ELBOW TENDINOPATHIES: LATERAL EPICONDYLITIS
Lateral epicondylitis, universally recognized by the colloquialism "tennis elbow," represents a myriad of symptoms localized to the lateral aspect of the elbow. Despite its moniker, the condition occurs with significantly higher frequency in non-athletes than in the athletic population. Epidemiological data demonstrate a peak incidence in the early fifth decade of life, with a nearly equal distribution between genders.
The pathomechanics of lateral epicondylitis are intimately tied to activities requiring repetitive supination and pronation of the forearm while the elbow is in near-full extension. First described as a distinct clinical entity by Runge in 1873, the etiology of tennis elbow has been the subject of extensive debate, with nearly 30 different conditions proposed over the past century.
Pathophysiology and Biomechanics
Although historically mischaracterized as an inflammatory process (hence the suffix "-itis"), modern histopathological consensus confirms that lateral epicondylitis is a degenerative tendinopathy. The process is initiated by microtears, predominantly within the origin of the extensor carpi radialis brevis (ECRB).
Clinical Pearl: The ECRB is uniquely susceptible to microtrauma due to its biomechanical role. It is the primary stabilizer of the wrist during power grip, and its origin intimately overlies the capitellum, subjecting it to sheer forces during repetitive elbow flexion and extension.
Microscopic evaluation of the diseased tissue reveals an aborted healing response characterized by immature reparative tissue, disorganized collagen, and a lack of traditional inflammatory cells. This histological picture was famously termed angiofibroblastic hyperplasia by Nirschl. While the pathological epicenter is the ECRB origin, the degenerative process can extend to involve the anterior margins of the extensor digitorum communis (EDC) and, less frequently, the extensor carpi radialis longus (ECRL).
CLINICAL EVALUATION AND DIAGNOSIS
The diagnosis of lateral epicondylitis is primarily clinical, relying on a meticulous history and targeted physical examination.
Physical Examination
The hallmark of tennis elbow is exquisite point tenderness localized precisely to the origin of the ECRB. This anatomical focal point is typically found approximately 5 mm distal and anterior to the midpoint of the lateral epicondyle.
Provocative testing is highly sensitive:
* Cozen's Test: Pain is exacerbated by resisted wrist dorsiflexion with the elbow in extension.
* Mill's Test: Passive stretching of the extensor mass (elbow extension, forearm pronation, and wrist flexion) reproduces lateral pain.
* Maudsley's Test: Resisted extension of the long finger reproduces pain, highlighting the involvement of the ECRB and EDC.
* Grip Strength: Patients frequently report sharp pain and subjective weakness when grasping objects, often quantified by a significant discrepancy in grip strength testing with the elbow extended versus flexed.
Imaging Modalities
Plain radiographs are typically unremarkable but are essential to rule out intra-articular pathology, such as osteochondritis dissecans of the capitellum or lateral compartment arthrosis. Occasionally, dystrophic calcification (calcific tendinitis) may be visualized within the extensor origin.
Magnetic Resonance Imaging (MRI) is the gold standard for advanced imaging. It typically demonstrates tendon thickening with increased signal intensity on both T1 and T2-weighted sequences.
Evidence-Based Insight: Literature indicates a strong correlation between surgical success and the presence of a high-signal intensity focus on T2-weighted MRI within the ECRB at the lateral epicondyle. This focal hyperintensity represents the core of the angiofibroblastic degeneration.
Differential Diagnosis
A rigorous differential diagnosis is paramount, as misdiagnosis is the leading cause of failed tennis elbow surgery. The differential includes:
1. Osteochondritis Dissecans (OCD) of the Capitellum: Common in adolescent throwing athletes or gymnasts.
2. Lateral Compartment Arthrosis: Characterized by mechanical symptoms, crepitus, and radiographic joint space narrowing.
3. Posterolateral Rotatory Instability (PLRI): Often secondary to prior trauma or iatrogenic injury to the lateral ulnar collateral ligament (LUCL).
