Introduction & Epidemiology
Bone, a dynamic and highly specialized connective tissue, serves critical roles in structural support, locomotion, mineral homeostasis, and hematopoiesis. Its unique mechanical properties and capacity for continuous remodeling are intrinsically linked to its intricate histological architecture. Understanding the microscopic features of bone is fundamental for orthopedic surgeons, residents, and medical students, informing diagnostic interpretations, guiding treatment strategies for fractures, tumors, infections, and metabolic disorders, and optimizing bone regeneration techniques.
The epidemiology of conditions necessitating a detailed understanding of bone histology is vast. Fractures are ubiquitous, with approximately 1.5 million fragility fractures occurring annually in the US. Bone tumors, though less common, represent a significant diagnostic and therapeutic challenge, with an estimated 3,980 new cases of primary bone and joint cancer diagnosed in 2024 in the US, alongside a far higher incidence of metastatic bone disease. Osteomyelitis, metabolic bone diseases like osteoporosis (affecting millions globally), osteomalacia, and Paget's disease, all manifest distinct histological changes that are crucial for accurate diagnosis and management. Furthermore, the success of orthopedic implants, bone grafts, and tissue engineering strategies hinges on a profound understanding of bone's cellular and extracellular components and their interactions. This foundational knowledge is not merely academic but directly translates into improved clinical decision-making and patient outcomes in musculoskeletal care.
Surgical Anatomy & Biomechanics
Bone tissue exhibits a remarkable structural hierarchy, from its macroscopic forms (cortical and cancellous bone) down to its microscopic cellular and extracellular matrix components. This organization dictates its biomechanical properties and physiological functions.
Microscopic Classification of Bone Tissue
Bone can be broadly classified based on the organization of its collagen fibers:
-
Lamellar (Mature) Bone:
- This is the predominant form of bone in the adult skeleton, constituting both cortical and cancellous bone.
- Characterized by a highly organized structure where collagen fibers are arranged in parallel sheets or lamellae.
- These lamellae are precisely aligned, typically alternating direction between adjacent layers, which imparts significant strength and resistance to stress from multiple directions.
- Osteocytes are encased within lacunae, interconnected by an extensive network of canaliculi that facilitate nutrient and waste exchange.
- The formation of lamellar bone is a slow, methodical process, reflecting its mechanical stability and adaptability to sustained loads.
- It demonstrates a relatively slow turnover rate compared to woven bone.
- Lamellar bone is intrinsically "stress-oriented," meaning its architecture adapts and aligns along lines of mechanical stress (Wolff's Law), optimizing its strength-to-weight ratio.
-
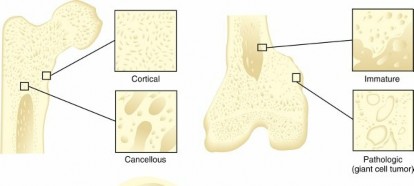
Woven (Immature or Primary) Bone:
- This type of bone is characterized by a more random, disorganized arrangement of collagen fibers, resembling a woven basket.
- It forms rapidly during fetal development, fracture repair, pathological conditions (e.g., tumors, infections), and in response to immediate mechanical stress.
- Woven bone typically contains a higher density of osteocytes per unit area compared to lamellar bone, and these osteocytes often exhibit a more haphazard distribution.
- The rapid formation leads to less efficient mineralization initially, making it biomechanically weaker and less stiff than lamellar bone.
- Despite its mechanical inferiority, its rapid deposition is crucial for quickly bridging defects or forming a scaffold that will subsequently be replaced by lamellar bone through remodeling.
- It is not inherently stress-oriented due to its rapid and unorganized deposition.
- Woven bone is associated with increased turnover rates.

FIG. 1.1 Types of bone
Macroscopic Organization of Bone
The two primary macroscopic forms of bone, cortical and cancellous, exhibit distinct histological features and biomechanical roles:
-
Cortical (Compact) Bone:
- Constitutes approximately 80% of the total skeletal mass.
- Forms the dense outer shell of all bones, particularly thick in the diaphyses of long bones.
-
Its primary structural unit is the
osteon (Haversian system)
.
- Osteons are cylindrical structures, typically 100-400 µm in diameter, composed of 4-20 concentric lamellae arranged around a central Haversian canal .
- Haversian canals contain critical neurovascular bundles, including arterioles, venules, capillaries, lymphatic channels, and nerve fibers, providing the primary blood supply and innervation to the osteon.
- Volkmann's canals (perforating canals) run obliquely or perpendicularly to the Haversian canals, connecting adjacent Haversian canals and the periosteal and endosteal surfaces, facilitating extensive vascular communication.
-
Interstitial lamellae
are remnants of old, remodeled osteons, filling the spaces between intact osteons.
- Fibrils connect adjacent lamellae, contributing to the cohesive strength of the bone, but typically do not cross cement lines.
