INTRODUCTION TO FAILED SPINE SURGERY
One of the most formidable challenges in both orthopaedic surgery and neurosurgery is the management of failed spine surgery, clinically referred to as Failed Back Surgery Syndrome (FBSS) or Post-Laminectomy Syndrome. Despite advancements in minimally invasive techniques, neuroimaging, and spinal instrumentation, a subset of patients continues to experience persistent, recurrent, or worsened pain following lumbar spine surgery.
The socioeconomic burden of FBSS is immense, leading to prolonged disability, psychological distress, and escalating healthcare costs. The success rates for reoperation are historically variable, reported to range from 31% to 80%. Crucially, as the frequency of repeat back surgeries increases, the probability of a satisfactory clinical outcome decreases precipitously. Therefore, the decision to reoperate must be approached with extreme caution, rigorous diagnostic workup, and a profound understanding of spinal biomechanics and pathoanatomy.
Clinical Pearl: Patients undergoing revision spine surgery must be counseled to expect an improvement in the severity of their symptoms and an enhancement in functional capacity, rather than the complete and total relief of pain. Managing preoperative expectations is as critical as the surgical execution itself.
ETIOLOGY OF SURGICAL FAILURE
Numerous reasons for the failure of index spinal procedures have been advanced in the literature. Spengler et al. and Long et al. observed that the single major cause of failure is improper patient selection. Failures can be broadly categorized into preoperative, intraoperative, and postoperative factors.
Preoperative Factors
- Poor Patient Selection: Operating on patients with predominantly axial back pain without instability, or those with severe psychosocial comorbidities.
- Adverse Psychological Factors: Depression, anxiety, secondary gain, and active workers' compensation claims significantly correlate with poor outcomes.
- Misdiagnosis: Failure to identify the true pain generator (e.g., operating on a radiographic disc bulge when the pain is originating from the sacroiliac joint or hip pathology).
Intraoperative Factors
- Inadequate Decompression: Retained disc fragments or failure to decompress the lateral recess and neural foramen.
- Wrong-Level Surgery: A catastrophic error resulting from inadequate intraoperative fluoroscopic localization.
- Iatrogenic Instability: Excessive resection of the pars interarticularis or facet joints (typically >50% bilateral resection) leading to postoperative spondylolisthesis.
Postoperative Factors
- Recurrent Disc Herniation: Occurs in 5% to 15% of primary discectomies.
- Epidural Fibrosis: Excessive scar tissue formation tethering the nerve root.
- Pseudarthrosis: Failure of fusion mass consolidation, leading to hardware loosening and micromotion.
- Adjacent Segment Pathology (ASP): Accelerated degeneration of the disc and facets at the levels immediately cranial or caudal to a fusion construct.
CLINICAL EVALUATION AND PREDICTORS OF SUCCESS
The recurrence or intensification of pain in the subacute or late period after disc surgery should initially be treated with standard conservative modalities, including physical therapy, NSAIDs, and targeted epidural steroid injections. If these methods fail to relieve the pain, the patient requires a comprehensive reevaluation.
The Pain-Free Interval
A meticulous history is paramount. Waddell et al. reported that the best results from repeat surgery occur under specific historical conditions:
1. The patient experienced 6 months or more of complete pain relief after the first procedure.
2. The patient's radicular (leg) pain exceeds their axial (back) pain.
3. A definite, recurrent disc herniation can be identified on imaging.
Conversely, Waddell, Lehmann, LaRocca, and Finnegan et al. identified several adverse factors that portend a poor prognosis for revision surgery. These include extensive epidural scarring, previous postoperative infection, repair of pseudarthrosis, and adverse psychological factors.
Physical Examination
A repeat physical examination must differentiate between radiculopathy, myelopathy, and mechanical back pain.
* Neurological Deficits: Document any new or progressive motor weakness, sensory deficits, or reflex asymmetry.
* Tension Signs: A positive straight leg raise (SLR) or femoral nerve stretch test strongly suggests recurrent herniation or severe root tethering.
* Palpation and ROM: Assess for palpable step-offs, severe paraspinal spasm, and pain elicited on extension (suggestive of facet arthropathy or pseudarthrosis).
ADVANCED DIAGNOSTIC IMAGING AND MODALITIES
A distinct, surgically correctable, anatomical problem must be identified before revision surgery is contemplated. The surgery must be tailored specifically to the anatomical problem identified.
Magnetic Resonance Imaging (MRI)
MRI with and without intravenous gadolinium contrast is the gold standard for evaluating the failed spine.
* Scar vs. Disc: Gadolinium enhances vascularized epidural fibrosis (scar tissue), whereas a recurrent avascular disc fragment will not enhance (though it may show a peripheral rim of enhancement).
