Comprehensive Guide to Lateral Femoral Condyle Pathologies: Anatomy, Biomechanics & Management
Key Takeaway
Lateral femoral condyle injuries encompass diverse knee pathologies, from osteochondral lesions to articular fractures. They significantly impair knee function, stability, and load distribution. Understanding the LFC's surgical anatomy and biomechanics is paramount for accurate diagnosis and effective management, aiming to restore alignment, articular integrity, and long-term knee function.
Introduction & Epidemiology
Injuries involving the lateral femoral condyle (LFC) represent a diverse spectrum of knee pathologies, ranging from isolated osteochondral lesions and subchondral bone bruises to complex articular fractures and ligamentous avulsions. As a critical component of the tibiofemoral and patellofemoral articulations, the integrity of the LFC is paramount for normal knee kinematics, stability, and load distribution. Disruption of this anatomical region can lead to significant functional impairment, chronic pain, instability, and accelerated degenerative changes if not appropriately diagnosed and managed.
The epidemiology of LFC injuries varies with the specific pathology. Articular fractures of the distal femur, while less common than tibial plateau fractures, often involve the condyles, with LFC involvement frequently seen in high-energy trauma, particularly those with a valgus component. Supracondylar-intercondylar distal femoral fractures (AO/OTA 33-C type) frequently involve the LFC, accounting for a significant proportion of surgically managed distal femur fractures. Isolated LFC fractures are rarer but can result from direct impact or avulsion mechanisms. Osteochondral lesions, on the other hand, are commonly encountered in younger, active populations following acute traumatic events (e.g., patellar dislocation, pivot-shift injury) or as manifestations of osteochondritis dissecans (OCD). The lateral femoral condyle is a recognized site for OCD, though less common than the medial condyle. Impaction injuries, such as the characteristic deep sulcus sign or lateral condyle bone bruise seen on MRI after an acute ACL tear (Segond fracture equivalent), underscore the LFC's vulnerability during knee instability events.
These injuries demand a thorough understanding of their anatomical basis, biomechanical consequences, and a meticulous approach to diagnosis and treatment. Given the LFC's role in knee stability and articular congruence, optimal management aims to restore the anatomical alignment, articular surface integrity, and ultimately, long-term knee function while minimizing the risk of post-traumatic osteoarthritis.
Surgical Anatomy & Biomechanics
Surgical Anatomy
The lateral femoral condyle forms the lateral aspect of the distal femur, articulating with the lateral tibial plateau and contributing to the patellofemoral joint via the lateral trochlear facet.
- Articular Surface: The articular cartilage of the LFC is thicker than its medial counterpart posteriorly, reflecting its specific load-bearing patterns. It possesses a distinct trochlear groove anteriorly for patellar articulation and a more convex tibiofemoral articular surface.
-
Ligamentous and Tendinous Attachments:
- Fibular Collateral Ligament (FCL): Originates from the lateral femoral epicondyle, a palpable bony prominence superior and posterior to the LFC articular cartilage. It is a primary static stabilizer against varus stress.
- Popliteus Tendon: Originates from the lateral aspect of the lateral femoral condyle, anterior and inferior to the FCL origin, within a distinct groove. It plays a crucial role in internal rotation of the tibia on the femur, unlocking the knee, and provides posterior and lateral stability.
- Lateral Gastrocnemius Head: Originates from the posterior aspect of the lateral femoral condyle, forming part of the posterolateral corner (PLC).
- Posterolateral Capsule: Firmly attached to the LFC.
- Vascular Supply: The distal femur receives its blood supply from the genicular arteries, branches of the popliteal artery. The superior and inferior lateral genicular arteries contribute significantly to the vascularization of the lateral femoral condyle and surrounding soft tissues.
- Neural Structures: The common peroneal nerve is located superficially on the lateral aspect of the fibular neck, posterior to the biceps femoris tendon. While not directly adjacent to the LFC, it is vulnerable during posterolateral surgical approaches or extensive dissection of the posterolateral corner structures.
- Cortical Bone: The subchondral bone of the LFC is compact, transitioning to cancellous bone within the metaphysis. Fracture patterns often involve this cancellous bone and can extend into the articular surface.
