Surgical Management of Rickets, Osteomalacia, and Renal Osteodystrophy
Key Takeaway
Rickets, osteomalacia, and renal osteodystrophy present complex orthopaedic challenges requiring a multidisciplinary approach. Surgical intervention, primarily corrective osteotomies for severe long-bone deformities like genu varum and genu valgum, must be meticulously timed with metabolic stabilization. This guide details the indications, preoperative medical optimization, and operative techniques necessary to achieve durable alignment and prevent recurrence in patients with impaired calcium and phosphorus homeostasis.
Pathophysiology and Clinical Evaluation
Rickets represents the bony manifestation of altered vitamin D, calcium, and phosphorus metabolism in the growing skeleton of a child, whereas osteomalacia is the adult equivalent occurring after physeal closure. Mankin’s extensive reviews of the metabolic defects underlying rickets, osteomalacia, and renal osteodystrophy highlight a multitude of etiologies, ranging from nutritional deficiencies to complex genetic and renal tubular disorders. Regardless of the specific metabolic derangement, children afflicted with rickets present with remarkably similar long-bone and trunk deformities.
Because nutritional vitamin D deficiency has become less prevalent in developed nations, rickets and osteomalacia are frequently overlooked in the differential diagnosis of pediatric patients presenting with extremity pain, bowing, or gait abnormalities. However, the orthopaedic surgeon must remain highly vigilant and familiar with the classic radiographic and laboratory findings that accompany these metabolic bone diseases.
The orthopaedist’s primary responsibilities encompass establishing an accurate diagnosis and managing the osseous complications. These complications frequently include pathologic fractures, slipped capital femoral epiphysis (SCFE), and severe multiplanar bowing of the extremities. When formulating a treatment plan for patients with rickets, osteomalacia, or renal osteodystrophy, the surgeon must perpetually consider the profound impact that surgical intervention and subsequent immobilization may have on an already impaired calcium homeostasis.
Non-Operative Management and Early Intervention
In very young children (typically under the age of 5) presenting with early deformity, aggressive treatment of the underlying metabolic defect supplemented by corrective splinting or bracing can frequently correct the deformity without the need for surgical intervention. The Hueter-Volkmann principle dictates that correcting the mechanical axis through bracing relieves asymmetric physeal compression, allowing the newly mineralizing bone to resume normal longitudinal growth.

Fig. 29-38 Vitamin D–deficient rickets. A, Standing radiograph of a young child with nutritional rickets resulting from severe vitamin D deficiency, demonstrating classic metaphyseal flaring and varus bowing. B, The same child 18 months later, demonstrating profound spontaneous correction of the mechanical axis following targeted treatment with vitamin D supplementation and corrective bracing.
Evans et al. reviewed a cohort of patients with primary hypophosphatemic rickets, noting that early medical intervention drastically reduced the surgical burden. In patients who received early treatment (age < 5 years) with inorganic phosphate and vitamin D, surgical requirements were minimal, typically limited to relatively simple proximal metaphyseal tibial osteotomies to correct residual varus deformities. Conversely, patients whose deformities went undiagnosed or untreated in early childhood required extensive, multi-level surgical reconstructions.
Surgical Indications and Timing
In prepubertal children or adolescents, medical management and bracing are generally insufficient to correct an established, structural deformity. Ferris et al. reported on patients with hypophosphatemic rickets, including teenagers and young adults presenting with debilitating knee pathology.
⚠️ Surgical Warning: The Natural History of Untreated Deformity
Joint problems typically develop in these patients during their early teens, often manifesting as osteochondritis dissecans-like lesions in the knee. By their 20s, late degenerative changes become apparent, characterized by the shedding of articular cartilage. Older patients frequently develop incapacitating stiffness and immobility secondary to the calcification of ligamentous structures. Early corrective osteotomy is strongly recommended to ensure that the joints remain in a functional position, mitigating the onset of premature osteoarthritis.
Osteotomy Selection: Diaphyseal vs. Metaphyseal
The location and fixation of the osteotomy depend heavily on the patient's skeletal maturity and the apex of the deformity:
* Diaphyseal Deformities: Best managed with multi-level osteotomies stabilized over an intramedullary nail. This provides load-sharing biomechanics and protects the entire length of the structurally compromised bone from future fractures.
* Metaphyseal Deformities: Most successfully addressed using a fixed-angle device, such as a blade plate or locking plate, performed at or near skeletal maturity to prevent physeal injury and ensure rigid fixation.
