Anterior Cruciate Ligament (ACL): Comprehensive Guide to Anatomy, Biomechanics & Epidemiology
Key Takeaway
The Anterior Cruciate Ligament (ACL) is a crucial intra-articular knee ligament, acting as the primary restraint to anterior tibial translation and rotational instability. Comprising anteromedial and posterolateral bundles, it originates from the lateral femoral condyle and inserts onto the anterior tibia, vital for knee stability and often injured in pivoting sports.
Introduction & Epidemiology
Anterior Cruciate Ligament (ACL) injury represents a significant and prevalent pathology within orthopedic sports medicine, impacting a diverse demographic, particularly active individuals and athletes. The incidence of ACL ruptures in the general population ranges from 30 to 78 per 100,000 person-years, with higher rates observed in specific athletic cohorts. Athletes involved in pivoting and contact sports such as soccer, basketball, football, volleyball, and skiing are at considerably elevated risk. Epidemiological data consistently demonstrate a higher incidence in female athletes compared to male athletes in comparable sports, a disparity attributed to a complex interplay of anatomical, hormonal, neuromuscular, and biomechanical factors.
The economic burden associated with ACL injuries is substantial, encompassing surgical reconstruction, prolonged rehabilitation, lost productivity, and potential long-term sequelae such as post-traumatic osteoarthritis (PTOA). Early identification and appropriate management are crucial to mitigate these impacts. While the primary goal of ACL reconstruction is to restore knee stability and facilitate a safe return to sport, ongoing research continues to refine our understanding of its role in preventing subsequent meniscal and chondral injuries, and ultimately, the progression of PTOA. The historical perspective has evolved from rudimentary repair techniques to sophisticated arthroscopic anatomical reconstruction, with a continuous focus on optimizing biomechanical function and long-term joint health.
Surgical Anatomy & Biomechanics
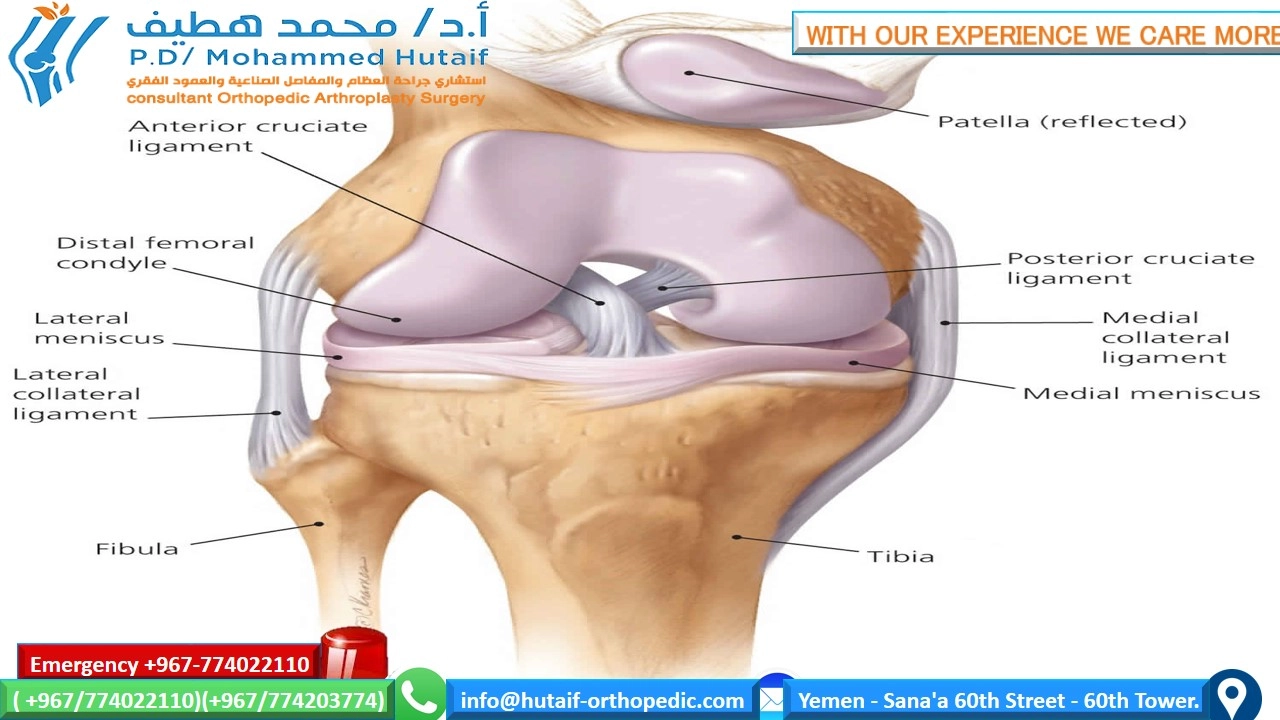
The knee joint, a modified hinge joint, is composed of the distal femur, proximal tibia, and patella. Articulating surfaces are covered by hyaline cartilage, facilitating smooth motion. The patella, a sesamoid bone, enhances the quadriceps lever arm. Stability is conferred by both static restraints (ligaments, menisci, joint capsule) and dynamic restraints (musculotendinous units).
