INTRODUCTION AND HISTORICAL CONTEXT
The evolution of the free vascularized bone transplant represents a monumental leap in reconstructive orthopaedic surgery, offering a reliable solution for massive skeletal defects that historically resulted in amputation. Ostrup and Fredrickson, alongside Doi et al., were the first to report experimental success using microvascular techniques to transfer ribs as bone grafts to the mandibles of canine models. Their pioneering work demonstrated a critical biological principle: vascularized bone grafts remain entirely viable based on their preserved medullary and periosteal circulation. Consequently, these grafts heal primarily at the host-graft junction without undergoing the protracted and mechanically vulnerable process of "creeping substitution" seen in conventional non-vascularized autografts.
In 1975, Taylor, Miller, and Ham reported the first successful clinical case of a free vascularized bone graft, utilizing a fibula to reconstruct a massive tibial defect where conventional bone grafting techniques had repeatedly failed. Today, this technique is a cornerstone in the treatment of complex orthopaedic pathologies, including massive long bone defects secondary to high-energy trauma, heavy irradiation, wide excision for malignant tumors, and recalcitrant congenital pseudarthrosis of the tibia.
BIOLOGICAL AND BIOMECHANICAL RATIONALE
Conventional non-vascularized cortical bone grafts function primarily as osteoconductive scaffolds. They undergo avascular necrosis, followed by osteoclastic resorption and subsequent osteoblastic bone deposition—a process known as creeping substitution. During this phase, the graft becomes significantly weaker, predisposing it to fatigue fractures.
Conversely, a free vascularized bone transplant maintains its intrinsic blood supply. Osteocytes within the lacunae survive, and the graft retains its original biomechanical strength. Union at the host-graft interface occurs at a rate comparable to normal fracture healing. Furthermore, living bone is capable of hypertrophy in response to mechanical loading (Wolff’s Law), making vascularized grafts particularly suited for lower extremity reconstruction where weight-bearing demands are high. The preserved vascularity also provides superior resistance to infection, making it the gold standard for reconstructing infected nonunions or irradiated tissue beds.
Clinical Pearl: The ability of a vascularized fibula to hypertrophy is remarkable. Within 12 to 18 months of weight-bearing, a transferred fibula can double in diameter, radiographically mimicking the native tibia.
INDICATIONS AND PATIENT SELECTION
The transfer of a vascularized bone graft is indicated when traditional bone grafting techniques are biologically or mechanically insufficient.
Primary Indications
- Massive Bone Defects: Segmental bone loss exceeding 6 cm.
- Hostile Tissue Beds: Defects situated in heavily irradiated areas or zones of chronic osteomyelitis where local vascularity is severely compromised.
- Oncologic Reconstruction: Limb salvage following wide local excision of primary bone sarcomas (e.g., osteosarcoma, Ewing sarcoma) or aggressive benign lesions (e.g., fibrous dysplasia).
- Congenital Deformities: Congenital pseudarthrosis of the tibia or forearm, where intrinsic biological failure prevents normal union.
- Recalcitrant Nonunions: Avascular nonunions of the scaphoid, clavicle, humerus, or tibia that have failed multiple conventional grafting attempts.
Contraindications
Absolute contraindications include severe peripheral vascular disease that precludes safe microvascular anastomosis, active untreated systemic sepsis, and patient inability to tolerate prolonged anesthesia. Relative contraindications include profound venous insufficiency or a lack of adequate recipient vessels.
PREOPERATIVE PLANNING AND IMAGING
Meticulous preoperative planning is the bedrock of microsurgical success. The selection of the donor site is dictated by the precise geometric and biological needs of the recipient site.
Defect Size Algorithm
- Defects ≤ 6 cm: Traditional non-vascularized cancellous or corticocancellous bone grafting techniques generally suffice, provided the soft tissue envelope is well-vascularized.
- Defects 6 to 10 cm: The vascularized iliac crest or a short segment of the vascularized fibula is appropriate.
- Defects > 10 cm: The free vascularized fibula is the undisputed graft of choice due to its linear geometry and available length (up to 25 cm).
Angiography and Templating
High-resolution CT angiography (CTA) or conventional angiography of both the donor and recipient limbs is mandatory. This identifies anomalous vascular anatomy (e.g., a peronea arteria magna where the peroneal artery is the dominant supply to the foot) and confirms the patency of recipient vessels outside the zone of injury.
