs. Non-Operative Indications
| Indication Category | Non-Operative Management | Operative Management |
|---|---|---|
| Factor Deficiency | Prophylactic factor replacement for all severe hemophiliacs | Pre/intra/post-operative factor administration essential for all surgeries |
| Early Arthropathy | On-demand factor for acute bleeds, RICE, physiotherapy | Radiosynovectomy for recurrent hemarthrosis (Pettersson I-II) |
| Chronic Synovitis | Arthroscopic or Open Synovectomy for persistent synovitis, pain, ROM loss (Pettersson II-III) | |
| Mechanical Symptoms | Arthroscopic debridement for loose bodies, osteophytes, adhesions | |
| Joint Deformity | Bracing, physical therapy for mild contractures | Osteotomy for axial malalignment (e.g., HTO) |
| End-Stage Arthropathy | Palliative pain management, adaptive equipment | Total Joint Arthroplasty (TJA/TKA/TEA/TAA) for severe pain, dysfunction (Pettersson IV-V) |
| Failed Arthroplasty | Arthrodesis for failed TJA, recurrent infection, severe bone loss |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is paramount for orthopedic surgery in hemophilia patients, focusing on coagulation management, infection prevention, and surgical optimization.
Coagulation Management
- Hematology Consultation: Essential to establish factor baseline, assess inhibitor status, and develop a comprehensive factor replacement plan. This typically involves elevating factor levels pre-operatively to 80-100% of normal.
-
Factor Administration:
- Pre-operative Dose: Administered 30-60 minutes before skin incision to achieve target factor levels.
- Intra-operative Monitoring: Factor levels may be monitored, especially in prolonged cases.
- Post-operative Dosing: Maintained at therapeutic levels (e.g., 50-80% for 3-5 days, then 30-50% for 10-14 days) to prevent bleeding during the critical healing and rehabilitation phases. The duration and intensity depend on the invasiveness of the surgery and patient response.
- Inhibitor Management: If inhibitors are present, specialized agents (e.g., bypassing agents like activated prothrombin complex concentrate (aPCC) or recombinant factor VIIa (rFVIIa)) are used. Immune tolerance induction (ITI) may be considered pre-emptively for elective surgery, but its effectiveness for acute surgical needs is limited.
- Blood Products: Availability of blood products (packed red blood cells, fresh frozen plasma, cryoprecipitate) should be ensured, although intraoperative transfusion rates have decreased significantly with effective factor replacement.
- Antifibrinolytics: Tranexamic acid or ε-aminocaproic acid may be used adjunctively, especially for oral or mucocutaneous bleeding risk, but should be used cautiously with aPCC due to thrombotic risk.
Imaging & Evaluation
- Radiographs: Standard AP/Lateral views are essential to assess joint space narrowing, subchondral cysts, osteophytes, and bone destruction (Pettersson or Arnold-Hilgartner classification). Specific views like skyline patellar views or stress views for deformity assessment.
- MRI: Provides superior soft tissue detail, visualizing synovial hypertrophy, cartilage integrity, hemosiderin deposition (low signal on T1/T2), and early subchondral changes. Crucial for surgical planning in synovectomy cases.
- CT Scan: Useful for complex bony deformities, severe bone loss, or pre-operative templating for arthroplasty, especially for component sizing and placement.
Infection Prophylaxis
- Antibiotics: Standard surgical prophylactic antibiotics (e.g., Cefazolin) administered pre-operatively. Prolonged post-operative antibiotics may be considered for arthroplasty due to the immunocompromised state sometimes associated with chronic illness and the high consequence of periprosthetic joint infection.
- Skin Preparation: Strict aseptic technique and comprehensive skin preparation are crucial.
Patient Positioning
- General Principles: Standard positioning for the specific joint approach. Meticulous padding of all pressure points is essential due to increased friability of tissues and potential for hematoma formation.
- Tourniquet Use: Generally safe and recommended for extremity surgery in hemophilia to reduce intraoperative bleeding and improve visibility. Inflation pressure and duration should be minimized. Factor replacement ensures adequate hemostasis upon tourniquet release.