Biomechanics
The LFC is integral to the complex biomechanics of the knee joint.
- Tibiofemoral Kinematics: During flexion and extension, the LFC exhibits a "roll-back" phenomenon, particularly in higher degrees of flexion, allowing for a larger range of motion without impingement. It influences screw-home mechanism during terminal extension. Its shape and orientation dictate the path of the lateral meniscus during knee movement.
- Load Bearing: Under axial loading, the lateral compartment of the knee typically experiences slightly less load than the medial compartment in healthy alignment. However, in certain activities or deformities (e.g., valgus alignment), the LFC can be subjected to increased compressive and shear forces.
- Stability: The LFC, in conjunction with its ligamentous and tendinous attachments (FCL, popliteus, PLC), provides crucial stability against varus angulation, external rotation, and posterior translation of the tibia. Impairment of the LFC (e.g., depression fracture, large osteochondral defect) can lead to altered joint mechanics, increased contact pressures, and subsequent chondral damage or instability.
- Patellofemoral Tracking: The lateral trochlear facet guides patellar motion. Fractures or significant osteochondral defects in this region can lead to patellar instability or maltracking, causing anterior knee pain and potential chondrosis.
- Associated Injuries: Understanding the biomechanics is crucial for recognizing associated injuries. A common example is the "pivot-shift" injury in ACL tears, where a valgus and internal rotation force causes the lateral tibia to subluxate anteriorly, impacting the LFC. This often results in a bone bruise of the posterior LFC and an anterior-central lateral tibial plateau, or even an osteochondral fracture of the LFC.

Indications & Contraindications
The decision for operative versus non-operative management of LFC injuries is multifactorial, depending on the type and severity of the injury, patient factors (age, activity level, comorbidities), and the presence of associated pathologies.
Operative Indications
-
Displaced Articular Fractures:
- Articular step-off or gap >2 mm.
- Significant depression of the articular surface (>5 mm).
- Involvement of a large portion of the condyle, potentially leading to instability or altered load transmission.
- Open fractures.
- Fractures with associated neurovascular compromise or compartment syndrome.
- Fractures involving the weight-bearing axis that compromise knee stability.
- Fractures with significant metaphyseal comminution or displacement requiring buttress support.
-
Osteochondral Lesions:
- Large, unstable, or symptomatic osteochondral fragments (e.g., Outerbridge Grade III-IV lesions, ICRS Grade 3-4).
- Loose bodies causing mechanical symptoms (locking, catching).
- Full-thickness cartilage defects (ICRS Grade 3-4) >1-2 cm² in high-demand patients.
- Failed non-operative management of symptomatic lesions.
- Osteochondritis dissecans (OCD) lesions that are unstable, displaced, or failing conservative treatment.
-
Avulsion Fractures:
- Displaced avulsion fractures involving ligamentous or tendinous attachments (e.g., FCL, popliteus, lateral retinaculum) resulting in knee instability.
-
Associated Injuries:
- Ligamentous instability requiring concomitant repair/reconstruction (e.g., ACL tear with LFC impaction fracture, PLC injury).
- Meniscal tears amenable to repair.
Non-Operative Indications
-
Non-Displaced or Minimally Displaced Articular Fractures:
- Articular step-off <2 mm and gap <5 mm.
- Stable fractures without significant instability.
- Minimally symptomatic or incidental osteochondral lesions.
-
Small, Stable Osteochondral Lesions:
- Focal, stable lesions without loose bodies or significant mechanical symptoms (e.g., ICRS Grade 1-2).
- Bone bruises without obvious fracture lines or articular disruption.
-
Low-Demand Patients:
- Elderly or sedentary individuals with stable injuries where surgical risks outweigh potential benefits.
-
Contraindications to Surgery:
- Severe comorbidities precluding safe anesthesia and surgery.
Contraindications
-
Absolute Contraindications:
- Active local or systemic infection.
- Severe, irreversible comorbidities (e.g., unstable cardiac disease, uncontrolled diabetes) that significantly increase perioperative risk.
- Extensive, unsalvageable soft tissue damage.
- Patient refusal.
-
Relative Contraindications:
- Significant osteoporosis potentially compromising fixation.
