Comprehensive Bone Biology: Cellular Mechanisms, Remodeling, & Orthopedic Pathologies
Key Takeaway
Bone biology centers on osteoblasts, bone-forming cells originating from mesenchymal stem cells. They synthesize bone matrix under regulation by RUNX2, BMPs, and Wnt pathways. This cellular orchestration drives bone remodeling, vital for skeletal integrity, mechanical adaptation, and effective orthopedic fracture healing.
Understanding the Biology of Bone: Cells, Origin, and Function
Introduction & Epidemiology
Bone, a dynamic and highly specialized connective tissue, serves critical mechanical and metabolic functions within the human body. As orthopedic surgeons, a profound understanding of bone biology, from its cellular constituents to its adaptive responses, is paramount to optimizing patient outcomes. The remarkable capacity of bone for self-repair and remodeling differentiates it from many other tissues, yet this capacity is tightly regulated by intricate cellular and molecular mechanisms. Disruptions to these processes underpin a vast array of orthopedic pathologies, including fracture nonunion, osteoporosis, osteonecrosis, and tumor formation.
The fundamental unit of bone remodeling is the basic multicellular unit (BMU), a coordinated consortium of bone-resorbing osteoclasts and bone-forming osteoblasts. The equilibrium between bone resorption and formation dictates overall bone mass and structural integrity. A comprehensive grasp of this cellular interplay is essential for the effective management of both acute trauma and chronic degenerative conditions. Furthermore, age-related decline in bone repair capabilities, influenced by changes in stem cell niches and hormonal signaling, contributes to the epidemiological burden of fragility fractures in the elderly. Understanding the precise cellular and molecular pathways that govern bone anabolism and catabolism allows for targeted therapeutic interventions, ranging from enhancing fracture healing to mitigating bone loss.
Surgical Anatomy & Biomechanics
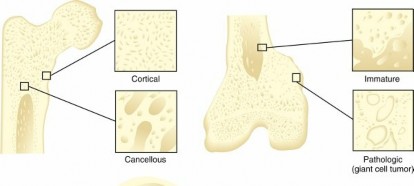
Bone macrostructure comprises cortical (compact) bone, providing strength and rigidity, and cancellous (trabecular) bone, offering porosity and metabolic activity. At the microscopic level, these structures are organized into osteons in cortical bone and trabeculae in cancellous bone. The mechanical properties of bone, including its stiffness, strength, and toughness, are directly influenced by its hierarchical organization, composition, and cellular activity. The bone matrix, primarily composed of type I collagen and a diverse array of non-collagenous proteins, is mineralized by hydroxyapatite crystals, providing the material's compressive strength. The arrangement of collagen fibers and mineral crystals, influenced by mechanical loading and cellular secretion, dictates anisotropic mechanical behavior.
The cellular biology of bone dictates its adaptive response to mechanical stimuli, a principle fundamental to Wolff's Law. Mechanical stress and strain are transduced into biochemical signals, regulating the activity of osteocytes, osteoblasts, and osteoclasts.

Osteoblasts
Osteoblasts are the primary bone-forming cells, responsible for synthesizing and secreting the organic components of the bone matrix, followed by its mineralization.
*
Morphology and Location:
They appear as cuboid cells aligned in layers along immature osteoid, typically found on bone surfaces undergoing active formation.
*
Origin and Differentiation:
Osteoblasts are derived from undifferentiated mesenchymal stem cells (MSCs). These MSCs reside in critical niches such as the haversian canals, endosteum, and periosteum. Their differentiation into osteoblasts is a tightly regulated process, critically influenced by the local mechanical and biochemical environment.
* Under conditions of low strain and increased oxygen tension, MSCs are directed towards the osteoblast lineage.
* The transcription factor RUNX2 (Runt-related transcription factor 2), also known as Core-binding factor α-1 (CBFα1), is a master regulator of osteoblast differentiation, essential for skeletal development.
* Bone morphogenetic proteins (BMPs), particularly BMP-2, -4, and -7, are potent osteoinductive factors that signal through SMAD pathways to direct mesenchymal cells toward the osteoblast phenotype.
* β-catenin, a key component of the Wnt signaling pathway, also plays a crucial role in stimulating osteoblast differentiation and bone formation.