Ligamentous structures are paramount for knee stability. There are four primary ligaments:
-
Cruciate Ligaments:
Located intra-articularly but extrasynovially, crossing each other to form an "X." They prevent anterior-posterior translation and rotational instability.
- Anterior Cruciate Ligament (ACL): Originates from the posteromedial aspect of the lateral femoral condyle, within the intercondylar notch, and inserts onto the anterior intercondylar area of the tibia, between the medial and lateral tibial spines. It runs obliquely inferiorly, anteriorly, and medially.
- Posterior Cruciate Ligament (PCL): Originates from the medial femoral condyle and inserts onto the posterior intercondylar area of the tibia.
-
Collateral Ligaments:
Located extra-articularly, controlling varus and valgus stability.
- Medial Collateral Ligament (MCL): Superficial and deep layers, connecting the medial femoral epicondyle to the medial tibial condyle.
- Lateral Collateral Ligament (LCL): Cord-like structure, connecting the lateral femoral epicondyle to the fibular head, distinct from the joint capsule.
The ACL is the primary restraint to anterior tibial translation, accounting for approximately 87% of the total anterior load at 30° and 90° of flexion. It also plays a critical role in resisting internal rotation of the tibia relative to the femur and secondary resistance to varus and valgus stresses.

Detailed ACL Anatomy:
The ACL is typically described as a two-bundle structure:
*
Anteromedial Bundle (AMB):
Taut in flexion.
*
Posterolateral Bundle (PLB):
Taut in extension.
The femoral footprint is crescent-shaped, situated on the posteromedial aspect of the lateral femoral condyle, forming part of the "resident's ridge" (lateral intercondylar ridge) and "over-the-top" line. The tibial footprint is larger, anterior to the tibial spine, immediately posterior to the anterior horn of the medial meniscus. Precise identification of these footprints is crucial for anatomical reconstruction, aiming to replicate the native ACL's biomechanical function.
Neurovascular Supply:
The ACL receives its primary blood supply from the middle geniculate artery, a branch of the popliteal artery. It is innervated by branches of the posterior articular nerve (from the tibial nerve), which contains mechanoreceptors contributing to proprioception and kinesthesia. This neurosensory component is a critical aspect of knee function, often compromised after injury and difficult to fully restore with current reconstruction techniques.

Biomechanical Considerations:
The ACL's collagen fibers are arranged predominantly longitudinally, providing high tensile strength. Its viscoelastic properties allow it to absorb energy and dissipate forces. ACL failure typically occurs due to excessive anterior tibial translation combined with internal rotation, often during non-contact deceleration and pivoting movements. The "pivot shift" phenomenon, a pathognomonic sign of ACL deficiency, demonstrates anterior subluxation of the lateral tibial plateau relative to the lateral femoral condyle as the knee approaches extension, which then reduces with further flexion. This dynamic instability underscores the ACL's role in controlling tibiofemoral kinematics.
Graft choice for reconstruction also significantly impacts biomechanics. Autografts (bone-patellar tendon-bone, hamstrings) provide biological integration and mechanical properties similar to native ACL, though with donor site morbidity. Allografts (e.g., Achilles, tibialis anterior) avoid donor site morbidity but carry theoretical risks of disease transmission and slower incorporation. The biomechanical integrity of the reconstructed ACL relies heavily on tunnel placement, graft tensioning, and fixation strength, all of which must withstand physiological loads during the healing and rehabilitation phases.
Indications & Contraindications
The decision for operative versus non-operative management of an ACL injury is multifactorial, requiring careful consideration of patient-specific factors, injury characteristics, and functional goals. The primary indication for surgical reconstruction is symptomatic knee instability (giving way) that significantly impairs activities of daily living or participation in desired sports.
Operative Indications
- Young, active individuals: Patients with high athletic demands, particularly those involved in pivoting, cutting, or contact sports, who desire to return to their pre-injury activity level.
- Symptomatic instability: Recurrent episodes of "giving way" or subluxation during routine activities or sport, even after a trial of rehabilitation.
