Principles of Microvascular Free Tissue Transfer and Postoperative Management
Key Takeaway
Microvascular free tissue transfer requires meticulous surgical execution and rigorous postoperative care. Success hinges on a coordinated two-team approach for donor and recipient site preparation, precise microvascular anastomoses, and strict environmental controls to prevent vasospasm. Postoperative management demands intensive monitoring of flap perfusion, optimized hemodynamics, tailored anticoagulation protocols, and immediate re-exploration if vascular compromise is suspected.
PRINCIPLES OF MICROVASCULAR FREE TISSUE TRANSFER
The reconstruction of complex orthopaedic defects—often resulting from high-energy trauma, oncologic resection, or severe infection—frequently necessitates microvascular free tissue transfer. The success of these procedures relies not only on flawless microsurgical technique but also on meticulous preoperative planning, coordinated intraoperative execution, and rigorous postoperative care. The margin for error in microvascular surgery is exceptionally narrow; a single technical oversight or a lapse in postoperative monitoring can precipitate irreversible flap failure.
This comprehensive guide delineates the standard protocols for executing free tissue transfers, focusing on the two-team surgical approach, microvascular anastomotic techniques, and the critical tenets of postoperative flap management.
INTRAOPERATIVE STRATEGY: THE TWO-TEAM APPROACH
To minimize operative time and reduce the physiological burden on the patient, free tissue transfers are optimally performed using a synchronized two-team approach. This is particularly critical for large composite transfers where prolonged anesthesia and extended ischemia times can compromise both patient survival and flap viability.
Recipient Site Preparation
The primary objective of the recipient team is to prepare a biologically sound bed for the incoming tissue and to isolate healthy, pulsatile recipient vessels outside the zone of injury.
- Radical Débridement: The recipient bed must be meticulously débrided of all scar tissue, devitalized fascia, and necrotic bone. Free flaps will not survive—nor will they clear infection—if placed over a bed of compromised, avascular tissue.
- Vessel Exposure and Selection: All potential recipient vessels must be exposed to ensure that arterial and venous pedicles of appropriate caliber and length are available. Dissection must proceed well proximal to the zone of trauma to ensure the vessels possess healthy intima.
- Handling of the Vasculature: Extreme care must be taken during perivascular dissection.
Surgical Warning: Avoid stripping the recipient vessels completely clean of their surrounding adventitia over long segments. Aggressive stripping damages the vasa vasorum and provokes refractory, cold-induced or mechanically-induced vasospasm, which can preclude the planned tissue transfer.
- Anastomotic Geometry: In the extremities, particularly the lower limb, if distal circulation depends on a single continuous artery (e.g., a single-vessel runoff leg), the surgeon must critically evaluate whether to utilize an end-to-side anastomosis to preserve distal flow, or whether an alternative recipient vessel should be sought.
- Vein Grafting: If a tension-free anastomosis cannot be achieved due to a spatial gap between healthy recipient vessels and the flap pedicle, interpositional vein grafts must be utilized. These should be harvested before the donor tissue is detached to minimize the ischemia time of the flap.
Donor Site Dissection and Harvest
Simultaneously, the second surgical team dissects the donor area. The approach to the free flap typically begins at the vascular pedicle, utilizing the identified course of the donor artery as the central axis for the outlined tissue.
- Pedicle Isolation: The dissection proceeds only after suitable arteries and veins are definitively identified and protected.
- Contingency Planning: If anatomical anomalies or iatrogenic injuries render the vessels on the primary donor side unsatisfactory, the contralateral side may be explored, provided patient positioning and preoperative consent permit.
FLAP TRANSFER AND MICROVASCULAR ANASTOMOSIS
Pedicle Transection and Ischemia Time Management
Once the flap is fully elevated, it must remain attached to its native vascular pedicle until the recipient site is completely prepared. The pedicle is only transected when the recipient team confirms that the recipient vessels are ready and capable of supplying sufficient inflow and outflow.