* Conversely, MSCs exhibit plasticity; under intermediate strain and low oxygen tension, they may differentiate into chondrocytes, forming cartilage. High strain conditions can lead to differentiation into fibrous tissue. This mechanotransduction is critical in fracture healing, where initial instability often leads to cartilage or fibrous tissue formation, preceding endochondral ossification.
*
Cellular Machinery:
Osteoblasts possess a highly developed endoplasmic reticulum, Golgi apparatus, and numerous mitochondria, reflecting their intensive synthetic and secretory activity required for matrix production and mineralization.
*
Activity Levels:
Bone surfaces are lined by a heterogeneous population of osteoblasts. More differentiated, metabolically active cells are responsible for active bone formation, while less active, quiescent cells in resting regions help maintain the ionic milieu of bone. Disruption of the active lining cell layer can activate these quiescent cells.
*
Secretory Products:
Osteoblasts produce a variety of critical molecules:
*
Alkaline phosphatase (ALP):
An enzyme essential for mineralization, hydrolyzing pyrophosphate, an inhibitor of calcification.
*
Osteocalcin:
A vitamin K-dependent protein, its synthesis is stimulated by 1,25-dihydroxyvitamin D [1,25(OH)2D3]. It binds to hydroxyapatite and is a marker of osteoblast activity.
*
Type I collagen:
The predominant organic component of the bone matrix, providing tensile strength.
*
Bone sialoprotein (BSP):
A non-collagenous protein involved in initial mineralization and cell attachment.
*
Receptor Activator of Nuclear Factor (NF)-κβ Ligand (RANKL):
A crucial cytokine that promotes osteoclast differentiation and activation.
*
Osteoprotegerin (OPG):
A soluble decoy receptor that binds to RANKL, thereby inhibiting its interaction with RANK on osteoclast precursors, thus limiting osteoclastogenesis and bone resorption. The RANKL/OPG ratio is a key determinant of bone remodeling balance.

Osteocytes
Osteoblasts that become entrapped within the mineralized matrix differentiate into osteocytes, which are the most abundant cells in mature bone. Osteocytes reside in lacunae and extend dendritic processes through canaliculi, forming an intricate cellular network throughout the bone matrix. This network enables communication between osteocytes and with cells on the bone surface. Osteocytes are recognized as the primary mechanosensors of bone, detecting mechanical strain and initiating signaling cascades that regulate bone remodeling in response to loading. They orchestrate the activity of osteoblasts and osteoclasts via molecules such as sclerostin and RANKL.
Osteoclasts
Osteoclasts are large, multinucleated cells responsible for bone resorption. They are derived from hematopoietic stem cells of the monocyte/macrophage lineage. Their differentiation and activity are tightly regulated by osteoblasts, primarily through the RANKL/OPG system. Osteoclasts create an acidic microenvironment beneath their ruffled border, dissolving the mineralized matrix, and then secrete proteases (e.g., cathepsin K) to degrade the organic matrix.

Indications & Contraindications
A thorough understanding of bone biology informs the indications and contraindications for numerous orthopedic interventions. Surgical decisions often hinge on the bone's intrinsic capacity for healing, its current metabolic state, and the physiological response to various stressors.
Operative Indications
Operative intervention is typically indicated when the biological environment or mechanical forces preclude adequate non-operative healing, or when a pathological process mandates surgical extirpation or stabilization. Examples include:
*
Fracture Fixation:
Unstable fractures, displaced intra-articular fractures, open fractures, and fractures with neurovascular compromise. The goal is to provide a stable mechanical environment conducive to secondary bone healing, which relies heavily on osteoblast activity and adequate vascular supply.
*
Nonunion/Malunion:
Failed fracture healing (nonunion) or healing in an unacceptable anatomical position (malunion) often require surgical intervention, including debridement, bone grafting (autograft or allograft), and rigid internal fixation to stimulate dormant osteoblasts and provide appropriate mechanical cues.
*
Osteotomies:
Corrective osteotomies for deformity or joint alignment rely on inducing controlled fracture healing, with precise cuts and stable fixation to guide new bone formation.
*
Joint Arthroplasty:
In cases of severe degenerative joint disease or inflammatory arthritis, joint replacement necessitates stable bone-implant interfaces, where osteoblasts contribute to osseointegration.
*
Bone Tumors:
Resection of primary or metastatic bone tumors often requires extensive reconstruction, utilizing biological principles of bone regeneration and stability.