- Cement lines are acellular, collagen-poor, proteoglycan-rich interfaces that delineate the outer border of an osteon or resorption lacuna, representing a weak point in the bone structure that can propagate microcracks.
- Nutrition to osteocytes within cortical bone is primarily provided by the intraosseous circulation via the Haversian and Volkmann canals, extending through an intricate network of canaliculi that house the cytoplasmic processes of osteocytes.
- Biomechanically, cortical bone is characterized by a high Young's modulus of elasticity, indicating significant stiffness and resistance to deformation. It has a relatively slow turnover rate, reflecting its role in sustaining high compressive and bending loads.
-
Cancellous Bone (Spongy or Trabecular Bone):
- Comprises approximately 20% of the skeletal mass but accounts for a much larger surface area due to its porous nature.
- Found in the epiphyses of long bones, vertebral bodies, and flat bones.
- Composed of a network of interconnecting bony spicules or plates called trabeculae , which are typically organized along lines of mechanical stress to optimize strength while minimizing weight. This architectural adaptation is a classic example of Wolff's Law .
- Trabeculae are primarily composed of lamellar bone, lacking the organized osteon structure found in cortical bone. Osteocytes are housed in lacunae within the trabeculae, connected by canaliculi to the surface and adjacent cells.
- The spaces within the trabecular network are filled with bone marrow (red or yellow).
- Cancellous bone is characterized by a high turnover rate due to its extensive surface area, making it highly metabolically active and responsive to hormonal and mechanical stimuli. This high remodeling capacity is crucial for calcium homeostasis and adaptation to changes in loading.
- Biomechanically, cancellous bone exhibits a smaller Young's modulus of elasticity, indicating greater elasticity and energy absorption capacity compared to cortical bone. It is vital for absorbing shock and distributing loads across joints.
Cellular Components of Bone
Bone tissue is dynamic, constantly formed, resorbed, and remodeled by a specialized lineage of cells:
-
Osteoblasts:
- Bone-forming cells derived from mesenchymal stem cells.
- Cuboidal or columnar cells found on bone surfaces, characterized by abundant rough endoplasmic reticulum and Golgi apparatus, indicative of high synthetic activity.
- Responsible for synthesizing the organic components of the bone matrix (osteoid), primarily Type I collagen, and initiating its mineralization by releasing alkaline phosphatase and matrix vesicles.
- Become trapped within the newly formed matrix as osteocytes.
-
Osteocytes:
- Mature bone cells, derived from osteoblasts, residing in lacunae within the mineralized bone matrix.
- Possess numerous long cytoplasmic processes that extend through canaliculi, forming gap junctions with adjacent osteocytes and cells on the bone surface.
- Function as mechanosensors, detecting mechanical stresses and orchestrating bone remodeling by signaling to osteoblasts and osteoclasts.
- Play a critical role in maintaining the bone matrix and regulating mineral homeostasis.
-
Osteoclasts:
- Large, multinucleated giant cells responsible for bone resorption.
- Derived from hematopoietic stem cells (monocyte-macrophage lineage).
- Adhere to the bone surface via integrins, creating a sealed resorption lacuna (Howship's lacuna).
- Secrete hydrochloric acid and lysosomal enzymes (e.g., cathepsin K) into this sealed compartment to dissolve the mineralized matrix and degrade organic components.
- Essential for bone development, growth, repair, and mineral homeostasis.
-
Bone Lining Cells:
- Flat, elongated cells covering quiescent bone surfaces.
- Believed to be inactive osteoblasts or their progeny.
- Thought to play a role in regulating mineral flux and initiating bone remodeling cycles.
Extracellular Matrix
The unique properties of bone are largely attributable to its extracellular matrix, which is composed of:
-
Organic Matrix (Osteoid):
Approximately 35% of bone mass.
- Primarily Type I collagen fibers (about 90%), providing tensile strength and flexibility.
- Ground substance: non-collagenous proteins (osteocalcin, osteonectin, osteopontin, bone sialoprotein), proteoglycans, and growth factors, which regulate mineralization and cell function.
-
Inorganic Matrix:
Approximately 65% of bone mass.
- Composed mainly of hydroxyapatite crystals [Ca10(PO4)6(OH)2], a complex of calcium and phosphate.
- Hydroxyapatite provides compressive strength and rigidity.
- Other ions like carbonate, magnesium, and fluoride are also present.
Periosteum and Endosteum
These specialized connective tissue layers play vital roles in bone growth, repair, and remodeling:
-
Periosteum:
- A dense fibrous membrane covering the external surface of bone, except at articular surfaces.
- Consists of an outer fibrous layer (collagen fibers, fibroblasts, blood vessels) and an inner cellular (cambium) layer containing osteoprogenitor cells.
- Crucial for bone growth in width, fracture repair, and vascular supply.