-
Concomitant injuries:
- Meniscal tears: Especially repairable meniscal tears (e.g., peripheral longitudinal tears, bucket handle tears) in an unstable knee, as an unstable knee significantly increases the risk of subsequent meniscal injury.
- Articular cartilage injuries: While not a direct indication, ACL reconstruction may be performed concurrently to stabilize the knee and protect articular surfaces.
- Multi-ligamentous injuries: Combined ACL and MCL, LCL, or PCL injuries often necessitate surgical stabilization.
- Patients with specific occupational demands: Individuals whose profession requires high levels of knee stability (e.g., military personnel, first responders).
- Failed non-operative management: Persistent instability or functional limitation despite adherence to a structured rehabilitation program.
Non-Operative Indications
- Older, low-demand individuals: Patients with sedentary lifestyles or those willing to modify their activity level to avoid provocative maneuvers.
- Asymptomatic instability: Individuals who experience no subjective instability or functional limitation despite a complete ACL tear.
- Significant medical comorbidities: Patients with conditions that increase surgical risk (e.g., severe cardiovascular disease, poorly controlled diabetes) or contraindicate anesthesia.
- Inability or unwillingness to comply with rehabilitation: Post-operative rehabilitation is crucial for successful outcomes; lack of adherence can compromise results.
- Skeletally immature patients: While increasingly being challenged, traditional approaches often favor delayed reconstruction until physeal closure to prevent growth disturbances. However, physeal-sparing techniques are now routinely employed for active, unstable skeletally immature patients.
Contraindications
Absolute contraindications for ACL reconstruction are rare and primarily revolve around patient health or inability to participate in the recovery process:
* Active systemic infection.
* Severe medical conditions precluding surgery or anesthesia.
* Inability to comply with post-operative rehabilitation.
* Fixed knee flexion contracture unresponsive to conservative measures.
* Severe osteoarthritis where arthroplasty is a more appropriate treatment.
Table 1: Operative vs. Non-Operative Indications for ACL Injury
| Factor | Operative Management | Non-Operative Management |
|---|---|---|
| Activity Level/Goals | High-demand athletics, pivoting sports, desire to return to pre-injury level | Low-demand activities, sedentary lifestyle, willingness to modify sports |
| Symptomatic Instability | Recurrent "giving way," positive pivot shift | Asymptomatic, no perceived instability |
| Age | Young (typically <40-50 years), skeletally mature (or physeal-sparing in immature) | Older (typically >50-60 years), low functional demands |
| Concomitant Injuries | Repairable meniscal tears, significant chondral lesions, multi-ligament injury | Isolated ACL tear without other significant intra-articular pathology |
| Functional Outcome | Desire for high-level function, prevent secondary meniscal/chondral injury | Acceptance of modified activity, potentially increased risk of secondary injury |
| Medical Comorbidities | Few/controlled | Significant, increasing surgical/anesthesia risk |
| Rehabilitation Adherence | High likelihood of compliance | Unlikely to comply fully with intensive rehabilitation |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is essential for a successful ACL reconstruction. This involves a comprehensive patient evaluation, selection of graft type, detailed imaging review, and meticulous surgical setup.
Pre-Operative Evaluation
- History: Document mechanism of injury, acute symptoms (pop, swelling, hemarthrosis), episodes of "giving way," previous knee injuries, activity level, and specific athletic goals.
-
Physical Examination:
- Inspection: Effusion, atrophy (especially quadriceps).
- Palpation: Joint line tenderness, patellar tracking.
- Range of Motion: Assess for flexion contracture or extension deficit.
-
Ligamentous Stability Tests:
- Lachman Test: Most sensitive for ACL integrity. Assesses anterior tibial translation at 20-30° flexion.
- Anterior Drawer Test: Performed at 90° flexion, less sensitive in acute setting due to hamstring spasm.
- Pivot Shift Test: Gold standard for dynamic ACL instability. Recreates the anterolateral rotatory subluxation seen with ACL deficiency.
- Varus/Valgus Stress Tests: Assess collateral ligament integrity.
-
Imaging:
- Radiographs: Weight-bearing AP, lateral, and Merchant views to assess alignment, exclude fractures, identify osteochondral lesions, and evaluate for early arthritic changes. May show Segond fracture (avulsion of lateral tibial plateau), indicative of ACL injury.
- Magnetic Resonance Imaging (MRI): Confirms ACL tear, identifies concomitant meniscal, chondral, or other ligamentous injuries, and can guide decision-making regarding surgical technique (e.g., stump preservation).
Graft Selection
The choice of graft material is critical and depends on surgeon preference, patient factors (age, activity level, previous surgeries), and graft availability.