- Poor patient compliance with rehabilitation protocols.
- Severe peripheral vascular disease.
- Morbid obesity.
| Indication Type | Operative Indications | Non-Operative Indications |
|---|---|---|
| Fractures | Displaced articular fractures (>2mm step-off, >5mm gap) | Non-displaced or minimally displaced fractures (<2mm step-off, <5mm gap) |
| Open fractures | Stable stress fractures without articular involvement | |
| Fractures with neurovascular compromise | Low-demand patients with stable injuries | |
| Osteochondral Lesions | Large (>1-2 cm²), unstable, or symptomatic lesions (Grade III-IV) | Small, stable, asymptomatic lesions (Grade I-II) |
| Loose bodies causing mechanical symptoms | Bone bruises without articular disruption | |
| Unstable OCD lesions failing conservative treatment | ||
| Ligamentous/Avulsion | Displaced avulsion fractures causing instability | Minimally displaced avulsions with stable knee |
| Concomitant ligamentous instability requiring reconstruction | Isolated ligamentous sprains (Grades I-II) without bony avulsion |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is essential for successful outcomes in LFC injuries, considering the complex anatomy and potential for associated pathology.
Imaging
-
Radiographs:
- Standard AP, lateral, and oblique views are crucial for initial assessment of fractures, alignment, and joint space.
- Weight-bearing AP views can assess mechanical axis and joint line congruity if stability allows.
- Specialized views (e.g., notch view, Merchant view for patellofemoral tracking) may be beneficial.
- For OCD, Tunnel (notch) view may better visualize the lesion.
-
Computed Tomography (CT) Scan:
- Indications: Complex articular fractures, comminuted fractures, evaluation of fracture extension, quantification of articular step-off/gap, assessment of fragment size and location.
- Utility: 3D reconstructions are invaluable for understanding fracture morphology, planning reduction strategies, and selecting appropriate implants. It helps identify occult fractures not clearly visible on plain radiographs.
-
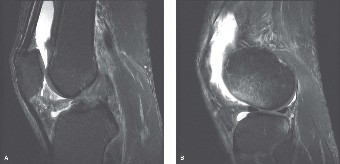
Magnetic Resonance Imaging (MRI):
- Indications: Suspected osteochondral lesions, occult fractures, bone bruises, meniscal tears, ligamentous injuries (e.g., ACL, PCL, FCL, PLC), and other soft tissue pathology.
- Utility: Excellent for assessing cartilage integrity, subchondral bone health, identifying edema patterns, and detailing soft tissue involvement, which is common with LFC injuries. Critical for pre-operative assessment of OCD lesions.
Surgical Plan
- Determine Surgical Approach: Based on imaging and injury type (arthroscopic, mini-open, formal open).
-
Implant Selection:
- Fractures: Cannulated lag screws (partially threaded), headless compression screws, small fragment plates (e.g., pre-contoured lateral distal femoral locking plates), K-wires for temporary fixation.
- Osteochondral Lesions: Osteochondral autograft transfer system (OATS), allograft plugs, matrix-induced autologous chondrocyte implantation (MACI) patches, microfracture instruments, debridement tools.
- Anesthesia: General or regional anesthesia, often combined with a femoral nerve block for post-operative analgesia.
- Tourniquet: Essential for a bloodless field, usually placed high on the thigh.
- Antibiotics: Pre-operative prophylactic antibiotics are standard.
- Blood Products: Type and cross-match may be considered for complex fracture cases with anticipated significant blood loss.
Patient Positioning
- Supine Position: The patient is positioned supine on the operating table.
- Operating Table Setup: A radiolucent table is often preferred to allow fluoroscopic imaging during the procedure, especially for fracture fixation.
- Leg Holder/Post: A lateral post or leg holder can be used to stabilize the thigh and allow free flexion/extension of the knee. This facilitates access to the lateral aspect of the knee and allows for traction or manipulation as needed.
- Foot Position: The foot is usually free to allow the surgeon to manipulate the lower leg for various views or to assess stability. For some approaches, the leg may be draped out free.