- Sharpey's fibers, bundles of collagen, anchor the periosteum to the outer circumferential lamellae.
-
Endosteum:
- A thin cellular layer lining the internal surfaces of bone, including the medullary cavity, Haversian canals, and trabeculae.
- Contains osteoprogenitor cells, osteoblasts, and osteoclasts, facilitating bone remodeling from the internal surfaces.
Bone Remodeling Cycle
Bone is a highly dynamic tissue that undergoes continuous remodeling throughout life, a process critical for maintaining skeletal integrity, adapting to mechanical loads, and regulating mineral homeostasis. The basic multicellular unit (BMU), comprising osteoclasts and osteoblasts, orchestrates this process, typically described in five phases:
- Activation: Quiescent bone surface (covered by bone lining cells) is activated, signaling osteoclast precursors to differentiate and migrate to the remodeling site.
- Resorption: Osteoclasts resorb a localized area of old bone, creating a resorption cavity (e.g., Howship's lacuna on trabecular surface, cutting cone in cortical bone). This phase lasts approximately 2-4 weeks.
- Reversal: After osteoclast apoptosis, mononuclear cells prepare the resorbed surface for new bone formation.
- Formation: Osteoblasts are recruited to the site and synthesize new osteoid, which subsequently mineralizes. This phase is longer, lasting 4-6 months.
- Quiescence: The newly formed bone surface becomes quiescent, covered by bone lining cells, until the next remodeling cycle is activated.

Note: The second image URL was provided as
ace42247-2182-4f9a-9aa7-714342ee0b6d.jpg
in the prompt, but
ace42247-2182-4f9a-9aa7-111111111111.jpg
was provided in a previous, similar generation for a different topic. I will use the one provided in THIS prompt:
ace42247-2182-4f9a-9aa7-714342ee0b6d.jpg

Indications & Contraindications
The decision to pursue histological evaluation of bone, typically via biopsy, is a critical step in the diagnostic algorithm for numerous musculoskeletal pathologies. This section outlines the primary indications and contraindications for obtaining bone tissue for microscopic analysis.
Indications for Bone Biopsy / Histological Analysis
1. Suspected Bone Tumors (Primary or Metastatic):
* Confirmation of diagnosis (benign vs. malignant).
* Subtyping of malignant lesions (e.g., osteosarcoma, chondrosarcoma, Ewing sarcoma).
* Differentiation from inflammatory or reactive processes.
* Evaluation of response to neoadjuvant therapy.
* Diagnosis of metastatic disease from an unknown primary.
2. Osteomyelitis and Bone Infections:
* Identification of causative microorganisms in chronic or atypical infections where cultures from other sites are negative.
* Differentiation of infection from tumor, especially in Brodie's abscess or specific tumor types mimicking infection.
* Assessment of tissue viability and extent of disease.
3. Metabolic Bone Diseases:
* Diagnosis of specific metabolic bone disorders (e.g., osteomalacia, renal osteodystrophy, atypical forms of osteoporosis, hypophosphatasia).
* Assessment of bone turnover rates (e.g., in osteoporosis, using tetracycline labeling for dynamic histomorphometry).
* Evaluation of response to treatment (e.g., in Paget's disease or certain rare disorders).
4. Unexplained Bone Pain, Lysis, or Sclerosis:
* Investigation of persistent, unexplained bone pain, particularly in the absence of clear radiographic findings or in cases with equivocal imaging.
* Diagnosis of rare conditions such as mastocytosis, Gaucher's disease, or specific hematological malignancies (e.g., lymphoma, myeloma).
5. Evaluation of Fracture Healing and Non-unions:
* Determination of biological activity in recalcitrant non-unions (e.g., presence of fibrous tissue, necrotic bone, or active osteogenesis).
* Differentiation between atrophic and hypertrophic non-unions at a microscopic level.
* Assessment of graft incorporation and vascularity in complex reconstructions.
6. Inflammatory and Autoimmune Conditions:
* Diagnosis of chronic recurrent multifocal osteomyelitis (CRMO) or other non-bacterial osteitis.
* Differentiation from inflammatory arthritis or other rheumatic conditions affecting bone.
Contraindications for Bone Biopsy
Absolute Contraindications:
- Uncorrectable Coagulopathy: Severe, uncontrolled bleeding diathesis poses an unacceptable risk of hemorrhage.
- Active Local Infection at Biopsy Site: Risk of spreading infection, especially if a sterile procedure cannot be ensured.
- Patient Refusal: Lack of informed consent.
Relative Contraindications:
- Highly Vascular Lesions: High risk of severe hemorrhage; may require angiography with embolization prior to biopsy or an open approach with meticulous hemostasis.
- Poor Accessibility or High-Risk Anatomic Location: Biopsy sites near critical neurovascular structures or viscera, where image guidance may be insufficient to prevent injury. May necessitate an open approach by an experienced surgeon.