Computed Tomography (CT)
Reformatted CT scans (sagittal and coronal planes) are essential for evaluating bony anatomy.
* Indications: Assessing for areas of foraminal stenosis, lateral extra-foraminal herniations, retained osteophytes, and evaluating the integrity of a previous fusion mass (pseudarthrosis).
Diagnostic Injections and Blocks
When imaging is equivocal, diagnostic injections help isolate the pain generator:
* Selective Nerve Root Blocks (SNRB): Can confirm if a specific compressed root is the source of radicular pain.
* Facet Blocks / Medial Branch Blocks: Useful for diagnosing facet-mediated axial pain.
* Discography: Provocative discograms can help identify internal disc derangement or symptomatic adjacent segment disease, though their use remains controversial.
* Differential Spinal Blocks: Can help differentiate somatic pain from sympathetic or psychogenic pain.
Surgical Warning: The presence of abnormal psychological test results or an abnormal differential spinal block should serve as a strict modifier to any suggested surgical treatment indicated by other testing. In these scenarios, multidisciplinary pain management is preferred over reoperation.
BIOMECHANICS AND MOTION PRESERVATION: TOTAL DISC REPLACEMENT
In cases where the primary cause of failure is internal disc derangement or adjacent segment disease without gross instability, motion-preserving technologies have been utilized as an alternative to rigid fusion. The goal is to restore disc height, decompress the neural elements indirectly, and maintain segmental kinematics to protect adjacent levels.
The Charité Total Disc Replacement (TDR) represents a historical milestone in this biomechanical approach. It consists of two metallic endplates (typically cobalt-chromium-molybdenum) and a mobile ultra-high-molecular-weight polyethylene (UHMWPE) sliding core.
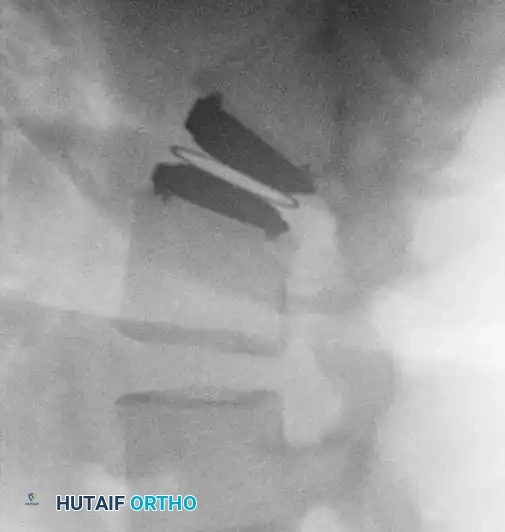
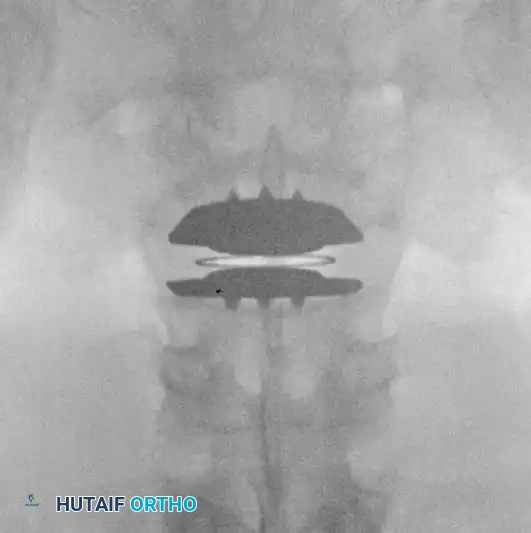
Fig. 39-47 A and B, Anteroposterior and lateral views of a patient with internal disc derangement treated with Charité total disc replacement.
Biomechanical Rationale for TDR
- Restoration of Lordosis: The angled endplates help recreate the natural lumbar lordosis, which is often lost in degenerative disc disease.
- Kinematic Preservation: By allowing flexion, extension, lateral bending, and axial rotation, TDR theoretically reduces the abnormal stress transferred to adjacent segments, a common cause of late failure in rigid fusions.
- Indications: Strictly limited to patients with isolated 1- or 2-level discogenic pain who have failed at least 6 months of conservative care, without significant facet arthropathy, osteoporosis, or instability.
SURGICAL APPROACH TO THE PREVIOUSLY OPERATED SPINE
Revision spine surgery is technically demanding due to the loss of normal anatomical landmarks and the presence of dense epidural fibrosis.
1. Positioning and Preparation
- The patient is positioned prone on a radiolucent Jackson table or Wilson frame.
- The abdomen must hang completely free to decrease intra-abdominal pressure, thereby reducing epidural venous engorgement and intraoperative bleeding.
