Flexor Hallucis Longus (FHL) Tendon Transfer for Chronic Achilles Rupture: Comprehensive Review
Key Takeaway
Flexor Hallucis Longus (FHL) transfer is a gold standard surgical technique for chronic Achilles tendon ruptures, particularly for large defects. The FHL tendon is harvested, often proximal to the Knot of Henry, and used to reconstruct the Achilles, restoring plantarflexion strength. This method leverages the FHL's expendability and anatomical benefits to bridge tendon gaps.
Introduction and Epidemiology
Chronic Achilles tendon rupture represents a significant clinical challenge, distinct from acute ruptures in its pathology, treatment complexity, and often, prognosis. While acute ruptures are generally amenable to direct repair, chronic ruptures, defined typically as those presenting more than 4-6 weeks after injury, are characterized by substantial tendon gap formation, significant scar tissue, tendon retraction, and muscle atrophy. These factors preclude primary end-to-end repair without excessive tension, necessitating reconstructive techniques to bridge the defect and restore the functional integrity of the triceps surae complex.
The incidence of Achilles tendon ruptures has been reported to be increasing, particularly among middle-aged populations participating in recreational sports. While the majority present acutely, a notable percentage of ruptures are either initially misdiagnosed, neglected, or suffer re-rupture after initial treatment, leading to a chronic presentation. Misdiagnosis rates for acute Achilles ruptures can exceed 20% in emergency department settings, often masquerading as ankle sprains due to the preservation of weak plantarflexion through accessory muscles (tibialis posterior, flexor hallucis longus, flexor digitorum longus, and peroneals). Such chronic cases result in persistent pain, weakness in plantarflexion, inability to perform a single heel raise, an altered gait cycle lacking terminal stance push-off, and significant functional disability.
Reconstructive options for chronic Achilles ruptures range from local tendon transfers (e.g., gastrocnemius-soleus V-Y advancement, turndown flaps) to synthetic grafts or allografts. However, for large defects exceeding 3-6 cm, autologous tendon transfer, specifically utilizing the Flexor Hallucis Longus (FHL) tendon, has emerged as a gold standard. The FHL offers several distinct advantages: it is a robust, expendable tendon, anatomically proximal to the Achilles insertion, and its harvest typically results in minimal functional deficit due to synergistic action with the Flexor Digitorum Longus (FDL). Furthermore, the FHL muscle belly is low-lying, providing a highly vascularized tissue bed to the relatively avascular posterior ankle, thereby enhancing graft incorporation and wound healing. This extensive review will delve into the surgical considerations, detailed technique, and evidence supporting FHL transfer for the management of chronic Achilles rupture.
Surgical Anatomy and Biomechanics
A thorough understanding of the regional anatomy is paramount for successful FHL transfer and minimization of iatrogenic complications.
Achilles Tendon Anatomy
The Achilles tendon, the strongest and thickest tendon in the human body, is formed by the confluence of the gastrocnemius and soleus muscles (triceps surae). It inserts onto the posterior aspect of the calcaneal tuberosity. The tendon typically undergoes a 90-degree twist, with fibers from the medial gastrocnemius rotating laterally and those from the lateral gastrocnemius rotating medially before insertion. This anatomical twist is important as it influences biomechanics and potential rerupture patterns.
The Achilles tendon possesses a relatively tenuous blood supply, particularly in its watershed zone (2-6 cm proximal to its insertion), which is considered the most common site for degenerative rupture. Vascularization is primarily derived from the posterior tibial artery through the paratenon and to a lesser extent from the peroneal artery. Motor innervation to the gastrocnemius and soleus is provided by the tibial nerve (S1, S2). Sensory innervation around the tendon is predominantly from the sural nerve, which runs superficially along the lateral border of the Achilles tendon. Iatrogenic injury to the sural nerve is a common complication during surgical approaches to the posterior ankle.
Flexor Hallucis Longus Anatomy
The FHL muscle originates from the posterior surface of the fibula and the interosseous membrane. Its tendon is robust, providing strong plantarflexion of the hallux and contributing secondary plantarflexion force to the ankle joint.
The FHL tendon runs in a fibro-osseous groove on the posterior aspect of the talus, flanked by the medial and lateral tubercles, then courses beneath the sustentaculum tali before passing into the plantar aspect of the foot. It crosses dorsal to the FDL tendon at the Master Knot of Henry, where there are often interconnecting tendinous slips, before inserting on the plantar aspect of the distal phalanx of the great toe.
In the posterior compartment of the leg, the FHL lies deep to the soleus and the Achilles tendon. It is immediately adjacent to the neurovascular bundle. Specifically, the posterior tibial artery and tibial nerve lie medial to the FHL muscle belly in the deep posterior compartment. The close proximity of the FHL to the Achilles tendon makes it an ideal local transfer option.