-
Specific Positioning:
- Knee: Supine, with leg draped free to allow full ROM. A pneumatic tourniquet is typically used.
- Elbow: Lateral decubitus or supine with arm abducted on a hand table.
- Ankle: Supine with bump under ipsilateral hip, foot draped free.
Detailed Surgical Approach / Technique
Surgical interventions for hemophilic arthropathy range from synovectomy to complex joint arthroplasty. The choice depends on the stage of arthropathy, patient age, and functional goals.
1. Radiosynovectomy (Chemical/Radiation Synovectomy)
This is a non-incisional procedure, typically performed by an interventional radiologist in consultation with an orthopedic surgeon and hematologist.
*
Mechanism:
Intra-articular injection of a beta-emitting radioisotope (e.g., Yttrium-90 for large joints, Phosphorus-32 or Rhenium-186 for medium joints, Erbium-169 for small joints). The beta particles penetrate only a few millimeters, causing selective necrosis of the hypertrophic synovial lining with minimal radiation exposure to surrounding tissues.
*
Indications:
Chronic synovitis with recurrent hemarthrosis refractory to prophylactic factor, Pettersson stages I-II, where articular cartilage is largely preserved.
*
Technique:
1. Pre-operative factor replacement to achieve target levels.
2. Sterile preparation and draping.
3. Joint aspiration to remove effusions and confirm hemarthrosis if present.
4. Intra-articular injection of the radioisotope, often guided by fluoroscopy or ultrasound to ensure accurate placement.
5. A small amount of corticosteroid may be added to reduce immediate post-injection synovitis.
6. Temporary immobilization (e.g., splinting for 24-48 hours) to prevent leakage of the isotope into extra-articular tissues.
*
Post-operative:
Continued factor replacement for a few days, rest, and gentle mobilization.
2. Arthroscopic Synovectomy
This is the preferred method for surgical synovectomy due to its minimally invasive nature, reduced post-operative pain, and faster recovery compared to open synovectomy.
*
Indications:
Chronic, hypertrophic synovitis with recurrent hemarthrosis, pain, and ROM limitations (Pettersson stages II-III) unresponsive to factor prophylaxis and radiosynovectomy.
*
General Principles:
* Tourniquet use is standard for optimal visualization and hemostasis.
* Meticulous hemostasis is paramount throughout the procedure.
* Aggressive removal of all hypertrophic synovium is the goal, particularly from recesses and areas prone to bleeding.
* Maintain high infusion pressure to flush out blood and maintain a clear field.
*
Knee Synovectomy:
1.
Portals:
Standard anteromedial and anterolateral portals. Additional posteromedial or posterolateral portals may be used for posterior compartment access.
2.
Technique:
Arthroscope inserted. Systemic inspection of the joint. Using an arthroscopic shaver, hypertrophic synovium is meticulously resected from the suprapatellar pouch, medial and lateral gutters, intercondylar notch, and infrapatellar fat pad (Hoffa's fat pad). Posteromedial and posterolateral approaches may be required for complete posterior synovectomy, which is crucial for preventing further bleeding. The aim is near-total synovectomy.
3.
Debridement:
Concomitant debridement of unstable meniscal tears, chondral flaps, or loose bodies may be performed.
4.
Closure:
Portals closed with sutures. A drain may be left if significant oozing is anticipated, particularly if tourniquet time was prolonged.
*
Elbow Synovectomy:
1.
Portals:
Anterolateral, anteromedial, posterolateral, direct posterior. Care must be taken to avoid neurovascular structures (ulnar nerve medially, radial nerve laterally).
2.
Technique:
Synovium is resected from the olecranon fossa, coronoid fossa, and radiocapitellar joint. Osteophytes causing impingement may also be resected (osteophyte cheilectomy). Release of contractures may involve capsular release.
*
Ankle Synovectomy:
1.
Portals:
Anteromedial, anterolateral, posterolateral, posteromedial. Careful attention to neurovascular structures (dorsalis pedis artery, superficial peroneal nerve, sural nerve, posterior tibial nerve and vessels).
2.
Technique:
Resection of hypertrophic synovium from the anterior and posterior compartments. Debridement of anterior tibiotalar osteophytes (anterior impingement osteophytes) if present.
