Osteochondritis Dissecans: Comprehensive Surgical Management
Key Takeaway
Osteochondritis dissecans (OCD) is a focal idiopathic alteration of subchondral bone with risk for instability and disruption of adjacent articular cartilage. It is the most common source of intra-articular loose bodies in the knee. Management depends on physeal status and lesion stability, ranging from nonoperative immobilization in juveniles to surgical fixation, autologous chondrocyte implantation, or osteochondral grafting in adults.
Introduction and Pathophysiology
Osteochondritis dissecans (OCD) represents a focal, idiopathic alteration of subchondral bone with a subsequent risk for instability and disruption of the adjacent articular cartilage. It is internationally recognized as the most common source of loose bodies within the knee joint. While other etiologies for intra-articular loose bodies exist—namely synovial chondromatosis, osteophytes, fractured articular surfaces, and damaged menisci—OCD remains the primary differential in the adolescent and young adult populations presenting with mechanical joint symptoms.
The exact pathophysiology of OCD remains multifactorial and heavily debated, though it is generally accepted to involve a combination of repetitive microtrauma, localized ischemia, and genetic predisposition. In osteochondritis dissecans, a demarcated area of subchondral bone becomes necrotic. Consequently, degenerative changes usually occur in the articular cartilage overlying this ischemic zone. During the natural course of the disease, unless interrupted by surgical intervention, targeted nonoperative treatment, or spontaneous healing, the necrotic bone and its overlying cartilage gradually separate from the adjacent healthy osseous and chondral bed. Together, this separated fragment becomes a free-floating loose body within the joint space, leading to mechanical locking, catching, and accelerated osteoarthritis.
Clinical Pearl: The critical prognostic factor in OCD is the patency of the distal femoral physis. Juvenile Osteochondritis Dissecans (JOCD) occurs in patients with open physes and carries an excellent prognosis for spontaneous healing. Adult Osteochondritis Dissecans (AOCD) occurs after physeal closure, is often driven by a vascular phenomenon, and rarely heals without surgical intervention.
Epidemiological Profile and Anatomic Distribution
While OCD can manifest in any synovial joint—including the elbow (capitellum), ankle (talar dome), shoulder, and hip—it occurs most frequently in the knee joint. The condition exhibits a strong male predominance and typically presents during adolescence, coinciding with periods of rapid skeletal growth and increased participation in high-impact athletics. Furthermore, osteochondritis dissecans is bilateral in 20% to 30% of patients, mandating a thorough evaluation of the contralateral asymptomatic joint during the initial diagnostic workup.
Topographic Distribution in the Knee
The anatomic distribution of OCD lesions in the knee is highly predictable and follows specific biomechanical wear patterns.

According to classical topographical mapping by Hefti et al. and Aichroth, the distribution is as follows:
* Medial Femoral Condyle (77% - 85%): The vast majority of lesions are located on the lateral aspect of the medial femoral condyle, directly adjacent to the attachment of the posterior cruciate ligament (PCL). This is often referred to as the "classic" location. Biomechanically, this area is subjected to repetitive impingement from the tibial spine during internal rotation of the tibia.
* Lateral Femoral Condyle (15% - 17%): Lesions here typically occur on the inferocentral or posterolateral aspect of the condyle. These lesions are often larger, carry a higher risk of instability, and are frequently associated with discoid lateral menisci.
* Patella (5% - 6%): Occurring primarily in the inferomedial quadrant, patellar OCD is rare but presents significant reconstructive challenges due to the high shear forces of the patellofemoral articulation.
Clinical Presentation and Diagnostic Imaging
Patients typically present with an insidious onset of poorly localized, activity-related knee pain. As the lesion progresses to instability, mechanical symptoms such as catching, locking, and giving way become prominent. A joint effusion is frequently present in unstable lesions. Wilson's test (pain with knee extension while the tibia is internally rotated, relieved by external rotation) may be positive in classic medial condyle lesions, though its sensitivity is limited.
