Acute Compartment Syndrome Diagnosis in Open Tibial Fractures: A Clinical Case Study
Key Takeaway
Acute compartment syndrome after tibial fracture presents with pain disproportionate to injury, unremitting despite analgesia. Key red flags include tense, 'wood-hard' compartments, severe pain on passive stretch, progressive neurological deficits (weakness, paresthesia), and evolving vascular compromise (diminished pulses, delayed capillary refill). Early recognition is vital.
Patient Presentation and History
A 28-year-old previously healthy male presented to the Level I trauma center emergency department following a high-energy motor vehicle collision. The patient was the unrestrained driver of a sedan that struck a stationary object (a tree) head-on at an estimated velocity of 70 km/h. On scene, he required a prolonged extrication of approximately 35 minutes by emergency medical services due to severe dashboard intrusion into the driver-side footwell.
Initial Advanced Trauma Life Support assessment in the field revealed a Glasgow Coma Scale of 15. Vital signs demonstrated a hyperdynamic state consistent with acute pain and sympathetic activation, though hemodynamically stable: heart rate 88 to 105 beats per minute, blood pressure 125/78 mmHg, respiratory rate 16 breaths per minute, and oxygen saturation 99% on ambient air. The primary pre-hospital complaint was severe, intractable right lower extremity pain. There was no reported history of significant medical comorbidities, congenital anomalies, known drug allergies, or prior surgical interventions. His social history was non-contributory, noting only occasional social ethanol consumption and denying illicit substance use or tobacco smoking.
The initial primary survey in the trauma bay proceeded according to standard ATLS protocols. The airway was patent and protected, breathing was symmetric and unlabored, and circulation was intact with strong central pulses. The secondary survey identified an obvious, gross clinical deformity of the right lower leg. A 2 cm transversely oriented laceration was noted over the anterior aspect of the distal third of the tibia, actively weeping serosanguinous fluid with visible subcutaneous adipose tissue and underlying fascial disruption. Significant, rapidly progressive soft tissue swelling was noted circumferentially around the lower leg. Distal pulses (dorsalis pedis and posterior tibial) were palpable and symmetric to the contralateral limb, and capillary refill was brisk at less than two seconds. Initial gross neurological assessment indicated intact light touch sensation in the superficial peroneal, deep peroneal, tibial, sural, and saphenous nerve distributions. A provisional diagnosis of a Gustilo-Anderson Type II open right tibial diaphyseal fracture was established. The remainder of the secondary survey was unremarkable, with focused assessment with sonography for trauma yielding negative results for intraperitoneal free fluid or pericardial effusion. Pelvic stability was confirmed, and there were no other acute traumatic orthopedic or visceral injuries identified.
Upon transfer to the definitive care bay, the clinical picture began to evolve rapidly. The patient reported excruciating, unremitting pain in his right lower leg, rated as 10/10 on the Visual Analog Scale. This pain severity persisted despite the administration of 10 mg of intravenous morphine sulfate by EMS en route, followed by an additional 50 mcg of intravenous fentanyl in the trauma bay. The patient described the pain as a deep, throbbing, and constant pressure that was completely unrelieved by repositioning, elevation, or immobilization in a temporary radiolucent splint. This pain profile was noticeably worse and out of proportion to the initial assessment in the field, raising immediate clinical suspicion for an evolving acute compartment syndrome.
Clinical Examination Findings
Upon definitive evaluation by the orthopedic trauma service, the patient was alert, oriented, but in significant systemic distress secondary to the localized pain. The right lower extremity examination was meticulously conducted to differentiate between standard fracture-related pain and ischemic pain indicative of compartment syndrome.
Inspection
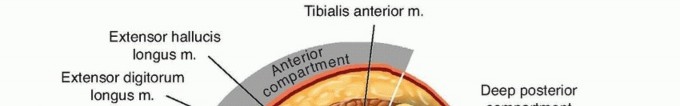
Marked angulation and circumferential swelling of the mid-to-distal diaphyseal region of the right tibia were immediately apparent. The 2 cm transversely oriented laceration over the anterior aspect of the distal third of the tibia was actively weeping a mixture of hematoma and serosanguinous fluid. The surrounding soft tissue envelope was exceedingly tense, shiny, and erythematous. While no fracture blisters or gross ecchymosis had yet formed—consistent with the acute timeline of less than two hours post-injury—the swelling was visibly and rapidly progressive.