3. Total Joint Arthroplasty (TJA)
This is the definitive treatment for end-stage hemophilic arthropathy with severe pain, loss of function, and radiographic evidence of joint destruction.
*
General Considerations:
*
Factor Management:
Absolutely critical. Factor levels maintained at 80-100% pre-op and intra-op, then gradually tapered over 2-3 weeks post-op.
*
Bone Loss:
Hemophilic joints often have severe osteolysis and cysts, necessitating specialized implants (e.g., augments, stems, constrained components) and bone grafting.
*
Contractures:
Pre-existing severe flexion or extension contractures require aggressive soft tissue releases to achieve adequate ROM and component alignment.
*
Infection Risk:
Historically higher in hemophiliacs, meticulous sterile technique and prophylactic antibiotics are crucial.
*
Total Knee Arthroplasty (TKA):
1.
Approach:
Standard medial parapatellar approach.
2.
Releases:
Extensive soft tissue releases may be required for severe flexion contractures or varus/valgus deformities, especially posterior capsular release and deep MCL release for flexion contractures.
3.
Bone Resection:
Careful attention to bone cuts, as bone quality can be poor. Use of constrained or semi-constrained components (e.g., posterior stabilized or varus/valgus constrained) is often preferred due to ligamentous laxity and bone loss. Stems may be needed for femoral or tibial bone loss.
4.
Patellar Resurfacing:
Typically performed.
5.
Closure:
Meticulous layered closure. Drains are often used for 24-48 hours to monitor bleeding, but factor levels should ideally control post-operative oozing.
*
Total Elbow Arthroplasty (TEA):
1.
Approach:
Posterior approach with ulnar nerve identified and protected or transposed. Triceps-sparing or triceps-splitting approaches.
2.
Bone Resection:
Less bone is resected than in knee or hip arthroplasty. Cemented, linked (constrained) components are often preferred due to inherent instability and significant bone loss around the elbow.
3.
Complications:
High rates of aseptic loosening, infection, and ulnar neuropathy.
*
Total Ankle Arthroplasty (TAA):
1.
Approach:
Standard anterior approach.
2.
Challenges:
Significant bone loss and soft tissue contractures can complicate component placement. Three-component designs are common.
3.
Alternative:
Arthrodesis is often considered for severe ankle arthropathy due to lower revision rates and higher long-term success in younger, active patients.
4. Arthrodesis (Joint Fusion)
- Indications: Severely destroyed, painful, unstable joints where arthroplasty is contraindicated or has failed (e.g., recurrent infection, significant bone loss), particularly in younger, high-demand patients.
-
General Principles:
- Stable fixation is crucial for successful fusion.
- Correction of any significant deformity.
- Bone grafting may be necessary in the presence of large cysts or bone loss.
-
Technique:
- Joint Preparation: All articular cartilage and hypertrophic synovium are resected down to bleeding subchondral bone. Cysts are curetted and grafted.
- Positioning: Joint positioned in optimal functional alignment (e.g., knee 10-15 degrees flexion, ankle 0-5 degrees dorsiflexion).
- Fixation: Internal fixation (e.g., plates, screws, intramedullary nails) or external fixation, often combined with compression.
- Post-operative: Prolonged immobilization (cast/brace) for 8-12 weeks, non-weight bearing or protected weight bearing until fusion is confirmed radiographically. Factor support is critical throughout the healing process.
Complications & Management
Complications in orthopedic surgery for hemophilia patients are often exacerbated by the underlying coagulopathy, although modern factor replacement has significantly improved outcomes.