*
Autografts:
*
Bone-Patellar Tendon-Bone (BPTB):
Gold standard, provides bone-to-bone healing, high primary fixation strength, excellent outcomes. Donor site morbidity includes patellofemoral pain, patellar fracture risk, and extensor mechanism weakness.
*
Hamstring Tendon Autograft (Semitendinosus and Gracilis - ST/G):
Common choice, less anterior knee pain than BPTB, potential for quicker rehabilitation. Disadvantages include slower graft incorporation, potential hamstring weakness, and theoretical risk of increased anterior tibial translation due to hamstring harvest. Quadriceps tendon autograft is an increasingly popular alternative, offering robust graft with less donor site morbidity than BTB.
*
Quadriceps Tendon Autograft (QT):
A robust graft with high ultimate load to failure, particularly useful for revision cases or larger patients. Can be harvested with or without a bone block.
*
Allografts:
(e.g., Achilles tendon, tibialis anterior, ST/G)
* Advantages: No donor site morbidity, shorter operative time.
* Disadvantages: Slower incorporation, potential for immune response, risk of disease transmission (though rigorously screened), higher re-rupture rates in younger, active individuals. Generally reserved for older, less active patients, or revision cases.
Pre-Operative Protocols
- Prehabilitation: Encourage pre-operative physical therapy to reduce swelling, restore full range of motion (especially extension), and strengthen the quadriceps. This has been shown to improve post-operative outcomes.
- Patient Education: Discuss the surgical procedure, graft options, risks, expected recovery, and rehabilitation commitment.
- Antibiotic Prophylaxis: Administer pre-operatively (e.g., cefazolin) to reduce infection risk.
- Thromboprophylaxis: Consider based on patient risk factors, though routinely not required for isolated ACL reconstruction.
Patient Positioning
- Supine Position: The patient is placed supine on the operating table.
- Tourniquet: A pneumatic tourniquet is applied high on the thigh, ensuring adequate pressure control throughout the procedure.
- Leg Holder: A lateral post or leg holder is typically used to stabilize the thigh, allowing the knee to be flexed and extended freely. Some surgeons prefer a foot block or an assistant to hold the foot, providing dynamic control over knee position.
- Foot Block/Pillow: A bump or pillow beneath the ipsilateral buttock may be used to allow for full knee extension and prevent external rotation of the limb.
- Preparation and Draping: The entire limb, from the tourniquet down to the toes, is meticulously prepped with an antiseptic solution (e.g., povidone-iodine or chlorhexidine) and draped in a sterile fashion, ensuring unrestricted access to the knee and potential graft harvest sites.
- Arterial Line/Foley Catheter: Consider for longer cases or patients with comorbidities.
Detailed Surgical Approach / Technique
ACL reconstruction is predominantly performed arthroscopically, allowing for minimally invasive access, enhanced visualization, and reduced post-operative morbidity compared to open techniques. The following outlines a common approach, acknowledging variations based on surgeon preference, graft choice, and specific patient anatomy.
1. Diagnostic Arthroscopy
- Portals: Standard anterolateral (AL) and anteromedial (AM) portals are established. The AL portal is typically used for the arthroscope, and the AM for instrumentation. An accessory medial portal may be used for better visualization or working angle.
-
Systematic Evaluation:
- Patellofemoral Joint: Assess articular cartilage, patellar tracking.
- Medial Compartment: Inspect medial meniscus, medial femoral condyle, tibial plateau, and MCL integrity.
- Lateral Compartment: Inspect lateral meniscus, lateral femoral condyle, tibial plateau, and LCL/popliteal complex integrity.
- Intercondylar Notch: Evaluate the integrity of the PCL, remnants of the torn ACL. Assess for notch osteophytes or impingement.
- ACL Stump Assessment: Note the amount of remaining ACL tissue. Some surgeons advocate for stump preservation where possible, hypothesizing improved graft revascularization and proprioception.
2. Graft Harvest (if autograft)
The specific harvest technique depends on the chosen graft:
-
Hamstring (Semitendinosus and Gracilis):
- A 2-3 cm oblique incision is made anteromedially, approximately 2-3 cm distal to the joint line and 2 cm medial to the tibial tubercle.
- Subcutaneous dissection to identify the sartorius fascia. The fascia is incised longitudinally.
- Identify the semitendinosus and gracilis tendons. The semitendinosus is typically more superficial and robust.
- Use a tendon stripper to harvest the tendons. Ensure the stripper follows the tendon closely to avoid injury to surrounding neurovascular structures (saphenous nerve and its infrapatellar branch).