Surgical Warning: Never harvest a fibula without confirming three-vessel runoff to the foot. Harvesting the peroneal artery in a patient with an aberrant vascular tree can result in catastrophic acute limb ischemia.
The templating technique recommended by Taylor involves superimposing the image of the donor bone (with its vascular pedicle) from the angiogram onto the recipient site radiograph. This allows the surgeon to precisely plan the osteotomy sites, estimate the required pedicle length, and determine the optimal geometry for the microvascular anastomoses.
DONOR SITE SELECTION AND ANATOMY
Three primary sites are considered the best sources of vascularized bone. More recently, the medial femoral condyle has emerged as a highly specialized option for smaller defects.
1. The Free Fibula Flap
The fibula is the workhorse for long bone reconstruction. It provides a long, straight, dense cortical strut.
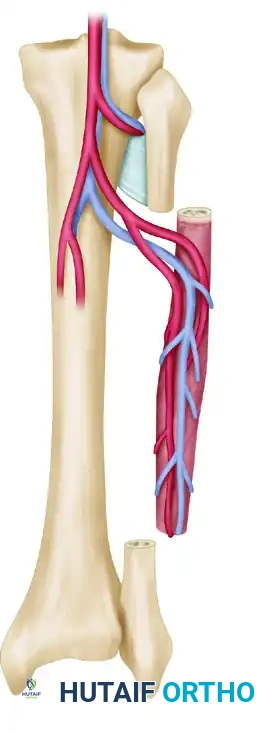
- Blood Supply: The peroneal artery and its venae comitantes. The artery provides both endosteal (nutrient artery) and periosteal (segmental musculoperiosteal branches) blood supply.
- Advantages: Yields up to 25 cm of bone. Can be harvested as an osteocutaneous flap by including a skin paddle based on septocutaneous perforators, allowing for simultaneous soft tissue coverage and postoperative flap monitoring.
- Harvest Considerations: A 0.7 to 1.0 cm muscle cuff (flexor hallucis longus, tibialis posterior, soleus) is retained around the bone to protect the periosteal blood supply. The proximal and distal 6 to 8 cm of the fibula must be preserved to maintain knee and ankle stability, respectively.
2. The Vascularized Iliac Crest
The iliac crest is preferred for shorter bone defects (6 to 10 cm) and is particularly useful for mandibular reconstruction or metaphyseal defects requiring a large volume of cancellous bone.
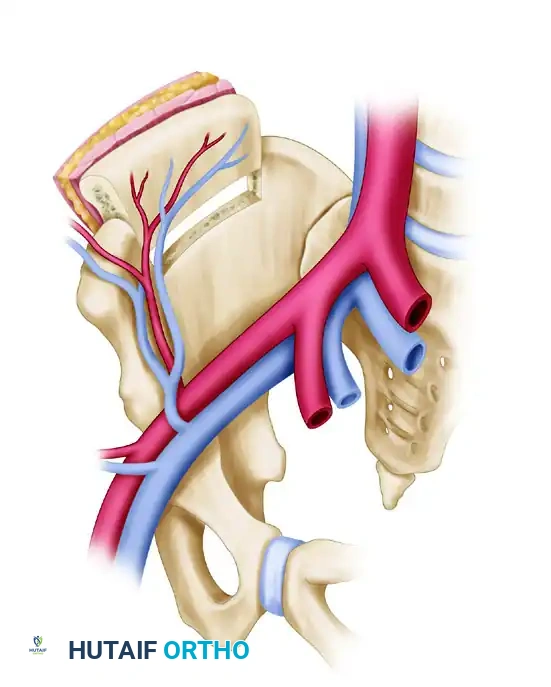
- Blood Supply: The deep circumflex iliac artery (DCIA) and vein.
- Advantages: Provides a natural curve (ideal for the hemimandible or pelvis) and incorporates a massive volume of osteogenic cancellous bone. It can be harvested as a composite flap with the overlying internal oblique muscle or skin.
- Harvest Considerations: The dissection can be tedious, and closure must be meticulous to prevent postoperative abdominal hernias.
3. The Vascularized Rib
The rib is historically significant but less commonly used today for extremity reconstruction due to donor site morbidity. It remains applicable for mandibular reconstruction and composite extremity injuries requiring thin, pliable bone.