Clinical Pearl: When transecting the donor pedicle, the artery should be clamped and transected first. This allows a brief period for the venous system to drain the flap, preventing engorgement and toxic metabolite accumulation within the microcirculation before the veins are subsequently clamped and transected.
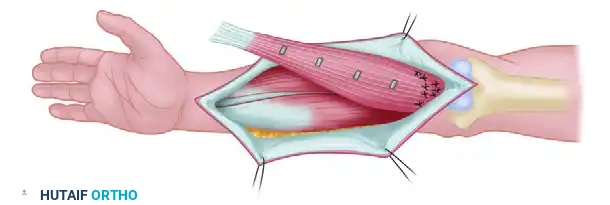
Upon delivery of the flap to the recipient team, the donor team immediately proceeds with hemostasis and closure of the donor site. While direct primary closure is preferred, split-thickness skin grafting may be required depending on the size and location of the donor defect.
Flap Inset and Stabilization
Before the microscope is brought into the field, the recipient team must loosely attach the flap to the recipient bed.
- Mechanical Stabilization: Sutures are placed at widely spaced intervals around the periphery. This critical step prevents mechanical shear, torsion, or tension on the delicate vascular pedicle during and after the anastomosis.
- Spatial Orientation: The flap must be positioned so that the vascular pedicle lies naturally, allowing the anastomoses to be performed ergonomically and without kinking.
Microvascular Anastomotic Technique
The operating microscope, sterilely draped, is introduced into the field.
- Adventitectomy: The perivascular adventitia and loose areolar tissue are gently dissected away from the immediate anastomotic site. This prevents adventitial tissue from being dragged into the lumen by the needle, which is a primary catalyst for microvascular thrombosis.
- Sequence of Anastomosis: The arterial anastomosis is typically performed first, followed by the venous anastomoses.
- Inflow Management:
Pitfall: It is highly advisable to keep the microvascular clamp on the repaired artery until at least one venous anastomosis is fully completed. Releasing the arterial clamp before venous outflow is established will cause immediate, severe flap congestion and endothelial damage. However, clamps must not be left on the vessels longer than absolutely necessary to avoid crush injury to the intima.
Anastomoses should be performed on as many suitable veins as are available to ensure redundant outflow, as venous congestion is the most common cause of free flap failure.
GENERAL POSTOPERATIVE CARE AND ENVIRONMENTAL CONTROLS
The postoperative phase is as critical as the intraoperative execution. The primary goals are to maintain optimal hemodynamics, prevent vasospasm, and ensure immediate detection of any vascular compromise.
The Postoperative Setting
Patients should ideally be transferred to an Intensive Care Unit (ICU) or a highly specialized microsurgical step-down unit.
* Monitoring Capabilities: The ICU ensures continuous monitoring of core vital signs, fluid balance, and flap vascularity.
* Staff Expertise: If the patient is to be managed on a standard surgical ward, it is an absolute prerequisite that the nursing and house staff are specifically trained in microvascular postoperative care. Ignorance of the subtle signs of flap failure will lead to catastrophic outcomes.
Environmental and Systemic Controls
The microvasculature of a newly transferred flap is denervated and exquisitely sensitive to circulating catecholamines and temperature fluctuations.
- Temperature Regulation: The patient's room must be kept consistently warm. Excessive cooling induces profound peripheral vasoconstriction and vasospasm. Forced-air warming blankets should be utilized to maintain patient normothermia.
- Sympathetic Tone Reduction: The environment must be kept quiet, and visitors should be restricted to minimize emotional stress, which triggers catecholamine release and subsequent vasospasm.
- Strict Prohibitions: Cigarette smoking (by both the patient and visitors) is strictly prohibited. Nicotine is a potent vasoconstrictor and promotes platelet aggregation. Caffeinated and cold beverages are similarly avoided to prevent sympathetically mediated vasospasm.
HEMODYNAMICS AND LIMB POSITIONING
The positioning of the reconstructed extremity plays a profound role in the hydrostatic pressures across the microvascular anastomoses.
- Baseline Positioning: The involved extremity is typically maintained at the level of the heart or slightly elevated. This promotes adequate venous drainage without compromising arterial inflow.