- Prepare the graft on a back table, removing muscle remnants and sizing it to the desired diameter (typically 7.5-10 mm). Create whipstitches at each end for fixation.
-
Bone-Patellar Tendon-Bone (BPTB):
- A 5-7 cm midline longitudinal incision is made over the patellar tendon, extending from the inferior pole of the patella to the tibial tubercle.
- Dissect through subcutaneous tissue. Identify and protect the infrapatellar branch of the saphenous nerve.
- Harvest a central 10 mm wide strip of patellar tendon with a 20-25 mm long bone block from the inferior pole of the patella and a 25-30 mm long bone block from the tibial tubercle.
- Smooth the bone blocks, drill holes for suture passage, and prepare the graft.
-
Quadriceps Tendon (QT):
- A 3-5 cm longitudinal incision is made over the distal quadriceps tendon, superior to the patella.
- Harvest a 10-11 mm wide full-thickness or partial-thickness strip of quadriceps tendon, typically 70-80 mm long, with or without an associated patellar bone block.
- The quadriceps tendon provides a large, strong graft with less anterior knee pain than BPTB, though it involves proximal donor site morbidity.
3. Tibial Tunnel Creation
- ACL Footprint Preparation: Use a shaver and curette to debride the remnants of the torn ACL from the tibial footprint, exposing the native anatomical insertion site. Avoid over-resection, especially of the posterior cruciate ligament fibers.
- Tibial Guide Placement: An ACL tibial aim guide is used. The tip is placed anatomically at the center of the native tibial footprint, ensuring it is posterior to the anterior horn of the medial meniscus. The angle of the guide typically positions the wire exit anteromedially on the tibia.
- Drilling: A guide wire is drilled, followed by a cannulated drill bit matching the prepared graft diameter. Ensure the tibial tunnel is parallel to the Blumensaat's line and avoids posterior wall blow-out.
4. Femoral Tunnel Creation
Femoral tunnel placement is arguably the most critical step for achieving anatomical and biomechanical reconstruction. Common techniques include transtibial, anteromedial (AM) portal, and outside-in. An AM portal technique is generally preferred for anatomical single-bundle reconstruction.
-
Anatomical Single-Bundle via AM Portal:
- Hyperflex the knee to 110-120° to expose the lateral femoral condyle.
- Use the AM portal for instrumentation. Visualize the lateral intercondylar ridge ("resident's ridge") and the posterior wall of the notch.
- Identify the femoral footprint of the native ACL, which is typically located between the resident's ridge and the posterior cartilage margin of the lateral femoral condyle, closer to the posterior margin.
- Use a femoral aiming guide (e.g., offset guide) or a free-hand technique with a burr to create a starting point.
- A guide wire is drilled from the lateral aspect of the femur (or through the AM portal, depending on guide) into the desired femoral footprint.
- A cannulated drill bit, typically reverse-cutting, is then used to create the femoral socket to the appropriate depth (usually 25-30 mm). Ensure a stable posterior wall and avoid "killer turn" (excessive angle of graft in tunnel) by aiming the tunnel appropriately.
- Double-Bundle Reconstruction (Optional): If performing a double-bundle technique, separate tunnels for the AMB and PLB are created. The AMB tunnel is typically more anterior and superior, and the PLB tunnel more posterior and inferior on the femoral footprint.
5. Graft Passage and Fixation
- Graft Passage: A suture leader is passed through the femoral tunnel, then through the tibial tunnel. The graft is then pulled into the joint through the tibial tunnel and into the femoral socket. Ensure the graft is oriented correctly and is not twisted.
-
Femoral Fixation:
- Suspension Devices (e.g., Endobutton, adjustable loop devices): Most common for hamstring grafts. The device is passed through the femoral tunnel, flipped on the lateral femoral cortex, and then tensioned.
- Interference Screws: Used for BPTB grafts (bone block into bone tunnel) and often for hamstrings (graft directly into bone tunnel). Screws are inserted from the outside-in, engaging the graft within the tunnel.
- Cross-Pin Fixation: Pins are drilled perpendicular to the femoral tunnel, engaging the graft.
-
Tibial Fixation:
- Interference Screws: Most common, engaging the graft firmly within the tibial tunnel.
- Suture Posts/Washers: Suture from the graft is tied over a post or washer on the anteromedial aspect of the tibia.
- Staples: Can be used in conjunction with screws or alone.
-
Graft Tensioning:
This is critical.
- For single-bundle grafts, the graft is typically tensioned at 20-30° of knee flexion to balance tension between the AMB and PLB functions, and minimize impingement. Some prefer tensioning closer to full extension.
- Cyclic loading of the graft (e.g., 20 cycles) prior to final tensioning and fixation can minimize post-fixation creep.