4. Radial Tunnel Syndrome: The most common confounding diagnosis.
Radial Tunnel Syndrome vs. Lateral Epicondylitis
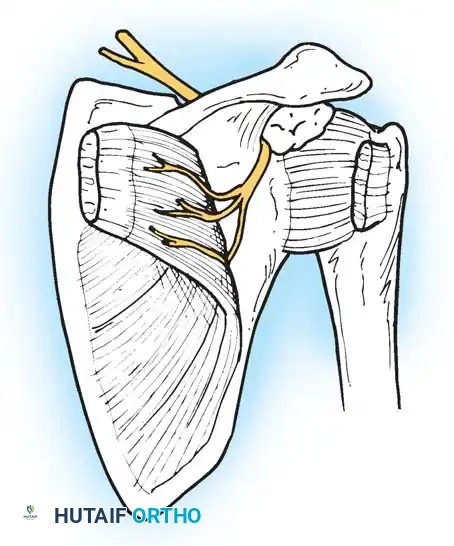
Radial tunnel syndrome is a compressive neuropathy of the posterior interosseous nerve (PIN). Compression can occur at four distinct anatomical structures within the radial tunnel:
1. A fibrous band anterior to the radiocapitellar joint.
2. The vascular leash of Henry (recurrent radial artery).
3. The fibrous distal margin of the ECRB.
4. The arcade of Frohse (proximal edge of the superficial supinator).
Clinically, the pain of radial tunnel syndrome is located 3 to 4 cm distal to the lateral epicondyle, deep within the mobile wad. While resisted long finger extension may reproduce pain, this finding is inconsistent. Electromyography (EMG) is notoriously unreliable for confirming radial tunnel syndrome. It is critical to note that true lateral epicondylitis and radial tunnel syndrome coexist in approximately 5% of patients.
NONOPERATIVE MANAGEMENT
Regardless of the underlying etiology, conservative management is highly efficacious, yielding successful outcomes in up to 95% of patients.
First-Line Modalities
Initial treatment focuses on mitigating pain and promoting tissue healing:
* Activity Modification: Rest and avoidance of repetitive gripping/pronation.
* Counterforce Bracing: Application of a strap distal to the epicondyle to disperse mechanical forces away from the ECRB origin.
* Physical Therapy: Modalities include ultrasound, iontophoresis (often with dexamethasone), high-voltage galvanic stimulation, and deep friction massage. A structured program of stretching followed by eccentric strengthening is the cornerstone of rehabilitation.
Injections and Orthobiologics
- Corticosteroid Injections: One or two local injections of a steroid preparation directed at the area of maximal tenderness can provide profound short-term relief. Porretta and Janes reported that 40% of patients obtained complete and permanent relief. However, multiple injections are discouraged due to the risk of tendon atrophy and skin depigmentation.
- Autologous Blood and PRP: Autologous blood injections and Platelet-Rich Plasma (PRP) have shown significant benefit in recalcitrant cases. Edwards and Calandruccio reported a 79% success rate in relieving pain in patients who had failed all other nonoperative treatments.
- Extracorporeal Shock Wave Therapy (ESWT): While early studies were promising, recent randomized controlled trials have shown conflicting results, with some studies finding no meaningful difference between ESWT combined with stretching versus stretching alone.
Surgical Warning: Patients who fail to respond to a comprehensive 6- to 12-month nonoperative regimen should be carefully scrutinized. Coonrad and Hooper strongly recommended evaluating for sources of secondary gain (e.g., worker's compensation, litigation) using tools like the Minnesota Multiphasic Personality Inventory (MMPI) before offering surgical intervention.
OPERATIVE MANAGEMENT
Surgical intervention is indicated for the 5% to 10% of patients who remain highly symptomatic after 6 to 12 months of exhaustive conservative care. When patients are properly selected, operative treatment is effective in 90% of cases.
Historically, procedures such as manipulation under anesthesia (advocated by Mills and Wadsworth) were utilized to forcibly rupture the diseased tendon, eliciting an audible snap. Similarly, the Boyd and McLeod technique involved a massive release of the entire extensor origin, excision of the annular ligament, and resection of radiocapitellar synovium.
Currently, the gold standard is a targeted, limited approach focusing on the excision of the pathological ECRB tissue and stimulation of a localized healing response.
🔪 Surgical Technique 44-11: Open ECRB Debridement and Repair (Nirschl Technique)
Positioning and Anesthesia: The patient is placed supine with the operative arm draped free on a hand table. A regional block (supraclavicular or axillary) combined with monitored anesthesia care is preferred. A sterile tourniquet is applied to the proximal arm.