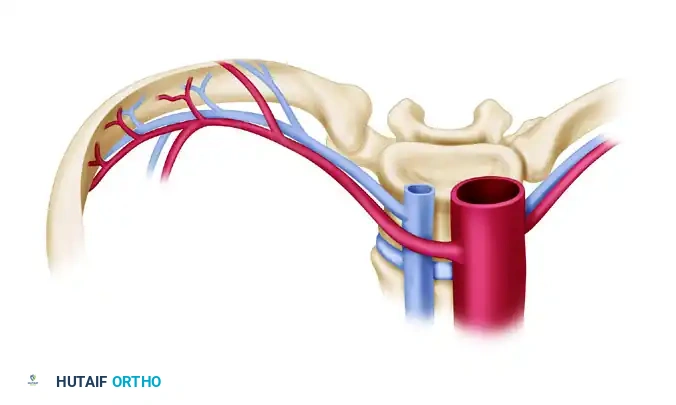
- Blood Supply: The posterior intercostal artery and vein. The vein is typically 1.2 to 2.5 mm in diameter and the pedicle may be 3 to 5 cm in length.
- Advantages: Can include the intercostal nerve, overlying skin, muscle, and an articular surface.
- Disadvantages: The dissection is deep and technically demanding. A physis is unavailable. Complications frequently include pneumothorax, and a tube thoracotomy is often required postoperatively.
4. The Medial Femoral Condyle (MFC) Flap
More recently, free vascularized grafts from the medial femoral condyle, harvested as either thin periosteal or corticoperiosteal grafts, have proven highly efficacious. Based on the descending genicular artery and vein, the MFC flap is the premier choice for treating recalcitrant nonunions of the scaphoid, ulna, metacarpals, clavicle, and humerus.
CONCURRENT SOFT TISSUE AND MUSCLE RECONSTRUCTION
In high-energy trauma, massive bone loss is frequently accompanied by devastating soft tissue and muscle destruction. In these scenarios, a vascularized bone graft alone is insufficient; functional muscle transfer is required to restore motor deficits and provide a robust soft tissue envelope.
The Gracilis muscle is the most commonly utilized functioning free muscle transfer. It can be transferred independently or in conjunction with a bone graft to reconstruct composite defects.

Isolated Gracilis muscle prepared for transfer, demonstrating the neurovascular bundle (medial circumflex femoral artery branches and anterior branch of the obturator nerve).
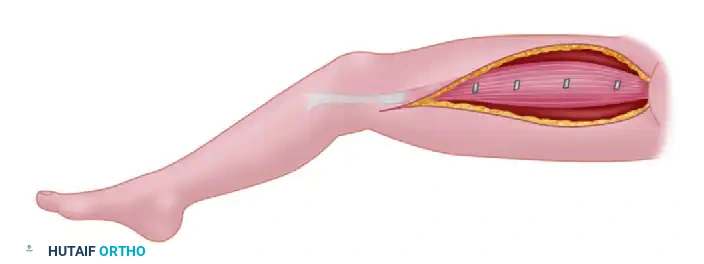
Harvest of the Gracilis muscle from the medial thigh. The muscle is carefully dissected to preserve its primary neurovascular pedicle.
When reconstructing the upper extremity (e.g., restoring elbow flexion or finger flexion), the Gracilis is transferred and revascularized.
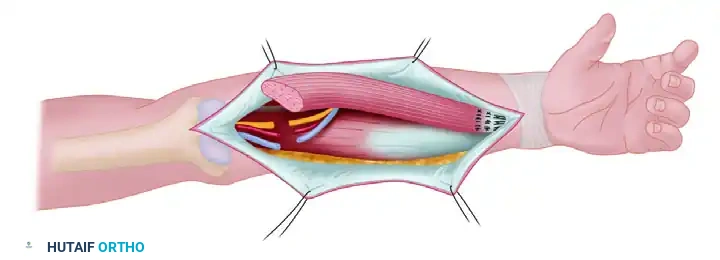
Scheme of transfer: The functioning gracilis muscle is transferred to the recipient forearm. If harvested from the contralateral thigh, it is reversed to match the neurovascular repairs optimally.
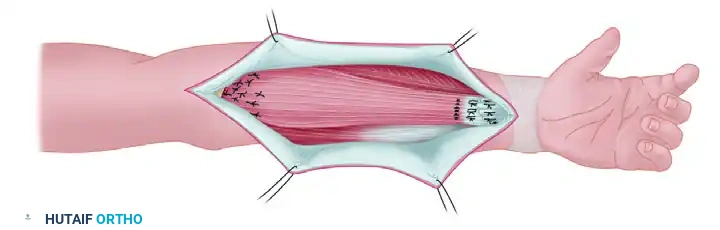
Completed transfer: The muscle is anchored to the fascia and periosteum proximally, while the distal tendon is interwoven with the recipient flexor tendons to restore dynamic function.