The anterior and lateral compartments of the right lower leg appeared overtly tense and distended. The skin over these specific fascial compartments felt stretched, losing normal resting skin tension lines. This visual tenseness was distinctly asymmetric when compared to the uninjured contralateral limb and appeared disproportionately severe relative to the superficial and deep posterior compartments.
Palpation
Palpation elicited exquisite, poorly localized tenderness along the entire osseous length of the tibia, which was expected given the fracture. However, this tenderness was dramatically exacerbated by specific palpation over the muscular bellies of the anterior and lateral compartments.
Assessment of compartment turgor revealed that the anterior compartment felt "wood-hard" and entirely non-yielding to deep digital palpation. The lateral compartment demonstrated similar, though slightly less pronounced, firmness. The superficial and deep posterior compartments, while exhibiting post-traumatic edema, were palpably less tense and retained some degree of compressibility.
The most critical and diagnostic finding was the assessment of pain with passive stretch of the ischemic muscle groups:
* Anterior compartment: Passive plantarflexion of the ankle and passive flexion of the great toe and lesser toes (selectively stretching the tibialis anterior, extensor hallucis longus, and extensor digitorum longus) elicited severe, excruciating pain that caused the patient to physically withdraw. This pain response was entirely disproportionate to the mechanical movement of the fracture site.
* Lateral compartment: Passive inversion of the ankle (stretching the peroneus longus and brevis) resulted in severe, sharp pain along the lateral fibular shaft.
* Posterior compartments: Passive dorsiflexion of the ankle and passive extension of the toes (stretching the gastrocnemius, soleus, flexor hallucis longus, and flexor digitorum longus) produced mild to moderate discomfort, but this was notably less severe than the pain elicited from the anterior and lateral compartments.
* The skin overlying the affected compartments was warm to the touch, ruling out late-stage arterial occlusion but consistent with the hyperemic response of early tissue distress.
Range of Motion
Active range of motion of the ankle and toes was severely limited and functionally absent due to pain inhibition, particularly for active dorsiflexion and eversion. Passive range of motion was mechanically restricted by the tense soft tissue envelope and elicited the disproportionate pain cascade described above.
Neurological and Vascular Assessment
Motor function assessment revealed rapid deterioration. Within the anterior compartment, active dorsiflexion of the ankle (tibialis anterior, innervated by the deep peroneal nerve) and toe extension (EHL, EDL, innervated by the deep peroneal nerve) demonstrated profound weakness. The patient was unable to overcome gravity. Serial examinations over a 30-minute window showed a rapid decline in motor strength from an initial 3/5 to 1/5 (flicker of contraction). Within the lateral compartment, active eversion of the ankle (peroneus longus and brevis, innervated by the superficial peroneal nerve) declined from 3/5 to 2/5. The superficial posterior compartment (gastrocnemius/soleus complex, innervated by the tibial nerve) maintained 4/5 strength for plantarflexion.
Sensory examination revealed diminished two-point discrimination in the first dorsal webspace, indicating early ischemic neurapraxia of the deep peroneal nerve. Sensation over the lateral dorsum of the foot (superficial peroneal nerve) was altered, characterized by hyperesthesia progressing to early paresthesia.
Crucially, the dorsalis pedis and posterior tibial pulses remained robust and palpable. Capillary refill in the digits remained under two seconds. This highlighted the classic physiological principle that peripheral pulses are maintained until the intra-compartmental pressure exceeds the systolic arterial pressure, making pulselessness an unreliable and exceedingly late sign of acute compartment syndrome.
Imaging and Diagnostics
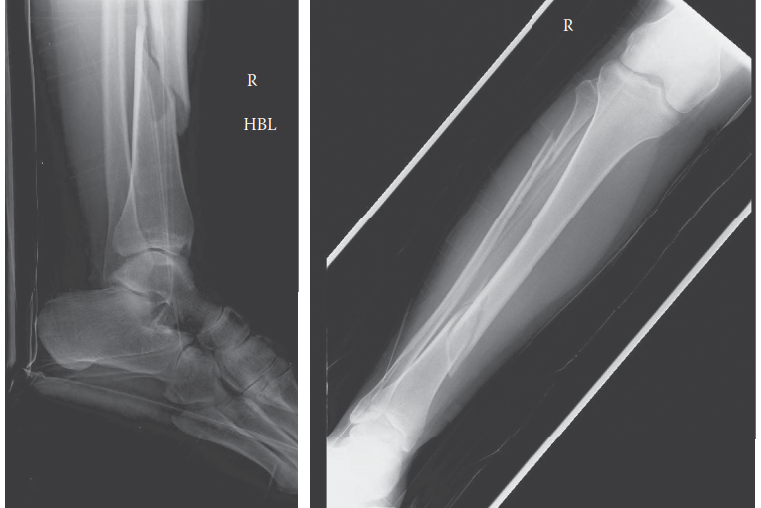
Standard trauma radiographic series were obtained, including orthogonal anteroposterior and lateral projections of the right tibia and fibula, as well as dedicated views of the ipsilateral knee and ankle joints to rule out contiguous intra-articular extension or associated injuries.