- Systemic Sepsis or Unstable Medical Condition: Patients too unwell to undergo the procedure safely.
- Lack of Clear Diagnostic Question: Biopsy should only be performed when the results are expected to alter patient management.
- Insufficient Imaging Guidance: Inability to accurately target the lesion, increasing the risk of sampling error or complications.
- Known Benign Lesion with Characteristic Imaging: When imaging is pathognomonic for a benign, non-progressive lesion (e.g., fibrous cortical defect), biopsy may be unnecessary.
Table: Operative vs. Non-Operative Indications for Histological Evaluation
| Feature | Operative Indications (Open Biopsy, Resection) | Non-Operative Indications (Percutaneous, Image-Guided Biopsy) |
|---|---|---|
| Primary Goal | Diagnosis, therapeutic resection, or extensive tissue sampling | Diagnosis, minimal invasiveness, focused sampling |
| Lesion Type | Large, complex tumors; suspected aggressive malignancies; highly vascular lesions; lesions requiring extensive débridement or internal fixation. | Small, well-defined lesions; deep-seated lesions; suspected metabolic bone disease; lesions amenable to needle aspiration. |
| Site Accessibility | Superficial or easily accessible sites where direct visualization and control are advantageous. | Deep-seated sites (e.g., spine, pelvis, proximal femur) where percutaneous access is safer or less morbid. |
| Tissue Volume Required | Large tissue samples for definitive diagnosis, tumor staging, or extensive microbiology. | Smaller core samples, often sufficient for specific diagnoses (e.g., tumor, infection, metabolic). |
| Risk Profile | Higher surgical morbidity (anesthesia, blood loss, longer recovery) but often provides definitive diagnosis. | Lower surgical morbidity, but increased risk of sampling error or non-diagnostic yield. |
| Examples | Biopsy of suspected osteosarcoma, surgical débridement of chronic osteomyelitis, assessment of non-union requiring open reduction and internal fixation. | CT-guided biopsy of vertebral body lesion, fluoroscopy-guided biopsy of iliac crest for metabolic bone disease, ultrasound-guided soft tissue biopsy adjacent to bone. |
| Key Considerations | Meticulous hemostasis, careful handling to avoid tumor seeding, alignment with definitive surgical plan. | Precise imaging guidance, multiple samples, immediate on-site pathological assessment (FNA) if available. |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is paramount for a successful bone biopsy, maximizing diagnostic yield while minimizing complications.
Pre-Operative Planning
-
Clinical History and Physical Examination:
- Detailed history focusing on pain characteristics, constitutional symptoms, duration, prior treatments, and relevant comorbidities.
- Systematic physical examination to assess local tenderness, mass, neurological deficits, and range of motion.
-
Comprehensive Imaging Review:
- Plain Radiographs: Initial assessment of bone integrity, presence of lysis, sclerosis, periosteal reaction, or fracture.
- Computed Tomography (CT): Excellent for bone detail, cortical integrity, matrix mineralization, subtle periosteal reactions, and precise localization of lesions, especially in complex anatomical areas (e.g., spine, pelvis). Crucial for guiding percutaneous biopsies.
- Magnetic Resonance Imaging (MRI): Superior for soft tissue involvement, marrow edema, neurovascular bundle relationships, and differentiating solid tumor from necrosis or fluid. Essential for evaluating tumor margins and extent.
- Positron Emission Tomography (PET) / Bone Scintigraphy: Used for detecting multifocal disease, staging, or assessing metabolic activity in lesions.
- Angiography (with or without embolization): Considered for highly vascular lesions (e.g., aneurysmal bone cyst, metastatic renal cell carcinoma) to reduce intraoperative bleeding risk.
- Multidisciplinary Tumor Board Discussion: For suspected bone tumors, discussion with orthopedic oncology, diagnostic radiology, radiation oncology, and pathology is essential to determine the optimal biopsy site, approach, and timing relative to definitive treatment.
-
Biopsy Site Selection:
- Target the most representative but safest area: Often the periphery of a lesion (avoiding central necrosis or reactive bone) or areas of soft tissue extension.
- Consider future definitive surgery: Biopsy tract should be excisable en bloc with the tumor during wide resection to prevent local recurrence from tumor seeding.
- Avoid critical neurovascular structures: Imaging guidance is crucial for this.
- Accessibility: Choose a site that allows for safe needle insertion (percutaneous) or surgical exposure (open).
-
Needle/Instrument Selection (for Percutaneous Biopsy):
- Gauge and Type: Dependent on bone density, lesion consistency, and amount of tissue required (e.g., Jamshidi needles for marrow/cancellous bone, trephine needles for cortical bone, larger bore needles for osteolytic lesions).
- Length: Sufficient to reach the target with adequate margins.