- Preparation and Draping: Standard sterile preparation from the mid-thigh to the foot, ensuring the entire knee region is accessible. Draping should allow for knee flexion and extension throughout the procedure, and if fluoroscopy is planned, the draping should accommodate the C-arm.
Detailed Surgical Approach / Technique
Surgical management of LFC injuries varies significantly based on the specific pathology (fracture, osteochondral lesion, avulsion) and its severity. The following describes general principles and common techniques.
General Principles
- Arthroscopic Assessment: For many LFC pathologies, particularly osteochondral lesions or impaction injuries, initial diagnostic arthroscopy is invaluable. It allows for thorough assessment of the articular surface, menisci, ligaments, and provides an opportunity for lavage and removal of loose bodies. Arthroscopy can often be combined with an open procedure (arthroscopically assisted fixation) or be the primary treatment modality.
- Tourniquet Use: A pneumatic tourniquet applied to the proximal thigh is routinely used to maintain a bloodless field, crucial for precise anatomical reduction and fixation, especially in articular fractures and osteochondral repairs.
- Fluoroscopy: Intraoperative fluoroscopy is essential for confirming reduction and hardware placement in fracture fixation.
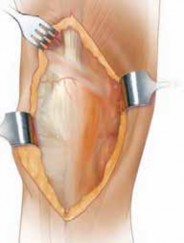
Surgical Approach to the Lateral Femoral Condyle
For open reduction and internal fixation (ORIF) of LFC fractures, a direct lateral approach or a lateral parapatellar approach offers good visualization.
- Incision: A longitudinal incision is made slightly lateral to the patella, extending proximally and distally as needed. Alternatively, a more posterior lateral incision can be used if posterior condylar involvement is significant.
- Subcutaneous Dissection: Dissect through subcutaneous fat. Identify and protect the terminal branches of the lateral femoral cutaneous nerve and saphenous nerve branches.
- Fascial Layer: The iliotibial band (ITB) is encountered. Depending on the extent of exposure needed, the ITB can be split longitudinally or incised anteriorly and retracted posteriorly, or vice versa. For most condylar exposures, an anterior reflection of the ITB, or an incision in its posterior aspect with anterior reflection of the anterior portion, provides adequate access.
-
Vastus Lateralis:
Deep to the ITB, the vastus lateralis muscle is encountered.
- Subvastus Approach: For less extensive exposures, the vastus lateralis can be reflected anteriorly off the lateral intermuscular septum. This preserves the muscle belly and minimizes quadriceps disruption.
- Transvastus Approach: For more extensive exposure, especially if plating is required, a longitudinal split can be made through the vastus lateralis muscle belly. Care must be taken to identify and preserve the motor nerve supply to the vastus lateralis, which typically enters the muscle from its medial aspect.
- Lateral Intermuscular Septum: Identify the lateral intermuscular septum, which attaches from the ITB to the linea aspera. This septum forms a natural plane.
- Capsular Incision: Once the LFC is exposed, a longitudinal capsular incision is made, carefully reflecting it to expose the articular surface and fracture fragments. Protect the lateral meniscus and avoid unnecessary detachment.
- Internervous Plane: The approach to the LFC is primarily between the vastus lateralis (innervated by the femoral nerve) anteriorly and the biceps femoris and associated structures of the posterolateral corner (innervated by the common peroneal nerve, a branch of the sciatic nerve) posteriorly. The plane of dissection is typically anterior to the common peroneal nerve.
Detailed Surgical Technique for Fractures
-
Reduction:
- Direct Visualization: Achieve direct visualization of the articular surface and fracture lines.
- Elevation of Depressed Fragments: For depressed articular fragments, use an osteotome or blunt elevator to carefully elevate the fragment back to its anatomical position. This may require creating a small cortical window on the lateral non-articular surface if the fragment cannot be manipulated adequately from the fracture site.
- Bone Grafting: If there is a metaphyseal void beneath an elevated articular segment, it should be filled with autologous cancellous bone graft (e.g., from the iliac crest or proximal tibia) or an allograft substitute to prevent re-collapse and provide structural support.
- Fragment Manipulation: Use reduction clamps, pointed reduction forceps, or K-wires as joysticks to align fracture fragments.