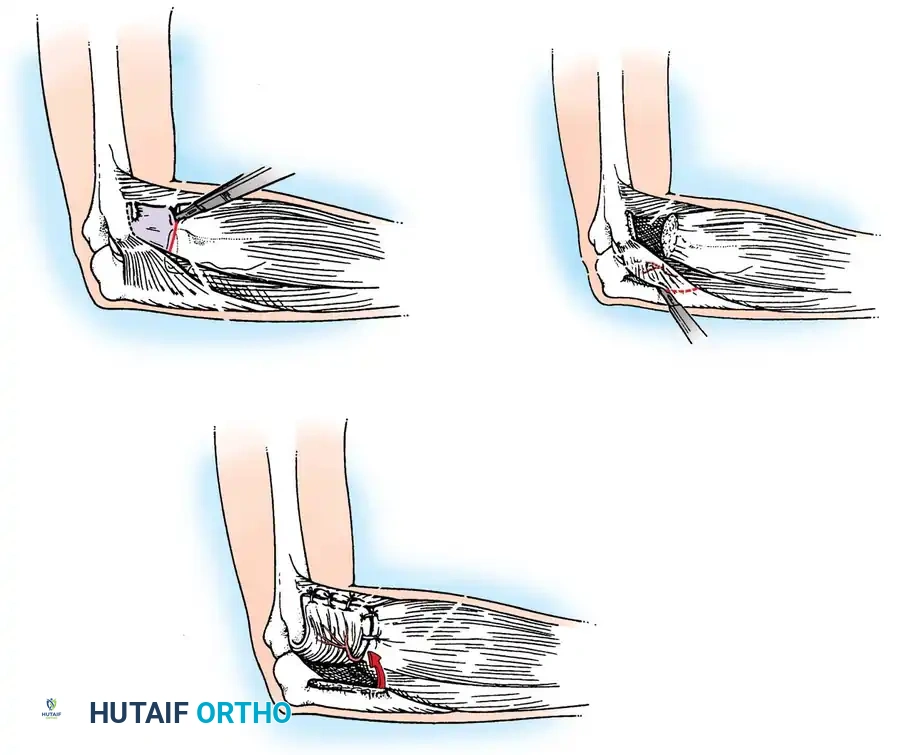
Step 1: Surgical Approach
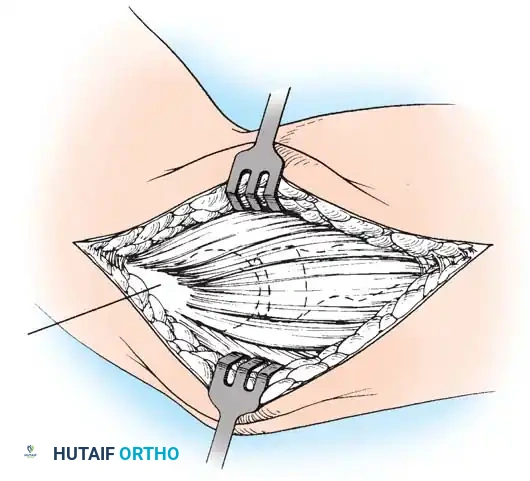
* Make a gently curved incision, approximately 5 cm in length, centered directly over the lateral epicondyle.
* Incise the deep fascia in line with the skin incision and retract the fascial flaps.
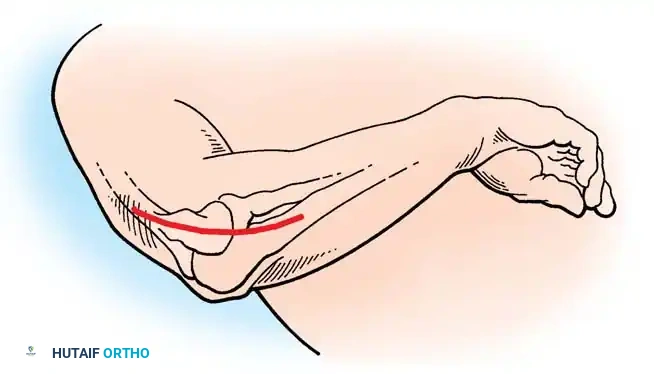
Step 2: Interval Identification
* Identify the interval between the extensor carpi radialis longus (ECRL) and the origin of the extensor digitorum communis (EDC). The EDC partially obscures the deeper ECRB origin.