- Final Assessment: Perform Lachman, Anterior Drawer, and Pivot Shift tests to confirm stability. Ensure full range of motion without impingement.
6. Wound Closure
- Close graft harvest site incisions in layers.
- Close arthroscopy portals with a single suture or sterile strips.
- Apply sterile dressings.
Complications & Management
Despite advancements in surgical technique, ACL reconstruction is not without potential complications. Recognizing and managing these effectively is crucial for optimizing patient outcomes.
Table 2: Common Complications of ACL Reconstruction, Incidence, and Salvage Strategies
| Complication | Incidence (%) | Etiology | Management / Salvage Strategy |
|---|---|---|---|
| Infection | 0.1 – 2.0 | Contamination during surgery, hematogenous spread | I&D, debridement, IV antibiotics; graft retention vs. removal (based on severity/timing) |
| Arthrofibrosis/Stiffness | 5 – 25 | Insufficient ROM post-op, poor graft placement, inflammatory response, impingement | Intensive PT, MUA, arthroscopic lysis of adhesions (cyclops lesion excision), notchplasty |
| Graft Failure/Re-rupture | 2 – 10 (primary) | Technical error, early return to sport, inadequate rehab, concomitant ligamentous laxity | Revision ACL reconstruction (often 2-stage), addressing underlying causes |
| Persistent Instability | 5 – 20 | Incomplete reconstruction, technical error (non-anatomic tunnels), concomitant ligament injury, medial/lateral laxity, meniscus tear | Revision ACL, lateral extra-articular tenodesis/ligament reconstruction |
| Anterior Knee Pain (AKP) | 10 – 40 (BPTB > HS) | Donor site morbidity (patellofemoral pain, patellar fracture), hardware irritation | PT, NSAIDs, steroid injection, hardware removal, debridement |
| Nerve Injury | < 1 (major) | Saphenous nerve (infrapatellar branch, for HS/BPTB), common peroneal nerve (traction/compression) | Observation, neurolysis, PT, bracing; usually transient |
| Vascular Injury | < 0.1 | Popliteal artery injury during drilling, excessive traction | Immediate vascular surgery consultation, repair |
| Deep Vein Thrombosis (DVT) | 0.5 – 5.0 | Hypercoagulability, stasis, endothelial injury | Anticoagulation (LMWH, oral), early mobilization |
| Complex Regional Pain Syndrome (CRPS) | < 1 | Multifactorial, sympathetic dysfunction | Multimodal pain management, PT, sympathetic blocks, gabapentin |
| Hardware-Related Issues | 2 – 5 | Prominent screws/buttons, migration, breakage | Hardware removal, revision fixation |
| Loss of Extension | 5 – 15 | Graft impingement (notch/roof), arthrofibrosis, cyclops lesion | PT, MUA, arthroscopic debridement/notchplasty |
| Patellar Fracture (BPTB) | 0.1 – 0.5 | Donor site weakness, stress riser | Open reduction internal fixation (ORIF), bracing, activity modification |
| Growth Plate Injury (Skeletally Immature) | Variable | Transphyseal tunnel placement | Physeal-sparing techniques, epiphysiodesis, guided growth, corrective osteotomy |
| Osteoarthritis Progression | Long-term (high) | Pre-existing damage, meniscal injury, residual instability, altered knee kinematics | Lifestyle modification, injections, eventual arthroplasty |
Key Management Principles:
*
Prevention:
Meticulous surgical technique, appropriate graft selection, rigorous pre-operative planning, and adherence to sterile protocols are paramount.
*
Early Recognition:
Vigilant post-operative monitoring for signs of complications.
*
Conservative Management:
Many complications (e.g., mild stiffness, AKP) can be initially managed with aggressive physical therapy, NSAIDs, and activity modification.
*
Surgical Intervention:
For severe or refractory complications, surgical revision, debridement, or hardware removal may be necessary.
*
Multidisciplinary Approach:
Involving pain specialists, infectious disease consultants, or vascular surgeons when appropriate.
Post-Operative Rehabilitation Protocols
A structured and progressive post-operative rehabilitation protocol is as critical to the success of ACL reconstruction as the surgical technique itself. The protocol is typically divided into phases, with clear goals and criteria for progression. It aims to protect the healing graft, restore range of motion (ROM), regain muscle strength and neuromuscular control, and facilitate a safe return to sport.
General Principles
- Individualization: Protocols are guidelines; progression must be tailored to the individual patient's healing response, pain, swelling, and functional milestones.