- Restore Articular Congruity: The primary goal is to restore a smooth, congruent articular surface. This is assessed visually and often confirmed with fluoroscopy.
- Temporary Fixation: Once anatomical reduction is achieved, use multiple K-wires to temporarily secure the fragments. Check knee range of motion to ensure no impingement or instability.
-
Definitive Fixation:
- Lag Screws: For large, single or few fragments, fully or partially threaded cannulated lag screws (e.g., 4.0 mm or 4.5 mm) can be inserted perpendicular to the fracture plane. Screws can be placed in an anterior-to-posterior (AP) or posterior-to-anterior (PA) direction depending on fragment size and surgeon preference, ensuring the screw heads are countersunk and do not violate the articular surface or exit the posterior cortex excessively.
- Buttress Plating: For comminuted fractures, those with significant metaphyseal extension, or when lag screws alone are insufficient for stability, a pre-contoured lateral distal femoral locking plate is often employed. The plate acts as a buttress to support the reduced articular fragments and provides stability to the metaphyseal and diaphyseal components. Screws are placed through the plate into the condylar fragments (non-locking or locking) and into the femoral shaft (locking screws).
- Combined Techniques: Often, a combination of lag screws for articular reduction and a buttress plate for overall stability is used.
- Hardware Placement: Ensure hardware does not impinge on the patellofemoral joint, popliteus tendon, or FCL origin. Use fluoroscopy in multiple planes (AP, lateral, oblique) to confirm screw length and position, avoiding intra-articular penetration.
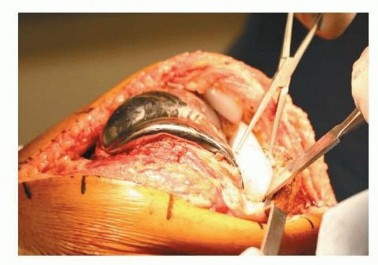
Detailed Surgical Technique for Osteochondral Lesions
- Arthroscopic Debridement: Via standard arthroscopic portals (anterolateral, anteromedial), unstable cartilage flaps are debrided to a stable rim. Loose bodies are removed.
- Microfracture: For contained, full-thickness defects (<2-3 cm²) with healthy surrounding cartilage and viable subchondral bone, microfracture can be performed. The calcified cartilage layer is removed, and multiple perforations (3-4 mm apart) are made in the subchondral bone using an awl to allow mesenchymal stem cells from the bone marrow to form a fibrocartilaginous repair tissue.
- Osteochondral Autograft Transfer System (OATS - Mosaicplasty): For larger, focal defects (>1 cm²), cylindrical osteochondral plugs can be harvested from non-weight-bearing areas of the knee (e.g., superior aspect of the intercondylar notch, lateral edge of the medial femoral condyle) and press-fit into the prepared defect. This technique aims to restore hyaline cartilage to the defect.
- Osteochondral Allograft: For very large defects (>3-4 cm²) or when autograft harvest is not feasible, fresh osteochondral allografts can be utilized. This involves precise sizing and implantation of a cadaveric plug or shell, offering the advantage of true hyaline cartilage replacement. Requires careful donor matching and handling.
- Autologous Chondrocyte Implantation (ACI) / Matrix-Associated ACI (MACI): A two-stage procedure where chondrocytes are harvested, cultured, and then reimplanted. MACI involves seeding chondrocytes onto a biodegradable matrix, which is then implanted into the defect and sealed. These techniques are typically reserved for larger, full-thickness defects in younger patients.
Closure
- Capsular Repair: The joint capsule is repaired to restore the knee's integrity.
- Muscle and Fascial Repair: The vastus lateralis, if incised, and the ITB are repaired.
- Subcutaneous and Skin Closure: Standard layered closure.
Complications & Management
Complications following surgical intervention for lateral femoral condyle injuries, while not universally occurring, can significantly impact patient outcomes. Vigilant monitoring and timely management are paramount.
Early Complications
-
Infection (0.5-5%):
- Presentation: Localized pain, redness, swelling, warmth, fever, purulent drainage.
- Management: Superficial infections may respond to oral antibiotics. Deep infections often require surgical irrigation and debridement, intravenous antibiotics, and potentially hardware removal. Culture-guided antibiotic therapy is essential.
