Compartment Syndrome: Pathophysiology and Surgical Management
Key Takeaway
Compartment syndrome is a surgical emergency characterized by elevated interstitial pressure within a closed osseofascial compartment, leading to microvascular compromise and tissue ischemia. Diagnosis relies on clinical signs—primarily pain out of proportion to injury—and intracompartmental pressure monitoring. Prompt surgical intervention via decompressive fasciotomy is the definitive treatment to prevent irreversible muscle necrosis and neurological deficit.
Introduction to Compartment Syndrome
Mubarak defined compartment syndrome as an elevation of the interstitial pressure within a closed osseofascial compartment that results in microvascular compromise. This condition represents a true orthopedic emergency. Compartments bounded by relatively noncompliant fascial or osseous structures are most frequently involved. In clinical practice, the anterior and deep posterior compartments of the lower leg, alongside the volar compartment of the forearm, represent the most common anatomical sites for this pathology.

However, compartment syndrome can develop in any anatomical region where skeletal muscle is enveloped by substantial, unyielding fascia. Documented sites include the buttock, thigh, shoulder, hand, foot, arm, and lumbar paraspinous musculature.
Compartment syndromes are broadly classified into acute and chronic forms, dictated by the etiology of the elevated pressure and the temporal profile of the symptoms.
Acute Compartment Syndrome
The most prevalent etiologies of acute compartment syndrome include high-energy fractures (particularly tibial shaft fractures), severe soft-tissue crush injuries, arterial injuries with subsequent reperfusion edema, prolonged limb compression during states of altered consciousness, and circumferential full-thickness burns. Iatrogenic causes, such as intravenous fluid extravasation and complications related to anticoagulant therapy, must also be considered in the differential diagnosis.
Chronic Exertional Compartment Syndrome (CECS)
Chronic exertional compartment syndrome (CECS) is characterized by a recurrent, exercise-induced elevation of intracompartmental pressure, most frequently localizing to the anterior or deep posterior compartments of the leg. It has also been documented in the forearms of elite weightlifters and motocross riders. Physiologically, vigorous exercise can increase skeletal muscle volume by up to 20%. In the presence of a noncompliant fascial envelope, this volume expansion precipitates a pathological rise in pressure. CECS is predominantly observed in long-distance runners and military recruits subjected to physical demands exceeding their normal functional tolerance.
Clinical Pearl: The pathophysiology of CECS may fundamentally differ from the acute syndrome. Nuclear blood flow studies by Amendola et al. suggest that the pathophysiology of exertional compartment syndrome may not be strictly related to absolute ischemia, though the exact mechanism of pain generation remains a subject of ongoing academic debate.
Pathophysiology and Biomechanics
The pathophysiology of acute compartment syndrome is driven by a catastrophic insult to normal local tissue homeostasis. This insult initiates a vicious cycle: increased tissue pressure leads to decreased capillary blood flow, which in turn causes local tissue hypoxia, further edema, and eventual cellular necrosis.
Local blood flow (LBF) is mathematically defined by the arteriovenous gradient divided by the local vascular resistance:
LBF = (Pa − Pv) / R
Under ischemic conditions, local autoregulatory mechanisms minimize vascular resistance (R), maximizing arterial blood flow. However, as intracompartmental pressure rises, venous pressure (Pv) must rise concurrently to maintain venous outflow. When Pv exceeds the capillary perfusion pressure, capillary collapse occurs, halting microvascular perfusion despite the presence of a palpable distal arterial pulse.
Experimental evidence demonstrates that significant, irreversible muscle necrosis can occur even in patients with normal systemic blood flow if the intracompartmental pressure is sustained above 30 mm Hg for longer than 8 hours. The relationship between pressure and time is inversely proportional; higher pressures precipitate neuromuscular compromise in significantly shorter durations.
Canine models have elucidated these ischemic thresholds:
* A pressure of 40 mm Hg sustained for 14 hours did not arrest nerve conduction.
* A pressure of 50 mm Hg arrested nerve conduction in less than 7 hours.