Imaging Modalities
- Radiographs: A standard knee series must include weight-bearing anteroposterior (AP), lateral, skyline, and notch (tunnel) views. The notch view is critical, as classic lesions on the posterolateral aspect of the medial femoral condyle are often obscured on standard AP projections.
- Magnetic Resonance Imaging (MRI): MRI is the gold standard for evaluating lesion size, depth, and, most importantly, stability. Signs of instability on MRI include:
- High T2 signal intensity rim surrounding the lesion (fluid tracking behind the fragment).
- Cysts measuring >5 mm beneath the lesion.
- A focal defect in the overlying articular cartilage.
- A high T2 signal line breaching the subchondral bone plate.
Nonoperative Management
Nonoperative management is the first-line treatment for stable JOCD lesions in patients with open physes. The goal is to promote spontaneous revascularization and ossification of the necrotic fragment by eliminating repetitive shear and compressive forces.
Protocols and Outcomes
Treatment consists of activity modification, protected weight-bearing, and occasionally immobilization. In highly non-compliant patients or those with highly symptomatic lesions, a period of casting or bracing may be utilized.
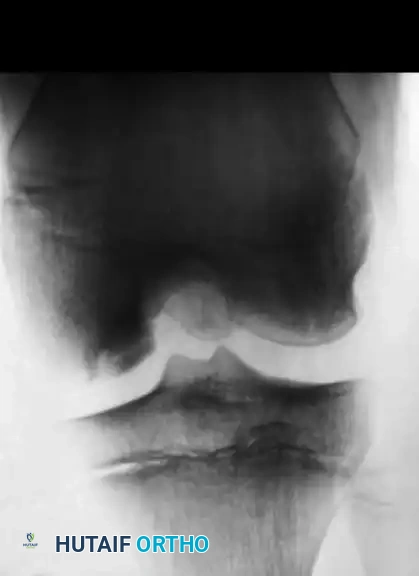
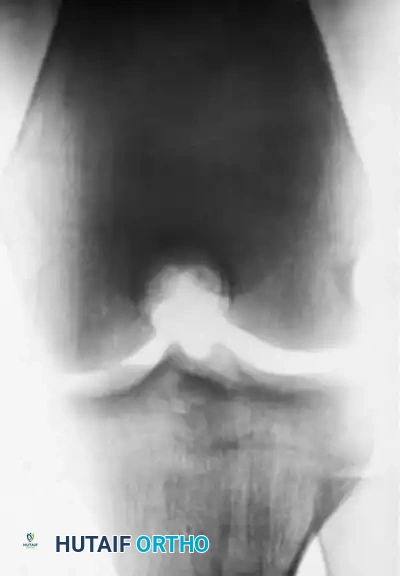
As demonstrated in the clinical case above, an adolescent with an open physis and a stable lesion was treated nonoperatively in a cast for 9 months. Several years later, complete radiographic healing is apparent, and the knee is entirely asymptomatic. However, prolonged immobilization must be balanced against the risks of muscle atrophy and joint stiffness. Contemporary protocols favor hinged knee braces and strict cessation of sports rather than rigid casting.
Surgical Indications and Preoperative Planning
Surgical intervention is indicated under the following circumstances:
1. Failure of nonoperative management in JOCD (typically after 6 months of compliant conservative care).
2. Evidence of lesion instability (clinical, radiographic, or MRI).
3. Presence of an intra-articular loose body.
4. AOCD (closed physes), as these lesions have a negligible rate of spontaneous healing.
Preoperative planning must account for the patient's skeletal maturity, lesion size, anatomic location, and the presence of any coexisting pathology. With any of these procedures, malalignment (e.g., varus or valgus deformity), ligament instability (e.g., ACL deficiency), patellar instability, or meniscal pathology must be addressed concurrently. Failure to correct the underlying biomechanical environment will inevitably lead to failure of the cartilage restoration procedure.
