Single-Stage Tissue Transfer: Principles and Techniques of Free Flaps in Orthopaedics
Key Takeaway
Single-stage tissue transfer, or free flap surgery, is a cornerstone of complex orthopaedic reconstruction. By utilizing microvascular anastomosis, surgeons can transfer composite tissues—including skin, muscle, and bone—to reconstruct severe traumatic, oncologic, or congenital defects. This technique provides robust, well-vascularized coverage, allowing for early mobilization and single-stage resolution of complex extremity wounds that preclude the use of traditional random or axial pedicle flaps.
INTRODUCTION TO SINGLE-STAGE TISSUE TRANSFER
The evolution of soft tissue reconstruction in orthopaedic surgery has been fundamentally transformed by the advent of microvascular techniques. Before the development of reliable microsurgery, the coverage of major soft tissue defects relied heavily on remote pedicle flaps, which often required multiple staged procedures, prolonged immobilization, and significant patient morbidity.
In 1946, Shaw and Payne reported their extensive experience with tubed pedicle flaps based on the superficial epigastric and superficial circumflex arterial circulations. Building upon that foundational report, their analysis of the deltopectoral flap of Bakamjian, and their own extensive experience with the groin pedicle flap, McGregor and Morgan elucidated the critical hemodynamic differences between random pattern and axial pattern flaps. This distinction remains the bedrock of modern reconstructive microsurgery, dictating flap design, harvest, and survival.
Today, single-stage transfers of composite tissue grafts (free flaps) are indispensable for the repair and reconstruction of traumatic, infectious, neoplastic, congenital, and developmental anomalies in both the upper and lower extremities.
VASCULAR ANATOMY AND FLAP CLASSIFICATION
Understanding the intrinsic vascular anatomy of donor tissues is paramount. Flaps are broadly categorized based on their vascular supply into random pattern and axial pattern flaps.
Random Pattern Flaps
The random pattern flap relies on no specific, established pattern of circulation. Instead, its viability depends entirely on the rich, interconnecting subdermal vascular plexus. Because the blood supply is not driven by a named, high-pressure arterial system, the perfusion pressure drops precipitously as the distance from the flap base increases.
Biomechanics of Random Flaps: A length-to-width ratio of greater than 2:1 exponentially increases the risk of distal ischemia and subsequent failure of a random flap. Consequently, their utility in covering large or complex orthopaedic defects is severely limited.
Axial Pattern Flaps
An axial pattern flap relies on a definite and usually consistent arterial supply centered on one or more named arteries running longitudinally within the flap. Because the tissue is perfused by a direct arterial system, the perfusion pressure is maintained over a much greater distance. Therefore, there are no rigid length-to-width ratio requirements for axial pattern flaps, provided the distal tissue remains within the angiosome of the central vessel.
These flaps are generally classified as cutaneous or myocutaneous, depending on the specific architectural pattern of their arterial circulation.
Cutaneous Flaps
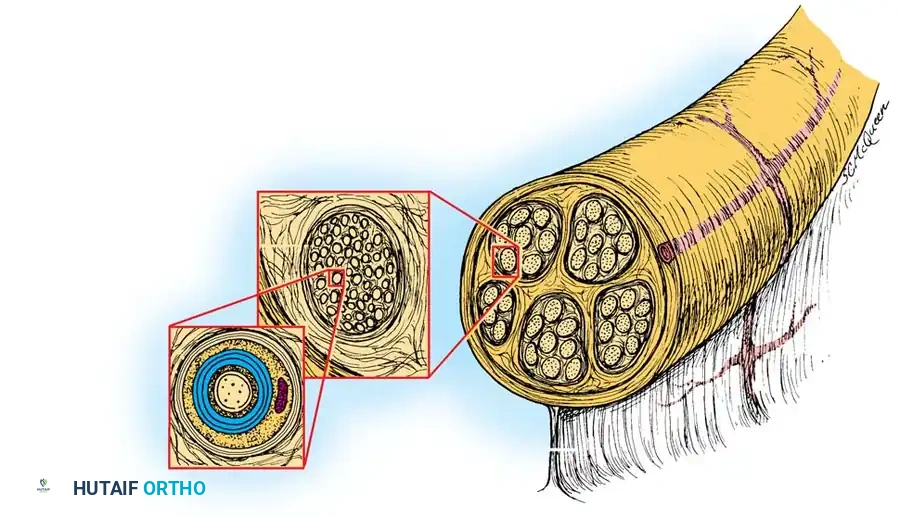
Cutaneous flaps rely on a constant circulation from a single artery passing through the underlying subcutaneous tissue. This central vessel supplies the overlying skin through a network of dermal and subdermal vessels.

