Emergency Department Management of Acute Spinal Cord Injury
Key Takeaway
The initial emergency management of acute spinal cord injury requires a multidisciplinary approach prioritizing hemodynamic stability and neurological preservation. Orthopaedic surgeons must rapidly identify neurogenic shock—characterized by hypotension, bradycardia, and hypothermia in injuries above T6. This guide details advanced trauma life support protocols, incomplete spinal cord syndromes, surgical indications, and the current evidence-based consensus regarding high-dose corticosteroid administration in spinal trauma.
EMERGENCY DEPARTMENT MANAGEMENT AND RESUSCITATION
The initial evaluation and resuscitation of a trauma patient with a suspected acute spinal cord injury (SCI) demand a highly coordinated, multidisciplinary approach. Upon arrival at a Level I trauma center, the patient is simultaneously assessed by specialists in general surgery, anesthesiology, emergency medicine, neurosurgery, and orthopaedic surgery. The overarching goals in the emergency department (ED) are to secure the airway, maintain adequate oxygenation, restore hemodynamic stability, and prevent secondary spinal cord ischemia while strictly maintaining spinal precautions.
According to Advanced Trauma Life Support (ATLS) protocols, the spine must be protected using a rigid cervical collar and a rigid backboard during extrication and initial transport. However, prolonged use of rigid backboards is associated with pressure ulcerations; therefore, patients should be transferred to a firm, pressure-relieving trauma mattress as soon as clinically feasible, utilizing a coordinated log-roll technique to maintain spinal alignment.
Hemodynamic Instability: Neurogenic vs. Hypovolemic Shock
While cardiovascular, respiratory, and neurological functions are generally co-managed by the trauma team, the consulting orthopaedic surgeon must be acutely aware of the physiological derangements specific to spinal trauma.
Clinical Pearl: The "Triad of Neurogenic Shock" consists of hypotension, bradycardia, and hypothermia. This triad is a hallmark of cervical or upper thoracic spinal cord injuries occurring above the level of T6.
Neurogenic shock results from the disruption of descending sympathetic pathways within the spinal cord, leading to unopposed vagal parasympathetic tone. This manifests as profound peripheral vasodilation (distributive shock) and an inability to mount a tachycardic response to hypotension.
It is imperative to differentiate neurogenic shock from hypovolemic (hemorrhagic) shock, which is common in polytrauma patients. Hypovolemic shock typically presents with hypotension accompanied by tachycardia and cold, clammy extremities. In contrast, patients in neurogenic shock often have warm, flushed extremities due to peripheral vasodilation.
Hemodynamic Management Protocol:
* Fluid Resuscitation: Initial management involves judicious administration of isotonic crystalloids to ensure adequate intravascular volume.
* Vasopressors: If hypotension persists after volume resuscitation, vasopressors with both alpha- and beta-adrenergic activity (e.g., Norepinephrine or Dopamine) are indicated to restore vascular tone and chronotropy.
* MAP Goals: Current guidelines mandate maintaining a Mean Arterial Pressure (MAP) between 85 and 90 mm Hg for 7 days post-injury to optimize spinal cord perfusion and minimize secondary ischemic penumbra.
NEUROLOGICAL EVALUATION AND INCOMPLETE CORD SYNDROMES
Once life-threatening injuries are addressed, a meticulous neurological examination must be performed and documented using the American Spinal Injury Association (ASIA) Impairment Scale (AIS). This involves testing key motor myotomes and sensory dermatomes (both pinprick and light touch), as well as assessing sacral sparing (voluntary anal contraction and deep anal sensation), which differentiates a complete from an incomplete spinal cord injury.
Recognizing specific incomplete spinal cord syndromes is critical for determining the mechanism of injury, guiding imaging, and establishing a prognosis.