Biomechanical Considerations
Biomechanically, the FHL is the second strongest plantarflexor of the ankle, following the triceps surae, generating approximately 30% of the force of the intact Achilles. Crucially, the FHL is an "in-phase" muscle, meaning it naturally fires synchronously with the gastrocnemius and soleus during the push-off phase of the gait cycle. This in-phase firing eliminates the need for extensive postoperative neuromuscular retraining, a significant advantage over out-of-phase transfers. The transfer of the FHL to the calcaneus effectively restores the functional axis of plantarflexion, bridging massive defects while introducing a vascularized muscle belly to the healing environment.
Indications and Contraindications
The decision to proceed with an FHL transfer is dictated by the chronological age of the tear, the size of the tendon gap after debridement, patient functional demands, and the quality of the remaining host tissue. The Kuwada classification is frequently utilized to guide surgical decision-making based on gap size. Kuwada Type III defects (3-6 cm) often require tendon flaps or V-Y advancements, while Kuwada Type IV defects (>6 cm) definitively necessitate tendon transfer or allograft reconstruction.
Operative Versus Non Operative Indications
| Management Strategy | Primary Indications | Key Clinical Considerations |
|---|---|---|
| Non-Operative Management | Elderly, low-demand patients; severe medical comorbidities precluding anesthesia; severe peripheral vascular disease; active local infection; non-ambulatory status. | Focuses on bracing (e.g., AFO or CAM boot) to provide stability. Does not restore push-off strength. High risk of persistent weakness and altered gait mechanics. |
| Primary Repair / V-Y Advancement | Chronic ruptures with gaps < 3 cm after debridement; distinct proximal and distal tendon stumps with adequate collagenous integrity. | Requires robust tissue quality. V-Y advancement of the gastrocnemius aponeurosis can bridge small defects but may weaken the triceps surae if over-lengthened. |
| FHL Tendon Transfer | Chronic ruptures with gaps > 5-6 cm; failed prior Achilles repairs; severe tendinosis requiring extensive debridement; significant fatty infiltration/atrophy of the triceps surae. | Gold standard for large gaps. Restores in-phase plantarflexion. Muscle belly provides localized vascularity. |
| Allograft Reconstruction | Massive defects where autograft is insufficient or contraindicated; revision scenarios lacking local autograft options. | Carries risks of delayed incorporation, disease transmission, and higher cost. Lacks the dynamic contractile property of an FHL transfer. |
Contraindications for FHL transfer include pre-existing hallux rigidus or severe first metatarsophalangeal (MTP) joint pathology, as the loss of FHL function can exacerbate first ray biomechanical dysfunction. Additionally, severe peripheral neuropathy (e.g., advanced Charcot neuroarthropathy) and compromised soft tissue envelopes are relative contraindications requiring careful preoperative optimization.
Pre Operative Planning and Patient Positioning
Thorough preoperative evaluation relies on a combination of clinical examination and advanced imaging.
Clinical Evaluation
Patients typically present with a history of a remote "pop" or acute pain episode followed by a period of perceived improvement, ultimately plateauing with persistent weakness. Clinical examination reveals a palpable defect in the Achilles tendon, though this may be obscured by organized hematoma or fibrotic scar tissue. The Thompson test may be equivocal or falsely negative in chronic settings due to the compensatory hypertrophy and action of the FHL, FDL, and tibialis posterior. The hallmark clinical sign is the inability to perform a single-limb heel rise, accompanied by increased resting ankle dorsiflexion compared to the contralateral side.
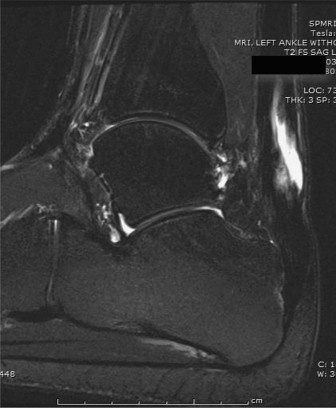
Imaging Modalities
Weight-bearing radiographs of the foot and ankle are obtained to rule out calcaneal avulsion fractures, Haglund's deformity, or degenerative joint disease. Magnetic Resonance Imaging (MRI) is the gold standard for preoperative planning. MRI allows for precise measurement of the tendon gap, assessment of the degree of tendinosis in the remaining stumps, and evaluation of the triceps surae muscle bellies for fatty infiltration and atrophy. Severe fatty infiltration of the gastrocnemius and soleus further supports the use of a dynamic tendon transfer like the FHL, as the native muscle may no longer be capable of generating adequate contractile force even if continuity is restored.