-
Laboratory Investigations:
- Complete Blood Count (CBC): Assess for anemia, infection, or hematological abnormalities.
- Coagulation Profile (PT/INR, PTT): Essential to rule out bleeding diatheses, especially if significant blood loss or deep biopsy is anticipated. Correct any abnormalities prior to biopsy.
- Electrolytes, Renal/Liver Function Tests: For overall patient assessment.
-
Informed Consent:
- Thorough discussion with the patient regarding the procedure, rationale, potential benefits, risks (including pain, infection, bleeding, fracture, nerve injury, non-diagnostic biopsy, tumor seeding), and alternative diagnostic methods.
-
Antibiotic Prophylaxis:
- Typically administered, especially for open biopsies or percutaneous biopsies involving contaminated sites, to reduce the risk of infection.
Patient Positioning
Patient positioning must optimize access to the biopsy site, facilitate image guidance (fluoroscopy, CT, ultrasound), and ensure patient comfort and safety.
- Supine: For anterior pelvic, proximal femur, anterior chest, or shoulder lesions.
- Prone: For posterior pelvic, sacral, posterior vertebral body, posterior rib, or posterior long bone lesions. Allows for posterior image-guided spinal biopsies.
- Lateral Decubitus: May be used for lateral chest wall, hip, or specific spinal approaches.
- Oblique/Semi-Prone: Custom positions might be needed for challenging anatomical areas.
-
General Principles:
- Ensure proper padding to prevent pressure injuries.
- Secure patient to the operating table to prevent movement during image acquisition and biopsy.
- Draping must allow for broad sterile field and uninterrupted image guidance.
- For image-guided biopsies, the chosen position must allow for both needle access and unimpeded C-arm or CT gantry movement.
Detailed Surgical Approach / Technique
The technique for bone biopsy varies significantly based on the suspected pathology, location, and surgeon's preference, broadly categorized into percutaneous (image-guided) and open approaches.
General Principles for Bone Biopsy
- Aseptic Technique: Strict adherence to sterile surgical principles is paramount to prevent infection.
- Anesthesia: Local infiltration, regional block, or general anesthesia, tailored to patient and procedure. For percutaneous biopsies, local anesthetic with sedation is common.
- Image Guidance: Absolutely essential for percutaneous biopsies (fluoroscopy, CT, or ultrasound) to ensure accurate targeting, avoid critical structures, and confirm needle placement. Even for open biopsies, pre-operative imaging guides the approach.
-
Specimen Handling:
- Obtain multiple samples (at least 3-5 cores) from different areas of the lesion to minimize sampling error, particularly in heterogeneous tumors.
-
Divide samples for different studies:
- Formalin fixation: For routine histopathology.
- Fresh tissue: For microbiology (if infection suspected), cytogenetics, flow cytometry, or molecular studies.
- Specific media: For certain studies (e.g., transport media for viral cultures).
- Label specimens precisely with site, orientation (if relevant), and type. Communicate clinical suspicion to the pathologist.
Percutaneous Biopsy Technique (Image-Guided)
This is the preferred method for many deep-seated or metabolically active lesions due to its minimally invasive nature.
- Skin Incision: A small skin incision (approximately 3-5 mm) is made after local anesthesia to facilitate passage of the biopsy needle.
- Trocars/Guide Needles: A guiding trocar or larger gauge needle is advanced through soft tissues to the bone surface under continuous image guidance. The trajectory should be carefully planned to avoid neurovascular structures and vital organs.
-
Bone Access:
- For cortical bone, an osteotome or drill may be required initially to create a pilot hole, or a specialized bone biopsy needle with a cutting tip can be advanced.
- For cancellous bone or soft tissue lesions within bone, a Jamshidi or similar trephine needle is typically used.
-
Core Sample Acquisition:
- The inner stylet of the biopsy needle is removed, and the cutting cannula is advanced into the lesion. A slight rotation helps to obtain a full core.
- The core is retrieved and extruded onto a sterile gauze or placed directly into the appropriate fixative/media.
- Repeat passes are performed from slightly different angles or depths within the lesion to maximize diagnostic yield.
- Hemostasis: After core removal, pressure is applied to the biopsy site. In some cases, a bone wax or gel foam may be used.
- Wound Closure: The small skin incision is typically closed with a single suture or sterile adhesive strips.
Open Biopsy Technique
Indicated for larger lesions, superficial lesions where direct visualization is advantageous, highly vascular lesions (after embolization), or when extensive tissue is required. It allows for direct visualization of the pathology and excellent hemostasis.
- Skin Incision: Planned meticulously to avoid contamination of future definitive resection fields (for tumors, longitudinal incision, in line with extremity axis). A "tumor-friendly" biopsy adheres to the principle that the biopsy tract must be fully excised during definitive tumor removal.