Rorabeck and Clarke further demonstrated that the duration of elevated pressure is the critical determinant for the return of neurological function. Pressures between 40 and 80 mm Hg sustained for 4 hours caused no permanent nerve dysfunction; however, when these pressures were maintained for 12 hours or more, irreversible neurological deficits ensued. Human volunteer studies corroborate these findings, though they highlight significant inter-individual variations in tolerance to elevated compartmental pressures.
Clinical Diagnosis of Acute Compartment Syndrome
The diagnosis of acute compartment syndrome is primarily clinical, demanding a high index of suspicion. The classic "Ps" (Pain, Pallor, Pulselessness, Paresthesia, Paralysis) are often taught; however, relying on pallor and pulselessness is a dangerous pitfall, as these are late signs indicating established arterial occlusion rather than early microvascular compromise.
The earliest and most reliable physical signs include:
* Tightness: A tense, "wood-like" feeling of the involved compartment upon palpation.
* Pain with Passive Stretch: Exquisite pain elicited by passive motion of the muscles traversing the affected compartment (e.g., severe pain in the anterior leg upon passive plantar flexion of the toes).
* Pain Out of Proportion: The hallmark symptom is pain that is disproportionate to the anticipated severity of the primary injury and is refractory to standard opioid analgesia.
* Neurological Deficits: Hypesthesia or paresthesia in the distribution of the nerves traversing the compartment. This must be meticulously evaluated using pinprick, light touch, and two-point discrimination testing.
Surgical Warning: The clinical diagnosis of acute compartment syndrome can be severely delayed or masked in polytrauma patients, individuals with altered states of consciousness (e.g., traumatic brain injury, intubation), and pediatric populations where physical findings cannot be reliably documented. In these high-risk cohorts, objective pressure measurement is mandatory.
Intracompartmental Pressure Monitoring
When compartment syndrome is suspected but an adequate clinical examination is precluded, or when clinical findings are equivocal, direct measurement of intracompartmental pressure is indicated. Continuous monitoring is particularly valuable in patients with altered neurological function secondary to vascular injuries, continuous epidural anesthesia, peripheral nerve injury, or prolonged tourniquet application.
Measurement Techniques
Several modalities exist for measuring tissue pressure. If sophisticated equipment is unavailable, the classic technique described by Whitesides et al. can be employed using standard hospital supplies: intravenous tubing, a three-way stopcock, a syringe, and a mercury manometer.
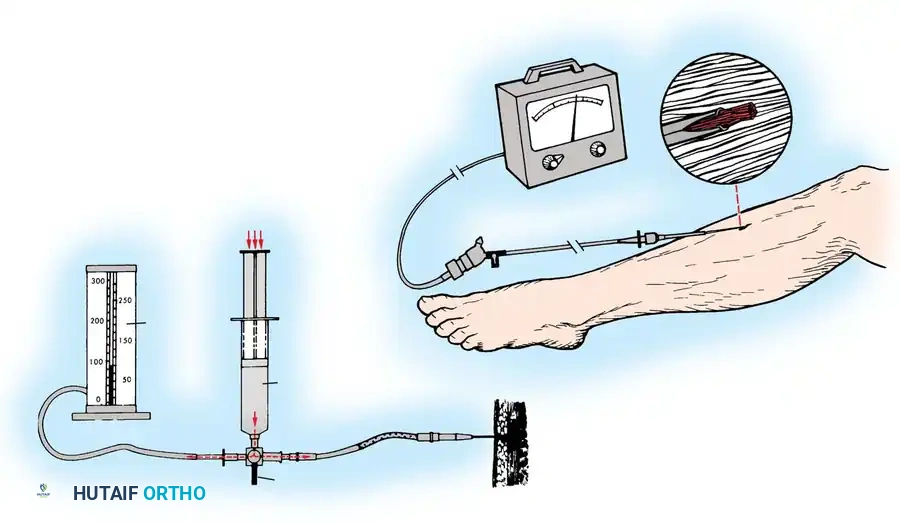
Modern, dedicated devices offer superior accuracy and ease of use. The hand-held monitor is widely utilized for single, rapid pressure readings in the emergency department or operating theater.