Patient Positioning and Preparation
The patient is placed in the prone position on a radiolucent operating table. Chest rolls and appropriate padding are utilized to protect pressure points. The operative extremity is prepped and draped in a standard sterile fashion to above the knee, allowing for full visualization of the posterior leg and unhindered manipulation of the knee and ankle joints. A thigh tourniquet is applied to provide a bloodless surgical field. Prophylactic intravenous antibiotics are administered prior to tourniquet inflation.
Detailed Surgical Approach and Technique
The surgical technique for FHL transfer can be performed via a single-incision posterior approach or a double-incision approach (incorporating a medial midfoot incision for extended graft harvest). The single-incision approach is generally preferred to minimize morbidity, provided adequate graft length can be obtained for calcaneal fixation.
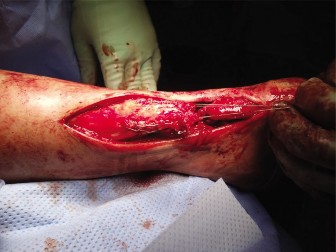
Incision and Dissection
A longitudinal incision is made slightly medial to the midline of the Achilles tendon, extending from the musculotendinous junction to the calcaneal insertion. A slightly medial approach reduces the risk of iatrogenic injury to the sural nerve, which crosses from medial to lateral in the proximal third of the calf and runs along the lateral border of the distal Achilles.
Full-thickness fasciocutaneous flaps are developed to preserve the delicate vascular supply to the skin. The paratenon is incised longitudinally and preserved for later closure if possible. The fibrotic scar tissue bridging the rupture site is meticulously excised until healthy, parallel collagen fibers are identified in both the proximal and distal tendon stumps. The resultant gap is measured with the ankle in neutral dorsiflexion to confirm the necessity of the FHL transfer.
Flexor Hallucis Longus Tendon Harvest
Attention is directed to the deep posterior compartment. The deep fascia covering the FHL muscle is incised. The FHL muscle belly is identifiable by its low-lying nature, extending distally almost to the level of the ankle joint.
To harvest the tendon, the dissection proceeds distally along the medial aspect of the calcaneus. The fibro-osseous tunnel of the FHL (posterior to the medial malleolus and deep to the flexor retinaculum) is identified. Care must be taken to protect the posterior tibial neurovascular bundle, which lies immediately medial and superficial to the FHL tendon in this region. The retinaculum is incised, and the FHL tendon is traced as far distally as possible into the plantar vault.
In a single-incision technique, maximum plantarflexion of the hallux and ankle allows for the tendon to be transected as distally as possible, usually yielding sufficient length (approximately 5-7 cm) for calcaneal tunnel fixation. If additional length is required, a secondary medial midfoot incision is made over the Master Knot of Henry. The FHL is identified dorsal to the FDL, tagged, and transected. Tenodesis of the distal FHL stump to the FDL can be performed to preserve distal hallux plantarflexion, though biomechanical studies suggest this may not significantly alter clinical outcomes. The proximal FHL tendon is then pulled through into the posterior ankle wound.
Graft Preparation
Once harvested, the distal end of the FHL tendon is prepared using a heavy non-absorbable suture (e.g., #2 FiberWire) utilizing a locking Krackow or whipstitch technique. The diameter of the prepared graft is measured using sizing blocks to determine the appropriate calcaneal tunnel diameter, typically ranging from 6 to 8 mm.
Calcaneal Tunnel Preparation and Fixation
A guide pin is placed into the superior aspect of the calcaneal tuberosity, anterior to the native Achilles insertion, directing it plantarward and slightly anteriorly to avoid breaching the plantar cortex. The tunnel is reamed to a depth of approximately 25-30 mm, matching the diameter of the prepared FHL graft.

The FHL tendon is then passed into the calcaneal tunnel. Fixation is most commonly achieved using a bioabsorbable or polyetheretherketone (PEEK) interference screw.
Tensioning and Final Repair
Proper tensioning of the FHL graft is a critical step. The ankle must be positioned in approximately 15 to 20 degrees of plantarflexion to match the resting tension of the contralateral limb. With the ankle held in this position, the interference screw is deployed over a guidewire, firmly securing the tendon within the calcaneal tunnel.
Following rigid bony fixation, the remaining proximal Achilles stump is advanced and sutured to the FHL muscle belly and tendon using interrupted non-absorbable sutures. This side-to-side anastomosis augments the construct and allows the vascularized FHL muscle belly to drape over the repair site. If a distal Achilles stump remains, it is also incorporated into the repair.
Closure
The paratenon is closed over the repair if viable. The subcutaneous tissues and skin are closed in a layered fashion. Meticulous hemostasis is verified prior to closure. A sterile dressing is applied, and the limb is immobilized in a well-padded short-leg splint with the ankle in 20 degrees of plantarflexion.
Complications and Management
While FHL transfer is highly successful, it carries inherent risks associated with posterior ankle surgery and tendon harvest.