Preoperative Metabolic Optimization
The cornerstone of surgical success in metabolic bone disease is preoperative optimization. Surgery must never be undertaken in a metabolically uncontrolled patient. If the disease is not metabolically stabilized, the deformity is highly likely to recur rapidly following corrective osteotomy due to the continued presence of soft, unmineralized osteoid.
Management of the metabolic defect with vitamin D, phosphorus, and calcium must be optimized for several months prior to any surgical intervention.
Managing Hypercalcemia Risk During Immobilization
A critical perioperative consideration is the management of high-dose vitamin D therapy. Large-dose vitamin D treatment must be discontinued for at least 3 weeks prior to surgery. Failure to do so, combined with postoperative immobilization, creates a severe risk of life-threatening hypercalcemia.
If a water-soluble preparation of vitamin D (such as dihydrotachysterol) is utilized instead of cholecalciferol (which is heavily stored in the liver), the mandatory washout period before surgery can be safely shortened. In cases of hypophosphatemic vitamin D–resistant rickets controlled with inorganic phosphate and low-dose vitamin D (≤ 50,000 U/day), the risk of postoperative hypercalcemia is reduced. Evans et al. reported successful procedures without discontinuing low-dose vitamin D; however, the standard of care remains a 3-week cessation to absolutely prevent hypercalcemic symptoms, which include severe anorexia, nausea, vomiting, weight loss, confusion, and seizures.
Postoperatively, rapid mobilization of the patient is imperative. Early weight-bearing and resumption of medical treatment prevent delayed mineralization of the healing osteotomy and avoid the recurrence of deformity during continued growth.
Special Considerations: Azotemic (Renal) Osteodystrophy
Patients with azotemic osteodystrophy require an expert, multidisciplinary approach. Preoperative and postoperative medical management must be directed by a specialized nephrology team trained in chronic renal failure.
According to Cattell et al., the safe administration of anesthesia requires meticulous correction of anemia, adequate hydration, strict uremia control, and precise electrolyte balance. Peritoneal dialysis or hemodialysis is frequently required immediately prior to surgery.
💡 Clinical Pearl: Requisites for Surgery in Renal Osteodystrophy
Surgery in azotemic patients is highly feasible but demands strict criteria:
1. A reasonable overall life expectancy.
2. Highly intelligent and motivated patients and parents.
3. Documented radiographic improvement of bone lesions under medical management.
4. Deformities that are correctable within one or two surgical procedures.
5. A high probability that the intervention will significantly reduce functional disability.
Oppenheim et al. emphasized that patients requiring repeated osteotomies often exhibited poor metabolic control during their initial surgeries, objectively measured by persistently elevated alkaline phosphatase levels.
Contraindications: Patients with resistant hypertension generally have shortened life expectancies and are poor surgical candidates. Furthermore, if parathyroid autonomy (tertiary hyperparathyroidism) is present and uncontrolled by medical management or parathyroidectomy, orthopaedic surgery is strictly contraindicated.
Operative Techniques for Deformity Correction
The deformities most frequently requiring surgical correction are genu varum and genu valgum. Sheridan et al. demonstrated in a 40-year follow-up study that lower extremity deformities in these patients are permanent without intervention, and that corrective osteotomies are highly protective against degenerative arthritis of the knee.
Genu Varum Correction
In severe genu varum, the femur, tibia, and fibula are all typically deformed. The tibia and fibula often exhibit the most severe pathology, presenting with both lateral bowing and internal tibial torsion.
* Approach: Osteotomy of the tibia and fibula is performed near the apex of the most severe bowing (Center of Rotation of Angulation - CORA).
* Concomitant Femoral Deformity: Frequently, a distal femoral osteotomy is also required to fully correct the mechanical axis. These osteotomies can be performed bilaterally during a single anesthetic event if the patient is medically optimized.
Genu Valgum Correction
In genu valgum, the primary apex of bowing is usually located in the distal femur. Severe deformities in older children and adults are best corrected via a medial closing-wedge or dome supracondylar osteotomy of the femur.
Surgical Goal: The ultimate biomechanical goal of both tibial and femoral osteotomies is the precise correction of the mechanical axis deviation (MAD) so that the joint orientation angles (mLDFA and MPTA) are restored, rendering the plane of each knee joint perfectly horizontal when the patient is standing.