- Protection of Graft: The graft undergoes a process of "ligamentization," which involves avascular necrosis followed by revascularization and cellular repopulation. During this time, the graft is weakest at approximately 6-12 weeks post-op. Activities must respect this biological healing process.
- Early Motion: Early, controlled ROM is crucial to prevent arthrofibrosis and promote articular cartilage health.
- Quadriceps Reactivation: Regaining quadriceps strength and control is a primary focus throughout rehabilitation.
- Neuromuscular Control and Proprioception: Essential for dynamic knee stability and reducing re-injury risk.
- Weight-Bearing: Typically initiated early, often protected, depending on concomitant injuries (e.g., meniscal repair).
- Bracing: Post-operative bracing is controversial. Functional bracing may be considered for return to sport in high-risk individuals, but its efficacy in preventing re-injury is not definitively proven.
Phases of Rehabilitation
Phase 1: Immediate Post-operative (Weeks 0-2)
*
Goals:
Protect graft, control pain and swelling, achieve full knee extension, initiate quadriceps activation, immediate protected weight-bearing.
*
Focus:
*
Pain & Swelling:
RICE (Rest, Ice, Compression, Elevation).
*
ROM:
Achieve 0-90° flexion by week 1. Emphasize full extension immediately with passive extension exercises, prone hangs.
*
Weight-Bearing:
Partial weight-bearing with crutches (PWB) as tolerated, typically progressing to full weight-bearing (FWB) within the first 1-2 weeks, unless meniscal repair or other contraindications.
*
Strengthening:
Quadriceps setting, straight leg raises (SLR) in multiple planes, ankle pumps.
*
Exercises:
Ankle pumps, quad sets, passive knee extension (prone hangs, towel stretch), heel slides (active-assisted flexion).
Phase 2: Early Strengthening & Full ROM (Weeks 2-6)
*
Goals:
Restore full knee ROM (0-120°+), progress weight-bearing, normalize gait pattern, improve quadriceps and hamstring strength.
*
Focus:
*
ROM:
Continue to work towards full flexion while maintaining full extension.
*
Strengthening:
Closed kinetic chain (CKC) exercises (mini-squats, wall slides, leg press - avoiding deep flexion initially), open kinetic chain (OKC) hamstring curls. OKC knee extension is typically limited or avoided initially due to anterior shear forces on the graft.
*
Balance & Proprioception:
Single leg stance, balance board (initially bilateral).
*
Exercises:
Stationary bike (light resistance), elliptical, step-ups, hamstring curls (OKC), calf raises, balance training.
Phase 3: Intermediate Strengthening & Neuromuscular Control (Weeks 6-12)
*
Goals:
Regain >75% strength compared to contralateral limb, improve endurance, initiate sport-specific light activities, progress neuromuscular control.
*
Focus:
*
Strength:
Progress CKC exercises with increased resistance and depth. Incorporate OKC knee extension from 90°-45° (to minimize anterior shear) or avoid until later. Focus on gluteal and core strengthening.
*
Proprioception:
Advanced balance exercises (unstable surfaces, single-leg hopping).
*
Agility:
Begin linear agility drills (e.g., walking lunges, lateral shuffles, controlled backward walking) without cutting or twisting.
*
Exercises:
Stair master, lunges, squats (deeper), leg press, gentle plyometrics (box jumps - landing and soft controlled movements), treadmill walking/light jogging.
Phase 4: Advanced Strengthening & Return to Sport (Weeks 12-24+)
*
Goals:
Achieve near-normal strength, power, and endurance. Progress to sport-specific drills, prepare for return to full activity.
*
Focus:
*
Strength & Power:
High-intensity CKC and OKC exercises. Plyometric training (jumping, bounding), agility ladders, sport-specific drills.
*
Neuromuscular Control:
Advanced agility, cutting drills, reactive drills.
*
Psychological Readiness:
Address fear of re-injury.
*
Return to Sport (RTS) Criteria:
Typically not before 6-9 months, often closer to 9-12 months for competitive athletes, depending on sport and graft type. Criteria include:
* Full, pain-free ROM.
* No effusion.
* Quadriceps strength >90% of contralateral limb.
* Hamstring strength >90% of contralateral limb.
* Passing functional hop tests (>90% limb symmetry index - LSI).
* Passing sport-specific agility drills.
* Normal knee examination (negative Lachman, pivot shift).
* Psychological readiness.
Special Considerations
- Meniscal Repair: May require delayed weight-bearing or restricted ROM initially.
- Multi-ligamentous Injury: Rehabilitation is often prolonged and more conservative.
- Skeletally Immature: Physeal-sparing techniques necessitate careful monitoring for growth disturbances.
Consistent communication between the surgeon, physical therapist, and patient is essential to ensure safe and effective progression through rehabilitation.