- Managing Arterial Insufficiency: If the flap appears pale, cool, or exhibits sluggish capillary refill (signs of arterial ischemia), the extremity can be temporarily lowered (dependent position) to utilize gravity to augment arterial inflow.
- Managing Venous Congestion: If the flap becomes violaceous, swollen, and exhibits brisk but dark capillary refill (signs of venous congestion), the extremity should be elevated well above the level of the heart to facilitate venous drainage.
Surgical Warning: Positional maneuvers are temporizing measures. If a flap appears to be in jeopardy, do not waste valuable time awaiting spontaneous improvement through positioning alone. Irreversible no-reflow phenomenon can occur within hours. Early re-exploration is the safest and most definitive course of action.
PHARMACOLOGIC PROTOCOLS AND ANTICOAGULATION
The use of postoperative antithrombotic and spasmolytic agents varies significantly based on institutional protocols, the specific clinical scenario, and the surgeon's preference. The goal is to prevent microvascular thrombosis without causing catastrophic hematomas at the surgical site.
- Standard Medications: Broad-spectrum intravenous antibiotics, adequate analgesics (to prevent pain-induced sympathetic spikes), and mild sedatives are routinely administered.
- Anticoagulation Strategies:
- No Anticoagulation: In patients with a high risk of bleeding or those with pristine, high-flow anastomoses, some surgeons rely solely on meticulous surgical technique without systemic anticoagulation.
- Aspirin: Often utilized for its antiplatelet properties. A common regimen is 300 mg twice daily, though lower doses (81 mg daily) are frequently used in modern practice to inhibit thromboxane A2.
- Dextran: Low-molecular-weight dextran (Dextran 40) acts as a volume expander, reduces blood viscosity, and alters platelet adhesiveness. A historical but still utilized regimen is 500 mL administered intravenously every 24 hours for 3 to 5 days. Note: Dextran carries risks of anaphylaxis, pulmonary edema, and acute kidney injury, requiring careful patient selection.
- Heparin: Intravenous unfractionated heparin or subcutaneous low-molecular-weight heparin (LMWH) may be used, particularly in cases of crush injury, hypercoagulable states, or when intraoperative thrombosis was encountered and revised.
FLAP MONITORING AND SALVAGE PATHWAYS
The survival of a compromised free flap is inversely proportional to the time elapsed between the onset of ischemia and surgical re-exploration.
Clinical Evaluation
Regardless of the technological adjuncts available, regular, serial clinical evaluations by experienced surgical and nursing staff remain the gold standard. Flaps should be assessed hourly for the first 24 to 48 hours.
* Color: Should match the donor site. Pallor indicates arterial occlusion; a blue/purple hue indicates venous congestion.
* Temperature: The flap should feel warm to the touch. A sudden drop in temperature is a highly sensitive indicator of perfusion failure.
* Capillary Refill: Normal refill is 1 to 3 seconds. Refill > 3 seconds suggests arterial compromise. Refill < 1 second (often with dark blood) suggests venous congestion.
* Turgor: A congested flap will feel tense and swollen; an ischemic flap will feel empty and deflated.
Technological Adjuncts
While clinical exam is paramount, several technologies can assist in monitoring:
* Handheld Doppler Ultrasound: Used to auscultate the arterial signal over the marked pedicle.
* Implantable Venous/Arterial Doppler: Silicone cuffs placed directly around the anastomosed vessels provide continuous auditory and visual feedback of flow.
* Tissue Oximetry (NIRS): Near-infrared spectroscopy provides continuous, non-invasive monitoring of regional tissue oxygen saturation (StO2), offering early warning of both arterial and venous compromise before clinical signs become apparent.
The Decision to Re-explore
If vascular compromise is suspected, the threshold for returning to the operating room must be extremely low. Removing a few sutures at the bedside to relieve a hematoma may be attempted, but if perfusion does not immediately normalize, urgent surgical re-exploration is mandatory. Delaying re-exploration to "wait and see" is the most common cause of preventable free flap loss.
You Might Also Like