SURGICAL TECHNIQUE: THE FREE FIBULA TRANSFER
The procedure is highly complex and is best performed using a two-team approach to minimize ischemia time and overall operative duration.
Step 1: Recipient Site Preparation
The recipient site team radically debrides all necrotic bone, scar tissue, and infected material until healthy, bleeding bone margins are achieved (the "paprika sign"). The recipient artery and vein(s) are identified, dissected free of the zone of injury, and prepared for anastomosis. Rigid skeletal fixation (typically a bridging locking plate or an external fixator) is applied to stabilize the defect, leaving the exact gap ready to receive the graft.
Step 2: Fibula Harvest
The donor team utilizes a lateral approach to the leg.
1. The skin incision is made along the line of the fibula. If a skin paddle is required, it is centered over the posterior border of the fibula to capture the septocutaneous perforators.
2. The lateral compartment musculature (peroneus longus and brevis) is elevated anteriorly.
3. The anterior compartment is elevated, and the interosseous membrane is divided.
4. Proximal and distal osteotomies are performed using an oscillating saw, ensuring at least 6-8 cm of bone remains at the ankle and knee.
5. The fibula is retracted laterally, and the posterior tibialis muscle is divided, leaving a 1 cm cuff of muscle on the fibula to protect the peroneal vessels.
6. The peroneal artery and venae comitantes are traced proximally to their origin at the tibioperoneal trunk to maximize pedicle length.
Step 3: Ischemia and Transfer
Once the recipient site is fully prepared, the pedicle is ligated, and the fibula is transferred. Ischemia time is recorded; while bone is relatively tolerant of ischemia (up to 4-6 hours), minimizing this time reduces the risk of microvascular thrombosis.
Step 4: Inset and Fixation
The fibula is inset into the defect. It can be placed as an intramedullary dowel or onlaid, depending on the diameter of the host bone. Fixation is typically achieved with screws securing the fibula to the host bone, supplemented by the previously placed bridging plate.
Step 5: Microvascular Anastomosis
Under the operating microscope, the arterial anastomosis is performed first, typically using 8-0 or 9-0 nylon sutures in an end-to-end or end-to-side fashion. Venous anastomoses (usually two) are then completed. A venous coupler device is frequently utilized to expedite the venous repair and ensure a perfectly stented, intima-to-intima coaptation.
Pitfall: Redundant pedicle length can lead to kinking, while excessive tension will cause intimal tearing and thrombosis. The pedicle geometry must be perfect prior to clamp release.
POSTOPERATIVE PROTOCOL AND REHABILITATION
Postoperative care is critical to the survival of the free flap.
- Monitoring: The patient is monitored in a specialized microsurgical unit. If a skin paddle was included, its color, capillary refill, and turgor are assessed hourly. An implantable venous Doppler probe is often placed around the venous anastomosis to provide continuous auditory feedback of flow.
- Anticoagulation: Protocols vary by institution but generally include aspirin, low molecular weight heparin (for DVT prophylaxis), and occasionally dextran or heparin infusions, depending on the intraoperative flow characteristics.
- Weight-Bearing: The reconstructed limb is kept strictly non-weight-bearing. Radiographic union at the host-graft junctions typically occurs between 3 to 6 months.
- Hypertrophy Phase: Once union is confirmed, progressive, protected weight-bearing is initiated in a functional brace. This mechanical stress induces fibular hypertrophy. Full unprotected weight-bearing may take 12 to 18 months, depending on the extent of hypertrophy required to match the native bone strength.
COMPLICATIONS
Despite high success rates (>90% in experienced centers), complications can be severe.
* Vascular Thrombosis: Usually occurs within the first 48 hours. Requires emergent re-exploration and thrombectomy.
* Nonunion: Occurs in 10-15% of host-graft junctions, often requiring secondary cancellous bone grafting.
* Stress Fractures: The fibula is prone to stress fractures during the hypertrophy phase before it has achieved sufficient diameter. These are typically treated conservatively with bracing.
* Donor Site Morbidity: Includes transient peroneal nerve palsy, flexor hallucis longus weakness, and ankle instability if the distal osteotomy is performed too low.
===
📚 Medical References
- vascularized bone transplants in problematic nonunions of fractures, J Trauma 23:341, 1983.