*
Spinal Fusion:
Creation of a solid bony bridge between vertebrae in cases of instability or deformity, involving bone graft incorporation and osteoblast-mediated new bone formation.
Contraindications
Absolute contraindications are rare and typically relate to the patient's overall medical status. Relative contraindications often stem from factors that compromise bone biology and healing potential, increasing the risk of complications.
*
Active Infection:
Systemic or local infection is a strong relative contraindication for elective bone procedures, as it significantly impairs bone healing and increases implant failure rates.
*
Severe Comorbidities:
Uncontrolled diabetes, severe peripheral vascular disease, or poorly managed malnutrition can severely compromise bone blood supply and cellular metabolic activity, delaying or preventing healing.
*
Extremely Poor Bone Quality:
Severe osteoporosis or osteopetrosis can make successful fixation challenging, risking implant pull-out or periprosthetic fracture.
*
Patient Non-compliance:
Inability to adhere to post-operative weight-bearing or rehabilitation protocols can jeopardize fixation and healing.
Summary of Operative vs. Non-Operative Indications
| Indication Category | Operative Management | Non-Operative Management | Rationale Based on Bone Biology |
|---|---|---|---|
| Fractures | Unstable, displaced intra-articular, open, neurovascular compromise, pathological, nonunion/malunion | Stable, minimally displaced, stress fractures | Surgical stabilization provides optimal mechanical environment (low strain) for osteoblast activity and direct bone healing. Non-op relies on intrinsic healing with favorable biology. |
| Degenerative Joint Disease | Severe pain/dysfunction, failed conservative Rx, joint destruction, instability | Mild-moderate symptoms, early disease, activity modification, pharmacotherapy, injections | Joint arthroplasty addresses structural failure beyond bone's regenerative capacity. Conservative care manages symptoms. |
| Spinal Deformity/Instability | Progressive deformity, neurological deficit, intractable pain, spondylolisthesis | Mild, stable deformity, no neurological deficit, responsive to conservative care | Spinal fusion leverages osteoblast activity for bony bridging. Non-op manages symptoms without altering bone structure. |
| Bone Tumors | Malignant, aggressive benign, pathological fracture risk | Observation for incidental/benign lesions with low risk | Resection and reconstruction based on tumor biology and bone's regenerative capacity. |
| Infection (Osteomyelitis) | Chronic, sequestered bone, surgical debridement indicated | Acute, early, responsive to antibiotics, no significant bone destruction | Surgical debridement removes necrotic, infected bone to allow healthy tissue and osteoblasts to heal. |
| Metabolic Bone Disease | Pathological fracture, severe deformity (e.g., Paget's) | Systemic pharmacotherapy (e.g., bisphosphonates for osteoporosis) | Surgery addresses severe structural consequences; medical management targets cellular dysfunction (osteoclast/osteoblast imbalance). |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is critical for all orthopedic procedures involving bone. This extends beyond merely assessing anatomical features and includes evaluating the biological capacity of the patient's bone.
*
Patient Optimization:
Address factors that directly impact bone healing and host defense. Nutritional status, vitamin D levels, calcium homeostasis, and diabetic control must be optimized. Smoking cessation is crucial, given its detrimental effects on angiogenesis and osteoblast activity. Immunosuppression and steroid use should be considered in terms of their impact on osteoblast function and bone strength.
*
Imaging Review:
High-resolution radiographs, CT scans (with 3D reconstruction), and MRI provide detailed information on fracture patterns, bone defects, pre-existing bone quality, and vascularity. This guides implant choice, size, and placement. Poor bone stock, for instance, may necessitate augmented fixation techniques or bone grafting.
*
Surgical Strategy:
Define the implant type, size, and number based on fracture morphology, patient demographics, and bone quality. For complex cases, virtual surgical planning and 3D printing of anatomical models can enhance precision.
*
Patient Positioning:
Optimal patient positioning ensures adequate surgical exposure while protecting neurovascular structures. The specific approach dictates positioning, but general principles include maintaining physiological alignment and minimizing prolonged pressure points, which can compromise perfusion and cellular viability in the skin and subcutaneous tissues. For procedures involving extensive bone grafting or defect management, positioning should also facilitate access to donor sites (e.g., iliac crest).