Radiographic evaluation revealed a displaced, comminuted fracture of the middle and distal third junction of the right tibial diaphysis. The fracture pattern was classified as an OTA/AO 42-B3 (wedge fracture, fragmented wedge). Associated with this was a segmental fracture of the fibular diaphysis at a similar level. There was significant anterolateral angulation and approximately 1.5 cm of overriding shortening. The knee and ankle mortise appeared radiographically intact without evidence of syndesmotic widening or intra-articular fracture extension.
Given the unequivocal clinical presentation of acute compartment syndrome—specifically the escalating pain out of proportion, the wood-hard anterior compartment, and the excruciating pain with passive stretch—the diagnosis was primarily clinical. Advanced cross-sectional imaging such as Computed Tomography or Magnetic Resonance Imaging is strictly contraindicated in the setting of acute compartment syndrome, as the time delay required to obtain these studies inevitably leads to irreversible myonecrosis and neurological deficit.
However, in cases where the clinical picture is equivocal, particularly in obtunded, intubated, or polytraumatized patients, intra-compartmental pressure monitoring is an invaluable diagnostic adjunct. While not strictly required in this awake patient with classic findings, the academic discussion warrants its mention.
Using a solid-state transducer intra-compartmental pressure monitor (e.g., Stryker needle), pressures can be measured in all four compartments. The needle is introduced perpendicularly into the muscle belly, ensuring the side-port is not occluded by fascia.
The critical diagnostic metric is not the absolute compartment pressure, but rather the differential pressure, known as Delta P (ΔP).
ΔP = Diastolic Blood Pressure - Intra-compartmental Pressure.
A ΔP of less than 30 mmHg is the widely accepted threshold indicating inadequate tissue perfusion and absolute necessity for emergent fasciotomy. For example, if a patient has a diastolic blood pressure of 70 mmHg and an absolute anterior compartment pressure of 45 mmHg, the ΔP is 25 mmHg. This falls below the 30 mmHg threshold, confirming critical ischemia. Relying solely on an absolute pressure threshold (e.g., >30 mmHg) is a known pitfall, as a hypertensive patient with a diastolic pressure of 100 mmHg and a compartment pressure of 40 mmHg (ΔP = 60 mmHg) maintains adequate capillary perfusion pressure and may not require fasciotomy, whereas a hypotensive patient with a diastolic of 50 mmHg and a compartment pressure of 25 mmHg (ΔP = 25 mmHg) is actively undergoing ischemic necrosis.
In this clinical scenario, the decision was made to bypass pressure monitoring and proceed directly to the operating theater based on the unmistakable clinical triad of escalating pain, tense compartments, and severe pain with passive stretch.
Differential Diagnosis
The presentation of severe lower extremity pain following high-energy trauma requires a systematic differentiation between several limb-threatening pathologies. The following table outlines the primary differential diagnoses considered in this clinical scenario.