- Intraoperative neuromonitoring (Somatosensory Evoked Potentials [SSEPs] and Electromyography [EMG]) is highly recommended.
2. Incision and Exposure
- The previous surgical incision is utilized but must be extended proximally and distally to expose "virgin" (unoperated) anatomy.
- Dissection is carried down through the subcutaneous tissues. The fascial incision should be made cleanly, avoiding the scarred midline if possible.
- Subperiosteal Dissection: Muscle stripping must begin on normal, unoperated bone (e.g., the spinous process or lamina of the level above or below the previous surgery) and proceed toward the scarred defect.
3. Navigating Epidural Fibrosis (The "Outside-In" Approach)
- Never dissect directly into the center of the surgical scar, as the dura or nerve roots may be tethered superficially.
- Identify the normal dura proximally and distally to the previous laminectomy defect.
- Identify the medial border of the pedicle and the pars interarticularis; these are reliable, constant bony landmarks.
- Using micro-curettes and Kerrison rongeurs, carefully detach the scar tissue from the bony margins.
- Do not pull on the scar. Epidural fibrosis is often densely adherent to the dura. Sharp dissection with a #15 blade or micro-scissors is safer than blunt avulsion, which can easily cause a cerebrospinal fluid (CSF) leak.
4. Decompression and Root Mobilization
- Once the normal dura is identified, follow the nerve root out into the foramen.
- If a recurrent disc herniation is present, it is often located ventral to the scarred nerve root. The root must be gently retracted medially. If the root is immobile due to scarring, a wider bony decompression (complete facetectomy) may be required to access the disc fragment safely.
5. Revision Fusion and Instrumentation
If the revision is for pseudarthrosis, iatrogenic instability, or requires extensive facet resection for decompression, a stabilization procedure is mandatory.
* Pedicle Screw Fixation: In revision settings, previous hardware may need to be removed. If screws are loose, larger diameter "rescue" screws or a different trajectory (e.g., cortical bone trajectory) must be utilized.
* Interbody Fusion: Posterior Lumbar Interbody Fusion (PLIF) or Transforaminal Lumbar Interbody Fusion (TLIF) provides anterior column support, restores foraminal height, and increases fusion rates.
* Bone Grafting: Autologous iliac crest bone graft (ICBG) remains the gold standard, often supplemented with local bone and osteoinductive biologics (e.g., BMP-2) given the compromised healing environment of a previously operated bed.
POSTOPERATIVE PROTOCOLS
The postoperative management of a revision spine patient requires a multidisciplinary approach.
- Immediate Postoperative Care: Neurological checks are performed every 2-4 hours. A closed suction drain is typically left in place for 24-48 hours to prevent epidural hematoma formation.
- Mobilization: Early mobilization is encouraged to prevent deep vein thrombosis (DVT) and pulmonary complications. Patients are usually mobilized on postoperative day 1 with physical therapy.
- Bracing: Depending on the stability of the construct and bone quality, a rigid Thoracolumbosacral Orthosis (TLSO) may be prescribed for 6 to 12 weeks.
- Rehabilitation: Formal physical therapy begins at 6 weeks, focusing initially on isometric core strengthening and progressing to dynamic stabilization and functional restoration.
COMPLICATIONS AND MANAGEMENT
Revision spine surgery carries a significantly higher complication rate than primary surgery.
- Incidental Durotomy (Dural Tear): The incidence is markedly increased due to epidural scarring. If a tear occurs, it must be repaired primarily with 4-0 or 5-0 non-absorbable suture (e.g., Prolene or Nurolon). The repair is often augmented with a dural sealant or fascial patch. Postoperatively, the patient may be kept flat for 24-48 hours to reduce hydrostatic pressure on the repair.
- Nerve Root Injury: Excessive retraction of a scarred, tethered nerve root can lead to neuropraxia or permanent axonotmesis. Meticulous sharp dissection and adequate bony decompression are the best preventative measures.
- Infection: The risk of surgical site infection (SSI) is elevated due to compromised local vascularity and prolonged operative times. Strict adherence to sterile technique, copious intraoperative irrigation, and appropriate prophylactic antibiotics are essential.
CONCLUSION
The treatment of failed spine surgery demands a rigorous, evidence-based approach. The fundamental tenet of revision surgery is that a distinct, surgically correctable anatomical lesion must be identified that correlates directly with the patient's clinical symptoms. Through meticulous preoperative evaluation, advanced imaging, and masterful surgical technique, the orthopaedic surgeon can navigate the hostile environment of the previously operated spine to restore stability, decompress neural elements, and improve the patient's quality of life. However, conservative management and psychological optimization must always remain the first line of defense against the complexities of Failed Back Surgery Syndrome.