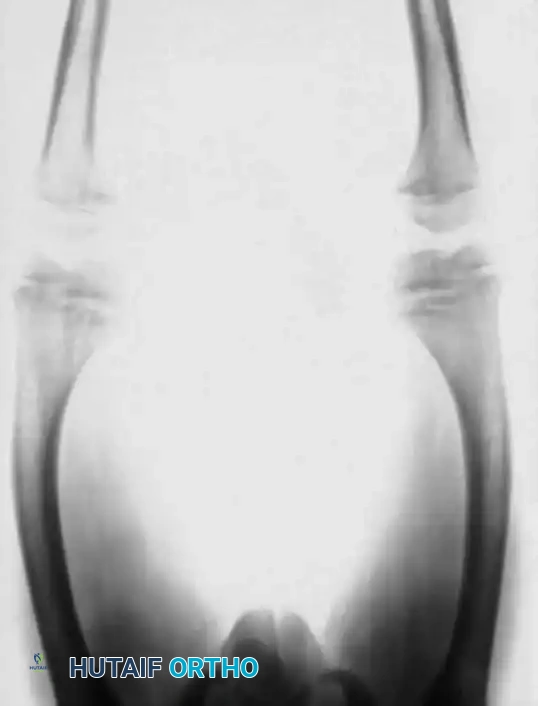
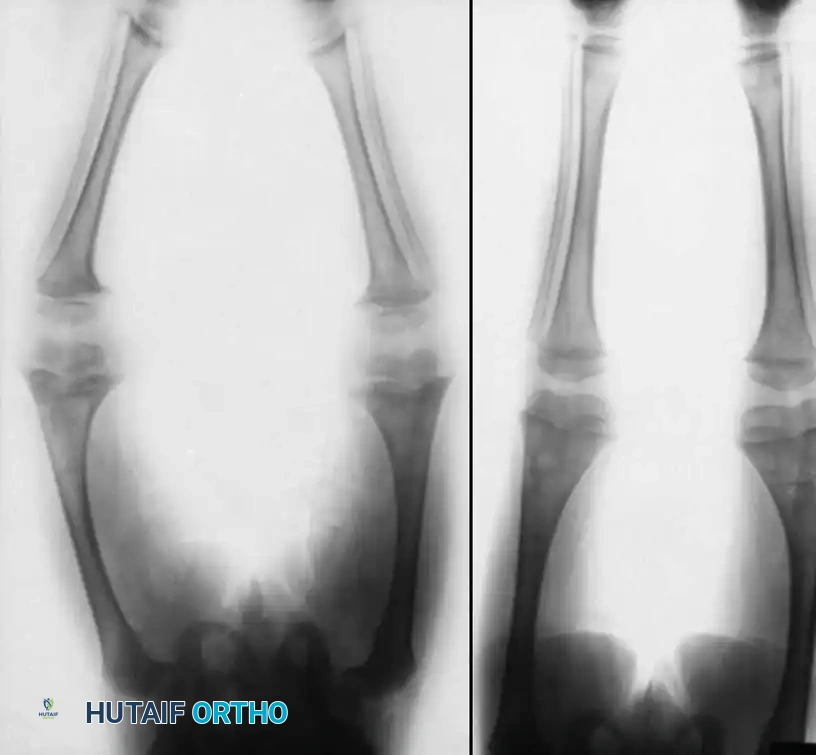
Fig. 29-39 A, Preoperative standing radiograph of a child with vitamin D–resistant rickets demonstrating severe bilateral valgus deformities localized primarily to the distal femurs.
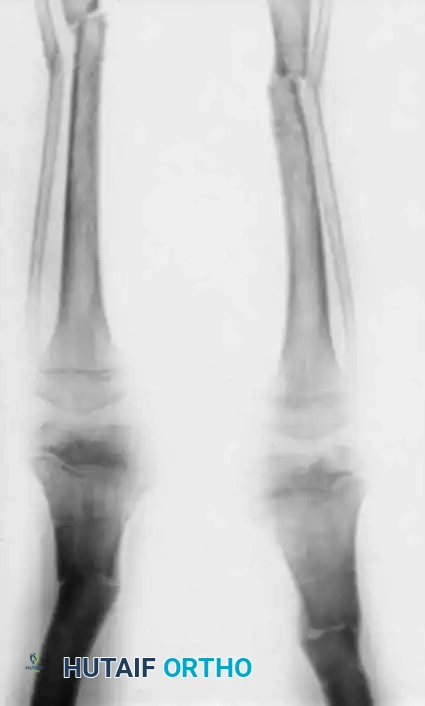
Fig. 29-39 B, Radiograph taken three months postoperatively following bilateral varus-producing osteotomies of the distal femurs and proximal tibias. Fixation was achieved using percutaneous pins incorporated into a plaster cast above and below the osteotomy sites.
Fig. 29-39 C, Two years post-osteotomy. The vitamin D–resistant rickets is now well controlled with high doses of vitamin D, calcium, and phosphorus. The mechanical axis remains corrected with no recurrence of the deformity.