Detailed Surgical Approach / Technique
While specific surgical techniques vary widely depending on the anatomical site and pathology, the underlying principles are universally guided by bone biology. The aim is always to create an environment that promotes efficient and robust bone healing, relying on the inherent capacity of osteoblasts and other bone cells to regenerate tissue.

Principles of Bone Handling and Fixation
-
Tissue Dissection and Preservation of Vascularity:
- Minimally Invasive Approaches: Where feasible, utilize approaches that minimize soft tissue stripping (e.g., percutaneous fixation, MIS plating). Extensive dissection compromises the periosteal blood supply, which is critical for osteoblast recruitment and survival, particularly in diaphyseal bone.
- Gentle Handling: Retraction and manipulation of muscle and fascial planes should be atraumatic to preserve their vascularity to the bone.
- Irrigation: Continuous irrigation with sterile saline throughout the procedure prevents desiccation of bone cells and periosteum, minimizing cellular damage.
-
Reduction:
- Anatomic vs. Biological Reduction: For intra-articular fractures, anatomical reduction is paramount to restore joint congruity and prevent post-traumatic arthritis. For simple diaphyseal fractures, biological reduction (indirect reduction with maintenance of fracture hematoma and periosteal attachments) is often preferred to preserve the intrinsic healing potential. The fracture hematoma is a rich source of growth factors and progenitor cells, crucial for the early stages of bone repair.
- Temporary Fixation: K-wires or reduction clamps can temporarily stabilize fragments while definitive fixation is prepared, ensuring alignment without excessive manipulation.
-
Fixation:
-
Stability:
The choice of fixation (plates, screws, intramedullary nails, external fixators) depends on the fracture pattern and desired healing mode.
- Absolute Stability (e.g., lag screws, compression plates): Achieves direct bone healing with minimal callus formation, requiring precise anatomical reduction and interfragmentary compression. This promotes osteoblast activity directly across the fracture gap.
- Relative Stability (e.g., bridging plates, intramedullary nails): Allows for controlled micromotion, promoting secondary bone healing through callus formation (endochondral and intramembranous ossification). This relies on the differentiation of mesenchymal stem cells into chondrocytes and osteoblasts under appropriate strain conditions.
- Bone Quality Considerations: In osteoporotic bone, augmented fixation (e.g., polymethylmethacrylate cement injection, locked plating systems with increased screw purchase) may be necessary to compensate for reduced bone mineral density and impaired osteoblast function, thus preventing implant loosening.
-
Stability:
The choice of fixation (plates, screws, intramedullary nails, external fixators) depends on the fracture pattern and desired healing mode.
-
Bone Grafting (as indicated):
- Osteoinductive Properties: Grafts containing osteoinductive factors (e.g., BMPs in demineralized bone matrix, DBM) stimulate differentiation of local mesenchymal stem cells into osteoblasts.
- Osteoconductive Properties: Scaffolds (e.g., allografts, synthetic ceramics) provide a framework for creeping substitution, allowing osteoblasts to migrate and lay down new bone.
- Osteogenic Properties: Autografts contain viable osteoblasts and mesenchymal stem cells, providing direct osteogenesis and growth factors. The iliac crest is a common source, providing cells that can become osteoblasts.
- Clinical Application: Bone grafting is employed for nonunions, large bone defects (e.g., after tumor resection), or spinal fusions to enhance osteoblast-mediated bone formation.

Illustrative image of bone repair mechanisms.
Complications & Management
Complications in orthopedic surgery involving bone are often rooted in a failure of the biological processes of bone healing, remodeling, or infection control. Understanding the underlying cellular and molecular mechanisms is crucial for prevention and effective management.