| Differential Diagnosis | Key Clinical Findings | Differentiating Factors from ACS | Diagnostic Modality |
|---|---|---|---|
| Acute Compartment Syndrome | Pain out of proportion, tense "wood-hard" compartments, severe pain with passive muscle stretch, paresthesia (late). | Pain escalates despite analgesia; passive stretch pain is hallmark. Pulses often remain palpable until late stages. | Clinical exam; Intra-compartmental pressure monitoring (ΔP < 30 mmHg). |
| Acute Arterial Occlusion / Ischemia | Pale, pulseless, poikilothermic (cold), paresthesia, paralysis, pain. | Limb is typically cold and pulseless early in the course. Compartments are generally soft, not tense. | Ankle-Brachial Index (ABI < 0.9); CT Angiography; Arterial Duplex Ultrasound. |
| Deep Vein Thrombosis (DVT) | Unilateral swelling, dull aching pain, erythema, positive Homan's sign (unreliable). | Swelling is usually generalized, not confined to specific fascial compartments. Pain is less severe, not "out of proportion." | Venous Duplex Ultrasound; D-dimer (low specificity in trauma). |
| Severe Fracture Pain / Hematoma | Localized pain at the fracture site, swelling, ecchymosis, gross deformity. | Pain is proportional to the injury and improves with immobilization/analgesia. No severe pain with passive stretch of adjacent joints. | Radiographs; Clinical response to splinting and standard analgesia. |
| Necrotizing Fasciitis | Erythema extending beyond wound margins, crepitus, hemorrhagic bullae, systemic toxicity (fever, tachycardia). | Pain is severe but accompanied by systemic sepsis signs early. Crepitus indicates gas-forming organisms. | LRINEC score; Surgical exploration (dishwater pus, lack of fascial bleeding). |
While acute arterial occlusion was considered due to the high-energy nature of the trauma, the presence of bounding distal pulses, brisk capillary refill, and a warm extremity made a major arterial disruption highly unlikely. Furthermore, the localized tenseness of the anterior and lateral compartments strongly pointed away from a generalized ischemic event and toward a localized pressure phenomenon. Deep vein thrombosis was ruled out given the acute timeline (minutes to hours post-injury), as DVT typically requires a longer period of stasis to develop. The primary differential remained standard fracture pain versus acute compartment syndrome. The failure of high-dose intravenous opioids to control the pain, combined with the exquisite pain upon passive stretch of the extensor hallucis longus and tibialis anterior, definitively shifted the diagnosis to acute compartment syndrome.
Surgical Decision Making and Classification
The patient presented with a Gustilo-Anderson Type II open tibial shaft fracture complicated by acute compartment syndrome. The fracture was classified as Type II because the laceration was greater than 1 cm (measuring 2 cm), without extensive soft tissue damage, avulsions, or degloving, and with adequate soft tissue coverage for the bone. However, the high-energy mechanism (70 km/h MVC) suggested significant underlying soft tissue injury not immediately visible at the skin level, which was corroborated by the rapid onset of compartment syndrome. The osseous injury was classified as an OTA/AO 42-B3.
The presence of an open fracture is a critical clinical pearl: an open fracture does not adequately decompress the fascial compartments. The fascial defect is rarely large enough to allow sufficient extrusion of hematoma and edematous muscle to lower the intra-compartmental pressure below the ischemic threshold. In many series, the incidence of acute compartment syndrome in open tibial fractures is equal to, or even slightly higher than, that in closed fractures, likely due to the higher energy imparted to the soft tissue envelope.
The diagnosis of acute compartment syndrome constitutes an absolute orthopedic emergency. The pathophysiology involves a localized increase in tissue pressure within a non-yielding osseous-fascial envelope. As pressure rises, it surpasses the venous outflow pressure, leading to venous congestion. This congestion further increases intra-compartmental pressure, which subsequently surpasses the capillary perfusion pressure, leading to capillary collapse and tissue ischemia. Ischemia causes cellular hypoxia, failure of the ATP-dependent Na+/K+ pump, and subsequent intracellular swelling, which exacerbates the pressure in a devastating positive feedback loop.
Skeletal muscle can tolerate ischemia for approximately 4 hours without permanent damage. Between 4 and 8 hours, irreversible myonecrosis and peripheral nerve damage begin to occur. Beyond 8 hours, the damage is essentially complete, and late fasciotomy carries a high risk of reperfusion injury, massive rhabdomyolysis, acute kidney injury, and potentially death from hyperkalemia. Given the patient's timeline (approximately 2.5 hours post-injury), emergent surgical intervention was indicated to halt the ischemic cascade.
The surgical strategy required two distinct but concurrent phases:
1. Emergent four-compartment fasciotomy to decompress the soft tissues.
2. Stabilization of the tibial fracture to protect the soft tissues from further mechanical trauma and to facilitate nursing care and wound management.