Summary of Key Literature / Guidelines
The landscape of ACL reconstruction has been shaped by decades of research, leading to an evidence-based approach to patient selection, surgical technique, and post-operative management.
1. Anatomical Reconstruction:
Modern ACL reconstruction emphasizes anatomical restoration of the native ACL footprint, particularly on the femur. The recognition of the two distinct bundles (AMB and PLB) led to the development of double-bundle techniques, aiming for superior rotational stability. However, systematic reviews and meta-analyses have largely shown no significant clinical superiority of double-bundle over well-performed single-bundle anatomical reconstruction in terms of objective stability or patient-reported outcomes, particularly regarding return to sport rates or re-rupture rates. Single-bundle anatomical reconstruction remains the gold standard, focusing on proper tunnel placement to mimic the native ACL's biomechanical role.
2. Graft Selection:
The choice between autograft and allograft, and within autografts (BPTB, hamstring, quadriceps), is a subject of ongoing debate.
*
Autografts:
Generally recommended for young, active patients due to superior re-rupture rates. The MOON (Multicenter Orthopaedic Outcomes Network) group studies have provided extensive data on various graft types, often showing comparable outcomes between BPTB and hamstring autografts in many parameters, though with different donor site morbidities. Patellofemoral pain is more common with BPTB, while hamstring weakness is a concern with hamstring grafts. Quadriceps tendon autograft is gaining popularity due to its robust size, low donor site morbidity compared to BPTB, and excellent biomechanical properties.
*
Allografts:
While avoiding donor site morbidity, allografts consistently show higher re-rupture rates, particularly in younger, high-demand patients. Their use is generally reserved for older, less active patients, revision cases, or multi-ligament injuries where autograft options are limited.
3. Timing of Surgery:
The prevailing consensus supports delaying ACL reconstruction until acute inflammation has subsided, and full knee range of motion, especially extension, has been regained. Early surgery in an acutely swollen, stiff knee significantly increases the risk of post-operative arthrofibrosis. Studies suggest surgery within 3-6 weeks, or even later, after achieving full pre-operative motion, yields better outcomes.
4. Rehabilitation Protocols:
Accelerated rehabilitation protocols, emphasizing early weight-bearing and full range of motion, have replaced prolonged immobilization, reducing complications like arthrofibrosis and quadriceps atrophy. However, the graft is biologically vulnerable in the first few months. Current guidelines emphasize objective criteria for return to sport (RTS), including functional hop tests, strength symmetry (>90% LSI), and psychological readiness, typically not before 6-9 months post-op, with many advocating for 9-12 months for high-level athletes to allow for sufficient graft maturation and neuromuscular adaptation.
5. Prevention of Post-Traumatic Osteoarthritis (PTOA):
Despite successful ACL reconstruction, the long-term risk of PTOA remains a significant concern, with incidence rates as high as 50% within 10-20 years. This risk is influenced by pre-existing cartilage damage, concomitant meniscal injuries (especially meniscectomy), and residual knee laxity or altered kinematics. Current research focuses on understanding the molecular mechanisms of PTOA and developing biologic augmentation strategies to mitigate its progression.
6. Lateral Extra-Articular Tenodesis (LET):
The role of adjuvant lateral extra-articular tenodesis (or ligament reconstruction) in primary ACL reconstruction is increasingly recognized, particularly in patients at high risk of failure (e.g., young athletes, high-grade pivot shift, generalized ligamentous laxity, revision cases). Studies have shown that adding an LET can significantly reduce re-rupture rates in these populations by augmenting rotational stability, especially against the pivot shift phenomenon.
7. Skeletally Immature Patients:
Managing ACL injuries in skeletally immature patients presents a challenge due to the risk of physeal arrest and growth disturbance. Physeal-sparing techniques (e.g., all-epiphyseal, partial transphyseal, or extra-physeal) are increasingly preferred over traditional transphyseal methods for active, unstable children, allowing for anatomical reconstruction while minimizing growth plate disruption.
8. Future Directions:
Ongoing research explores biologic augmentation strategies (e.g., PRP, mesenchymal stem cells), improved scaffold materials for ligament repair, personalized biomechanical analysis, and advanced imaging to predict and prevent PTOA. The goal remains to optimize functional outcomes, minimize re-injury rates, and preserve long-term knee health.
The American Academy of Orthopaedic Surgeons (AAOS) and the International Society of Arthroscopy, Knee Surgery and Orthopaedic Sports Medicine (ISAKOS) provide regularly updated clinical practice guidelines and consensus statements that serve as key references for contemporary ACL management.
You Might Also Like