Central Cord Syndrome
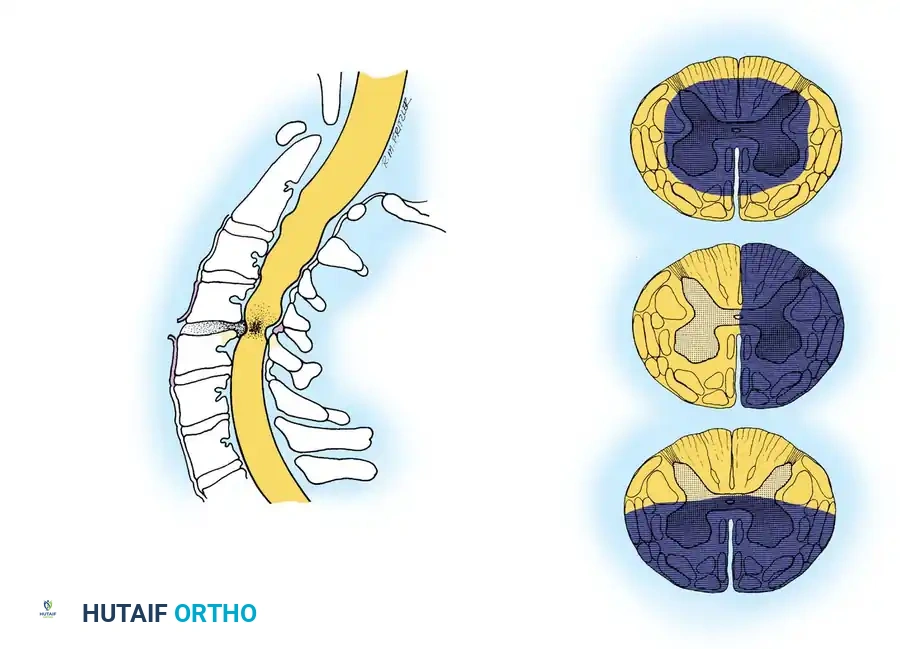
Central cord syndrome is the most common incomplete SCI, typically occurring in older patients with pre-existing cervical spondylosis who sustain a hyperextension injury. The spinal cord is pinched between the anterior osteophytes (or bulging disc) and the buckling posterior ligamentum flavum.
* Clinical Presentation: Motor weakness is disproportionately greater in the upper extremities than in the lower extremities. Sensory loss is variable, and bowel/bladder dysfunction may occur.
* Biomechanics: The cervical motor tracts for the upper extremities are located more centrally within the corticospinal tract, making them more susceptible to central hemorrhagic necrosis and edema.
Brown-Séquard Syndrome
This syndrome results from a functional hemisection of the spinal cord, often due to penetrating trauma (e.g., stab wounds) or severe unilateral facet dislocations.
* Clinical Presentation: Patients exhibit ipsilateral loss of motor function (corticospinal tract) and proprioception/vibratory sense (dorsal columns), combined with contralateral loss of pain and temperature sensation (spinothalamic tract) beginning one to two levels below the lesion.
Anterior Cord Syndrome
Anterior cord syndrome is caused by a hyperflexion injury resulting in anterior spinal artery compromise or direct anterior compression from a retropulsed bone fragment or massive disc herniation.
* Clinical Presentation: Complete loss of motor function, pain, and temperature sensation below the level of injury. Proprioception, vibratory sense, and light touch are preserved because the posterior columns (supplied by the posterior spinal arteries) remain intact.
* Prognosis: This syndrome carries the poorest prognosis for functional motor recovery among the incomplete syndromes.
THE METHYLPREDNISOLONE CONTROVERSY: EVIDENCE-BASED ANALYSIS
For decades, the pharmacological management of acute SCI was dominated by the administration of high-dose corticosteroids, specifically methylprednisolone (MP). This practice stemmed from the National Acute Spinal Cord Injury (NASCI) studies conducted in the 1990s. However, the routine use of MP has become one of the most heavily debated topics in orthopaedic and neurosurgical trauma.
