Mastering the Pathophysiology and Clinical Evaluation of Peripheral Nerve Injuries
Key Takeaway
Peripheral nerve injuries initiate a complex cascade of motor, sensory, and autonomic deficits. Understanding the timeline of Wallerian degeneration, muscle atrophy, and sensory recovery sequences is paramount for orthopedic surgeons. This guide details the pathophysiology, clinical evaluation using BMRC grading, and the recognition of complex regional pain syndrome (CRPS) to optimize surgical timing, microsurgical repair, and postoperative rehabilitation.
INTRODUCTION TO PERIPHERAL NERVE PATHOPHYSIOLOGY
The severance or severe crush of a peripheral nerve initiates a profound and highly predictable sequence of degenerative and regenerative events. For the reconstructive orthopedic surgeon or hand specialist, a masterful understanding of these physiological changes is not merely academic; it is the foundation upon which surgical timing, operative strategy, and prognostic accuracy are built. When a peripheral nerve is injured, the distal segment undergoes Wallerian degeneration, leading to a complete cessation of motor, sensory, and autonomic function within the nerve's specific anatomical distribution.
This comprehensive guide delineates the multifaceted effects of peripheral nerve injuries, providing an evidence-based framework for clinical evaluation, surgical decision-making, and postoperative management.
MOTOR EFFECTS AND MUSCLE DEGENERATION
When a peripheral nerve is severed at a specific anatomical level, all motor function mediated by the nerve distal to that level is instantaneously abolished. The skeletal muscles innervated by the distal branches become acutely paralyzed and flaccid (atonic).
Electromyographic (EMG) Evolution
The denervated muscle undergoes a series of electrophysiological changes that are critical for diagnostic mapping.
* 0 to 7 Days: Immediately following the injury, the muscle remains electrically silent at rest, though voluntary motor unit action potentials (MUAPs) are absent.
* 8 to 14 Days: Significant electromyography (EMG) changes begin to manifest. Transient fibrillation potentials upon needle insertion become apparent, reflecting the instability of the muscle fiber membrane as acetylcholine receptors begin to spread extrajunctionally.
* 2 to 4 Weeks: Spontaneous fibrillations and positive sharp waves become definitively evident. This electrophysiological milestone coincides with the onset of profound atrophic changes within the muscle fibers.
The Timeline of Muscle Atrophy
Denervation atrophy progresses at an alarming rate. According to Sunderland’s foundational research, the atrophy of muscle bulk progresses rapidly, losing 50% to 70% of its mass by the end of approximately two months.
Clinical Pearl: While muscle fiber atrophy is rapid initially, it continues at a much slower rate thereafter. Concurrently, the connective tissue component (fibrosis) within the muscle increases, leading to increased stiffness and loss of compliance.
Striations and motor end-plate configurations are remarkably resilient, often retained for longer than 12 months. However, the empty endoneurial tubes shrink to approximately one-third of their normal diameter, complicating delayed surgical coaptation. Complete disruption and fibrotic replacement of muscle fibers may not become absolute until after 3 years, establishing the critical "biological clock" for nerve repair or tendon transfer interventions.
Assessment of Motor Recovery
Evaluating motor return requires meticulous, serial examinations. Standardized assessments involve testing muscle strength against gravity and graded resistance, supplemented by pinch meters, grip dynamometers, and evaluations of endurance and speed.
The British Medical Research Council (BMRC) established the universally adopted grading system for assessing the return of muscle function:
* M0: No contraction has returned.
* M1: Perceptible contraction (flicker or trace) in proximal muscles has returned.
* M2: Perceptible contraction in proximal and distal muscles has returned, but cannot overcome gravity.
* M3: All important muscles act against resistance (can move through full range of motion against gravity).
* M4: All synergistic and independent movements are possible; muscle acts against moderate resistance.
* M5: Recovery is complete; normal strength.
SENSORY EFFECTS AND RECOVERY SEQUENCES
Sensory loss following nerve transection follows a definitive anatomical pattern. However, the phenomenon of nerve overlap from adjacent territories can frequently confound inexperienced examiners.
Anatomical Zones of Sensibility
To accurately map sensory deficits, the surgeon must understand three distinct zones:
1. The Autonomous Zone (Isolated Zone): A small area of complete sensory loss supplied exclusively by the severed nerve.
2. The Intermediate Zone: A larger area of tactile and thermal anesthesia that corresponds more closely to the gross anatomical distribution of the nerve.
3. The Maximal Zone: The absolute maximum area of sensibility supplied by the nerve, which only becomes apparent if all adjacent nerves are blocked or sectioned.
Surgical Warning: The autonomous zone often becomes smaller during the first few days or weeks after injury—long before actual axonal regeneration is possible. This is primarily due to the resumption of, or increase in, function in anastomotic branches from adjacent nerves (collateral sprouting). Inexperienced surgeons may misinterpret this shrinking zone as evidence of spontaneous regeneration, leading to a disastrous delay in necessary surgical exploration.