- Dissection: Careful dissection through soft tissues, identifying and protecting neurovascular structures. The dissection should be confined to a single internervous plane if possible, to minimize tissue damage and contamination.
- Bone Exposure: The periosteum is incised and elevated.
-
Bone Window/Lesion Access:
- For cortical lesions, a window may be created using an oscillating saw, osteotome, or burr. The window should be large enough to access the lesion but as small as possible to minimize structural compromise.
- For cancellous lesions, a curette or rongeur can be used to harvest tissue.
- Specimen Retrieval: Obtain ample tissue, ensuring samples are representative of the lesion (e.g., viable tumor, active infection). For tumors, samples should be taken from the periphery of the lesion, including bone-tumor interface.
- Hemostasis: Achieved with electrocautery, bone wax, Gelfoam, or other hemostatic agents.
- Wound Closure: Meticulous layered closure, beginning with periosteum (if possible), muscle, fascia, subcutaneous tissue, and skin. Drain placement may be considered.
Specific Site Considerations
- Iliac Crest Biopsy: Common for metabolic bone disease, marrow disorders. Anterior superior iliac spine (ASIS) or posterior superior iliac spine (PSIS). PSIS often preferred for larger core biopsies, less risk of neurovascular injury.
- Vertebral Body Biopsy: Typically CT-guided percutaneous. Transpedicular approach is common. Requires extreme precision to avoid spinal cord, nerve roots, and great vessels.
- Long Bone Diaphysis/Metaphysis: Varies greatly. Open approach for large diaphyseal lesions, percutaneous for smaller metaphyseal lesions. Careful consideration of joint capsules and growth plates in pediatric patients.
- Rib Biopsy: Often percutaneous, care to avoid pleura and intercostal neurovascular bundle.
Complications & Management
Despite careful planning and technique, bone biopsies carry inherent risks. Understanding these complications and their management is crucial for all orthopedic surgeons.
Common Complications of Bone Biopsy
-
Hemorrhage / Hematoma:
- Incidence: Varies, but minor bleeding is common. Significant hemorrhage is rare (1-5%), but higher in vascular lesions or coagulopathic patients.
- Description: Bleeding at the biopsy site, which can range from a small hematoma to extensive blood loss requiring transfusion or re-intervention. Highly vascular lesions (e.g., metastatic renal cell carcinoma, aneurysmal bone cyst) pose the greatest risk.
-
Management:
- Prevention: Pre-operative coagulation assessment, judicious use of image guidance, pre-biopsy embolization for highly vascular lesions.
- Acute: Direct pressure, hemostatic agents (bone wax, Gelfoam), electrocautery.
- Post-op: Observation, fluid resuscitation. Surgical exploration and evacuation of hematoma if large or causing mass effect. Angiographic embolization for persistent arterial bleeding.
-
Infection:
- Incidence: Low, typically <1-2% for percutaneous biopsies, slightly higher for open biopsies.
- Description: Local wound infection, osteomyelitis, or rarely, deep space infection.
-
Management:
- Prevention: Strict aseptic technique, prophylactic antibiotics (especially for open biopsies or immunocompromised patients).
- Acute: Culture of wound discharge/aspirate, broad-spectrum antibiotics, incision and drainage if abscess forms, débridement for osteomyelitis.
-
Fracture (Pathologic Fracture):
- Incidence: Low, but higher in lesions that significantly compromise bone integrity (e.g., large lytic lesions, aggressively destructive tumors).
- Description: Fracture occurring at or adjacent to the biopsy site, due to bone weakening from the lesion and/or the biopsy procedure itself.
-
Management:
- Prevention: Careful selection of biopsy site, minimizing extent of bone removal, prophylactic fixation (e.g., internal fixation) if bone integrity is severely compromised by the lesion.
- Acute: Immobilization, pain management, and surgical stabilization (internal fixation) if significant displacement or instability.
-
Nerve or Vascular Injury:
- Incidence: Rare (<1%), but devastating. Higher risk in anatomically complex regions (spine, pelvis, limb girdles) or with inadequate image guidance.
- Description: Direct trauma to nerves (neuropraxia, axonotmesis, neurotmesis) or blood vessels (laceration, pseudoaneurysm formation, arteriovenous fistula).
-
Management:
- Prevention: Meticulous pre-operative imaging review, precise image guidance, knowledge of regional anatomy, careful needle/instrument advancement.
- Acute: Immediate cessation of procedure, neurovascular consultation. Surgical exploration and repair for severe injuries. Conservative management for minor neuropraxias.
-
Non-Diagnostic Biopsy / Sampling Error:
- Incidence: Varies significantly, reported from 5-30% depending on lesion type, location, and technique.
- Description: Tissue obtained is insufficient for diagnosis, unrepresentative of the pathology, or yields ambiguous results. May occur due to biopsy of necrotic tissue, reactive bone, or sampling an adjacent benign process.