Common Complications and Salvage Strategies
| Complication | Incidence | Underlying Bone Biology/Mechanism | Salvage Strategies |
|---|---|---|---|
| Nonunion | 5-10% (higher for specific sites/risk factors) | Inadequate mechanical stability (high strain leading to fibrous tissue/cartilage instead of bone), compromised vascularity, insufficient osteoblasts/growth factors, infection, metabolic derangements | Revision fixation (achieve appropriate stability), debridement of fibrous tissue, bone grafting (autograft/allograft with osteoinductive agents like BMP), pulsed electromagnetic fields (PEMF), low-intensity pulsed ultrasound (LIPUS) to stimulate cellular activity. |
| Malunion | Varies widely | Inaccurate reduction, premature consolidation in poor alignment. | Corrective osteotomy and stable internal fixation, allowing for new, appropriately aligned bone formation by osteoblasts. |
| Infection (Osteomyelitis) | 0.5-5% (higher in open fractures/implants) | Bacterial colonization overwhelms host immune response, biofilm formation, inhibited osteoblast activity, increased osteoclast activity leading to bone lysis. | Surgical debridement of necrotic and infected bone, prolonged intravenous antibiotics, local antibiotic delivery (beads, cement spacers), soft tissue coverage, potential hardware removal. May require staged reconstruction and bone grafting once infection is controlled. |
| Implant Failure | Varies, often related to nonunion or osteoporosis | Mechanical overload, inadequate purchase in poor quality bone, micromotion leading to fibrous tissue encapsulation, stress shielding. | Revision surgery with stronger implants, different fixation principles (e.g., locked plating for osteoporosis), bone grafting to improve bone quality and implant-bone interface. Address underlying biological issues (e.g., vitamin D deficiency). |
| Avascular Necrosis (AVN) | Varies by site (e.g., femoral head: 10-30% with specific fractures) | Ischemia leading to osteocyte death, subsequent collapse of subchondral bone, disruption of osteoblast activity in repair. | Early stages: core decompression to promote revascularization, potentially bone grafting. Later stages: osteotomy or arthroplasty to replace necrotic bone and damaged joint. Understanding blood supply to bone is paramount for prevention. |
| Delayed Union | Common, but resolves | Similar to nonunion but less severe; slower cellular response, suboptimal mechanical environment. | Patient education and observation. Consider interventions to stimulate healing if progressing slowly (e.g., bone stimulators, activity modification) before defining as nonunion. Optimize systemic factors influencing bone biology. |
| Refracture | 1-5% | Incomplete bone remodeling and restoration of strength, premature hardware removal, underlying metabolic bone disease. | Re-fixation, potentially with bone grafting to reinforce the area. Address any systemic bone quality issues. |
| Heterotopic Ossification | 5-20% (higher in specific trauma/neurological conditions) | Ectopic differentiation of mesenchymal stem cells into osteoblasts in soft tissues due to inflammatory/traumatic stimuli. | Prophylaxis with NSAIDs or radiation therapy. Surgical excision after maturation (hot bone scan negative), with prophylaxis to prevent recurrence. Requires understanding of inappropriate osteoblast differentiation pathways. |

Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is not merely a process of regaining motion and strength; it is a meticulously planned interaction with bone biology and its response to mechanical loading. The application of controlled, progressive loading is crucial for stimulating osteoblast activity and guiding bone remodeling according to Wolff's Law.
Phases of Rehabilitation and Biological Rationale
-
Immobilization/Protection Phase (Inflammatory and Reparative Phases of Healing):
- Goal: Protect surgical repair, prevent excessive motion that could disrupt the delicate early cellular processes (hematoma formation, soft callus), and allow initial osteoblast recruitment and differentiation.
- Biological Rationale: The initial fracture hematoma and subsequent inflammatory response lay the groundwork for MSC migration and differentiation. Excessive strain during this phase can direct MSCs to form fibrous tissue or cartilage (leading to nonunion) rather than bone. Absolute stability in certain fractures (e.g., intra-articular) is critical to prevent shear forces that are detrimental to osteoblast activity.
- Activities: Non-weight-bearing or touch-down weight-bearing, controlled range of motion within protected limits, isometric exercises to maintain muscle tone without stressing the repair site.
-
Controlled Motion/Progressive Loading Phase (Remodeling Phase):
- Goal: Gradually increase mechanical stress to stimulate bone formation and consolidation, improve vascularity, and restore functional strength and range of motion.
- Biological Rationale: According to Wolff's Law, bone adapts to the loads placed upon it. Controlled, axial loading stimulates osteocytes to send signals that promote osteoblast activity and matrix deposition, increasing bone mineral density and structural integrity. Progressive loading also enhances blood flow, vital for oxygen and nutrient delivery to active osteoblasts.
- Activities: Gradual increase in weight-bearing, active and passive range of motion exercises, light resistance training, neuromuscular re-education. Monitoring radiographs for callus formation and bridging is essential.
-
Return to Activity/Sport Phase (Maturation and Long-term Remodeling):
- Goal: Achieve full functional recovery, maximal bone strength, and return to pre-injury activity levels.