The NASCI Trials and the Historical Protocol
The NASCI II study was a double-blind, randomized, controlled clinical trial evaluating very-high-dose methylprednisolone. The study reported that patients who received the MP infusion within 8 hours of injury demonstrated significantly more improvement in motor function, pinprick, and touch sensation at 6 weeks and 6 months post-injury compared to a control group. Conversely, MP infused 8 hours or more after the injury appeared to offer no benefit and, at 1-year follow-up, actually resulted in slightly lower functional scores than the placebo group.
The Historical NASCI Protocol:
* Bolus: 30 mg/kg body weight administered intravenously over 15 minutes.
* Pause: A 45-minute pause.
* Maintenance Infusion: A 23-hour continuous infusion of 5.4 mg/kg/h.
* Note: Bracken et al. reported a 55% overall improvement in motor function if this therapy was initiated within the 8-hour window.
The Shift in Paradigm: Critical Appraisals and Complications
Since the late 1990s, increasing controversy has surrounded the efficacy and safety of steroid use in spinal trauma. The massive dose of steroids administered over a 24- to 48-hour period significantly suppresses the immune system and impairs tissue healing.
Surgical Warning: High-dose methylprednisolone is associated with a drastically increased incidence of severe complications, including wound infections, gastrointestinal hemorrhage, sepsis, and pulmonary complications (especially in patients over 60 years of age).
Several critical appraisals have dismantled the routine use of MP:
* Statistical Flaws: Coleman et al. highlighted significant methodological flaws in the original NASCI studies, including issues with post-hoc statistical analysis, lack of access to raw data, and the fact that the FDA never approved MP for this specific indication.
* Lack of Clinical Benefit: Pollard and Apple found no long-term functional improvement in patients treated with the high-dose MP protocol. Similarly, McCutchon et al., in a review of 1,227 randomly selected SCI patients, found that those receiving MP had longer hospital stays and higher hospital charges without demonstrable long-term neurological benefits.
* High Complication Rates: Matsumoto et al. conducted a double-blind study revealing a complication rate of 56.5% in the MP group compared to 34.8% in the placebo group, with all gastrointestinal complications occurring in the steroid cohort.
* Histological Realities: Merola et al. demonstrated in animal models that while high-dose MP reduced severe edema and preserved adjacent architecture, it did not affect spinal cord necrosis or the astrocyte response at the primary zone of injury.
Current Institutional and Societal Guidelines
In an evidence-based analysis, Hurlbert definitively concluded that "methylprednisolone cannot be recommended for routine use in acute nonpenetrating spinal cord injury." Furthermore, prolonged administration (e.g., 48 hours) has been shown to be actively harmful.
Currently, major societies (including the AAOS and the AANS/CNS) classify high-dose steroids as a treatment option rather than a standard of care. In modern Level I trauma centers, this protocol is elective and utilized strictly on a case-by-case basis, determined collaboratively by the attending spinal surgeon and trauma surgeon, weighing the marginal potential neurological benefits against the substantial risks of life-threatening complications.
SURGICAL MANAGEMENT: INDICATIONS, TIMING, AND APPROACHES
The definitive management of acute spinal cord injury frequently requires surgical intervention. The primary goals of surgery are decompression of the neural elements, reduction of spinal deformity, and rigid mechanical stabilization to facilitate early mobilization and rehabilitation.
Surgical Indications and Timing
- Absolute Indications: Progressive neurological deficit in the setting of persistent compression, irreducible spinal dislocations (e.g., bilateral facet dislocations), and open/penetrating spinal injuries requiring debridement.
- Relative Indications: Static incomplete neurological deficits with radiographic evidence of cord compression, and highly unstable fracture patterns (e.g., flexion-distraction injuries, burst fractures with severe kyphosis).
- Timing of Surgery: The STASCIS (Surgical Timing in Acute Spinal Cord Injury Study) trial provided compelling evidence that early decompression (within 24 hours of injury) results in significantly better neurological outcomes compared to delayed decompression, particularly in cervical spine injuries.