Common Complications
| Complication | Incidence | Salvage Strategies |
|---|---|---|
| Hemorrhage (Intra/Post-Op) | Varies (historically high, now <5% with prophylaxis) | Immediate factor level optimization, surgical exploration & hemostasis, drainage of hematoma, blood transfusion, antifibrinolytics (with caution) |
| Infection (Superficial/Deep) | 1-5% for TJA | Aggressive antibiotics, surgical debridement, implant retention if superficial; staged revision, explantation, arthrodesis, or amputation for deep infection |
| Nerve Injury | <1% (e.g., ulnar nerve in TEA, peroneal nerve in TKA) | Early recognition, neurolysis, bracing, physical therapy, pain management |
| Stiffness/Contracture | 5-15% (more common post-synovectomy or arthroplasty) | Aggressive physical therapy, manipulation under anesthesia (with factor cover), judicious soft tissue release |
| Implant Loosening/Failure | 5-10% (TJA, especially TEA) | Revision arthroplasty, arthrodesis, excision arthroplasty |
| Fracture (Periprosthetic) | <1% | ORIF (Open Reduction Internal Fixation), revision arthroplasty with extended fixation |
| Avascular Necrosis (AVN) | Rare as a direct surgical complication; underlying hemarthrosis contributes to AVN of bone | Core decompression (early stage), osteotomy, arthroplasty |
| Development of Inhibitors | 5-10% of severe hemophilia A patients | Bypassing agents (rFVIIa, aPCC), immune tolerance induction (ITI) |
| Thromboembolism (DVT/PE) | Low with current factor regimens, but risk exists with bypassing agents | Prophylactic anticoagulation (LMWH post-op with factor cover), early mobilization, mechanical prophylaxis (SCDs) |
Management Strategies
-
Hemorrhage:
The most feared complication.
- Immediate Response: Suspect hematoma formation with sudden pain, swelling, and decreased hemoglobin. Immediately increase factor replacement to high therapeutic levels (100%).
- Diagnosis: Ultrasound or CT scan can confirm hematoma.
- Intervention: For expanding hematomas or compartment syndrome, surgical evacuation is necessary with full factor cover. Meticulous hemostasis is performed. Drains may be left in place.
-
Infection:
- Prophylaxis: Strict sterile technique, prophylactic antibiotics.
- Diagnosis: Aspiration of joint fluid for gram stain, cell count, and culture.
- Management: Superficial infections may respond to antibiotics and local wound care. Deep infections (e.g., periprosthetic joint infection) often require aggressive surgical debridement with retention of implant (DAIR - Debridement, Antibiotics, Implant Retention) for acute infections, or a two-stage revision arthroplasty for chronic infections. In severe cases or failed revisions, implant removal and arthrodesis or excision arthroplasty may be necessary.
-
Stiffness:
Common, particularly after synovectomy or arthroplasty due to scar tissue formation and pain.
- Prevention: Early, controlled range of motion exercises post-operatively with adequate pain control and factor coverage.
- Treatment: Aggressive physical therapy, dynamic splinting. For recalcitrant stiffness, manipulation under anesthesia with full factor cover may be considered.
-
Implant Loosening:
Can occur due to poor bone quality, repetitive microtrauma, or aseptic processes.
- Management: Revision arthroplasty. Severe bone loss may necessitate the use of constrained implants, custom components, or conversion to arthrodesis.
-
Nerve Injury:
Most commonly due to direct trauma during dissection, prolonged traction, or hematoma compression.
- Management: Early recognition, factor normalization for hematoma. Neurosurgical consultation for persistent deficits.
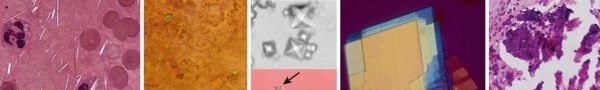
It is important to differentiate the crystal arthropathies from hemarthrosis, particularly in cases of acute joint pain and swelling. While hemophilic arthropathy is characterized by blood in the joint, other conditions present with crystalline deposits.

FIG. 1.38
Synovial fluid crystals. (A) Gout: yellow uric acid parallel to compensator, most common in first metatarsophalangeal joint. (B) Calcium pyrophosphate (dihydrate crystal) deposition disease (CPDD) or pseudogout crystals: blue rhomboid crystals
(arrow)
most common in knees and wrists. (C) Calcium oxalate crystals
(arrow)
are pyramidal and alm.