The Ilizarov Method and Leg Lengthening
For complex, multiplanar deformities or cases requiring concomitant limb lengthening, circular external fixation (the Ilizarov method) is highly effective. Kanel and Price advocated for external fixation as it allows for precise, gradual correction of deformities without interrupting ongoing medical management.
Choi et al. evaluated patients with hypophosphatemic rickets undergoing knee deformity correction and leg lengthening via the Ilizarov method. While nearly all patients achieved mechanical correction, the regenerate healing indices correlated directly with biochemical parameters.
⚠️ Surgical Warning: Phosphate Levels and Regenerate Bone
If deformity correction is to be combined with distraction osteogenesis (leg lengthening), the serum phosphate level must be maintained at > 2.5 mg/dL. Patients with serum phosphate levels below this threshold exhibit severely delayed regenerate consolidation, risking nonunion, regenerate collapse, or hardware failure.
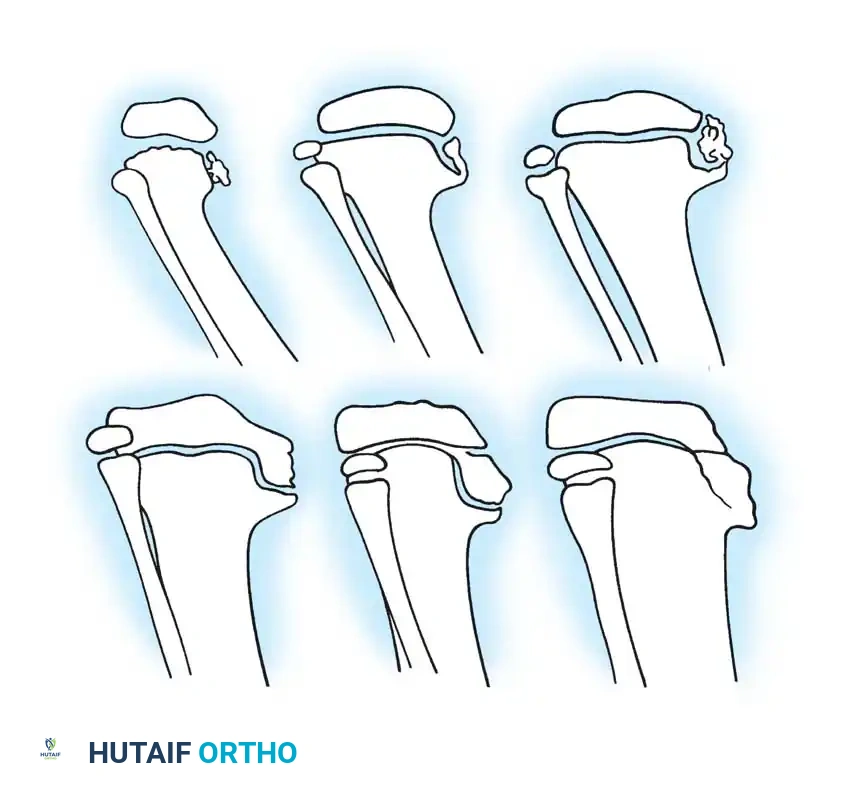
Diagram illustrating the stages of regenerate bone healing (I through VI) following osteotomy. Rapid progression through these stages in metabolic bone disease is entirely dependent on maintaining serum phosphate levels > 2.5 mg/dL.
Management of Concomitant Hemophilic Arthropathy: Ankle Synovectomy
Note: Patients with complex systemic or metabolic conditions may occasionally present with overlapping hematologic disorders, such as Factor VIII (Hemophilia A) or Factor IX (Hemophilia B) deficiencies. Recurrent hemarthrosis in these patients leads to severe synovial hypertrophy and rapid joint destruction. The following technique details a comprehensive ankle synovectomy for joint preservation.
🔪 Surgical Technique 29-14: Ankle Synovectomy
Anterior Approach and Debridement:
1. Incision: Make a 3-cm long anterolateral incision centered just lateral to the peroneus tertius tendon. Carefully retract this tendon medially to protect the neurovascular bundle.
2. Capsulotomy: Open the joint capsule longitudinally.
3. Synovial Resection: Excise the hypertrophic synovial tissue systematically. Remove all visible synovial tissue from the anterior compartment.
4. Gutter Clearance: Utilize small pituitary rongeurs to meticulously remove folds of synovial tissue that extend deep into the crypts between the talus and the medial malleolus, as well as the folds interposed between the talus and the lateral malleolus.