- Solonen KA: Free vascularized bone graft in the treatment of pseudarthrosis, Int Orthop 6:9, 1982.
- Soucacos PN, Dailiana Z, Beris AE, et al: Vascularised bone grafts for the management of non-union, Injury 37S:S41, 2006.
- Takagi K, Urist MR: The role of bone marrow in bone morphogenetic protein–induced repair of femoral massive diaphyseal defects, Clin Orthop Relat Res 171:224, 1982.
- Tillman RM, Smith RB: Successful bone grafting of fracture nonunion at the forearm radial fl ap donor site, Plast Surg 90:684, 1992.
- van der Werken C, Marti RK: Bone transplantations, Injury 13:271, 1982.
- Yang RS, Lin WL, Chen YZ, et al: Regulation by ultrasound treatment on the integrin expression and differentiation of osteoblasts, Bone 36:276, 2005.
- Electrical Stimulation Bassett CAL: The development and application of pulsed electromagnetic fi elds (PEMFs) for ununited fractures and arthrodeses, Orthop Clin North Am 15:61, 1984.
- Bassett CAL, Becker RO: Generation of electric potentials in bone in response to mechanical stress, Science 137:1063, 1962.
- Bassett CAL, Mitchell SN, Gaston SR: Treatment of ununited tibial diaphyseal fractures with pulsing electromagnetic fi elds, J Bone Joint Surg 63A:511, 1981.
- Bassett CAL, Mitchell SN, Gaston SR: Pulsing electromagnetic fi eld treatment in ununited fractures and failed arthrodeses, JAMA 247:623, 1982.
- Bassett CAL, Pawluk RJ, Pilla AA: Acceleration of fractures repair by electromagnetic fi elds (a surgically non-invasive method), Ann N Y Acad Sci 238:242, 1974.
- Bassett CAL, Pawluk RJ, Pilla AA: Augmentation of bone repair by inductively coupled electromagnetic fi elds, Science 184:575, 1974.
- Bassett CAL, Pilla AA, Pawluk RJ: A non-operative salvage of surgically resistant pseudarthroses and non-unions by pulsing electromagnetic fi elds: a preliminary report, Clin Orthop Relat Res 124:128, 1977.
- Brighton CT: Current concepts review: the treatment of nonunions with electricity, J Bone Joint Surg 63A:847, 1981.
- Brighton CT: Treatment of nonunion of the tibia with constant direct current (1980 Fitts Lecture, AAST), J Trauma 21:189, 1981.
- Brighton CT: The semi-invasive method of treating nonunion with direct current, Orthop Clin North Am 15:33, 1984.
- Brighton CT, Black J, Friedenberg ZB, et al: A multicenter study of the treatment of non-union with constant direct current, J Bone Joint Surg 63A:2, 1981.
- Brighton CT, Pollack SR: Treatment of nonunion of the tibia with a capacitively coupled electrical fi eld, J Trauma 24:153, 1984.
- Connolly JF: Selection, evaluation and indications for electrical stimulation of ununited fractures, Clin Orthop Relat Res 161:39, 1981.
- Connolly JF: Electrical treatment of nonunions: its use and abuse in 100 consecutive fractures, Orthop Clin North Am 15:89, 1984.
- Day L: Electrical stimulation in the treatment of ununited fractures, Clin Orthop Relat Res 161:54, 1981.
- De Haas WG, Beaupré A, Cameron H, et al: The Canadian experience with pulsed magnetic fi elds in the treatment of ununited tibial fractures, Clin Orthop Relat Res 208:55, 1986.
- De Haas WG, Watson J, Morrison DM: Non-invasive treatment of ununited fractures of the tibia using electrical stimulation, J Bone Joint Surg 62B:465, 1980.
- Esterhai JL Jr, Brighton CT, Heppenstall RB, et al: Detection of synovial pseudarthrosis by 99m-Tc scintigraphy: application to treatment of traumatic nonunion with constant direct current, Clin Orthop Relat Res 161:15, 1981.
- Esterhai JL Jr, Brighton CT, Heppenstall RB, et al: Nonunion of the humerus: clinical, radiographic, scintigraphic, and response characteristics to treatment with constant direct current stimulation of osteogenesis, Clin Orthop Relat Res 211:228, 1986.
- Fontanesi G, Giancecchi F, Rotini R, et al: Treatment of delayed union and pseudarthrosis by low-frequency pulsing electro- magnetic stimulation: study of 35 cases, Ital J Orthop Traumatol 9:305, 1983.