For fracture stabilization in the setting of acute compartment syndrome and an open fracture, the primary options are spanning external fixation or unreamed intramedullary nailing. While external fixation is the classic "damage control" orthopedics approach, providing rapid stabilization without further violating the medullary canal, modern trauma protocols often favor early definitive fixation with an unreamed intramedullary nail if the patient is physiologically stable (clear of the "lethal triad" of coagulopathy, hypothermia, and acidosis). Intramedullary nailing provides superior biomechanical stability and avoids the complications of pin-tract infections associated with external fixators, which can complicate definitive soft tissue coverage later. Given this patient's stable systemic status (ATLS clear, isolated extremity injury), the decision was made to proceed with a dual-incision four-compartment fasciotomy followed immediately by unreamed statically locked intramedullary nailing of the right tibia.
Surgical Technique and Intervention
The patient was transported emergently to the operating room and placed supine on a radiolucent flat Jackson table. General endotracheal anesthesia was induced. Regional anesthesia (e.g., popliteal block or epidural) was strictly avoided, as it causes sympathectomy leading to vasodilation (which can alter compartment dynamics) and, more importantly, masks the clinical symptoms of compartment syndrome, preventing accurate postoperative neurological assessment. A bump was placed under the ipsilateral hip to internally rotate the leg to a neutral position. A non-sterile pneumatic tourniquet was placed on the proximal thigh but was not inflated, as tourniquet ischemia exacerbates the existing ischemic insult of compartment syndrome; it was reserved strictly for catastrophic hemorrhage control.

The surgical intervention commenced with the Mubarak and Owen dual-incision four-compartment fasciotomy technique. This approach is preferred over the single-incision perifibular approach due to its superior visualization and lower risk of iatrogenic injury to the superficial peroneal nerve.
The Anterolateral Incision
A generous longitudinal incision, approximately 15 to 20 cm in length, was centered halfway between the palpable tibial crest and the fibular shaft. The incision extended from just distal to the tibial tubercle down to the level of the supramalleolar region. Subcutaneous dissection was performed carefully to avoid undermining the skin flaps, which compromises their vascularity.
The anterior intermuscular septum, which divides the anterior and lateral compartments, was identified. This is a critical landmark. The superficial peroneal nerve must be identified and protected; it typically exits the lateral compartment fascia to become subcutaneous in the distal third of the leg, approximately 10 to 12 cm proximal to the lateral malleolus.
A transverse nick was made in the fascia of the anterior compartment, midway between the tibial crest and the intermuscular septum. Metzenbaum scissors were then used to release the anterior fascia proximally toward the tibial plateau and distally toward the extensor retinaculum. Upon release, the tibialis anterior muscle immediately bulged through the defect, confirming the high intra-compartmental pressure. The muscle appeared dusky initially but demonstrated reactive hyperemia and contractility upon stimulation, indicating viability.
Next, a separate transverse nick was made in the fascia of the lateral compartment, posterior to the intermuscular septum. The lateral fascia was released proximally toward the fibular head (taking extreme care to avoid the common peroneal nerve as it wraps around the fibular neck) and distally toward the lateral malleolus. The peroneus longus and brevis muscles were inspected and found to be viable.
The Posteromedial Incision
A second longitudinal incision of similar length was made on the posteromedial aspect of the leg. This incision was placed approximately 2 cm posterior to the palpable posteromedial border of the tibia to avoid exposing the tibial bone, which could lead to desiccation and osteonecrosis.
The saphenous vein and nerve were identified in the subcutaneous tissues and retracted anteriorly to prevent injury. The superficial posterior compartment fascia was incised, releasing the gastrocnemius and soleus muscle bellies.
The most technically demanding portion of the procedure is the release of the deep posterior compartment. The soleus muscle was retracted posteriorly, exposing the deep transverse fascia that covers the flexor digitorum longus, tibialis posterior, and flexor hallucis longus. The soleus bridge (the fascial attachment of the soleus to the posteromedial tibia) was meticulously detached sharply from the tibia. The deep fascial envelope was then incised longitudinally down the length of the leg. The neurovascular bundle (posterior tibial artery, vein, and tibial nerve) was identified and protected. The deep muscles were inspected and showed signs of severe congestion but recovered normal color within minutes of decompression.
Fracture Stabilization and Wound Management
Following complete decompression of all four compartments, attention was turned to the OTA/AO 42-B3 open tibial fracture. The 2 cm traumatic laceration was incorporated into the surgical field, thoroughly irrigated with 9 liters of normal saline, and conservatively debrided of all devitalized subcutaneous tissue and bone fragments devoid of soft tissue attachments.