The direct cutaneous artery courses superficial to the deep fascia, sending perforating branches directly into the subdermal plexus. This allows for the harvest of relatively thin, pliable flaps ideal for areas requiring thin coverage, such as the dorsum of the hand or foot.
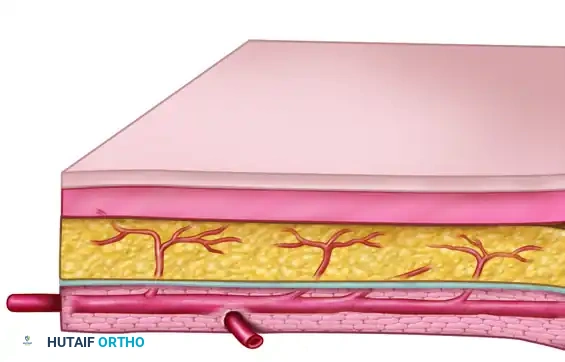
Myocutaneous Flaps
The myocutaneous flap receives its cutaneous arterial supply from deep vessels that first supply the underlying muscle. These vessels then send musculocutaneous perforators through the muscle belly and the overlying deep fascia to reach the skin and subcutaneous tissue.
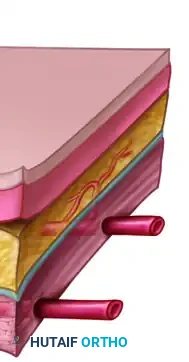
When harvesting a myocutaneous flap, the underlying muscle must be taken (either in whole or in part) to preserve these critical perforators, resulting in a bulkier flap that is excellent for obliterating dead space in deep traumatic or infectious defects.
INDICATIONS FOR FREE TISSUE TRANSFER
While the simplest procedures—including local and remote pedicle flaps—should always be considered first, microsurgical procedures are indicated when traditional techniques are precluded by the size, depth, or location of the defect. In certain complex scenarios, priority should be given immediately to the use of free flaps.
Primary indications in orthopaedic surgery include:
* Massive Bone and Soft Tissue Loss: High-energy trauma (e.g., Gustilo-Anderson Type IIIB and IIIC open fractures) requiring composite reconstruction.
* Infection and Osteomyelitis: Bringing well-vascularized tissue to an ischemic, infected bed to deliver systemic antibiotics and promote healing.
* Joint Reconstruction: Replacement of lost or destroyed joints in the fingers using vascularized joint transfers.
* Pediatric Reconstruction: Replacement of functioning epiphyses in the hand and forearm.
* Congenital Deformities: Correction of complex developmental anomalies, including radial clubhand and congenital pseudarthrosis of the tibia (often utilizing vascularized fibular grafts).
ADVANTAGES OF MICROVASCULAR FREE FLAPS
When compared to traditional staged pedicle techniques, free tissue transfer offers profound physiological and logistical benefits:
- Single-Stage Resolution: They are typically performed as single-stage procedures, drastically reducing the total number of operations.
- Donor Site Flexibility: The choice of a donor site is not as restrictive, allowing the surgeon to select tissue that perfectly matches the defect.
- Superior Tissue Matching: There is immense versatility regarding the matching of color, texture, thickness, and hair distribution of the donor area with the recipient site.
- Primary Closure: In many situations, the donor site can be closed primarily without resorting to secondary skin grafts.
