Achilles Tendon Rupture: Diagnosis & Surgical Management
Key Takeaway
Achilles tendon ruptures are common orthopedic injuries, particularly in middle-aged athletes. Management requires a nuanced understanding of tendon biomechanics, hypovascular zones, and patient-specific risk factors. This guide provides an evidence-based approach to clinical evaluation, including the Thompson and Matles tests, alongside comprehensive protocols for both nonoperative functional rehabilitation and advanced surgical repair techniques, ensuring optimal functional recovery and minimized rerupture rates.
RUPTURE OF MUSCLES AND TENDONS
EPIDEMIOLOGY AND DEMOGRAPHICS OF ACHILLES TENDON RUPTURE
Injuries of the Achilles tendon represent a significant and growing burden in orthopedic practice. They are relatively common in middle-aged athletes, and the epidemiological frequency of these injuries has seen a dramatic increase over the past few decades—rising from 2 per 100,000 in 1986 to approximately 10 per 100,000 in 2010. This surge is largely attributed to the "weekend warrior" phenomenon, where an aging population remains actively engaged in high-demand recreational sports for longer periods.
Tendinopathy spectrum disorders (tendinitis, tendinosis, and peritendinitis) account for up to 11% of lower extremity complaints at specialized sports and runners’ clinics. Furthermore, Achilles tendon ruptures are currently estimated to be the third most frequent tendon rupture in the human body. Clinical studies suggest that Achilles tendinopathy is present in 24% of competitive athletes and up to 50% of competitive runners, with frank tendon ruptures occurring in approximately 8% of this athletic cohort.
The peak age for an acute Achilles tendon rupture in both men and women is between 30 and 40 years. Patients who sustain an acute rupture must be counseled on the significant risk of future contralateral injury. Longitudinal surveys of patients with acute tendon ruptures demonstrate that at a 4-year follow-up, 6% sustain a rupture on the contralateral side. Statistically, patients with an acute Achilles tendon rupture possess a nearly 200-fold increased risk of a contralateral tendon rupture compared to the general population.
INTRINSIC AND EXTRINSIC RISK FACTORS
The etiology of Achilles tendon rupture is multifactorial, requiring a comprehensive understanding of both intrinsic anatomical predispositions and extrinsic environmental factors.
Intrinsic Factors (Anatomical Predisposition & Biomechanical Failure):
* Subtalar hyperpronation
* Excessive rearfoot/forefoot varus or valgus alignment
* Increased femoral anteversion
* Limb-length discrepancy
* Muscle weakness or gastrocnemius-soleus imbalance
* High body mass index (BMI)
* Aging (associated with decreased collagen turnover and increased stiffness)
Extrinsic Factors (Training Errors & Environmental Influences):
* Excessive running duration, frequency, and intensity
* Unfamiliar or unyielding running surfaces
* Pharmacological agents: Fluoroquinolone antibiotics (which upregulate matrix metalloproteinases, leading to collagen degradation) and local or systemic corticosteroids.
Clinical Pearl: Always review a patient's medication history. A recent course of fluoroquinolones (e.g., ciprofloxacin) combined with a sudden return to high-impact activity is a classic historical red flag for acute tendon rupture.
ANATOMY, BIOMECHANICS, AND PATHOPHYSIOLOGY
The pathophysiology of an Achilles tendon rupture is intimately related to its unique vascular anatomy. Angiographic studies have consistently demonstrated a relatively hypovascular "watershed" area located 2 to 6 cm proximal to the tendon's insertion into the calcaneus. The major blood supply to the tendon is delivered through its mesotendon, with the richest vascular network arising from the anterior mesentery. As a patient ages, this anterior mesenteric supply becomes progressively reduced.