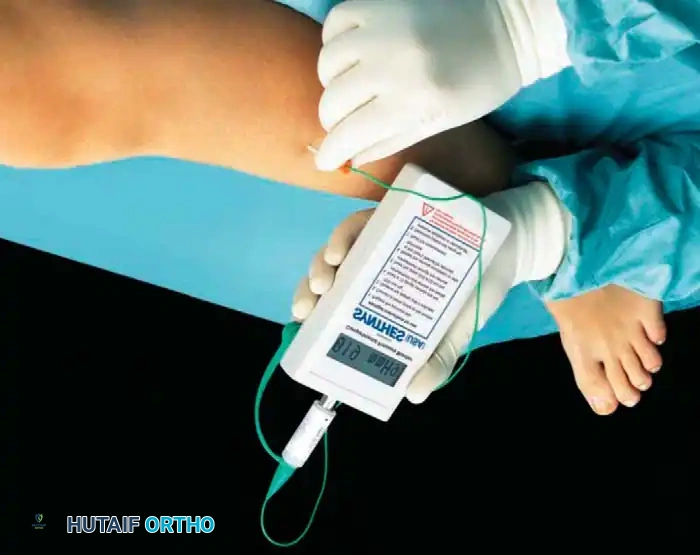
For continuous monitoring, the wick catheter (described by Scholander et al.) and the slit catheter provide accurate, reproducible data. The slit catheter is generally preferred for long-term monitoring as it is less susceptible to intraluminal coagulation.
Comparative studies by Boody and Wongworawat evaluated the Stryker Intracompartmental Pressure Monitor System, arterial line manometers, and the Whitesides apparatus. Their findings demonstrated that side-port needles and slit catheters are significantly more accurate than standard straight needles. While the arterial line manometer and the Stryker device proved highly accurate, the traditional Whitesides manometer apparatus was deemed to lack the precision required for critical clinical decision-making.
Diagnostic Thresholds and the Delta P Concept
Historically, an absolute compartmental pressure greater than 30 mm Hg was considered an absolute indication for fasciotomy. However, modern orthopedic trauma protocols emphasize the "Delta P" (ΔP) concept.
Heppenstall et al. demonstrated that cellular metabolic derangement in skeletal muscle correlates more strongly with the difference between the mean arterial pressure (or diastolic blood pressure) and the compartment pressure, rather than the absolute compartment pressure alone.
ΔP = Diastolic Blood Pressure − Compartment Pressure
A ΔP of less than 30 mm Hg is widely accepted as the threshold for surgical intervention. In severely traumatized muscle with higher metabolic demands, a ΔP of less than 40 mm Hg may be a safer threshold to prevent necrosis.
Bourne and Rorabeck developed a highly effective algorithm for patients with tibial fractures to synthesize clinical findings and pressure measurements into a cohesive treatment pathway.
Pitfall: Collinge and Person noted that erroneous pressure measurements occurred in up to 27% of clinical readings. Therefore, pressure measurements should never override unequivocal clinical signs. Treat the patient, not just the number.
Preoperative Management and Resuscitation
If an incipient compartment syndrome is suspected, immediate non-operative interventions must be initiated to mitigate the risk of progression to a full-blown syndrome.
- Release of Constriction: In isolated limb injuries treated with casting, splitting the cast and the underlying padding down to the skin can decrease compartment pressure by 50% to 85%. All circular constrictive bandages must be completely released.
- Limb Positioning: The limb must be positioned at the level of the heart. Elevating the limb above the heart decreases arterial inflow without significantly improving venous outflow, thereby exacerbating local ischemia and accelerating tissue necrosis.
- Systemic Resuscitation: Hypotension drastically reduces the perfusion gradient. Aggressive use of crystalloid or colloid fluids, blood replacement, and correction of coagulopathy are vital to maintaining adequate mean arterial pressure.
If symptoms do not resolve within 30 to 60 minutes following these interventions, pressure measurements should be repeated. If the ΔP remains critical, emergent fasciotomy is indicated.
Surgical Management: Decompressive Fasciotomy
Fasciotomy is the definitive treatment for acute compartment syndrome. The goal is the complete, unhindered release of all fascial envelopes surrounding the affected musculature.