- Aesthetic Outcomes: Most donor sites are left with a highly acceptable cosmetic appearance.
- Revascularization of Ischemic Beds: Well-vascularized tissue with a permanent, robust blood supply can replace ischemic or avascular tissue, which is critical in irradiated beds or chronic osteomyelitis.
- Composite Reconstruction: When indicated, a vascularized bone graft, functioning joints, epiphyses, and skeletal muscle can be electively included in a single composite graft.
- Freedom of Movement: Prolonged immobilization in awkward positions (e.g., arm attached to groin) is not required, allowing the patient immediate freedom in daily activities.
- Early Rehabilitation: Joints adjacent to the recipient area are mobilized much earlier than after conventional techniques, preventing debilitating joint stiffness and contractures.
- Reduced Hospitalization: Overall hospital stays and rehabilitation timelines are significantly shortened.
DISADVANTAGES AND LIMITATIONS
Despite their versatility, free flaps are technically demanding and carry significant risks. The reconstructive surgeon must carefully weigh the following disadvantages:
- Prolonged Operative Time: The initial operation is usually much longer than operations for conventional flaps. Free flap procedures typically take 4 to 10 hours, depending largely on the flap selected and the experience of the surgical team.
- Technical Difficulty: The operations are highly complex, tedious, and require specialized microsurgical training.
- Resource Intensive: Two complete teams of surgeons are usually required to work simultaneously (one at the donor site, one at the recipient site) to minimize ischemia time.
- Catastrophic Failure Risk: If vascular thrombosis occurs at the anastomotic site, the risk of complete, irreversible loss of the free flap is considerable.
- Higher Overall Failure Rates: Reportedly, the overall risk of free flap failure compared with conventional techniques is greater. A 10% to 30% failure rate for free flaps has been cited historically by Sharzer et al., though modern high-volume centers report success rates exceeding 95%. Additionally, the reoperation rate after free flap transfers (for hematoma, venous congestion, or arterial insufficiency) may be as high as 25%.
- Delayed Complications: Postoperative vascular complications, which most commonly occur in the first 24 hours, can still manifest up to 10 days after the procedure.
- Impact of Prior Surgery: If previous operative procedures have been done in the donor area, the donor vessels may have been inadvertently damaged, precluding the use of that specific donor site.
- Challenges of Obesity: Obesity makes the dissection of delicate vascular pedicles exceedingly difficult or impossible. Furthermore, bulky, obese flaps are awkward to manipulate and difficult to inset without causing tension, torsion, or disruption of the microvascular anastomoses. Excessive fat can also obstruct a clear view of the vascular pedicles, preventing the performance of satisfactory anastomoses.
PREOPERATIVE PLANNING AND FLAP SELECTION
The selection of one specific flap over another is a highly nuanced decision influenced by multiple patient and defect-specific factors.
Recipient Site Analysis
Specific tissue requirements at the recipient site dictate the reconstructive ladder:
* Is full-thickness coverage needed?
* Would a simple skin graft or conventional local flap suffice?
* Is a free flap strictly necessary?
* How thick and how large should the coverage be?
* Is skin sensibility, structural bone, a functional joint, nerve conduit, or functioning motor muscle needed?
Clinical Pearl: In general, free skin flaps (fasciocutaneous) are selected rather than free muscle flaps when dead space is minimal, and when skin and subcutaneous tissue must be matched to restore cutaneous sensibility and allow for future tendon gliding (e.g., over the dorsum of the hand). Conversely, muscle flaps with split-thickness skin grafts are preferred for deep, irregular defects with significant dead space, particularly in the setting of infection.
Vessel Assessment
The condition and availability of donor and recipient vessels are critical. Preoperative assessment often requires advanced imaging, such as CT angiography or conventional angiography, particularly in lower extremity trauma where the zone of injury may extend far beyond the visible wound. Handheld Doppler ultrasound is routinely used to map perforators at the donor site.
Generally, the simplest procedure that fulfills the tissue requirements should be chosen. The flap must be designed with a "lifeboat" in mind—if the primary free flap fails, a satisfactory salvage procedure must still be possible. Ultimately, a major factor in flap selection is the specific experience and comfort level of the individual surgeon.