* Bluntly separate the ECRL and EDC to expose the underlying ECRB.
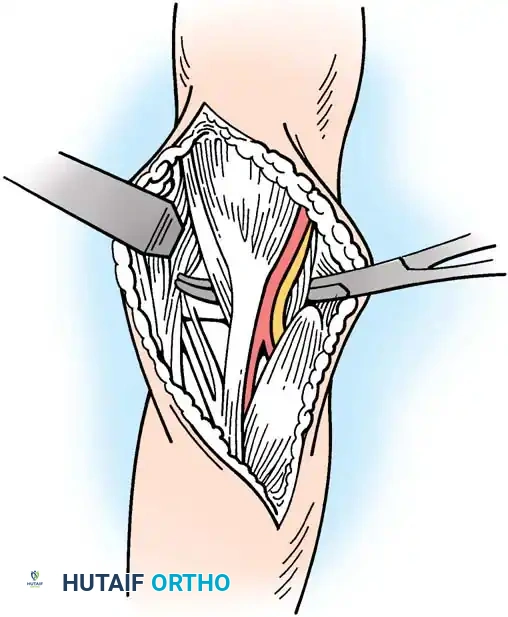
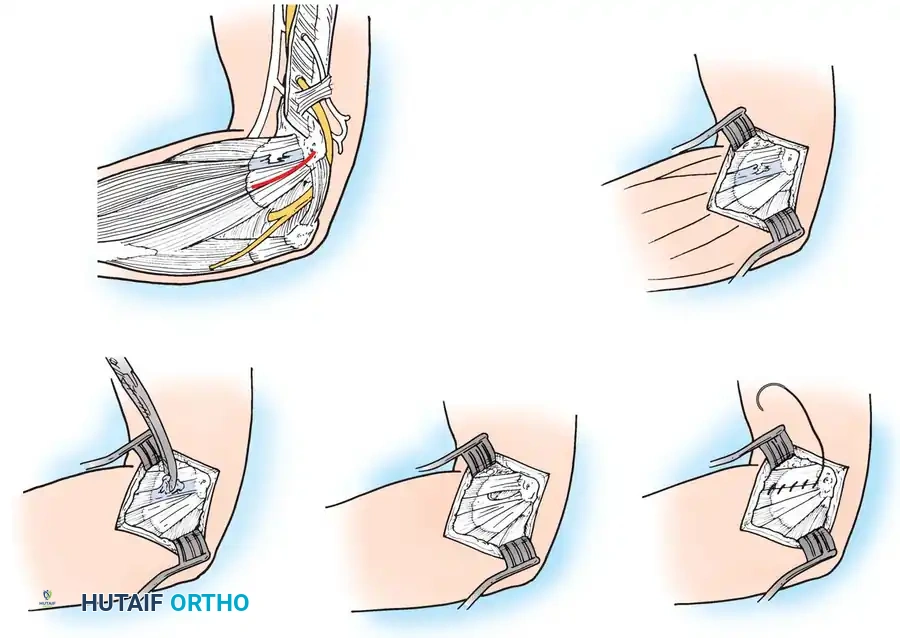
Step 3: Tendon Elevation and Excision
* Elevate the brevis portion of the conjoined tendon at the midportion of the lateral epicondyle, working proximally toward the elbow joint.
* Identify the pathological tissue. The diseased angiofibroblastic tissue typically appears grayish, fibrillated, edematous, and lacks the normal glistening appearance of healthy Sharpey's fibers. Calcium deposits may occasionally be encountered.
* Excise the abnormal-appearing tendon meticulously. If the degenerative process extends into the EDC origin, a portion of the anterior EDC may also be excised.
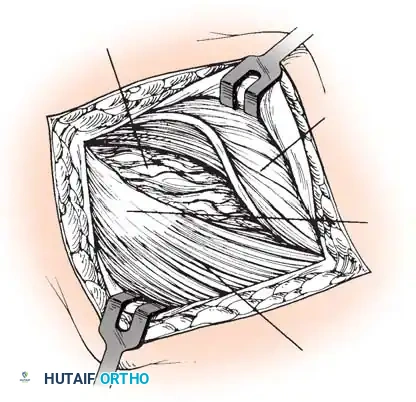
Surgical Pitfall: Do not arbitrarily enter the radiocapitellar joint. Capsulotomy is only indicated if preoperative imaging or clinical examination suggests intra-articular pathology, such as a loose body, synovial plica, or advanced degenerative joint disease.