-
Neurovascular Injury (Rare, <1%):
- Presentation: Common peroneal nerve palsy (foot drop, numbness lateral lower leg), vascular compromise (pallor, pulselessness, paresthesia, paralysis, pain).
- Management: Careful surgical dissection is key for prevention. Peroneal nerve injury may require exploration, neurolysis, or observation depending on severity. Vascular injury requires immediate surgical exploration and repair.
-
Compartment Syndrome (Rare, <1%):
- Presentation: Disproportionate pain, paresthesias, pain with passive stretch, tense compartment.
- Management: Immediate fasciotomy.
-
Deep Vein Thrombosis (DVT) / Pulmonary Embolism (PE) (1-5% for major knee surgery):
- Presentation: Swelling, pain in calf (DVT); dyspnea, chest pain, hypoxia (PE).
- Management: Prophylactic anticoagulation is standard. Treatment involves therapeutic anticoagulation.
-
Wound Healing Issues (1-5%):
- Presentation: Dehiscence, necrosis, hematoma formation.
- Management: Local wound care, débridement, delayed closure, or skin grafting if necessary.
Late Complications
-
Nonunion / Malunion (5-10% for complex fractures):
- Presentation: Persistent pain, instability, deformity, hardware failure, delayed weight-bearing.
- Management: Revision surgery with débridement of nonunion site, re-reduction, rigid fixation (often with locking plates), and bone grafting (autograft or allograft). Corrective osteotomy for malunion.
-
Post-Traumatic Arthritis (Common, up to 30-50% for articular fractures):
- Presentation: Chronic pain, stiffness, crepitus, loss of motion, swelling.
- Management: Conservative measures (NSAIDs, injections, physical therapy) initially. For severe, end-stage arthritis, surgical options include arthroscopy (débridement), osteotomy (to offload compartment), or partial/total knee arthroplasty, depending on age, activity level, and extent of arthritis.
-
Stiffness / Arthrofibrosis (5-15%):
- Presentation: Restricted range of motion, pain, reduced function.
- Management: Aggressive physical therapy, dynamic splinting. If conservative measures fail, arthroscopic or open arthrolysis may be required.
-
Instability (Variable, depending on associated ligamentous injury):
- Presentation: Feeling of giving way, recurrent effusions.
- Management: Physical therapy to strengthen dynamic stabilizers. If persistent, revision surgery to address underlying ligamentous laxity or bony malalignment.
-
Hardware Failure / Prominence (5-10%):
- Presentation: Pain, palpable hardware, loosening, fracture of implants.
- Management: Hardware removal, especially if symptomatic. If associated with nonunion, revision fixation.
-
Avascular Necrosis (AVN) of Fragments (Rare but devastating, <1%):
- Presentation: Progressive pain, collapse of articular segment, imaging evidence of osteonecrosis.
- Management: Often leads to post-traumatic arthritis. Small lesions may be managed with débridement. Larger lesions may require osteochondral grafting or eventually arthroplasty.
| Complication | Incidence (Approx.) | Salvage Strategies / Management |
|---|---|---|
| Infection | 0.5-5% | Debridement, IV antibiotics, hardware removal (if persistent/deep) |
| Neurovascular Injury | <1% | Immediate exploration & repair (vascular), observation/neurolysis (nerve) |
| Compartment Syndrome | <1% | Immediate fasciotomy |
| DVT/PE | 1-5% | Therapeutic anticoagulation, filter placement (for contraindications) |
| Wound Issues | 1-5% | Local wound care, debridement, delayed closure, skin grafting |
| Nonunion/Malunion | 5-10% | Revision ORIF with bone grafting, corrective osteotomy |
| Post-Traumatic Arthritis | 30-50% | Conservative, arthroscopy, osteotomy, TKA/UKA |
| Stiffness/Arthrofibrosis | 5-15% | Aggressive PT, dynamic splinting, arthroscopic/open arthrolysis |
| Instability | Variable | PT, revision ligamentous reconstruction/fixation |
| Hardware Failure/Prominence | 5-10% | Symptomatic hardware removal, revision fixation |
| Avascular Necrosis (AVN) | <1% | Débridement, osteochondral grafting, arthroplasty |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is a critical determinant of functional outcome after lateral femoral condyle surgery. Protocols are typically structured in phases, progressing from protection and early motion to strengthening and return to activity, tailored to the specific injury and surgical repair.