- Garland DE, Moses B, Salyer W: Long-term follow-up of fracture nonunions treated with PEMFs, Contemp Orthop 22:295, 1991.
- Heckman JD, Ingram AJ, Loyd D, et al: Nonunion treatment with pulsed electromagnetic fi elds, Clin Orthop Relat Res 161:58, 1981.
- Heppenstall RB, Brighton CT, Esterhai JL Jr, et al: Clinical and radiographic evaluation of nonunion of the forearm in relation to treatment with d.c. electrical stimulation, J Trauma 23:740, 1983.
- Jacobs RR, Luethi U, Dueland RT, et al: Electrical stimulation of experimental nonunions, Clin Orthop Relat Res 161:146, 1981.
- Lechner F, Ascherl R, Uraus W: Treatment of pseudarthroses with electrodynamic potentials of low frequency range, Clin Orthop Relat Res 161:71, 1981.
- Paterson D: Treatment of nonunion with a constant direct current: a totally implantable system, Orthop Clin North Am 15:47, 1984.
- Paterson DC, Lewis GN, Cass CA: Treatment of delayed union and nonunion with an implanted direct current stimulator, Clin Orthop Relat Res 148:117, 1980.
- Pienkowski D, Pollack SR, Brighton CT, et al: Comparison of asymmetrical and symmetrical pulse waveforms in electromagnetic stimulation, J Orthop Res 10:247, 1992.
- Pollack SR: Bioelectrical properties of bone: endogenous electrical signals, Orthop Clin North Am 15:3, 1984.
- Rijal KP, Kashimoto O, Sakurai M: Effect on capacitively coupled electric fi elds on an experimental model of delayed union of fracture, J Orthop Res 12:262, 1994.
- Scott G, King JB: A prospective, double-blind trial of electrical capacitive coupling in the treatment of non-union of long bones, J Bone Joint Surg 76A:820, 1994.
- Sharp KE, Lightwood R: Stimulation of bone by externally applied radio-frequency energy, Injury 14:523, 1983.
- Sharrard WJW: A double-blind trial of pulsed electromagnetic fi elds for delayed union of tibial fractures, J Bone Joint Surg 72B:347, 1990.
- Sharrard WJW, Sutcliffe ML, Robson MJ, et al: The treatment of fi brous non-union of fractures by pulsing electromagnetic stimulation, J Bone Joint Surg 64B:189, 1982.
- Simonis RB, Good C, Cowell TK: The treatment of non-union by pulsed electromagnetic fi elds combined with a Denham external fi xator, Injury 15:255, 1984.
- Valchanou VD, Michailov P: High energy shock waves in the treatment of delayed and nonunion of fractures, Int Orthop 15:181, 1991.
- Zichner L: Repair of nonunions by electrically pulsed current stimulation, Clin Orthop Relat Res 161:115, 1981.
- Zoltan JD: Electrical stimulation of bone: an overview, Semin Orthop 1:242, 1986.
- Ultrasound Busse JW, Bhandari M, Kulkarni AV, et al: The effect of lowintensity pulsed ultrasound therapy on time to fracture healing: a meta-analysis, Can Med Assoc J 166:437, 2002.
- Mayr E, Frankel V, Rüter A: Ultrasound—an alternative healing method for nonunions, Arch Orthop Trauma Surg 120:1, 2000.
- Rubin C, Bolander M, Ryaby JP, et al: The use of low-intensity ultrasound to accelerate the healing of fractures, J Bone Joint Surg 83A:259, 2001.
- Takikawa S, Matsui N, Kokubu T, et al: Low-intensity pulsed ultrasound initiates bone healing in rat nonunion fracture model, J Ultrasound Med 20:197, 2001.
- Xavier CAM, Duarte LR: Estimulaci ultra-sonica de callo osseo: applicaca clinica, Rev Brasileira Orthop 18:73, 1983.
- Factors Complicating Nonunion (Infection) Cabanela ME: Open cancellous bone grafting of infected bone defects, Orthop Clin North Am 15:427, 1984.
- Calhoun JH, Henry SL, Anger DM, et al: The treatment of infected nonunions with gentamicin-polymethylmethacrylate antibiotic beads, Clin Orthop Relat Res 295:23, 1993.
- Chen CE, Ko JY, Pan CC: Results of vancomycin-impregnated cancellous bone grafting for infected tibial nonunion, Arch Orthop Trauma Surg 125:369, 2005.
- De Oliveira JC: Bone grafts and