While the presence of these crystals (Gout, CPDD, Calcium Oxalate) is not typical for hemophilic arthropathy, diagnostic arthrocentesis remains a critical tool. If a hemophilic patient presents with acute monoarticular pain and swelling without a clear history of trauma, or with signs concerning for infection, synovial fluid analysis (cell count, differential, Gram stain, culture, and crystal analysis) is performed, always under full factor cover, to rule out other arthropathies or superimposed septic arthritis. The gross appearance of a hemarthrosis is bloody, distinguishing it from crystal-induced or septic arthritis unless an infection is superimposed on a bleed.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is crucial for optimizing surgical outcomes, restoring function, and preventing further complications. It must be carefully coordinated with hematology for continued factor coverage.
General Principles
- Factor Replacement: Continue prescribed factor regimen throughout rehabilitation, especially during periods of increased activity, to prevent re-bleeds.
- Pain Management: Aggressive pain control to facilitate early mobilization.
- Early Mobilization: Gentle, protected range of motion exercises are typically initiated soon after surgery to prevent stiffness and scar tissue formation, respecting soft tissue healing.
- Gradual Progression: Exercises gradually increase in intensity and resistance as healing progresses and pain subsides.
- Patient Education: Crucial for adherence to protocols and understanding activity restrictions.
Specific Joint Protocols
Post-Synovectomy (Arthroscopic/Open)
-
Immediate Post-Op (Days 0-7):
- Factor replacement as per hematology.
- Cryotherapy, elevation, and pain control.
- Gentle, passive, and active-assisted range of motion (ROM) exercises to prevent stiffness. Continuous passive motion (CPM) may be used for knee and elbow.
- Isometric muscle contractions (quadriceps sets, gluteal sets, elbow flexion/extension isometrics).
-
Early Phase (Weeks 1-6):
- Progressive ROM to achieve full pain-free motion.
- Gradual strengthening exercises (e.g., resistance bands, light weights).
- Proprioception and balance training.
- Protected weight-bearing (if lower extremity) with crutches, progressing to full weight-bearing as tolerated.
-
Intermediate Phase (Weeks 6-12):
- Advanced strengthening exercises, focusing on functional movements.
- Endurance training.
- Return to light activities of daily living.
- Long-Term: Maintenance exercises, joint protection strategies, avoidance of high-impact activities.
Post-Total Joint Arthroplasty (TJA)
Total Knee Arthroplasty (TKA)
-
Immediate Post-Op (Days 0-7):
- Factor replacement crucial for preventing hemarthrosis.
- CPM initiated immediately (0-90 degrees, gradually increasing).
- Ankle pumps, quadriceps sets, gluteal sets.
- Ambulation with walker or crutches, partial weight-bearing (PWB) or weight-bearing as tolerated (WBAT) per surgeon's protocol and bone quality.
-
Early Phase (Weeks 1-6):
- Focus on achieving 0-110+ degrees of flexion.
- Progressive strengthening (straight leg raises, knee flexion/extension with light resistance).
- Gait training, stair climbing.
-
Intermediate Phase (Weeks 6-12):
- Advanced strengthening, balance, and functional activities.
- Light recreational activities as tolerated.
- Long-Term: Avoidance of high-impact activities (running, jumping), heavy lifting. Regular low-impact exercises (swimming, cycling).
Total Elbow Arthroplasty (TEA)
-
Immediate Post-Op (Days 0-7):
- Splinting in a functional position (e.g., 90 degrees flexion with forearm neutral) for protection, often removed for supervised ROM exercises.
- Gentle passive and active-assisted ROM, avoiding valgus/varus stress.
- Isometrics for shoulder, wrist, and hand.
-
Early Phase (Weeks 1-6):
- Gradual increase in active ROM, aiming for functional arc (30-130 degrees flexion, 50 degrees pronation/supination).
- Light strengthening, protecting the implant from excessive loads.
-
Intermediate Phase (Weeks 6-12):
- Progressive strengthening, functional activities.
- Strict avoidance of lifting heavy objects (typically <1-2 kg indefinitely).
- Long-Term: Lifelong restrictions on lifting, pushing, and pulling heavy objects to protect the implant from loosening.
Total Ankle Arthroplasty (TAA)
-
Immediate Post-Op (Days 0-7):
- Initial non-weight-bearing (NWB) in a splint or cast.
- Factor replacement.