Posterior Approach and Debridement:
1. Positioning Adjustment: Remove the sandbag beneath the ipsilateral buttock and place it beneath the contralateral buttock to internally rotate the leg, facilitating posterior access.
2. Incision: Make a posterior incision approximately twice as long as the anterior incision, centering it precisely between the medial malleolus and the Achilles tendon.
3. Deep Dissection: Open the sheath of the posterior tibial tendon to allow adequate anterior retraction. Carefully dissect the remaining posterior tendons and the critical neurovascular structures (posterior tibial artery and tibial nerve) away from the posterior capsule of the ankle joint.
4. Retraction: Place a retractor lateral to the flexor hallucis longus muscle and medial to the posterior tibial tendon. This permits safe retraction of the soft-tissue structures located posterior to the ankle joint, providing full, unobstructed exposure of the posterior capsule.
5. Posterior Capsulotomy: Incise the capsule horizontally from the medial malleolus to the distal end of the fibula.
6. Posterior Synovectomy: Dissect the insertion of synovial tissue off the posterior talus and the distal tibia. Again, use pituitary rongeurs to clear any residual folds of synovial tissue lying in the posterior malleolar crypts.
* Surgical Pearl: If the synovium is intimately involved with the capsule and cannot be separated, en bloc removal of large sections of the capsule may be necessary. However, as noted by Greene, extensive resection can lead to severe posterior capsular scarring, which may impede postoperative rehabilitation and range of motion.
Closure and Hemostasis:
1. Tourniquet Deflation: Once the synovectomy is complete, deflate the pneumatic tourniquet. Meticulously secure hemostasis, as postoperative bleeding is the primary complication in this patient population.
2. Capsular Management: Repair the anterior portion of the capsule securely. Leave the posterior portion of the capsule open to decompress the joint, and place a deep closed-suction drain.
3. Immobilization: Close the subcutaneous tissues and skin in a standard layered fashion. Immobilize the ankle joint in a strictly neutral position (0 degrees of dorsiflexion) using a bulky compressive dressing augmented by a rigid plaster of Paris splint.
Postoperative Hematologic Protocol
The success of this procedure relies entirely on strict postoperative factor replacement:
* Factor VIII Deficiency: Patients must receive continuous intravenous transfusion therapy.
* Factor IX Deficiency: Patients require a bolus of Factor IX every 12 hours.
* Inpatient Phase: Transfusion protocols must be maintained continuously throughout the hospital stay (typically 7 to 10 days).
* Outpatient Phase: Following discharge, transfusions are administered three times a week for 4 weeks.
This rigorous regimen ensures that the deficient clotting factor level remains sufficiently elevated to minimize the risk of a catastrophic spontaneous hemarthrosis during the vulnerable immediate postoperative period, allowing for safe, early rehabilitation.
📚 Medical References
- Barrett IR, Papadimitriou DG: Skeletal disorders in children with renal failure, J Pediatr Orthop 16:264, 1996.
- Callenbach JC, Sheehan MB, Abramson SJ, et al: Etiologic factors in rickets of very low-birth-weight infants, J Pediatr 98:800, 1981.
- Cattell HS, Levin S, Kopits S, et al: Reconstructive surgery in children with azotemic osteodystrophy, J Bone Joint Surg 53A:216, 1971.
- Choi IH, Kim JK, Chung CY, et al: Deformity correction of knee and leg lengthening by Ilizarov method in hypophosphatemic rickets: outcomes and signifi cance of serum phosphate level, J Pediatr Orthop 22:626, 2002.
- Doppelt SH: Vitamin D, rickets, and osteomalacia, Orthop Clin North Am 15:671, 1984.
- Evans GA, Arulanantham K, Gage JR: Primary hypophosphatemic rickets: effect of oral phosphate and vitamin D on growth and surgical treatment, J Bone Joint Surg 62A:1130, 1980.
- Ferris B, Walker C, Jackson A, et al: The orthopedic manage ment of hypophosphatemic rickets, J Pediatr Orthop 11:367, 1991.
- Herman MJ, Gulthuis DB: Incidental diagnosis of nutri tional rickets after clavicle fracture, Orthopedics 22:254, 1999.
- Kanel JS, Price CT: Unilateral external fi xation for corrective osteotomies in patients with hypophosphatemic rickets, J Pediatr Orthop 15:232, 1995.
- Kanis JA: Vitamin D metabolism and its clinical application, J Bone Joint Surg 64B:542, 1982.
- Klein KL, Maxwell MH: Renal osteodystrophy, Orthop Clin North Am 15:687, 1984.
- Loder RT, Hensinger RN:
You Might Also Like