Surgical Techniques and Step-by-Step Approaches
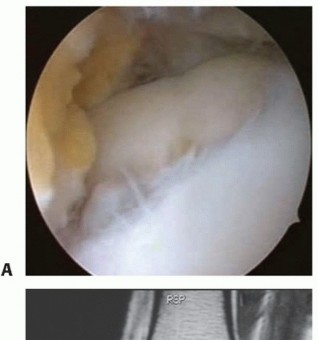
1. Diagnostic Arthroscopy and Lesion Probing
Every surgical intervention begins with a comprehensive diagnostic arthroscopy. The lesion is visualized and palpated with an arthroscopic probe to assess stability according to the International Cartilage Repair Society (ICRS) classification:
* Grade I: Stable, continuous but softened cartilage.
* Grade II: Partial discontinuity, stable on probing.
* Grade III: Complete discontinuity, "dead in situ" (not dislocated).
* Grade IV: Empty defect, loose body within the joint.
2. Subchondral Drilling (For Intact, Stable Lesions)
For stable lesions that have failed conservative care, drilling is performed to breach the sclerotic subchondral bone, creating channels for marrow-derived mesenchymal stem cells and angiogenic factors to promote healing.
* Transarticular Drilling: The drill passes through the articular cartilage into the subchondral bone. It is technically easier but violates the intact cartilage surface.
* Retroarticular Drilling: Under fluoroscopic guidance, a drill is advanced from the extra-articular cortex into the lesion, sparing the articular cartilage. This is preferred for large, pristine chondral surfaces but is technically demanding.
3. Internal Fixation of Unstable Lesions
For ICRS Grade II and III lesions, or salvageable Grade IV lesions, internal fixation is the treatment of choice. The goal is to achieve rigid compression of the fragment into its anatomic bed to facilitate osseous union.
Surgical Steps for Fixation:
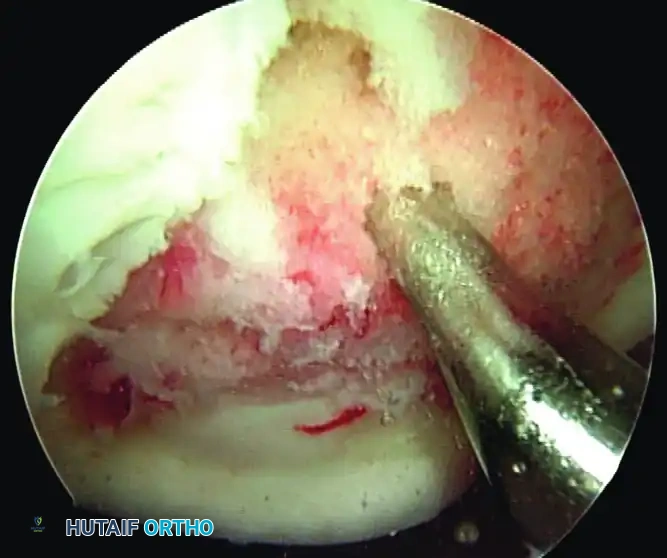
1. Debridement: The fragment is hinged open (if attached) or removed temporarily. The fibrous tissue at the base of the crater and on the undersurface of the fragment is meticulously debrided down to bleeding subchondral bone using a curette and motorized shaver.
2. Bone Grafting: If a volumetric bone void exists, autologous cancellous bone graft (often harvested from the proximal tibia or distal femur) is packed into the base of the defect.
3. Reduction and Fixation: The fragment is anatomically reduced. Fixation can be achieved using metallic headless compression screws, bioabsorbable pins/darts, or multiple Kirschner wires (K-wires).