Common Complications and Salvage Strategies
| Complication | Estimated Incidence | Etiology and Clinical Presentation | Management and Salvage Strategies |
|---|---|---|---|
| Wound Dehiscence / Infection | 5 - 10% | Poor vascularity of posterior skin flaps; excessive tension on closure. Presents as delayed healing, erythema, or purulent drainage. | Local wound care, oral/IV antibiotics. Deep infections may require surgical debridement and negative pressure wound therapy. Flap coverage (e.g., sural artery flap) for massive soft tissue defects. |
| Sural Nerve Injury | 3 - 8% | Iatrogenic traction or transection during lateral dissection. Presents as lateral foot numbness or painful neuroma. | Prevention via medialized incision. Management involves gabapentinoids, local corticosteroid injections, or surgical neuroma excision and burying into muscle. |
| Hallux Weakness / Cock-up Deformity | 10 - 15% | Loss of primary FHL function at the interphalangeal joint. Weakness in terminal push-off. | Generally well-tolerated. Tenodesis of FHL to FDL at the midfoot can mitigate this. Rigid orthotics with Morton's extension if symptomatic. |
| Deep Vein Thrombosis (DVT) | 1 - 5% | Prolonged immobilization, surgical trauma, prone positioning. Presents as calf swelling and pain. | Preoperative risk stratification. Chemical prophylaxis (e.g., LMWH or DOACs) in high-risk patients. Therapeutic anticoagulation if DVT is confirmed via duplex ultrasound. |
| Rerupture / Construct Failure | < 5% | Premature weight-bearing, failure of interference screw fixation, or aggressive early rehabilitation. | Revision surgery. May require massive allograft reconstruction, V-Y advancement, or pantalar arthrodesis in catastrophic salvage scenarios. |
Post Operative Rehabilitation Protocols
A structured, phased rehabilitation protocol is essential to protect the tendon transfer while preventing debilitating joint stiffness.
Phase 1: Maximum Protection (Weeks 0-2)
The patient is strictly non-weight-bearing (NWB) in a plantarflexed splint. Elevation and strict adherence to NWB status are critical to allow initial wound healing and mitigate edema.
Phase 2: Early Mobilization and Controlled Weight-Bearing (Weeks 2-6)
At two weeks, sutures are removed. The patient is transitioned to a Controlled Ankle Motion (CAM) boot equipped with a 2-3 cm heel lift (or multiple heel wedges) to maintain plantarflexion and offload the repair. Weight-bearing is progressively advanced from touch-down to weight-bearing as tolerated (WBAT). Active range of motion (ROM) exercises in plantarflexion are initiated, but active dorsiflexion past neutral is strictly prohibited to prevent stretching of the transfer.
Phase 3: Progressive Stretching and Strengthening (Weeks 6-12)
The heel wedges are sequentially removed (typically one wedge every 1-2 weeks) until the foot reaches a neutral position within the CAM boot. The patient is transitioned to regular footwear with a small silicone heel cup. Physical therapy focuses on progressive resistance exercises, isometric strengthening of the triceps surae and FHL, and gait retraining. Proprioceptive training on balance boards is introduced.
Phase 4: Return to Function (Months 3-6+)
Patients gradually resume normal activities. Plyometric exercises, light jogging, and sport-specific drills are introduced only after the patient demonstrates the ability to perform a single-limb heel rise without pain and achieves at least 80% strength compared to the contralateral limb. Maximum medical improvement may take up to 12-18 months.
Summary of Key Literature and Guidelines
The utilization of the FHL for chronic Achilles reconstruction is heavily supported by orthopedic literature.
Wapner et al. (1993) provided the foundational description of the FHL transfer for chronic Achilles ruptures, demonstrating excellent clinical outcomes, restoration of push-off power, and minimal donor site morbidity. Their study established the FHL as a superior dynamic graft due to its in-phase firing mechanics.
Subsequent investigations by Ostrander et al. evaluated the anatomical yields of single versus double-incision techniques. They concluded that while a double-incision approach yields a longer graft, a meticulously executed single posterior incision provides adequate length for calcaneal tunnel fixation in the vast majority of cases, while significantly reducing operative time and midfoot morbidity.
Long-term follow-up studies, such as those by Hahn et al., have confirmed the durability of the FHL transfer. Isokinetic testing in these cohorts frequently demonstrates restoration of plantarflexion strength to within 15-20% of the uninjured limb. While slight deficits in hallux plantarflexion are measurable, they rarely result in subjective patient dissatisfaction or significant gait alterations, validating the FHL as an expendable and highly effective transfer for massive Achilles defects.
Current academic guidelines advocate for FHL transfer as the procedure of choice for chronic Achilles ruptures with gaps exceeding 5 cm, emphasizing rigid interference screw fixation and a meticulous, phased postoperative rehabilitation protocol to optimize functional recovery.
You Might Also Like