- Elevation, cryotherapy.
-
Early Phase (Weeks 2-6, after wound healing):
- NWB to PWB in a CAM boot.
- Gentle non-impact ROM exercises.
- Isometrics for calf muscles.
-
Intermediate Phase (Weeks 6-12):
- Gradual progression to full weight-bearing in the boot, then transitioning to supportive footwear.
- Balance and proprioception training.
- Strengthening exercises (calf raises, resistance bands).
- Long-Term: Low-impact activities encouraged. Avoidance of uneven surfaces, running, and jumping.
Post-Arthrodesis
-
Immediate Post-Op (Weeks 0-8/12):
- Strict non-weight-bearing (NWB) in a cast or splint.
- Factor replacement for bone healing.
- Elevation, cryotherapy.
- Radiographic follow-up to monitor fusion.
-
Transition Phase (After confirmed early fusion):
- Gradual progression to protected weight-bearing in a walking boot or brace.
- Long-Term: Once fusion is solid, rehabilitation focuses on strengthening adjacent joints, gait training, and adaptation to altered biomechanics.
Summary of Key Literature / Guidelines
The management of hemophilic arthropathy has evolved significantly, largely driven by advancements in factor replacement therapy and surgical techniques. The World Federation of Hemophilia (WFH) and national hemophilia foundations provide comprehensive guidelines.
- Prophylaxis is Key: Strong evidence supports prophylactic factor replacement as the most effective strategy to prevent hemarthrosis and subsequent arthropathy, especially when initiated early in childhood. This has shifted the focus from managing end-stage disease to prevention.
- Radiosynovectomy Efficacy: Radiosynovectomy remains a valuable intervention for chronic synovitis and recurrent hemarthrosis in early-stage arthropathy (Pettersson stages I and II) where cartilage damage is minimal. Multiple studies demonstrate its efficacy in reducing bleeding episodes and improving joint function, with Yttrium-90 being the preferred isotope for large joints.
- Surgical Synovectomy: Arthroscopic synovectomy has largely replaced open synovectomy for many joints due to reduced morbidity, faster recovery, and comparable efficacy in reducing bleeding and improving pain for stages II and early III arthropathy. Complete synovectomy, as confirmed by histology, provides the best long-term outcomes.
-
Total Joint Arthroplasty Outcomes:
TJA, particularly TKA, offers significant pain relief and functional improvement for end-stage hemophilic arthropathy (Pettersson stages IV and V). Long-term survival rates for TKA in hemophilia are generally comparable to those in non-hemophilic patients, although there is a higher incidence of complications such as infection, aseptic loosening, and periprosthetic fracture.
- TKA: Studies show 10-year survival rates exceeding 85-90%. Challenges include significant bone loss necessitating augmented or constrained components, and higher rates of revision.
- TEA: Outcomes are generally less favorable than TKA, with higher rates of loosening, infection, and ulnar neuropathy. Long-term functional restrictions are often necessary.
- TAA: Outcomes are also improving, but arthrodesis remains a viable and often preferred option for severely diseased ankles in younger, active patients, offering predictable pain relief and durable results, albeit with loss of motion.
- Inhibitor Management: Patients with inhibitors pose a significant challenge. Surgical interventions in these patients require a multidisciplinary approach involving hematologists experienced in inhibitor management, often employing bypassing agents or other specialized treatments perioperatively. Outcomes for TJA in inhibitor patients can be poorer due to increased bleeding risk and potentially higher rates of infection.
- Multidisciplinary Approach: Consensus guidelines emphasize the importance of a specialized multidisciplinary team, including orthopedic surgeons, hematologists, physical therapists, and pain management specialists, for optimal care of hemophilia patients with arthropathy. This approach ensures comprehensive management from prophylaxis to rehabilitation.
- Ongoing Research: Research continues into gene therapy, novel clotting factor agents with extended half-lives, and improved surgical techniques and implant designs to further reduce the burden of hemophilic arthropathy. The goal is to prevent bleeding entirely and, failing that, to offer durable, limb-sparing surgical solutions with minimal complications.
Clinical & Radiographic Imaging

You Might Also Like

