Introduction & Epidemiology
Deep space infections of the hand represent a critical subset of musculoskeletal infections demanding prompt and accurate diagnosis, followed by judicious surgical intervention. Among these, infection of the medial midpalmar space, sometimes referred to as the hypothenar space in older literature or more precisely as the ulnar side of the midpalmar space, presents unique diagnostic and therapeutic challenges due to its complex anatomy and proximity to vital neurovascular structures. Unrecognized or inadequately treated infections in this compartment can lead to devastating consequences, including spread to other deep spaces, flexor tenosynovitis, septic arthritis, osteomyelitis, permanent hand dysfunction, and systemic sepsis.
The epidemiology of hand infections is broad, often linked to penetrating trauma (e.g., splinters, animal bites, intravenous drug use), spread from neglected superficial infections (e.g., paronychia, felon), or complications of flexor tenosynovitis. While the true incidence of isolated medial midpalmar space abscesses is challenging to delineate, they constitute a significant proportion of complex hand infections requiring operative drainage. Staphylococcus aureus, including Methicillin-Resistant Staphylococcus aureus (MRSA), remains the predominant pathogen, although polymicrobial infections, particularly in immunocompromised patients or those with specific exposures (e.g., animal bites), are not uncommon. Early recognition and understanding of the specific anatomical confines and potential dangers associated with this space are paramount for successful clinical outcomes.
Surgical Anatomy & Biomechanics
A thorough understanding of the intricate anatomy of the palm is fundamental to safely and effectively drain the medial midpalmar space. This deep compartment of the hand is situated between the palmar aponeurosis anteriorly and the deep palmar fascia overlying the interossei and metacarpals posteriorly.
Palmar Fascial Spaces
The palmar region is compartmentalized by fibrous septa extending from the palmar aponeurosis. These septa contribute to the formation of the deep palmar spaces:
*
Thenar space:
Located radial to the long flexor tendon of the index finger.
*
Midpalmar space:
The largest deep palmar space, divided into a radial and ulnar portion. The medial midpalmar space specifically refers to the ulnar portion of the midpalmar space, sometimes called the hypothenar space, though this term is less precise. It lies deep to the palmar aponeurosis and superficial to the interossei muscles and the adductor pollicis muscle.
The medial midpalmar space is bounded:
*
Anteriorly (superficial):
By the palmar aponeurosis.
*
Posteriorly (deep):
By the interosseous muscles and the metacarpals of the ring and little fingers.
*
Radially:
By the oblique septum connecting the palmar aponeurosis to the third metacarpal, effectively separating it from the radial midpalmar space (which contains the flexor tendons of the index and middle fingers).
*
Ulnarly:
By the hypothenar muscles (abductor digiti minimi, flexor digiti minimi brevis, opponens digiti minimi).
*
Proximally:
It is continuous with the carpal canal and potentially the forearm spaces via the lumbrical canals.
*
Distally:
It communicates with the web spaces and dorsal aspect of the fingers through the lumbrical canals of the ring and little fingers.
Contents of the Medial Midpalmar Space
The medial midpalmar space primarily contains:
* The flexor digitorum superficialis (FDS) and flexor digitorum profundus (FDP) tendons to the ring and little fingers.
* The lumbrical muscles of the ring and little fingers, which originate from the FDP tendons within this space and pass distally into the lumbrical canals.
* Neurovascular structures: Critically, the common digital nerves and vessels to the ring and little fingers traverse this area. The ulnar nerve and artery branches, including the deep palmar branch of the ulnar nerve, pass deep to the flexor tendons but are in close proximity. The superficial palmar arch lies anterior to the flexor tendons, within the deep subcutaneous fat but deep to the palmar aponeurosis.
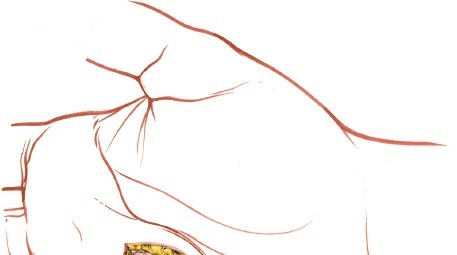
Figure 1: Illustration of the deep palmar fascial spaces, highlighting the medial midpalmar space and its relations to adjacent compartments and tendons.