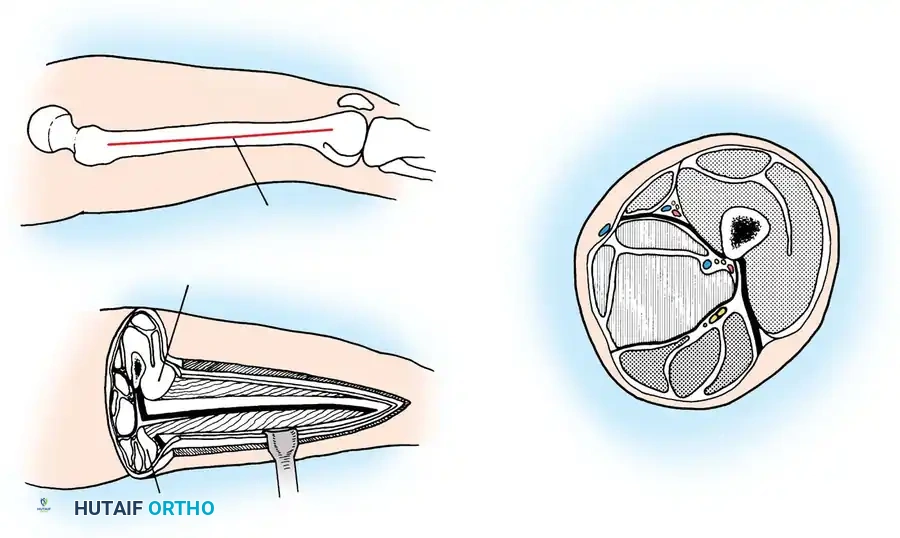
Fasciotomy for Acute Compartment Syndrome of the Thigh (Tarlow et al. Technique)
Compartment syndrome of the thigh is rare due to the large volume of the thigh compartments, which can accommodate significant swelling. However, it can occur following high-energy femur fractures, severe crush injuries, or prolonged external compression.
Step-by-Step Surgical Technique:
- Positioning and Preparation: Place the patient supine. Prepare and drape the entire thigh in a sterile fashion, ensuring exposure from the iliac crest proximally to the knee joint distally. Do not use a tourniquet, as assessing muscle viability and achieving hemostasis are critical.
- Lateral Incision: Make a generous lateral longitudinal incision beginning just distal to the intertrochanteric line and extending distally to the lateral epicondyle of the femur.
- Iliotibial Band Release: Utilize subcutaneous dissection to expose the iliotibial band. Make a straight, continuous incision in line with the skin incision through the entire length of the iliotibial band.
- Vastus Lateralis Reflection: Carefully reflect the vastus lateralis muscle anteriorly off the lateral intermuscular septum. Meticulously coagulate or ligate all perforating vessels encountered during this reflection to prevent postoperative hematoma.
- Septal Incision: Make a 1.5-cm incision in the lateral intermuscular septum. Using heavy Metzenbaum scissors, extend this incision proximally and distally along the entire length of the surgical exposure to decompress the posterior compartment.
- Medial Compartment Assessment: After the anterior and posterior compartments have been released, directly measure the pressure of the medial (adductor) compartment.
- Medial Release (If Indicated): If the medial compartment pressure remains elevated (ΔP < 30 mm Hg), make a separate medial longitudinal incision to release the adductor compartment fascia comprehensively.