SURGICAL TECHNIQUE: STEP-BY-STEP APPROACH
1. Positioning and Preparation
The patient is positioned to allow simultaneous access to both the donor and recipient sites. Two surgical teams operate concurrently. The room must be kept warm to prevent patient hypothermia, which induces peripheral vasospasm and compromises microvascular flow.
2. Recipient Site Preparation
The recipient site team begins with radical debridement of all necrotic, infected, or devitalized tissue. The success of a free flap is entirely dependent on the quality of the recipient bed.
Once debridement is complete, the recipient vessels are identified and dissected outside the zone of injury. Healthy vessels exhibit pulsatile arterial flow and brisk venous backbleeding. The vessels are stripped of their adventitia under the operating microscope in preparation for anastomosis.
3. Donor Tissue Harvest
Simultaneously, the donor team harvests the flap. Dissection proceeds carefully to isolate the vascular pedicle. In myocutaneous flaps, the muscle is elevated with care to protect the perforating vessels. In cutaneous or fasciocutaneous flaps, the deep fascia is included to protect the delicate subdermal plexus. The pedicle is not divided until the recipient site is fully prepared and ready to receive the tissue, thereby minimizing the critical ischemia time.
4. Microvascular Anastomosis
Once the flap is transferred, it is temporarily secured with a few tacking sutures to prevent traction on the pedicle. Under the operating microscope, the microvascular anastomosis is performed.
* Arterial Anastomosis: Typically performed first using 8-0 to 10-0 nylon sutures. End-to-end or end-to-side techniques are utilized depending on vessel size mismatch and the necessity of preserving distal flow in the recipient artery.
* Venous Anastomosis: One or two veins are anastomosed. Venous outflow is critical; venous congestion is the most common cause of free flap failure.
Surgical Warning: Meticulous technique is non-negotiable. A single misplaced suture catching the back wall of the vessel, or failure to adequately irrigate the lumen with heparinized saline to remove micro-thrombi, will lead to catastrophic anastomotic thrombosis.
5. Inset and Closure
After establishing flow, the flap is observed for 15 to 20 minutes to confirm robust perfusion (pink color, brisk capillary refill, bleeding from dermal edges). The flap is then meticulously inset. Tension over the pedicle must be strictly avoided. Drains are placed away from the anastomotic site, and the donor site is closed primarily or grafted.
POSTOPERATIVE PROTOCOL AND MONITORING
The postoperative period is as critical as the surgery itself. The majority of vascular complications occur within the first 24 hours, though the risk window extends up to 10 days.
- Flap Monitoring: Clinical observation is the gold standard. The flap is assessed hourly for color, temperature, capillary refill, and tissue turgor.
- Arterial Insufficiency: The flap becomes pale, cold, and empty, with absent capillary refill.
- Venous Congestion: The flap becomes swollen, tense, and violaceous (blue/purple), with excessively rapid capillary refill and dark bleeding on pinprick.
- Adjunct Monitoring: Implantable venous Doppler probes, surface temperature probes, or tissue oximetry (NIRS) are frequently utilized to detect early vascular compromise before clinical signs become apparent.
- Environment and Medications: The patient is kept warm and well-hydrated. The room temperature is elevated. Anticoagulation protocols vary by institution but often include aspirin, low molecular weight heparin (for DVT prophylaxis), or dextran, depending on the surgeon's preference and the complexity of the anastomosis.
- Emergency Re-exploration: If vascular compromise is suspected, there is no role for "watchful waiting." The patient must be returned to the operating room immediately for exploration, thrombectomy, and revision of the anastomosis. Time is tissue; delays beyond a few hours typically result in irreversible no-reflow phenomenon and complete flap loss.
Through meticulous patient selection, rigorous preoperative planning, and exacting microsurgical technique, single-stage tissue transfer provides unparalleled reconstructive capabilities, restoring form and function to the most severely compromised extremities.
You Might Also Like