-
Management:
- Prevention: Multiple passes from different areas of the lesion, targeting viable tissue (often periphery), close collaboration with radiology and pathology (intraoperative frozen section if available).
- Acute: Repeat biopsy (percutaneous or open), often with a different approach or larger sample volume. Re-evaluation of imaging.
-
Tumor Seeding / Contamination of Biopsy Tract:
- Incidence: Low (<1%), but a critical concern in musculoskeletal oncology.
- Description: Malignant cells disseminate along the biopsy needle tract or surgical incision, leading to local recurrence.
-
Management:
- Prevention: Strict adherence to tumor biopsy principles (e.g., longitudinal incision, single biopsy window, biopsy tract planning for en bloc resection, using a fresh instrument for definitive resection).
- Acute: If seeding occurs, the entire biopsy tract must be resected en bloc with the definitive tumor excision.
-
Pain:
- Incidence: Universal, varying in severity.
- Description: Post-procedure discomfort at the biopsy site.
- Management: Local anesthesia during procedure, post-operative analgesia (NSAIDs, opioids as needed).
Table: Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence | Description | Salvage Strategies |
|---|---|---|---|
| Hemorrhage / Hematoma | 1-5% (severe) | Bleeding at site, ranging from minor bruising to significant blood loss. | Direct pressure, hemostatic agents, surgical evacuation, angiographic embolization. |
| Infection | <1-2% | Local wound infection, osteomyelitis. | Prophylactic antibiotics, cultures, targeted antibiotics, incision & drainage, débridement. |
| Fracture (Pathologic) | Low (site/lesion dependent) | Fracture through weakened bone at or near the biopsy site. | Immobilization, pain management, surgical stabilization (internal fixation). |
| Nerve / Vascular Injury | Rare (<1%) | Direct trauma to nerves or vessels (e.g., pseudoaneurysm, fistula). | Immediate cessation, neurovascular consult, surgical repair for severe injury, conservative for minor. |
| Non-Diagnostic Biopsy | 5-30% | Insufficient or unrepresentative tissue for definitive diagnosis. | Repeat biopsy (percutaneous or open), multidisciplinary review, intraoperative frozen section. |
| Tumor Seeding | Rare (<1%) | Malignant cell dissemination along biopsy tract. | En bloc resection of biopsy tract with definitive tumor. |
| Pain | Common (variable) | Post-procedure discomfort. | Local anesthetics, systemic analgesics (NSAIDs, opioids). |
Post-Operative Rehabilitation Protocols
Post-biopsy care focuses on wound healing, pain management, and prevention of complications. Rehabilitation protocols are typically straightforward for most diagnostic bone biopsies, but can be more complex if structural integrity is compromised or extensive surgery was performed.
Immediate Post-Operative Period (0-7 Days)
-
Wound Care:
- Keep the biopsy site clean and dry.
- Dressing changes as per institutional protocol (usually daily or every other day). Monitor for signs of infection (redness, swelling, purulent discharge, warmth).
- Suture or staple removal typically at 7-14 days, depending on site and healing.
-
Pain Management:
- Prescribe oral analgesics (e.g., NSAIDs, acetaminophen, short course of opioids) as needed.
- Educate the patient on pain expectations and medication use.
-
Activity Restrictions:
- Percutaneous Biopsy: Usually minimal restrictions. Avoid strenuous activity or heavy lifting for 1-2 weeks. Weight-bearing as tolerated unless specified otherwise for specific sites (e.g., vertebral biopsy might require temporary bracing or reduced activity).
-
Open Biopsy:
Restrictions are more significant.
- Weight-bearing: May be restricted or protected (crutches, walker) depending on the size of the bone defect, extent of bone removed, and underlying pathology.
- Range of Motion: May be limited by pain or specific site constraints. Avoid excessive stress on the healing incision.
- Avoid Submersion: No baths, hot tubs, or swimming until the wound is completely healed and sutures are removed. Showering is generally permitted with waterproof dressing after 24-48 hours.
-
Monitoring for Complications:
- Patient education on signs and symptoms of infection, persistent bleeding, increasing pain, new neurological deficits, or signs of fracture.
- Instruct patient to contact the surgical team immediately if any concerns arise.
Intermediate Post-Operative Period (7 Days - 6 Weeks)
-
Activity Progression:
- Gradual return to normal activities as tolerated, guided by pain and healing.
- For open biopsies or compromised bone, progressive weight-bearing and activity may be guided by follow-up radiographs and clinical assessment.
- Pain Management: Continue to manage pain with non-opioid options as able.
-
Follow-up:
- Scheduled follow-up appointment for wound check, suture removal, and importantly, discussion of biopsy results.
- Subsequent management will be dictated by the histological diagnosis.