Fasciotomy of the Lower Leg (Double-Incision Technique)
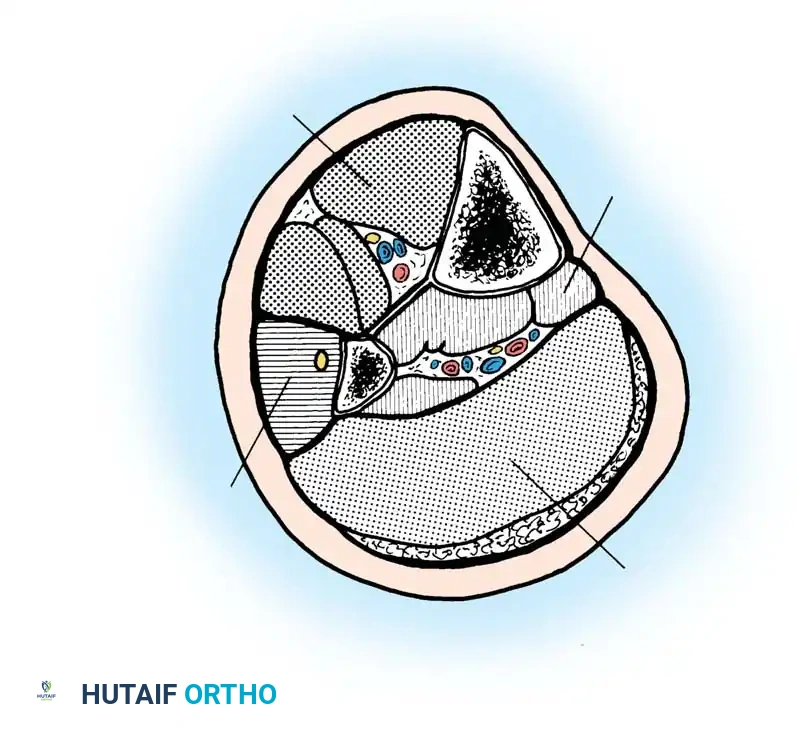
The lower leg contains four distinct compartments: anterior, lateral, superficial posterior, and deep posterior. A double-incision technique is the gold standard to ensure all four compartments are adequately decompressed.
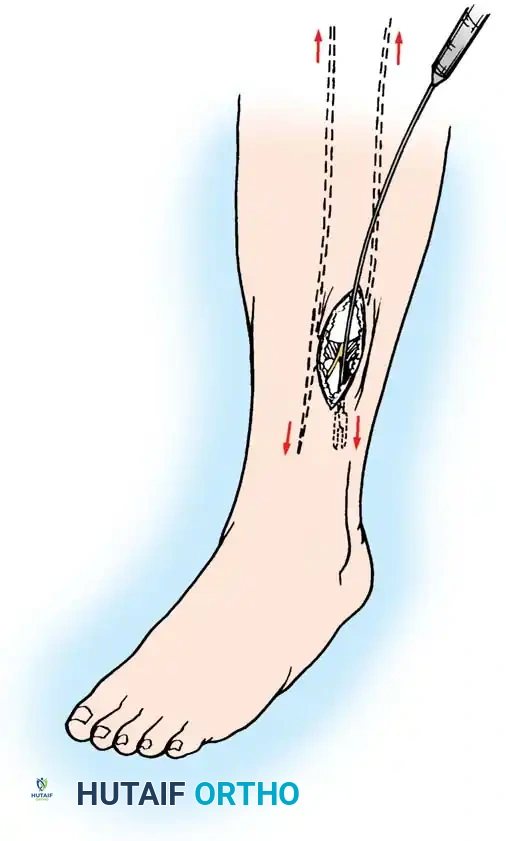
Step-by-Step Surgical Technique:
- Anterolateral Incision: Make a 15 to 20 cm longitudinal incision centered halfway between the tibial crest and the fibular shaft.
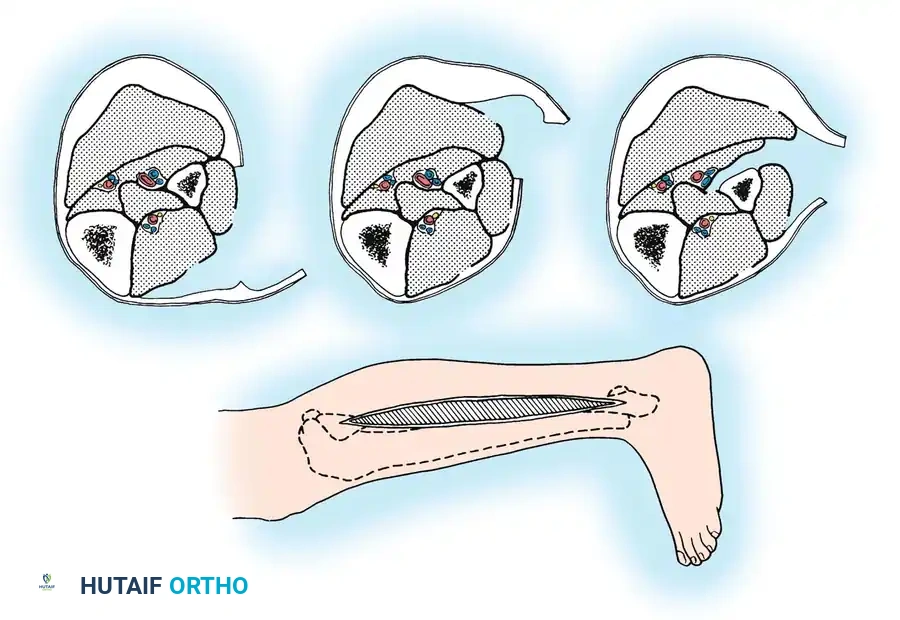
- Anterior and Lateral Release: Perform subcutaneous dissection to expose the fascia. Identify the anterior intermuscular septum dividing the anterior and lateral compartments.