Step 4: Epicondylar Decortication
* Once the diseased tendon is removed, decorticate a small, localized area of the anterior lateral epicondyle using a rongeur or a sharp osteotome.
* The goal is to expose bleeding cancellous bone to stimulate a vascular healing response. Take extreme care not to plunge distally into the joint, which could damage the capitellar articular cartilage.
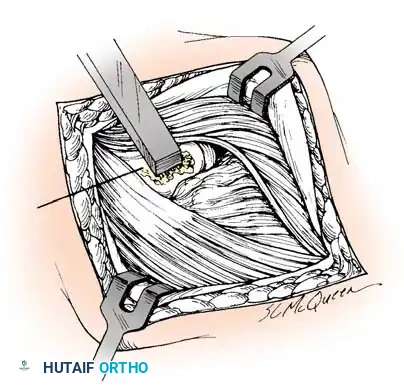
Step 5: Tendon Repair and Closure
* Suture the remaining healthy ECRB tendon margin to the adjacent extensor aponeurosis or periosteum.
* Alternatively, the tendon can be repaired directly to the decorticated bone using nonabsorbable sutures passed through transosseous drill holes or via small suture anchors. While suture anchors provide excellent biomechanical fixation, many surgeons find soft-tissue-to-soft-tissue closure sufficient.
* Close the ECRL and EDC interval with interrupted absorbable sutures. If anchors were used, this closure effectively buries the knots.
* Close the skin with a running subcuticular 4-0 nylon or Monocryl suture, reinforced with adhesive strips.
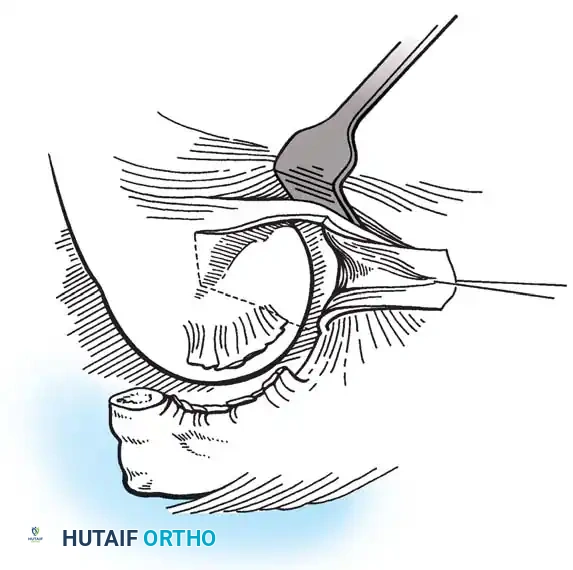
🔪 Surgical Technique 44-12: Epicondylar Resection and Anconeus Muscle Transfer (Almquist Procedure)
For patients with chronic, recalcitrant lateral epicondylitis who have failed previous surgical interventions, or those with massive tissue defects following debridement, Almquist et al. described an epicondylar resection combined with an anconeus muscle transfer. This technique provides robust, vascularized tissue to the lateral compartment and boasts a 94% good-to-excellent outcome rate in salvage scenarios.
Step 1: Exposure and Resection
* Under tourniquet control, make a 5-cm lateral incision over the epicondyle, extending slightly more distally than the standard Nirschl approach.
* Dissect subcutaneously to identify the anconeus muscle origin on the posterior border of the ulna and its insertion on the lateral epicondyle.
* Divide the aponeurosis longitudinally to define the deep structures.