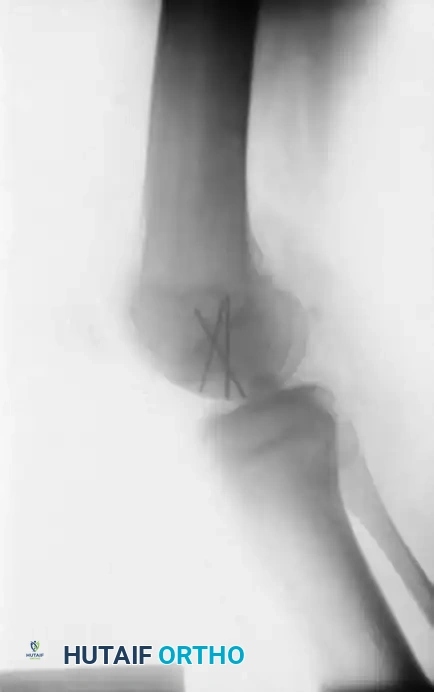
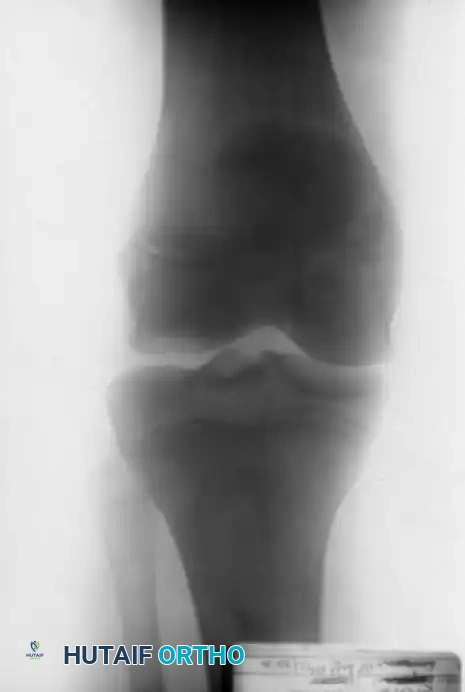
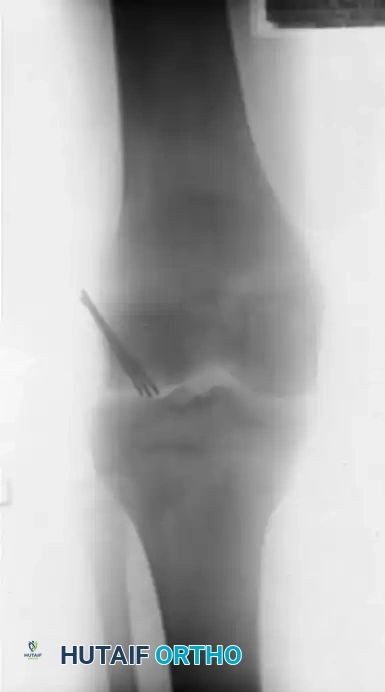
The radiographic series above illustrates a 15-year-old boy with a large osteochondritis dissecans lesion involving the weight-bearing portion of the lateral femoral condyle. The fragment was successfully reduced and internally fixed with multiple Kirschner wires. While K-wires provide excellent temporary fixation, they require subsequent removal. Modern practices frequently utilize bioabsorbable compression screws to avoid a secondary hardware removal surgery, though metallic screws remain the gold standard for maximal compression.
Surgical Warning: When using bioabsorbable implants, ensure the heads are countersunk at least 1-2 mm below the articular surface to prevent opposing "kissing" lesions on the tibial plateau during the degradation phase.
4. Cartilage Restoration Techniques (For Unsalvageable Lesions)
When the osteochondral fragment is fragmented, severely deformed, or missing, the defect must be treated with cartilage restoration techniques.
- Microfracture: Indicated for small defects (< 2 cm²). An awl is used to create holes in the subchondral plate, allowing marrow elements to form a fibrocartilage (Type I collagen) clot. While early results are good, fibrocartilage lacks the durability of hyaline cartilage.
- Osteochondral Autograft Transfer System (OATS): Indicated for medium defects (1-3 cm²). Cylindrical osteochondral plugs are harvested from non-weight-bearing areas (e.g., the intercondylar notch or periphery of the trochlea) and press-fit into the defect. This provides mature, hyaline cartilage but is limited by donor site morbidity.
- Osteochondral Allograft Transplantation: Indicated for large defects (> 3 cm²). Fresh allograft plugs are size-matched and implanted. This avoids donor site morbidity but carries logistical challenges regarding graft availability and cell viability.
Advanced Biologics: Autologous Chondrocyte Implantation (ACI)
For large, full-thickness defects where the subchondral bone is relatively intact, Autologous Chondrocyte Implantation (ACI) represents a sophisticated, cell-based restorative option.