Age-dependent changes in collagen crosslinking result in increased tendon stiffness and a critical loss of viscoelasticity, predisposing the tissue to mechanical failure. The prevailing pathophysiological theory posits that repetitive microtrauma to this hypovascular zone initiates an inflammatory reparative process. Because of the compromised blood supply, the tenocytes are unable to keep pace with the microstructural damage, leading to degenerative attrition (tendinosis).
Another biomechanical theory suggests the failure of Golgi tendon organ inhibiting mechanisms at the musculotendinous unit as a result of neuromuscular fatigue. This leads to an uninhibited eccentric overload. Ultimately, the rupture is the culmination of a relatively hypovascular area subjected to repetitive microtrauma, resulting in a weakened, degenerative tendon that fails catastrophically under a sudden mechanical overload.
The most common mechanisms of injury include:
1. Pushing off with the weight-bearing forefoot while simultaneously extending the knee (e.g., initiating a sprint or jump).
2. Sudden, unexpected dorsiflexion of the ankle (e.g., stepping into a hole).
3. Violent dorsiflexion of a plantar-flexed foot (e.g., falling from a height).
4. Direct blunt trauma to a contracted tendon or sharp laceration.
CLINICAL EVALUATION AND DIAGNOSTIC MANEUVERS
The diagnosis of an acute Achilles tendon rupture is primarily clinical and can be reliably made without advanced imaging in the majority of cases. The classic triad of physical examination findings includes:
1. A palpable tendon defect (gap) in the watershed area.
2. The inability to perform a single-leg toe raise on the affected side.
3. A positive Thompson "squeeze test."
The Thompson Test: Performed with the patient prone and the feet hanging off the edge of the examination table. The examiner squeezes the fleshy muscle belly of the calf (gastrocnemius-soleus complex) just distal to its maximal girth. In a normal, intact tendon, this maneuver mechanically pulls the Achilles, resulting in passive plantar flexion of the ankle. The test is positive for a complete rupture when squeezing the calf elicits no plantar flexion.
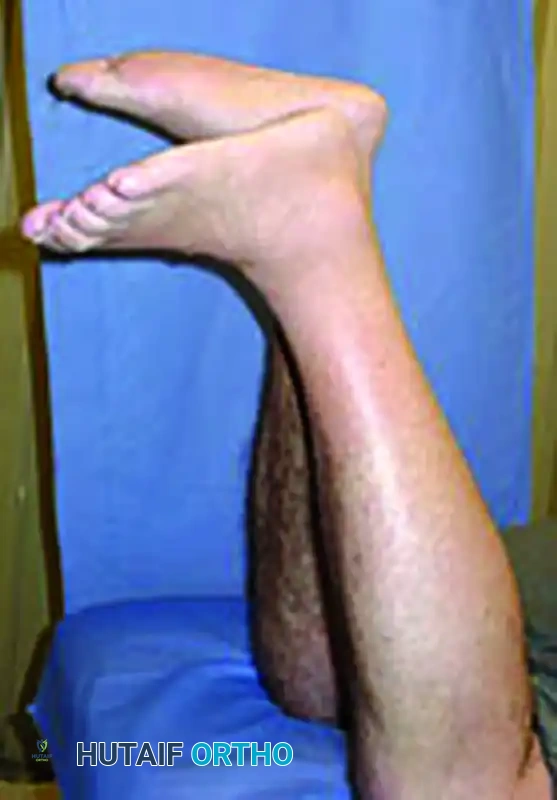
The Matles Test: The patient is placed prone with the lower legs extending off the bed and is asked to actively flex the knees to 90 degrees. Active flexion of the knee shortens the gastrocnemius, which normally causes the foot to rest in slight plantar flexion. If the foot falls into neutral or slight dorsiflexion compared to the contralateral side, the Achilles tendon is likely ruptured.

Figure: Matles test for Achilles tendon rupture. If the foot falls into neutral or slight dorsiflexion with this maneuver, the Achilles tendon is likely ruptured.