General Principles
- Weight-Bearing (WB) Restrictions: Strictly dictated by the stability of fixation (for fractures) or the nature of osteochondral repair (e.g., microfracture, OATS). Non-weight-bearing (NWB) or touch-down weight-bearing (TDWB) is common for 6-12 weeks in articular fractures or cartilage repair.
- Range of Motion (ROM): Initiated early but often with controlled limits to protect repairs.
- Pain and Swelling Management: Essential throughout recovery to facilitate participation in therapy.
- Bracing: A hinged knee brace is frequently used, initially locked in extension for protection, then gradually allowing controlled ROM.
Phase I: Protection and Early Motion (Weeks 0-6/8)
- Goals: Protect surgical repair, minimize pain and swelling, restore basic quadriceps control, prevent arthrofibrosis, initiate controlled motion.
-
Weight-Bearing:
- Fractures: NWB or TDWB with crutches for 6-8 weeks, gradually progressing to PWB based on radiographic healing and clinical stability.
- Osteochondral Repair: NWB for 6-8 weeks (microfracture, OATS, ACI/MACI) to protect cartilage integration.
-
Range of Motion:
- CPM (Continuous Passive Motion): Often initiated immediately post-op, aiming for 0-30° initially, progressing to 0-90° by week 4-6, depending on the repair stability.
- Active-Assistive ROM: Gentle knee flexion and extension within protected limits.
- Patellar Mobilization: Gentle superior, inferior, medial, lateral glides to prevent adhesions.
-
Strengthening (Isometric):
- Quadriceps setting, gluteal sets, ankle pumps.
- Straight leg raises (SLR) in supine position, avoiding hip flexion if hamstring activity could stress the knee.
- Modalities: Ice, compression, elevation to manage edema.
Phase II: Progressive Loading and Strengthening (Weeks 6/8 - 12/16)
- Goals: Restore full ROM, progress weight-bearing, initiate dynamic strengthening, improve proprioception.
- Weight-Bearing: Progress from PWB to FWB as tolerated, based on surgeon’s guidance and radiographic evidence of healing.
- Range of Motion: Actively work towards full flexion and extension. Continue patellar mobilization.
-
Strengthening (Isotonic/Isokinetic):
- Closed kinetic chain exercises: Mini-squats (within pain limits), wall slides, heel raises.
- Open kinetic chain exercises: Quadriceps and hamstring strengthening (e.g., leg extensions/curls with light resistance, avoiding terminal extension if patellofemoral issues).
- Core strengthening.
- Proprioception and Balance: Single-leg stance, wobble board, balance beam exercises.
- Gait Training: Focus on normal heel-toe gait pattern without limping. Discontinue crutches/cane when able to walk with normal mechanics.
Phase III: Advanced Strengthening and Return to Activity (Weeks 12/16 - 6+ Months)
- Goals: Maximize strength, power, endurance, agility; integrate sport-specific movements; prepare for return to full activity/sport.
-
Strengthening:
- Progressive resistance exercises, plyometrics (jumping, hopping), agility drills.
- Sport-specific training (cutting, pivoting, running).
- Interval training.
- Functional Progression: Gradual return to activities of daily living and work.
-
Return to Sport:
- Typically not before 6-9 months, and often longer (up to 12 months) for significant articular fractures or major cartilage repair.
- Requires achievement of full pain-free ROM, symmetrical strength (isokinetic testing >90% contralateral limb), good dynamic stability, and successful completion of sport-specific functional tests.
- Decision is made jointly by surgeon, physical therapist, and patient.
Specific Considerations
- Osteochondral Repairs: Often have more stringent and prolonged NWB restrictions. High-impact activities are avoided for longer periods.
- Associated Ligamentous Repairs: Protocols must integrate protection for both the LFC injury and the ligamentous repair (e.g., stricter bracing, specific ROM limits for ACL or PLC repair).
- Patient Compliance: Crucial for successful outcomes. Education on the importance of adhering to the protocol is vital.