ACI is a two-stage procedure:
1. Stage I (Harvest): An arthroscopic biopsy of healthy cartilage is taken from a minor weight-bearing area. The chondrocytes are enzymatically isolated and expanded in vitro over 4 to 6 weeks.
2. Stage II (Implantation): Through an arthrotomy, the defect is debrided to stable vertical margins. A periosteal patch (or, more commonly in modern generations, a Type I/III collagen membrane) is sutured over the defect and sealed with fibrin glue. The cultured chondrocytes are then injected beneath the membrane.
Challenges and Evolving Techniques:
Regardless of the specific technique used, a major disadvantage of autologous chondrocyte implantation is the prolonged postoperative rehabilitation that requires strict compliance of the patient with weight-bearing and activity restrictions. Furthermore, there is a need for the reduction of the high costs associated with the mechanical and logistical complexities of current methods of ACI.
To address these limitations, Matrix-Induced Autologous Chondrocyte Implantation (MACI) has been developed, where chondrocytes are seeded directly onto a collagen scaffold prior to implantation, eliminating the need for a liquid injection and a separate membrane cover. All of these techniques currently are investigational in some regions but may improve the results and cost-effectiveness of autologous chondrocyte implantation in the future.
One study, however, found that activity levels in patients with characterized chondrocyte implantation were comparable to those of patients with microfracture at 2 years after surgery. Interestingly, in both groups, a lack of low-load activities after surgery adversely affected functional outcomes, highlighting the necessity of controlled mechanical stimulus for optimal cartilage maturation.
Emerging Biologic Therapies and Future Directions
Another strategy currently being developed is the manipulation of the repair process at the cellular and molecular levels by the transfer of new genetic information to cells that contribute to the healing process (“gene therapy”). Research is heavily focused on finding the most efficient viral or non-viral vector for delivering this genetic information and identifying the candidate genes (such as TGF-β, BMPs, or IGF-1) most likely to improve cartilage repair and regeneration.
Cell-based therapy also is being investigated using mesenchymal stem cells (MSCs) derived from bone marrow, adipose tissue, or synovial fluid. The use of cell-based therapies allows the versatility of using scaffolds, growth factors, recombinant proteins, and gene therapy in various combinations. These "tissue engineering" triads aim to recreate the complex zonal architecture of native hyaline cartilage and the underlying subchondral bone plate simultaneously.
Postoperative Rehabilitation Protocols
Rehabilitation following OCD surgery is highly dependent on the procedure performed, the size of the lesion, and the security of the fixation.
General Protocol for Internal Fixation or ACI:
* Phase I (Weeks 0-6): Protection and Early Motion. Strict non-weight-bearing or touch-down weight-bearing. Continuous Passive Motion (CPM) machines are frequently utilized for 6-8 hours daily to promote cartilage nutrition and prevent adhesions.
* Phase II (Weeks 6-12): Progressive Weight-Bearing. Gradual progression to full weight-bearing. Closed kinetic chain exercises are initiated.
* Phase III (Months 3-6): Strengthening. Focus on quadriceps and hamstring hypertrophy, core stability, and proprioception. Avoid high-impact activities.
* Phase IV (Months 6-12+): Return to Sport. Progression to sport-specific drills. Return to play is permitted only when the patient has full, painless range of motion, >90% limb symmetry index on functional testing, and radiographic evidence of complete osseous integration.
Conclusion
Osteochondritis dissecans remains a complex pathology requiring a nuanced, patient-specific approach. While juvenile patients with stable lesions often respond well to conservative management, adult patients and those with unstable fragments demand precise surgical intervention. From arthroscopic drilling and internal fixation to advanced cellular therapies like ACI and emerging gene therapies, the orthopedic surgeon must possess a comprehensive armamentarium to restore joint congruity, alleviate symptoms, and prevent the early onset of degenerative joint disease. Strict adherence to biomechanical principles and rigorous postoperative rehabilitation are paramount to achieving successful, long-term outcomes.
You Might Also Like