The O’Brien Needle Test: A 25-gauge needle is placed percutaneously in the midline of the proximal tendon belly. The foot is then put through a passive range of motion. If the tendon is intact, the needle will tilt in the opposite direction of the foot's motion. If ruptured, the needle remains stationary, indicating a lack of continuity.
EVIDENCE-BASED TREATMENT ALGORITHMS
The decision to treat acute Achilles tendon ruptures nonoperatively versus operatively remains one of the most debated topics in orthopedic sports medicine. The American Academy of Orthopaedic Surgeons (AAOS) has published clinical practice guidelines to navigate this controversy.
AAOS Recommendations for Achilles Tendon Ruptures:
* Consensus: Diagnosis should rely on two or more clinical tests (Thompson test, decreased plantar flexion strength, palpable gap, increased passive dorsiflexion). Operative treatment should be approached with extreme caution in patients with diabetes, neuropathy, immunocompromised states, age > 65, tobacco use, obesity (BMI > 30), or peripheral vascular disease.
* Moderate Strength: Early (≤ 2 weeks) postoperative protected weight-bearing and the use of a protective functional device that allows mobilization by 2 to 4 weeks postoperatively.
* Weak Strength: Nonoperative treatment is a viable option. Open, limited open, and percutaneous techniques are all acceptable operative options.
* Inconclusive: Routine use of MRI or ultrasound for acute diagnosis; use of immediate functional bracing as nonoperative treatment; use of biologic adjuncts (PRP, allografts).
Nonoperative Management
Current nonoperative treatment has evolved significantly from prolonged cast immobilization. Modern protocols utilize functional bracing and aggressive rehabilitation that allows early, protected motion.
Ultrasound-based criteria for selecting nonoperative treatment include:
1. A tendon gap of less than 5 mm with maximal plantar flexion.
2. A gap of less than 10 mm with the foot in a neutral position.
3. More than 75% tendon apposition with the foot in 20 degrees of plantar flexion.
Historically, operative treatment boasted a significantly lower rerupture rate compared to nonoperative management, albeit with a higher risk of wound complications and infections. However, recent high-level randomized controlled trials demonstrate that when nonoperative treatment is paired with early functional rehabilitation, the rerupture rates (approximately 2.4% for bracing vs. 3.5% for operative) and functional outcomes are statistically similar.
Operative Management
Despite the success of functional bracing, operative repair is often preferred for high-demand athletes seeking to maximize push-off strength and minimize elongation of the musculotendinous unit.
Surgical Warning: Regardless of the treatment modality chosen, 30% to 50% of patients will experience long-term functional impairments, including mild pain, reduced peak torque strength, and decreased range of motion at 1 to 5 years post-injury. A study of NFL players revealed that only 64% returned to play, and those who did exhibited significant decreases in power ratings.
SURGICAL TECHNIQUES FOR ACUTE ACHILLES RUPTURE
A variety of techniques exist for the repair of acute ruptures, ranging from traditional open repair to minimally invasive percutaneous techniques.
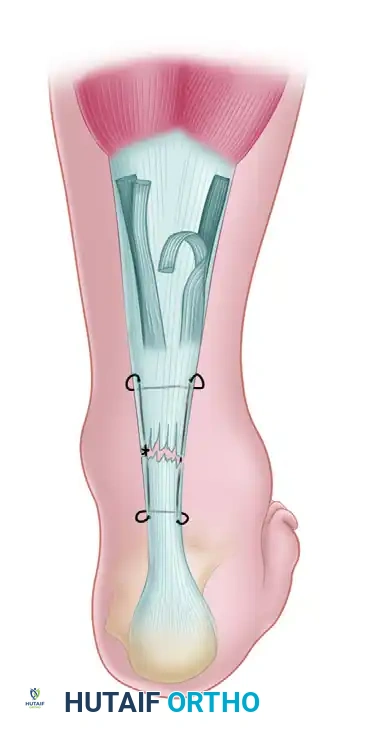
Open Repair Principles:
1. Positioning: The patient is placed prone. A thigh tourniquet is applied.
2. Incision: A longitudinal incision is made slightly posteromedial to the tendon to avoid injury to the sural nerve, which crosses from medial to lateral in the proximal calf.