The Sequence of Sensory Recovery
Dellon, Curtis, and Edgerton established the definitive sequence of sensory recovery using constant touch, moving touch, and graded vibratory stimuli. Their research demonstrated that sensory modalities return in a specific order based on fiber diameter and receptor maturation:
1. Pain and Temperature: Mediated by small-diameter, unmyelinated and thinly myelinated fibers. Perception of pinprick is the first to return.
2. 30 Hz Vibratory Stimulus: Mediated by quickly adapting fibers.
3. Moving Touch: Mediated by quickly adapting fibers and Pacinian corpuscles.
4. Constant Touch: Mediated by slowly adapting fibers and Merkel discs.
5. 256 Hz Vibratory Stimulus: The final modality to return, requiring high degrees of receptor reinnervation.
Assessment of Sensory Return
The BMRC sensory grading scale provides a standardized metric for recovery:
* S0: Absence of sensibility in the autonomous area.
* S1: Recovery of deep cutaneous pain within the autonomous area.
* S2: Return of some superficial cutaneous pain and tactile sensibility within the autonomous area.
* S3: Return of superficial cutaneous pain and tactile sensibility throughout the autonomous area with the disappearance of overreaction.
* S3+: Return of sensibility as in stage 3, plus some recovery of 2-point discrimination within the autonomous area.
* S4: Complete recovery.
Moberg revolutionized sensory evaluation by emphasizing functional sensibility over simple pinprick or cotton wool tests. He championed the Weber 2-point discrimination test, the picking-up test, and the Ninhydrin printing test, proving that 2-point discrimination directly correlates with actual hand function and object identification.
REFLEX AND AUTONOMIC CONSEQUENCES
Reflex Arc Disruption
Complete severance of a peripheral nerve abolishes all reflex activity transmitted by that nerve, whether the interruption is in the afferent or efferent arc. However, in partial nerve injuries, reflex activity is frequently abolished even when neither arc is completely interrupted. Therefore, the absence of a reflex is not a reliable guide to the absolute severity or completeness of the nerve lesion.
Autonomic Dysfunction
Interruption of a peripheral nerve immediately halts sympathetic transmission, leading to a loss of sweating (anhidrosis), loss of pilomotor response, and profound vasomotor paralysis in the autonomous zone.
Diagnostic Autonomic Tests:
* Starch-Iodine Test: Highlights areas of anhidrosis.
* Ninhydrin Printing Test (Aschan and Moberg): Detects amino acids in sweat; denervated skin will not produce a print.
* Wrinkle Test (O’Riain and Leukens): Normal skin immersed in warm water for 30 minutes will wrinkle due to sympathetic-mediated vasoconstriction. Denervated skin remains smooth and unwrinkled. As reinnervation occurs, wrinkling returns.
Vasomotor and Trophic Changes:
Initially, due to sympathetic disruption and resultant vasodilation, the denervated area is warmer and pinker than the rest of the limb. After 2 to 3 weeks, the affected area becomes colder than adjacent normal tissues. The skin may appear pale, cyanotic, or mottled.
Trophic changes are most evident in the hands and feet. The skin becomes thin, atrophic, and glistening. Minor trauma can lead to indolent ulcers that heal poorly. Fingernails become distorted, ridged, brittle, or may be lost entirely. Furthermore, severe localized osteoporosis often follows peripheral nerve injuries, particularly in incomplete lesions associated with pain (e.g., incomplete median nerve lesions affecting the distal phalanges).
COMPLEX REGIONAL PAIN SYNDROME (CRPS)
Reflex Sympathetic Dystrophy (RSD), now formally classified as Complex Regional Pain Syndrome (CRPS), is a devastating complication occurring in approximately 3% of major nerve injuries.
Historical Context and Terminology
During the American Civil War, Mitchell, Morehouse, and Keen first described painful sequelae following gunshot wounds to the median nerve. In 1867, Mitchell coined the term causalgia (Greek for "burning pain"). Historically, terms like Sudeck atrophy, shoulder-hand syndrome, and Leriche posttraumatic pain syndrome were used interchangeably.
Today, the International Association for the Study of Pain (IASP) divides the condition into:
* CRPS Type I: Occurs without a definable major nerve lesion (formerly RSD).
* CRPS Type II: Occurs secondary to a documented peripheral nerve injury (formerly Causalgia).
Pathophysiology and Lankford's Triad
The exact etiology remains debated, with theories including "short-circuiting" (ephaptic transmission) at the injury site where efferent sympathetic impulses irritate sensory afferent fibers, or abnormal feedback loops within the internuncial centers of the spinal cord.