Biomechanical Considerations
Infection within the medial midpalmar space can rapidly compromise hand function due to several factors:
*
Flexor Tendon Involvement:
Edema and pus accumulation can lead to increased pressure on the flexor tendons, potentially causing tenosynovitis, adhesions, and even necrosis if untreated. This directly impairs finger flexion.
*
Neurovascular Compression:
Swelling can compress the digital nerves and vessels, leading to paresthesias, sensory loss, and in severe cases, digital ischemia.
*
Spread of Infection:
The communication with other fascial spaces (thenar, dorsal subaponeurotic), lumbrical canals, and proximal forearm via the carpal tunnel, as well as potential extension to bone (osteomyelitis) or joints (septic arthritis), makes timely and complete drainage imperative to prevent widespread morbidity.
*
Scarring and Contracture:
Untreated infection results in significant inflammation and subsequent fibrosis, leading to palmar scarring, stiffness, and potentially fixed flexion deformities (e.g., Dupuytren-like contractures if the aponeurosis is severely affected).
Indications & Contraindications
The decision to proceed with surgical drainage of the medial midpalmar space is predicated on a careful clinical assessment, often supported by imaging and laboratory findings.
Indications for Operative Drainage
The primary indication is a confirmed or highly suspected abscess within the medial midpalmar space. This includes:
*
Clinical Presentation:
Localized pain, swelling, erythema, and tenderness over the medial midpalm, particularly with passive extension of the ring and little fingers. Fluctuation may be palpable in later stages, though deep-seated abscesses can be tense without overt fluctuation.
*
Failure of Conservative Management:
Lack of improvement or worsening of symptoms despite appropriate intravenous antibiotic therapy for 24-48 hours in cases without clear abscess formation initially.
*
Systemic Signs of Infection:
Fever, leukocytosis, elevated inflammatory markers (ESR, CRP) in conjunction with local findings.
*
Imaging Confirmation:
Ultrasound demonstrating a well-defined fluid collection consistent with an abscess. MRI can provide more detailed anatomical delineation in complex or chronic cases.
Contraindications for Operative Drainage
Absolute contraindications are rare, given the potential severity of untreated hand infections. Relative contraindications include:
*
Cellulitis Without Abscess:
In the absence of a drainable fluid collection, initial management with antibiotics and close observation may be appropriate. However, if cellulitis progresses or is refractory to antibiotics, re-evaluation for occult abscess is crucial.
*
Severe Coagulopathy:
While a relative contraindication, this must be corrected pre-operatively if possible. The risk of bleeding must be weighed against the risk of untreated infection.
*
Patient Instability/Comorbidity:
In critically ill patients, stabilization of life-threatening conditions takes precedence, though hand infection should not be neglected.
Table: Operative vs. Non-Operative Indications for Medial Midpalmar Space Infection
| Feature | Operative Indications | Non-Operative Indications |
|---|---|---|
| Clinical Findings | Localized fluctuant swelling, severe pain, erythema, limited ROM of digits (ring/little) due to pain/swelling. Signs of tenosynovitis. | Early cellulitis without fluctuance or clear abscess formation. |
| Systemic Symptoms | Fever, chills, leukocytosis, elevated inflammatory markers. | Afebrile, stable vital signs, mild leukocytosis. |
| Response to ABx | Worsening or no improvement after 24-48h of appropriate IV antibiotics. | Improving symptoms and signs with oral or IV antibiotics. |
| Imaging (Ultrasound) | Presence of a well-defined hypoechoic or anechoic fluid collection with internal debris (abscess). | Diffuse subcutaneous edema, no discrete fluid collection. |
| Pathogen | Any suspected deep space infection with abscess. | Superficial cellulitis responding to targeted antibiotics. |
Pre-Operative Planning & Patient Positioning
Careful pre-operative planning minimizes complications and optimizes surgical outcomes.
Diagnostic Workup
- History and Physical Examination: Detailed history of injury or onset, including comorbidities and immune status. Thorough hand examination, assessing pain, swelling, tenderness, range of motion, and neurovascular status.