3. Paratenon Preservation: The paratenon is meticulously incised longitudinally and preserved for later closure. This is critical, as the paratenon provides the extrinsic blood supply necessary for healing and prevents skin tethering.
4. Tendon Debridement: The ruptured ends (often resembling a "mop-end") are minimally debrided to interdigitating healthy tissue.
5. Suture Configuration: Heavy, non-absorbable or slowly absorbable sutures (e.g., #2 or #5 FiberWire/Ethibond) are used. Biomechanical cadaveric studies show no significant difference in ultimate load to failure between Krackow, Bunnell, and Kessler techniques, provided a double-suture technique is utilized. The Krackow locking-loop technique is highly favored for its resistance to pull-out.
6. Closure: The paratenon is closed meticulously with a fine absorbable suture (e.g., 3-0 Vicryl) to restore the gliding layer.
Augmentation and Biologics:
Historically, repairs were augmented with fascial "turn-down" flaps, plantaris tendon weaves, or peroneus brevis transfers. Contemporary literature strongly discourages routine augmentation for acute ruptures, as it requires larger incisions, increases operative time, and provides no proven clinical or biomechanical benefit over a robust end-to-end repair.
The use of Platelet-Rich Plasma (PRP) remains highly controversial. While animal models suggest PRP stimulates tenocyte proliferation, clinical trials in humans have yielded conflicting results. Some studies report faster recovery, while others suggest PRP may actually have a deleterious effect on tendon healing. Until standardized preparation protocols are established, PRP cannot be routinely recommended.
SURGICAL MANAGEMENT OF CHRONIC EXERTIONAL COMPARTMENT SYNDROME
While Achilles tendon pathology dominates the posterior leg, chronic exertional compartment syndrome (CECS) of the deep posterior compartment is another significant cause of posterior leg pain in athletes that requires precise surgical intervention when conservative measures fail.
Rorabek Two-Incision Release of the Deep Posterior Compartment
The deep posterior compartment is notoriously difficult to decompress adequately due to its deep anatomical location and proximity to critical neurovascular structures. The Rorabek two-incision technique provides safe and thorough decompression.
Surgical Steps:
- Incision and Superficial Dissection:
Two vertical incisions are made along the medial border of the tibia. The saphenous vein and nerve must be carefully identified and retracted anteriorly to prevent iatrogenic neuroma formation or venous bleeding.
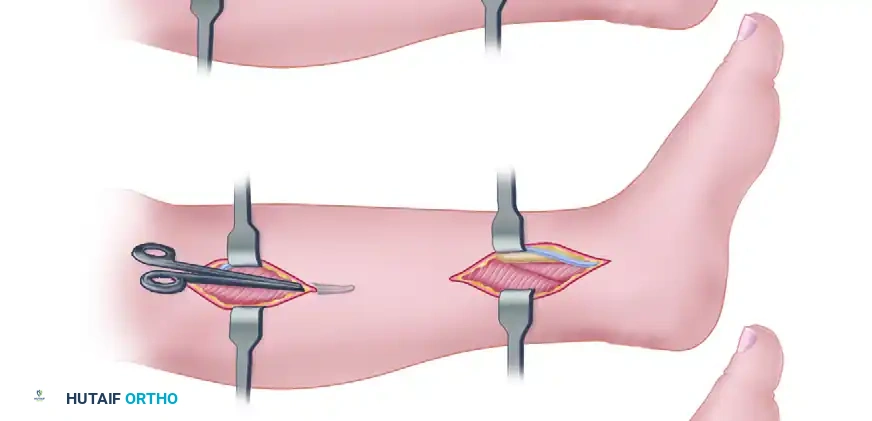
Figure A: Rorabek two-incision release. Two vertical incisions are made; the saphenous vein is identified and retracted anteriorly.