- Fascial Incision: Make a small transverse incision in the fascia to clearly identify the septum. Release the anterior compartment fascia longitudinally using fasciotomy scissors, extending the cut proximally toward the tibial tubercle and distally toward the ankle.
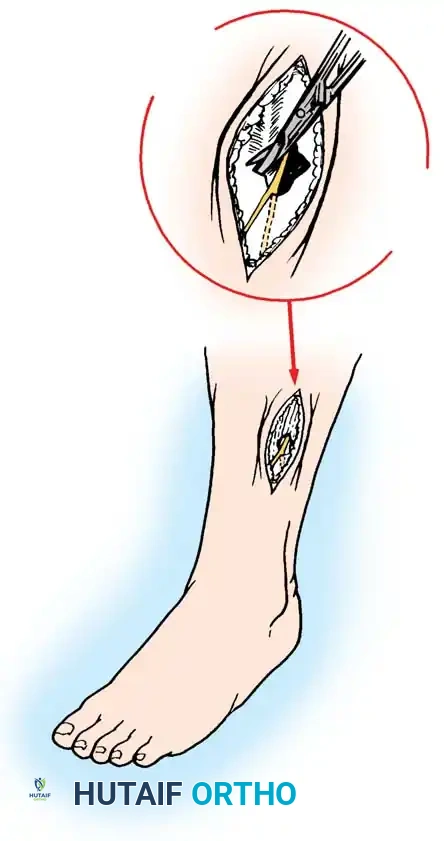
- Lateral Compartment Release: Similarly, release the lateral compartment fascia longitudinally, taking extreme care to identify and protect the superficial peroneal nerve, which exits the fascia in the distal third of the leg.
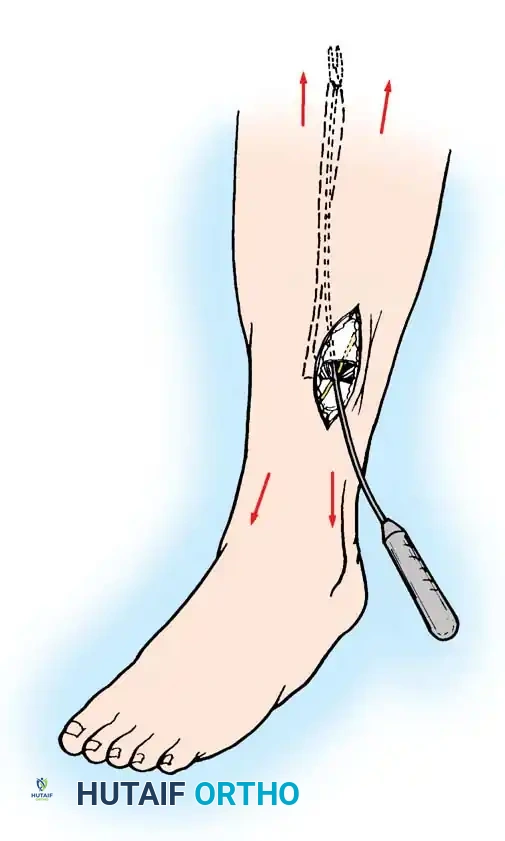
- Posteromedial Incision: Make a second 15 to 20 cm longitudinal incision on the medial aspect of the leg, approximately 2 cm posterior to the palpable posteromedial border of the tibia.