A standard infrapatellar approach was utilized for intramedullary nailing. The medullary canal was opened using an awl, and a ball-tipped guide wire was passed across the fracture site under fluoroscopic guidance. To minimize further endosteal vascular disruption and prevent additional elevation of intra-compartmental pressures, the canal was not reamed. A titanium unreamed intramedullary nail of appropriate diameter and length (templating performed intraoperatively) was inserted. The fracture was reduced using manual traction and percutaneous reduction forceps. The nail was statically locked with two proximal and two distal interlocking screws. Fluoroscopy confirmed excellent coronal and sagittal alignment, with restoration of length and rotation.
The fasciotomy wounds were left open. Primary closure is absolutely contraindicated as it recreates the compartment syndrome. The wounds were managed with a negative pressure wound therapy (NPWT) device (wound VAC). Reticulated open-cell polyurethane foam was cut to size and placed directly over the exposed muscle bellies. The system was sealed with adhesive drapes and set to continuous suction at -125 mmHg. This manages the significant expected exudate, reduces local tissue edema, and promotes microvascular blood flow. The traumatic open wound was also incorporated into the NPWT dressing.
Post Operative Protocol and Rehabilitation
Postoperatively, the patient was transferred to the Surgical Intensive Care Unit (SICU) for close hemodynamic and neurovascular monitoring. The immediate postoperative period is critical for managing the systemic manifestations of ischemia-reperfusion injury, specifically rhabdomyolysis.
Aggressive intravenous fluid resuscitation with lactated Ringer's solution was initiated to maintain a high urine output (target > 100 mL/hour) to flush myoglobin through the renal tubules and prevent acute tubular necrosis. Serum creatine kinase (CK) levels, basic metabolic panels (focusing on potassium and creatinine), and urine myoglobin were monitored every 6 hours. Alkalinization of the urine with sodium bicarbonate was held in reserve but ultimately not required, as the patient's CK levels peaked at 8,500 U/L at 24 hours post-op and subsequently trended downward, with normal renal function maintained throughout.
The injured extremity was elevated strictly at the level of the heart. Elevation above the heart decreases the local arterial perfusion pressure, which can exacerbate ischemia in borderline perfused tissues, while dependent positioning increases venous congestion and edema.
A planned "second look" surgery was scheduled for 48 to 72 hours post-index procedure. During this return to the operating room, the NPWT dressings were removed, and the muscle beds were meticulously re-evaluated for viability. Any delayed myonecrosis would require radical debridement. In this patient, all muscle bellies remained viable and contractile.
Closure of fasciotomy wounds is highly variable and depends entirely on the resolution of soft tissue edema. In this case, the anterolateral wound could not be closed primarily due to persistent tension. A vessel loop "shoelace" technique (Dermatotraction) was applied over the anterolateral wound to provide continuous, gentle tension on the skin margins, facilitating delayed primary closure. The posteromedial wound was partially closed, and a new NPWT dressing was applied.
By postoperative day 7, the edema had subsided sufficiently to allow for definitive soft tissue coverage. The patient was taken to the operating room for a third time. The posteromedial wound was closed primarily. The anterolateral wound, which still exhibited a 4 cm gap, was covered using a split-thickness skin graft (STSG) harvested from the ipsilateral anterior thigh. The graft was meshed 1.5:1 and secured with surgical staples, followed by a non-adherent dressing and a temporary NPWT dressing over the graft to promote adherence and prevent seroma formation.
Rehabilitation commenced immediately on postoperative day 1 with passive and active-assisted range of motion of the knee and ankle to prevent arthrofibrosis and tendon tethering. Weight-bearing status was restricted to touch-down weight-bearing (TDWB) on the right lower extremity for the first 6 weeks due to the comminuted nature of the diaphyseal fracture (OTA 42-B3) and the reliance on an unreamed nail construct. Progressive weight-bearing was advanced based on radiographic evidence of callus formation at the 6-week and 12-week follow-up intervals.
Clinical Pearls and Pitfalls
The management of acute compartment syndrome in the setting of open tibial fractures is fraught with diagnostic and therapeutic challenges. Several critical pearls and pitfalls must be highlighted for the orthopedic surgeon.
Pearl: Open Fractures Do Not Prevent Compartment Syndrome.
A common and dangerous misconception is that the fascial disruption associated with an open fracture provides auto-decompression of the compartment. As demonstrated in this case, a
You Might Also Like