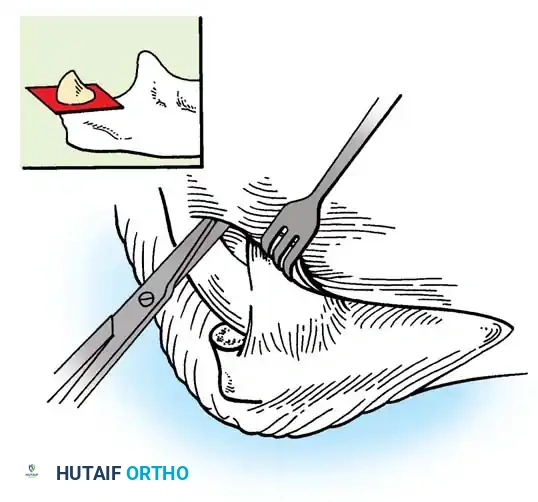
Step 2: En Bloc Excision
* Perform an en bloc excision of the superficial and deep pathological structures.
* The resection boundaries are: anteriorly to the fleshy border of the ECRL; distally to the fleshy portions of the ECRB and EDC at the level of the radial head; posteriorly to the firm aponeurosis of the extensor carpi ulnaris (ECU); and proximally to the lateral epicondyle.
* This excision typically removes approximately 4 cm² of diseased aponeurosis.
* Excise the superficial supinator aponeurosis. The radial nerve is not routinely exposed during this step.
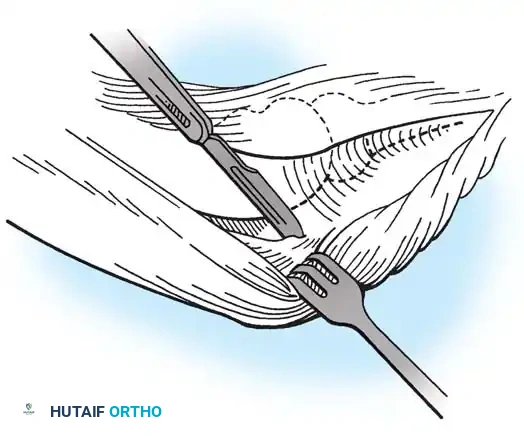
Critical Anatomy Warning: During the deep dissection and en bloc excision, meticulous care must be taken to avoid injury to the Lateral Ulnar Collateral Ligament (LUCL). Iatrogenic transection of the LUCL will result in devastating posterolateral rotatory instability (PLRI).
Step 3: Bone Preparation and Ligament Assessment
* If the lateral epicondyle is highly sclerotic, use a rongeur to decorticate it down to healthy, bleeding cancellous bone.
* If the LUCL is found to be completely avulsed or inadvertently compromised, it must be addressed prior to the muscle transfer. Utilize the firm fascia superficial to the anconeus and a portion of the triceps aponeurosis as a free graft. Suture this graft into the capsular margin of the exposed joint using heavy absorbable sutures to restore stability.
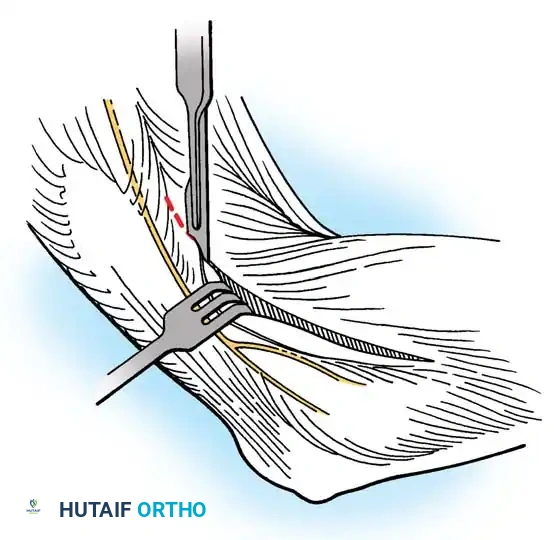
Step 4: Anconeus Mobilization and Transfer
* Mobilize the anconeus muscle by sharply releasing its broad origin on the ulna distally. Dissect proximally with extreme caution.
* Vascular Preservation: Leave the vascular leash (which runs parallel to and on the surface of the ulna, deep to the anconeus) attached to the periosteum. Elevate the muscle fibers from the ulna until the neurovascular bundle is encountered. This bundle is located just proximal and posterior to the orbicular (annular) ligament, between the radius and ulna.
* Mobilize the posterior margin of the anconeus to create a 5- to 7-cm mobile muscle pedicle.
* Rotate this vascularized anconeus flap anteriorly into the large defect created by the excision of the extensor aponeurosis. The anconeus muscle fibers will lie slightly oblique to the native wrist extensor fibers.