According to Lankford, a specific triad must exist for a patient to develop CRPS:
1. A Painful Lesion: A severe or prolonged painful injury (often a crush or partial nerve laceration).
2. Patient Diathesis: A psychological or physiological susceptibility within the patient's sympathetic nervous system.
3. Abnormal Sympathetic Reflex: An exaggerated, prolonged sympathetic response leading to the classic symptoms of burning pain, hyperhidrosis, stiffness, and profound osteoporosis.
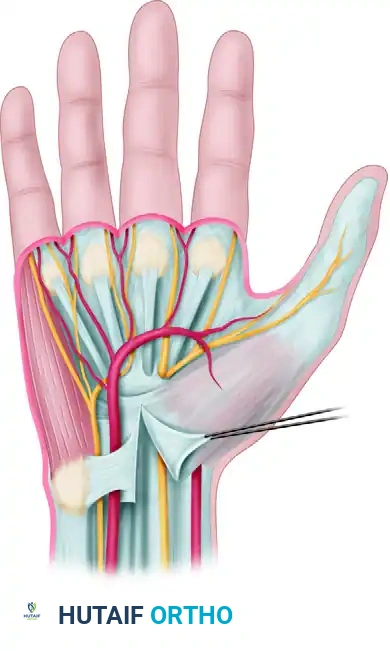
SURGICAL PRINCIPLES: EXPLORATION AND REPAIR
Understanding the pathophysiology of nerve injury dictates the surgical approach. The goal of operative intervention is to restore neural continuity, providing a tension-free conduit for regenerating axons to reach their target end-organs before irreversible muscle fibrosis occurs.
Indications for Surgical Exploration
- Open Wounds with Neurologic Deficit: Sharp lacerations (e.g., glass, knife) with distal deficits demand early exploration and primary repair (within 72 hours).
- Closed Injuries without Recovery: If clinical and EMG evaluations show no signs of reinnervation (e.g., advancing Tinel's sign, nascent motor units) by 3 to 4 months, exploration is indicated.
- Severe Crush or Gunshot Wounds: Usually managed with delayed exploration (at 3 weeks) to allow the zone of injury to demarcate, ensuring repair is performed in healthy neural tissue.
Patient Positioning and Preparation
- Upper Extremity: The patient is positioned supine with the arm extended on a radiolucent hand table. A pneumatic tourniquet is applied to the proximal arm to ensure a bloodless field, which is critical for microsurgical visualization.
- Lower Extremity: Depending on the nerve (e.g., sciatic vs. peroneal), the patient may be prone or supine.
- Magnification: Operating loupes (minimum 3.5x to 4.5x) or an operating microscope are mandatory for all peripheral nerve procedures.
Step-by-Step Surgical Approach
- Incision and Exposure: Utilize extensile incisions that cross flexion creases obliquely. Always identify the nerve in healthy, unscarred tissue proximal and distal to the zone of injury before tracing it into the fibrotic bed.
- Neuroma Resection: In delayed cases, a neuroma-in-continuity or a terminal neuroma will be present. The nerve ends must be serially sectioned using a fresh scalpel blade until healthy fascicles "pout" from the epineurium.
- Preparation of Nerve Ends: The epineurium is gently stripped back 1-2 mm to prevent it from interposing between the fascicles during repair.
- Coaptation:
- Primary Repair: If the nerve ends can be brought together without tension, an epineurial repair is performed using 8-0 or 9-0 non-absorbable monofilament sutures. The alignment of superficial epineurial blood vessels is used to prevent rotational malalignment.
- Nerve Grafting: If a tension-free repair is impossible, autologous nerve grafting (e.g., sural nerve, medial antebrachial cutaneous nerve) is the gold standard. The graft acts as a biological scaffold (providing Schwann cells and endoneurial tubes) for regenerating axons.
- Hemostasis and Closure: The tourniquet is deflated prior to closure to ensure absolute hemostasis, as a postoperative hematoma can cause devastating fibrosis and compression.
Postoperative Protocol and Rehabilitation
- Immobilization: The extremity is immobilized in a well-padded splint for 3 weeks to protect the micro-neurorrhaphy from tension. Joints are positioned to minimize stress on the repair (e.g., slight wrist flexion for a volar median nerve repair).
- Early Mobilization: At 3 weeks, the splint is removed, and a supervised nerve gliding and active range-of-motion program is initiated.
- Sensory Reeducation: As moving and constant touch return (guided by Dellon's sequence), formal sensory reeducation programs are instituted to help the cerebral cortex adapt to the altered sensory input.
- Motor Reeducation: Biofeedback and targeted strengthening begin as soon as M1/M2 function is detected clinically or electromyographically.
By synthesizing a deep understanding of Wallerian degeneration, precise clinical grading, and meticulous microsurgical technique, the orthopedic surgeon can maximize functional outcomes in the challenging arena of peripheral nerve reconstruction.
You Might Also Like