Preoperative Positioning and Biomechanics
Positioning a patient with an unstable spine requires extreme caution.
* Intubation: Awake fiberoptic intubation is preferred for unstable cervical injuries to avoid neck extension during direct laryngoscopy.
* Traction: Gardner-Wells tongs or cranial skeletal traction may be applied in the ED or operating room to achieve closed reduction of facet dislocations. Weight is added sequentially (typically 5 lbs per level, starting with 10 lbs) under continuous fluoroscopic and neurological monitoring.
* Positioning: For posterior approaches, the patient is carefully log-rolled onto a Jackson spinal table. The head is secured in Mayfield pins to maintain neutral alignment. All pressure points must be meticulously padded to prevent decubitus ulcers, as SCI patients lack protective sensation.
Step-by-Step Surgical Approaches
1. Anterior Cervical Discectomy and Fusion (ACDF)
The anterior approach is ideal for removing compressive disc material or bone fragments located ventral to the spinal cord, particularly in the setting of a traumatic disc herniation associated with a facet dislocation.
* Incision: A transverse incision is made within a natural skin crease on the anterior neck (typically left-sided to reduce the risk of recurrent laryngeal nerve injury).
* Dissection: The platysma is divided. Blunt dissection is performed between the visceral axis (trachea/esophagus) medially and the carotid sheath laterally.
* Decompression: The longus colli muscles are elevated. A complete discectomy is performed using curettes and pituitary rongeurs. The posterior longitudinal ligament (PLL) is often resected to ensure complete decompression of the ventral dura.
* Reconstruction: An interbody graft (allograft or autograft) is inserted to restore disc height and lordosis. An anterior cervical plate is secured to the adjacent vertebral bodies with screws.
2. Posterior Cervical Laminectomy and Fusion
The posterior approach is utilized for highly unstable injuries requiring robust biomechanical stabilization, irreducible facet dislocations, or multilevel posterior compression (e.g., central cord syndrome with congenital stenosis).
* Incision: A midline longitudinal incision is made over the spinous processes.
* Exposure: Subperiosteal dissection of the paraspinal musculature is performed laterally to the lateral mass margins.
* Reduction: If a facet dislocation is present, it is reduced under direct vision using gentle leverage with a Penfield elevator or by drilling the superior tip of the inferior facet.
* Decompression: A laminectomy is performed using a high-speed burr and Kerrison rongeurs, carefully lifting the lamina away from the underlying swollen spinal cord.
* Instrumentation: Lateral mass screws (using the Magerl or Roy-Camille trajectory) or pedicle screws are placed. Rods are contoured to match the physiological lordosis and secured. Decortication of the lateral masses is performed, and bone graft is applied to achieve arthrodesis.
POSTOPERATIVE PROTOCOLS AND REHABILITATION
The postoperative phase is critical for maximizing neurological recovery and preventing systemic complications.
- Hemodynamic Monitoring: Patients must be managed in an Intensive Care Unit (ICU). The MAP must be strictly maintained at >85 mm Hg for a full 7 days post-injury using intravenous fluids and vasopressors as needed.
- Respiratory Care: High cervical injuries (C3-C5) compromise diaphragmatic function. Aggressive pulmonary hygiene, incentive spirometry, and early tracheostomy (if prolonged ventilation is anticipated) are required to prevent pneumonia.
- Venous Thromboembolism (VTE) Prophylaxis: SCI patients are at an exceptionally high risk for deep vein thrombosis (DVT) and pulmonary embolism (PE). Chemical prophylaxis (e.g., Low Molecular Weight Heparin) should be initiated within 72 hours of injury, provided there is no evidence of active epidural hematoma or systemic bleeding. Mechanical prophylaxis (SCDs) should be used immediately.
- Rehabilitation: Early mobilization, physical therapy, and occupational therapy are initiated as soon as spinal stability is surgically achieved. Multidisciplinary rehabilitation focuses on maximizing residual motor function, managing spasticity, and training for activities of daily living (ADLs).