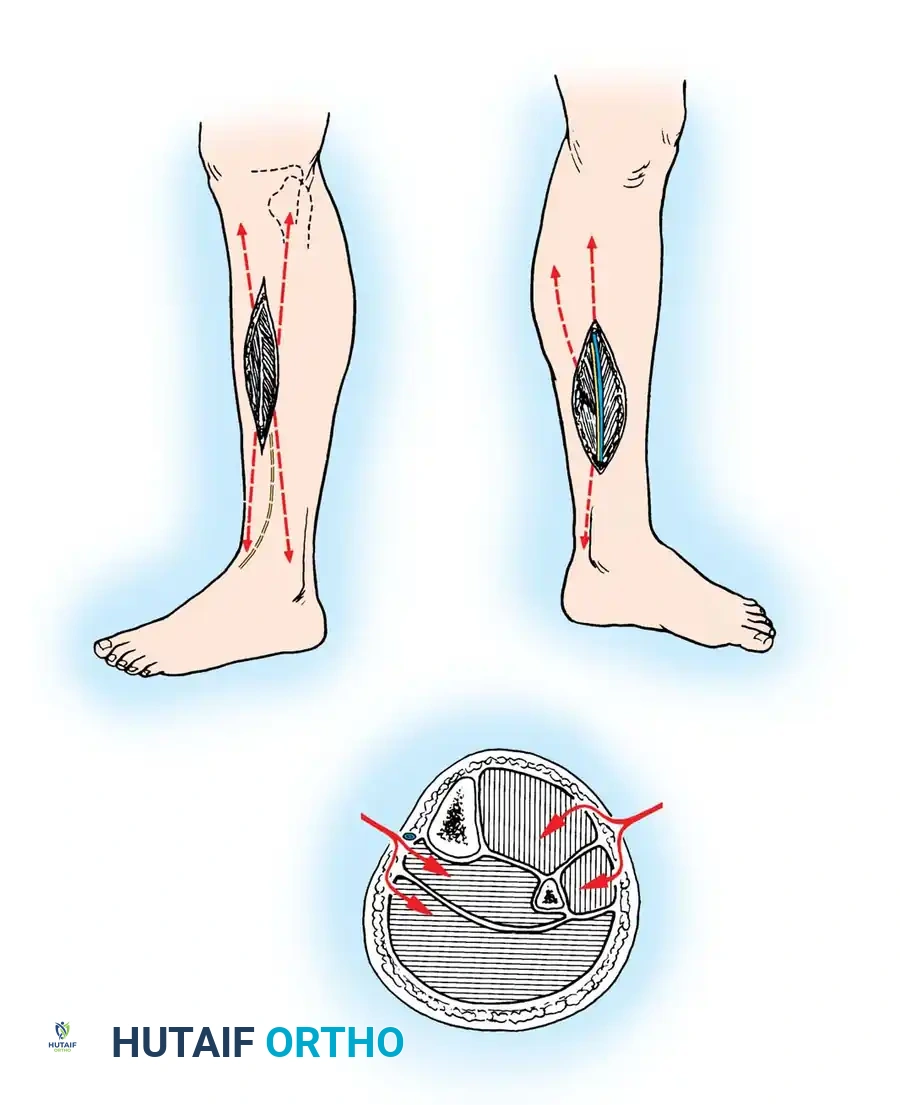
- Superficial and Deep Posterior Release: Retract the saphenous vein and nerve anteriorly. Incise the fascia over the superficial posterior compartment (gastrocnemius/soleus complex). Next, detach the soleus bridge from the posterior tibia to expose the fascia of the deep posterior compartment. Incise this fascia longitudinally to decompress the tibialis posterior, flexor hallucis longus, and flexor digitorum longus muscles, alongside the posterior tibial neurovascular bundle.