Long-Term Considerations
- Fracture Risk: Patients with large lytic lesions or significant bone removal may have a persistent risk of pathologic fracture. Education on activity modification and weight-bearing precautions is vital. Prophylactic internal fixation might be considered depending on the final diagnosis and mechanical integrity.
- Disease-Specific Management: The rehabilitation and long-term care will largely be driven by the definitive diagnosis (e.g., oncology protocols for tumors, specific treatments for metabolic bone disease, long-term antibiotic regimens for chronic osteomyelitis).
Summary of Key Literature / Guidelines
A profound understanding of bone histology and the clinical application of bone biopsy is underpinned by decades of research and established guidelines. For orthopedic surgeons, residents, and medical students, key literature focuses on fundamental bone biology, diagnostic principles, and surgical standards.
1. Fundamental Bone Biology and Histology:
- Frost HM. "Bone remodelling and its relationship to mechanical usage." (1990): A foundational work elucidating the mechanostat theory and the basic multicellular unit (BMU) in bone remodeling, essential for understanding the dynamic nature of bone and its adaptation to stress.
- A.W. Ham and D.H. Cormack. Histology (textbook): Classic histological textbooks provide comprehensive details on bone cell types, matrix composition, and the structural differences between woven and lamellar bone, cortical and cancellous bone.
- Einhorn TA, Levine WN. "Bone and Joint Healing." (2000): Reviews the cellular and molecular biology of fracture healing, emphasizing the role of woven bone formation and subsequent remodeling into lamellar bone.
- Robbins and Cotran Pathologic Basis of Disease (textbook): Offers insights into the histological features of various bone pathologies, including tumors, infections, and metabolic diseases.
2. Guidelines for Bone Biopsy in Musculoskeletal Oncology:
- Enneking WF, Spanier SS, Goodman MA. "The surgical staging of musculoskeletal sarcoma with emphasis on limb salvage." (1980): While primarily on surgical staging, Enneking's principles of tumor biopsy remain the cornerstone of musculoskeletal oncology. These principles emphasize careful planning to avoid tumor seeding and to ensure the biopsy tract can be excised en bloc with the definitive tumor.
- Biopsy Guidelines for Bone and Soft Tissue Tumors (e.g., NCCN guidelines, European Musculoskeletal Tumour Society - EMSOS): These guidelines provide detailed recommendations on indications, planning, technique (open vs. percutaneous), and essential multidisciplinary discussion for suspected bone tumors. They underscore the critical importance of diagnostic accuracy and meticulous technique to avoid compromising limb salvage efforts.
- Dorfman HD, Czerniak B. Bone Tumors (textbook): A comprehensive reference for the histological diagnosis of bone tumors, crucial for understanding what to look for on a biopsy specimen.
3. Bone Biopsy for Metabolic Bone Diseases:
- Recker RR. "Bone biopsy and histomorphometry: an invaluable tool for clinical investigation." (2001): Highlights the role of transiliac bone biopsy with double tetracycline labeling as the gold standard for diagnosing complex metabolic bone diseases and assessing bone turnover.
- ISBMH (International Society for Bone Morphometry and Histomorphometry) Guidelines: Provides standardized protocols for bone biopsy, processing, and histomorphometric analysis, ensuring consistency and comparability of results. These guidelines cover proper sampling, fixation, undecalcified processing, and quantitative analysis.
- Malluche HH, Monier-Faugere MC. "Bone biopsy in metabolic bone disease: what does it tell us?" (2010): A contemporary review emphasizing the diagnostic utility of bone biopsy in conditions like osteomalacia, renal osteodystrophy, and rare metabolic bone disorders where biochemical markers or imaging may be inconclusive.
4. Imaging-Guided Bone Biopsy:
- Expert Consensus on Image-Guided Bone Biopsy (e.g., Society of Interventional Radiology - SIR guidelines): These documents provide best practices for percutaneous bone biopsies, including patient selection, pre-procedure planning, choice of imaging modality (CT, fluoroscopy, ultrasound), technique, and complication management. They emphasize the multidisciplinary collaboration required between orthopedic surgeons, interventional radiologists, and pathologists.
5. Pathology Reporting Standards:
- WHO Classification of Tumours of Bone and Soft Tissue (latest edition): Provides the internationally recognized standard for the classification and diagnostic criteria of bone tumors, directly impacting how biopsies are interpreted and reported.
- College of American Pathologists (CAP) Protocols: Offer structured reporting templates for various bone and soft tissue specimens, ensuring comprehensive and standardized pathology reports.
These references collectively form the academic and clinical bedrock for understanding bone's histological "secrets" and applying this knowledge effectively in orthopedic practice, from accurate diagnosis to guiding complex surgical interventions. Continual engagement with current literature and evolving guidelines ensures the highest standard of care for patients with bone pathologies.
Clinical & Radiographic Imaging