The management of acute spinal cord injury remains a dynamic and evolving field. Surgeons and residents are strongly encouraged to continuously review the latest literature, societal guidelines, and institutional protocols to ensure the delivery of optimal, evidence-based care.
📚 Medical References
- acute spinal cord injury: an update of the randomized evidence, Spine 26(24 suppl):S47, 2001.
- Bracken MB, Holford TR: Effects of timing of methylprednisolone or naloxone administration on recovery of segmented and long-tract neurological function in NACSI II, J Neurosurg 79:500, 1993.
- Bracken MB, Shepard MJ, Collins WF, et al: A randomized controlled trial of methylprednisolone or naloxone in the treatment of acute spinal cord injury: results of the second National Study, N Engl J Med 322:1405, 1990.
- Brightman RP, Miller CA, Rea GL, et al: Magnetic resonance imaging of trauma to the thoracic and lumbar spine: the importance of the posterior longitudinal ligament, Spine 17:541, 1992.
- Brown MD, Eismont FJ, Quencer RM: Symposium: intraoperative ultrasonography in spinal surgery, Contemp Orthop 11:47, 1985.
- Chow YW, Inman C, Pollintine P, et al: Ultrasound bone densitometry and dual energy x-ray absorptiometry in patients with spinal cord injury: a cross-sectional study, Spinal Cord 34:736, 1996.
- Clark WK: Spinal cord decompression in spinal cord injury, Clin Orthop Relat Res 154:9, 1981.
- Cody DD, Goldstein SA, Flynn MJ, et al: Correlations between vertebral regional bone mineral density (rBMD) and whole bone fracture load, Spine 16:146, 1991.
- [Coleman WP, Benzel D, Cahill DW, et al: A critical appraisal of the reporting of the National Acute Spinal Cord Injury Studies
A
B
C

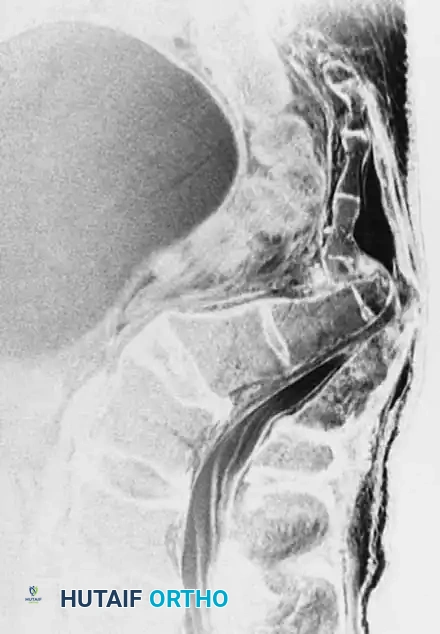
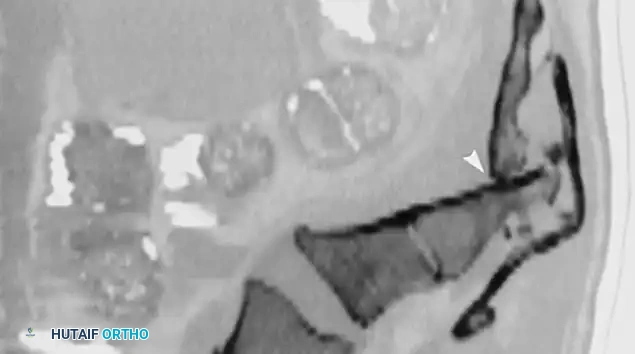
Fig. 35-62 A, Sagittal CT scan of transverse fracture through S3 resulting in transection of sacral nerve roots (arrow) . B, MRI of sacral fracture resulting in complete canal compromise. C, Postoperative CT scan shows decompression of sacral spinal canal after laminectomy (arrows) .