- Biological Rationale: Bone remodeling continues for months to years, with osteoclasts and osteoblasts working in concert to optimize bone architecture in response to long-term loading patterns. High-impact or repetitive loading must be introduced progressively to allow the bone to adapt, avoiding stress fractures or implant loosening.
- Activities: Advanced resistance training, plyometrics, sport-specific drills, gradual return to high-impact activities. Continued monitoring for symptoms of stress or fatigue.
Factors Influencing Protocols
- Patient Age and Bone Quality: Younger patients with robust osteoblast activity heal faster. Older patients or those with metabolic bone diseases (e.g., osteoporosis, which impairs osteoblast function and bone strength) require more cautious, prolonged protection and slower progression of loading.
- Fracture/Procedure Type: Intra-articular fractures often require early motion to prevent stiffness, balanced with protection for osteochondral repair. Long bone diaphyseal fractures may tolerate earlier weight-bearing with intramedullary nailing.
- Fixation Stability: Stable fixation allows for earlier, more aggressive rehabilitation. Less stable constructs (e.g., external fixators for open fractures) necessitate stricter adherence to weight-bearing restrictions.
- Systemic Factors: Optimal nutrition, vitamin D sufficiency, and absence of comorbidities like diabetes are critical for robust osteoblast activity and cellular healing.
Summary of Key Literature / Guidelines
Current orthopedic literature consistently emphasizes the critical interplay between mechanical stability, biological environment, and cellular function in achieving successful bone healing and reconstruction. Guidelines for fracture management, nonunion treatment, and implant selection are increasingly informed by a deep understanding of bone biology.
- AO Foundation Principles: The AO (Arbeitsgemeinschaft für Osteosynthesefragen) principles, while primarily mechanical, implicitly support optimal biological conditions. Their emphasis on anatomical reduction, stable internal fixation, preservation of blood supply, and early active mobilization directly facilitates cellular processes of bone healing. The concept of "biological fixation" (relative stability) for comminuted fractures acknowledges the importance of the periosteal blood supply and the contribution of primary bone-forming cells in the fracture hematoma.
- Bone Morphogenetic Proteins (BMPs): Clinical guidelines for nonunion management frequently include the consideration of recombinant human BMPs (rhBMP-2, rhBMP-7) as osteoinductive adjuncts, particularly for open tibial fractures or spinal fusions. This directly leverages the osteoblast-differentiation capabilities of BMPs as highlighted in the cellular biology.
- Vitamin D and Calcium Supplementation: Guidelines for managing osteoporosis and preventing fragility fractures uniformly recommend adequate vitamin D and calcium intake. This supports optimal osteoblast function, as 1,25(OH)2D3 stimulates osteocalcin production and is essential for calcium homeostasis and mineralization.
- Biologics and Regenerative Medicine: Emerging literature explores the use of autologous bone marrow aspirate concentrate (BMAC), platelet-rich plasma (PRP), and mesenchymal stem cell (MSC) therapies to augment bone healing. These approaches aim to deliver a concentrated source of osteoprogenitor cells and growth factors (including BMPs) directly to the site of injury or nonunion, thereby enhancing the cellular environment for osteoblast recruitment and activity.
- Mechanical Environment for Healing: Numerous studies reinforce the concept that the optimal mechanical environment for bone healing varies. Absolute stability promotes primary bone healing, while controlled micromotion encourages secondary healing via endochondral ossification, where MSCs differentiate into cartilage and subsequently bone. These findings directly relate to the varying differentiation pathways of MSCs under different strain conditions.
- RANKL/OPG Pathway Modulation: Research continues into therapeutic targets that modulate the RANKL/OPG pathway to influence bone remodeling, relevant for conditions like osteoporosis (denosumab, a RANKL inhibitor) and potentially for enhancing bone graft incorporation or preventing periprosthetic osteolysis.
- Smoking Cessation: Consensus guidelines universally recommend smoking cessation prior to elective orthopedic procedures and for nonunion treatment, underscoring the severe detrimental effects of nicotine and other toxins on angiogenesis, osteoblast proliferation, and matrix synthesis.
A comprehensive understanding of bone's cellular and molecular processes remains the cornerstone of modern orthopedic practice, guiding both innovative research and routine clinical decisions to restore musculoskeletal function.
Clinical & Radiographic Imaging