- Superficial Compartment Release:
The fascia overlying the superficial posterior compartment (gastrocnemius and soleus) is identified, entered, and released longitudinally.
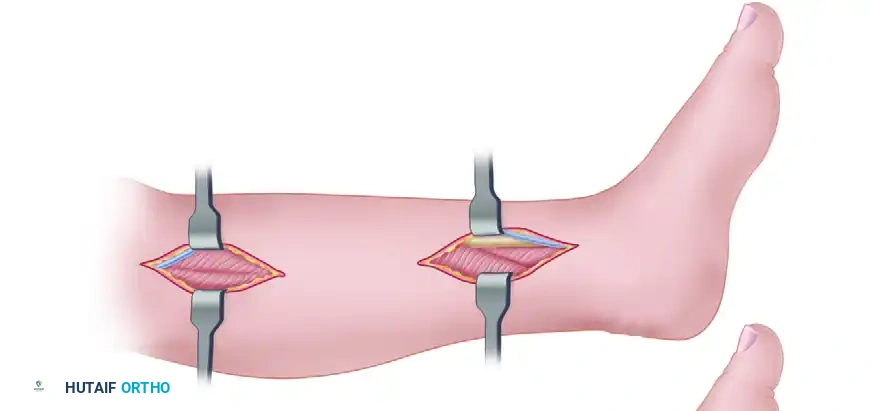
Figure B: The superficial compartment is entered and released.
-
Deep Posterior Compartment Decompression:
The deep fascia is then identified. The surgeon must locate the neurovascular bundle and the posterior tibial tendon. The fascia overlying the posterior tibial tendon is incised both proximally and distally.Surgical Pitfall: The posterior tibial tendon is the anatomical key to deep posterior compartment decompression. It is frequently constricted proximally between the two origins of the flexor hallucis longus. The surgeon must manually enlarge the opening between these two structures to ensure all constricting fascial bands are released.
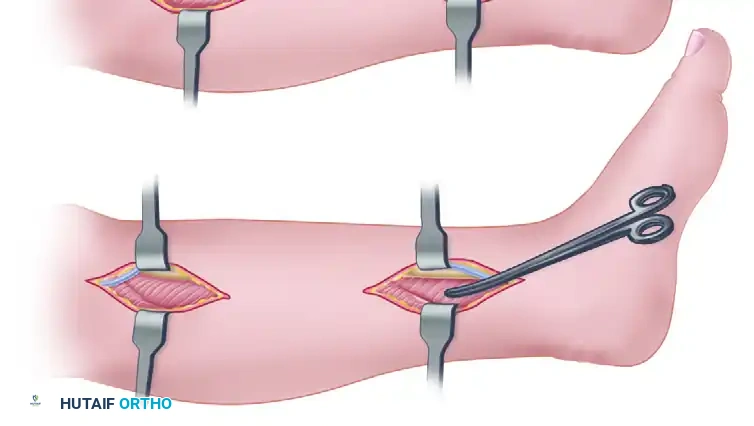
Figure C: The deep fascia is incised, and the deep posterior compartment is fully released.
- Hemostasis and Closure:
The tourniquet is released to obtain meticulous hemostasis. The skin is closed over a suction drain to minimize the risk of postoperative hematoma formation, which could precipitate an acute compartment syndrome.
Postoperative Care for Compartment Release:
Early range-of-motion exercises of the ankle and toes are encouraged immediately in the recovery room. Weight-bearing to tolerance on crutches is permitted the day after surgery. Crutches are discarded once the patient can ambulate without an antalgic gait. Light jogging may be initiated at 2 to 3 weeks postoperatively, provided that swelling and focal tenderness have completely resolved.
📚 Medical References
You Might Also Like