(II and III) of methylprednisolone in acute spinal cord injury, J Spinal Disord 13:165, 2000.](https://pubmed.ncbi.nlm.nih.gov/?term=Coleman%20WP%2C%20Benzel%20D%2C%20Cahill%20DW%2C%20et%20al%3A%20A%20critical%20appraisal%20of%20the%20reporting%20of%20the%20National%20Acute%20Spinal%20Cord%20Injury%20Studies%0A%0AA%0A%0AB%0A%0AC%0A%0A%21%5BFig.%2035-62%20A%2C%20Sagittal%20CT%20scan%20of%20transverse%20fracture%20through%20S3%20resulting%20in%20transection%20of%20sacral%20nerve%20roots%20%28arrow%29%20.%20B%2C%20MRI%20of%20sacral%20fracture%20resulting%20in%20complete%20canal%20compromise.%20C%2C%20Postoperative%20CT%20scan%20shows%20decompression%20of%20sacral%20spinal%20canal%20after%20laminectomy%20%28arrows%29%20.%5D%28/media/hutaifortho/hutaifortho-references-p1845-b1c190.webp%29%0A%0A%21%5BFig.%2035-62%20A%2C%20Sagittal%20CT%20scan%20of%20transverse%20fracture%20through%20S3%20resulting%20in%20transection%20of%20sacral%20nerve%20roots%20%28arrow%29%20.%20B%2C%20MRI%20of%20sacral%20fracture%20resulting%20in%20complete%20canal%20compromise.%20C%2C%20Postoperative%20CT%20scan%20shows%20decompression%20of%20sacral%20spinal%20canal%20after%20laminectomy%20%28arrows%29%20.%5D%28/media/hutaifortho/hutaifortho-references-p1845-cc5bfe.webp%29%0A%0A%21%5BFig.%2035-62%20A%2C%20Sagittal%20CT%20scan%20of%20transverse%20fracture%20through%20S3%20resulting%20in%20transection%20of%20sacral%20nerve%20roots%20%28arrow%29%20.%20B%2C%20MRI%20of%20sacral%20fracture%20resulting%20in%20complete%20canal%20compromise.%20C%2C%20Postoperative%20CT%20scan%20shows%20decompression%20of%20sacral%20spinal%20canal%20after%20laminectomy%20%28arrows%29%20.%5D%28/media/hutaifortho/hutaifortho-references-p1845-1e1957.webp%29%0A%0A%2AFig.%2035-62%20A%2C%20Sagittal%20CT%20scan%20of%20transverse%20fracture%20through%20S3%20resulting%20in%20transection%20of%20sacral%20nerve%20roots%20%28arrow%29%20.%20B%2C%20MRI%20of%20sacral%20fracture%20resulting%20in%20complete%20canal%20compromise.%20C%2C%20Postoperative%20CT%20scan%20shows%20decompression%20of%20sacral%20spinal%20canal%20after%20laminectomy%20%28arrows%29%20.%2A%0A%0A%28II%20and%20III%29%20of%20methylprednisolone%20in%20acute%20spinal%20cord%20injury%2C%20J%20Spinal%20Disord%2013%3A165%2C%202000.)
- Colterjohn NR, Bednar DA: Identifi able risk factors for secondary neurologic deterioration in the cervical spine-injured patient, Spine 20:2293, 1995.
- Connolly P, Yuan HA: Cervical spine fractures. In White AH, ed: Spine care: diagnosis and conservative treatment, St Louis, 1995, Mosby. Cooper C, Atkinson EJ, Fallon WM, et al: Incidence of clinically diagnosed vertebral fractures: a population-based study in Rochester, Minnesota, 1985-1989, J Bone Miner Res 7:221, 1992.
- Cotler JM, Herbison GJ, Nasti JF, et al: Closed reduction of traumatic cervical spine dislocation using traction weights up to 140 pounds, Spine 18:386, 1983.
- Coyne TJ, Fehlings MG, Wallace MC, et al: C1-C2 posterior cervical fusion: long-term evaluation of results and effi cacy, Neurosurgery 37:688, 1995.