* Secure the anconeus muscle into its new bed using interrupted absorbable mattress sutures.
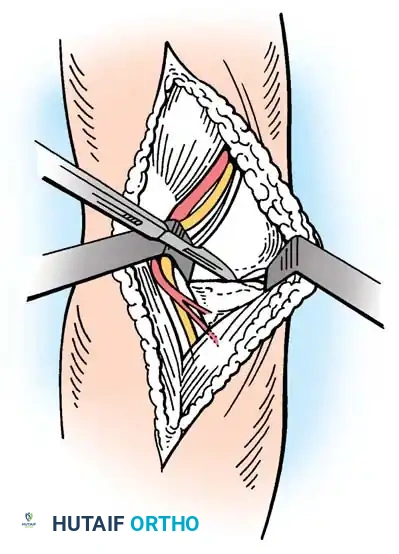
MANAGEMENT OF THE FAILED TENNIS ELBOW SURGERY
Despite high success rates, 1% to 2% of patients will fail surgical intervention. Morrey elegantly classified these failures into two distinct groups based on postoperative symptom presentation, which dictates the salvage strategy.
Group 1: Unchanged Symptoms
Patients in this cohort report symptoms identical to their preoperative state. The etiology of failure is almost universally an inadequate surgical release of the ECRB or an incorrect initial diagnosis. The most common missed diagnosis in this group is concomitant Radial Tunnel Syndrome.
Group 2: New Symptom Complex
Patients in this cohort report a different type of pain, often characterized by mechanical catching, clicking, or a sensation of the elbow "giving way" during extension and supination. The etiology here is iatrogenic capsular or ligamentous insufficiency—specifically, overzealous surgical release that violated the anterior band of the lateral collateral ligament complex (LUCL), resulting in Posterolateral Rotatory Instability (PLRI) or a synovial fistula.
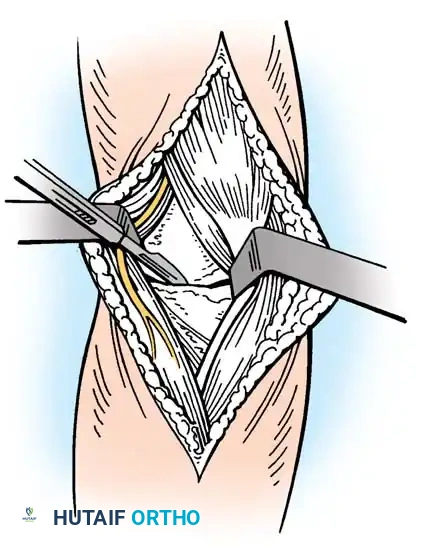
Revision Strategy:
Morrey emphasized the critical importance of a thorough history to distinguish between Group 1 and Group 2 failures. Physical examination must specifically assess for PLRI (Pivot-Shift test of the elbow) and PIN compression.
* Diagnostics should be supplemented with MR Arthrograms to detect synovial fistulas or capsular insufficiency.
* Diagnostic arthroscopy under anesthesia is invaluable for directly visualizing instability or occult arthrosis.
* Timing: Most patients who will improve postoperatively do so within 3 to 4 months. Posch et al. noted that improvement beyond 1 year is exceedingly rare. Therefore, 1 year is considered a reasonable threshold to offer revision surgery if symptoms remain debilitating. In Morrey’s series of 13 failed primary releases, revision surgery (addressing either the missed nerve compression or reconstructing the LUCL) was successful in 11 patients.
POSTOPERATIVE REHABILITATION PROTOCOL
The rehabilitation following lateral epicondyle surgery is goal-dependent rather than strictly time-dependent. Patients must meet specific clinical milestones before advancing to the next phase.
Phase 1: Acute Phase (Weeks 0–2)
- Immobilization: The elbow is placed in a posterior splint at 90 degrees of flexion with the wrist in neutral to slight extension. The splint is typically removed within the first 5 to 7 days.
- Goals: Reduce inflammation, protect the repair, and retard muscular atrophy.
- Interventions:
- Cryotherapy and edema control.
- Gentle active-assisted range of motion (AAROM) for wrist extension/flexion and elbow extension/flexion.
*