Postoperative Protocols and Wound Management
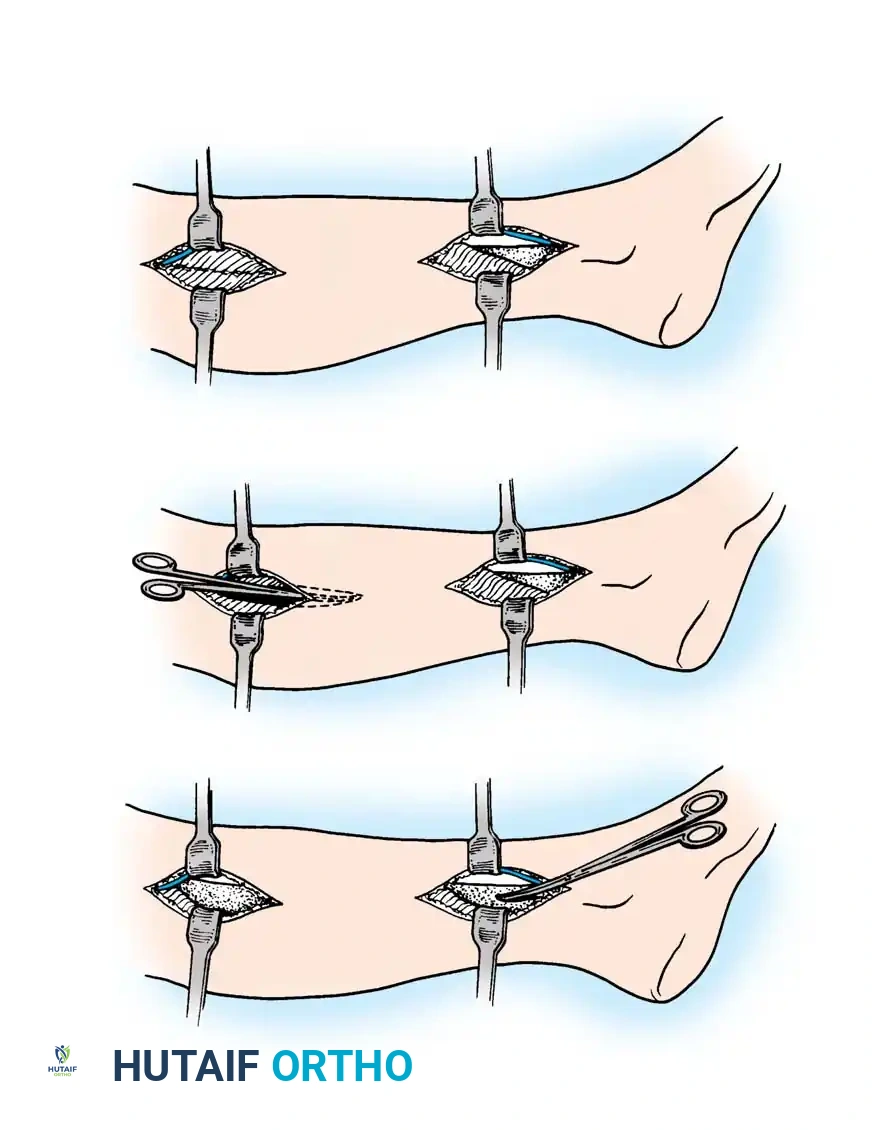
Following a decompressive fasciotomy, the surgical wounds must be left open. Primary closure is strictly contraindicated as it will recreate the compartment syndrome.
- Initial Dressing: The wounds are typically managed with sterile, non-adherent dressings or, preferably, Negative Pressure Wound Therapy (NPWT / VAC dressings) set to -75 to -125 mm Hg. NPWT helps manage exudate, reduces local tissue edema, and promotes the formation of healthy granulation tissue.
- Second Look: The patient should be returned to the operating room within 48 to 72 hours for a "second look" debridement. Any necrotic muscle identified at this stage must be aggressively excised until healthy, bleeding, contractile muscle is reached.
- Wound Closure: Delayed primary closure may be attempted using vessel loops or dynamic wound closure devices (e.g., dermatotraction) over several days. If the skin edges cannot be approximated without tension after 7 to 10 days, split-thickness skin grafting (STSG) is required to achieve definitive coverage.
- Rehabilitation: Early passive and active range of motion of the adjacent joints is instituted as soon as clinically permissible to prevent contractures and promote functional recovery. Weight-bearing status is dictated by the management of any concomitant fractures.
You Might Also Like