- Crowther ER: Missed cervical spine fractures: the importance of reviewing radiographs in chiropractic practice, J Manipulative Physiol Ther 18:29, 1995.
- Danisa OA, Shaffrey CI, Jane JA, et al: Surgical approaches for the correction of unstable thoracolumbar burst fractures: a retrospective analysis of treatment outcomes, J Neurosurg 83:977, 1995.
- Delamarter RB, Sherman J, Carr JB: Spinal cord injury: the pathophysiology of spinal cord damage and subsequent recovery following immediate or delayed decompression. Paper presented at Cervical Spine Research Society meeting, New York, 1993.
- De La Torre JC: Spinal cord injury: review of basic and applied research, Spine 6:315, 1981.
- Denis F: Spinal instability as defi ned by the three-column spine concept in acute spinal trauma, Clin Orthop Relat Res 189:65, 1984.
- Devilee R, Sanders R, deLange S: Treatment of fractures and dislocations of the thoracic and lumbar spine by fusion and Harrington instrumentation, Arch Orthop Trauma Surg 114:100, 1995.
- Dickman CA, Zabramski JM, Hadley MN, et al: Pediatric spinal cord injury without radiographic abnormalities: report of 26 cases and review of the literature, J Spinal Disord 4:296, 1991.
- Dorr LD, Harvey JP, Nickel VL: Clinical review of the early stability of spine injuries, Spine 7:545, 1982.
- Ebraheim NA, Rupp RE, Savolaine ER, et al: Posterior plating of the cervical spine, J Spinal Disord 8:111, 1995.
- Eismont FJ, Arena M, Green B: Extrusion of an intervertebral disc associated with traumatic subluxation or dislocation of cervical facets, J Bone Joint Surg 73A:1555, 1991.
- Eismont FJ, Clifford S, Goldberg M, et al: Cervical sagittal spinal canal size in spine injury, Spine 9:663, 1984.
- Fan RS, Schenk RS, Lee CK: Burst fracture of the fi fth lumbar vertebra in combination with a pelvic ring injury, J Orthop Trauma 9:345, 1995.
- Fontijne WPJ, DeKlerk LWL, Braakman R, et al: CT scan prediction of neurological defi cit in thoracolumbar burst fractures, J Bone Joint Surg 74B:683, 1992.
- Fowler BL, Dall BE, Rowe DE: Complications associated with harvesting autogenous iliac bone graft, Am J Orthop 24:895, 1995.
- Fricker R, Gächter A: Lateral fl exion/extension radiographs: still recommended following cervical spinal injury, Arch Orthop Trauma Surg 113:115, 1994.
- [Garfi n SR, ed: Complications of spine surgery, Baltimore, 1989, Williams & Wilkins.
Geisler FH, Dorsey FC, Coleman WP: Recovery of motor function after spinal cord injury: a randomized, placebocontrolled trial with GM-1 ganglioside, N Engl J Med 324:1829, 1991.](https://pubmed.ncbi.nlm.nih.gov/?term=Gar%EF%AC%81%20n%20SR%2C%20ed%3A%20Complications%20of%20spine%20surgery%2C%20Baltimore%2C%201989%2C%20Williams%20%26%20Wilkins.%0A%0AGeisler%20FH%2C%20Dorsey%20FC%2C%20Coleman%20WP%3A%20Recovery%20of%20motor%20function%20after%20spinal%20cord%20injury%3A%20a%20randomized%2C%20placebocontrolled%20trial%20with%20GM-1%20ganglioside%2C%20N%20Engl%20J%20Med%20324%3A1829%2C%201991.)
- Geisler FH, Dorsey FC, Coleman WP: GM-1 ganglioside in human spinal cord injury, J Neurotrauma 9(suppl):517, 1992.
- Gertzbein SD, Court-Brown CM, Marks P, et al: The neurological outcome following surgery for
You Might Also Like