Summary of Key Literature / Guidelines
The literature regarding lateral femoral condyle injuries provides a robust framework for diagnosis and management, though specific guidelines can vary based on the exact pathology.
Distal Femoral Fractures (Including LFC Fractures)
- AO/OTA Classification: This system is universally recognized for classifying distal femoral fractures (33-A: extra-articular supracondylar; 33-B: partial articular, unicondylar; 33-C: complete articular, bicondylar). LFC fractures typically fall into the 33-B type (e.g., 33-B2, lateral condyle fracture) or are components of 33-C type fractures.
-
Treatment Principles:
The primary goals for articular fractures are anatomical reduction of the articular surface and rigid internal fixation to allow early range of motion.
- Locking Plates: Lateral distal femoral locking plates have become the gold standard for many complex LFC and distal femoral fractures, providing angular stability and superior fixation in osteopenic bone compared to conventional plating. Studies by Kregor et al. (2001) and Schandelmaier et al. (2008) highlighted the advantages of indirect reduction techniques and biological fixation principles.
- Lag Screws: Articular fragments should be reduced and fixed with lag screws (e.g., 4.0 or 4.5 mm cannulated screws) prior to plate application, if possible.
- Outcomes: While good to excellent results are common, complications like nonunion (5-10%) and post-traumatic arthritis (up to 30-50%) remain significant, particularly in higher-energy injuries and those with initial severe articular comminution. Kienle et al. (2014) discussed factors influencing outcomes, emphasizing accurate reduction.
Osteochondral Lesions of the LFC
- Diagnosis: MRI is the imaging modality of choice, providing detailed information on lesion size, depth, stability, and subchondral bone integrity. Outerbridge (1961) and ICRS (International Cartilage Repair Society) classifications are commonly used to grade cartilage defects.
-
Treatment Algorithms:
- Microfracture: Indicated for smaller, contained full-thickness defects (<2-3 cm²). A meta-analysis by Mithoefer et al. (2009) showed good short- to mid-term outcomes, but acknowledged the formation of fibrocartilage rather than hyaline cartilage.
- Osteochondral Autograft Transfer (OATS/Mosaicplasty): Preferred for larger, focal defects (1-4 cm²) to restore hyaline cartilage. Hangody and Füles (2003) demonstrated favorable long-term results.
- Osteochondral Allograft: For large defects (>4 cm²) or failed autograft procedures. Bugbee (2011) reviewed the outcomes, showing good success rates but challenges related to donor availability and graft incorporation.
- Autologous Chondrocyte Implantation (ACI/MACI): A multi-stage procedure for large defects, often in younger patients. Peterson et al. (2003) provided landmark long-term follow-up demonstrating durable results with ACI. MACI has gained popularity for its simplified technique.
- Outcome Factors: Patient age, lesion size, previous surgeries, and concomitant pathologies (e.g., meniscal tears, ligamentous instability) significantly impact results. Addressing underlying instability is crucial for long-term success.
Associated Injuries
- ACL Tears: The LFC often exhibits a characteristic bone bruise (deep sulcus sign) or osteochondral impaction injury following an acute ACL tear due to the pivot-shift mechanism. Recognition of this MRI finding is important for confirming the diagnosis of ACL rupture and assessing potential chondral damage.
- Posterolateral Corner (PLC) Injuries: Avulsion fractures of the FCL or popliteus tendon from the LFC are critical to identify as they necessitate surgical repair or reconstruction to prevent chronic instability. Laprade et al. (2007) and Shon et al. (2010) have extensively detailed the diagnosis and management of PLC injuries, emphasizing anatomical repairs.
Guidelines & Future Directions
Current guidelines from professional societies (e.g., AAOS, ISAKOS) advocate for an individualized approach based on patient factors, injury characteristics, and surgeon experience. There is a continuous evolution towards less invasive techniques, improved biologics for cartilage repair, and personalized medicine approaches. Advanced imaging techniques and biomechanical modeling are aiding in better pre-operative planning and understanding of injury mechanisms. Long-term studies are continuously refining our understanding of the durability of various repair options, particularly for osteochondral defects.
You Might Also Like