- Laboratory Studies: Complete blood count (CBC) with differential, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP) to assess inflammatory response. Blood cultures are advisable in febrile patients or those with systemic signs.
- Microbiology: Whenever possible, aspiration of suspected abscess fluid for Gram stain, culture, and sensitivity should be performed pre-operatively. This guides initial empiric antibiotic selection.
-
Imaging:
- Plain Radiographs: Anteroposterior, lateral, and oblique views of the hand to rule out osteomyelitis, foreign bodies, or gas gangrene.
- Ultrasound: The modality of choice for confirming a fluid collection, differentiating cellulitis from abscess, and guiding aspiration. It can identify the precise location, size, and depth of the abscess.
- Magnetic Resonance Imaging (MRI): Reserved for complex cases, recurrent infections, or when osteomyelitis is strongly suspected and plain films are negative. MRI provides superior soft tissue contrast and anatomical detail.
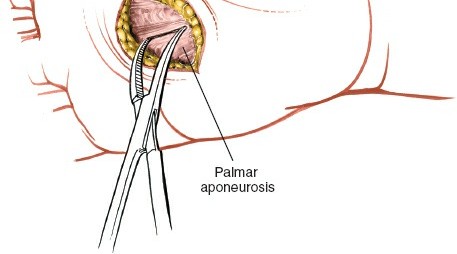
Figure 2: Pre-operative imaging demonstrating a deep palmar space infection (e.g., MRI T2-weighted image showing hyperintense fluid collection in the medial midpalmar space).
Antibiotic Prophylaxis/Therapy
Broad-spectrum intravenous antibiotics should be initiated empirically based on local resistance patterns and presumed pathogens. Common empiric regimens include vancomycin (for MRSA coverage) combined with a beta-lactamase inhibitor (e.g., piperacillin-tazobactam) or a carbapenem (e.g., meropenem) for broader Gram-negative and anaerobic coverage, especially in immunocompromised patients or those with severe infections. Antibiotics are then tailored once culture and sensitivity results are available.
Anesthesia
Regional anesthesia, such as an axillary or supraclavicular block, is often preferred as it provides excellent analgesia post-operatively and avoids the risks associated with general anesthesia. However, general anesthesia may be necessary for uncooperative patients, children, or those with extensive infections.
Patient Positioning and Preparation
- The patient is positioned supine with the affected arm abducted on a hand table.
- A tourniquet is applied to the upper arm. A sterile exsanguination technique using an Esmarch bandage should be performed prior to inflation to optimize visualization. The tourniquet pressure should be set to 250 mmHg or 100 mmHg above systolic blood pressure, not exceeding 1.5-2 hours of inflation time.
- The entire hand, wrist, and forearm should be prepped and draped in a sterile fashion, allowing for visualization of the entire palm and digits.
Detailed Surgical Approach / Technique
The goal of surgical drainage is complete evacuation of pus, débridement of necrotic tissue, and creation of a pathway for ongoing drainage while minimizing injury to vital structures.
Incision Design
The choice of incision for the medial midpalmar space is critical to provide adequate access for drainage while protecting neurovascular structures. The classical approach involves a transverse incision.
Incision
Make a curved transverse incision on the palm just proximal to the distal palmar crease and over the swelling. The length of the incision should be determined by the size of the abscess to be drained (Fig. 5-80). This transverse incision is typically located parallel to the distal palmar crease, positioned approximately at the level of the metacarpal heads of the ring and little fingers. It should be curved distally to align with the skin creases, minimizing scar contracture. The length must be sufficient to allow thorough exploration and débridement, often spanning 2-4 cm depending on the extent of the abscess.
Alternatively, in some cases, an oblique or longitudinal incision can be considered, though it must carefully navigate critical structures. For medial midpalmar space abscesses, the transverse approach is generally favored due to its lower risk to digital nerves if performed correctly, and better cosmetic outcome.
Figure 3: Depiction of the curved transverse incision commonly employed for drainage of the medial midpalmar space, positioned just proximal to the distal palmar crease and overlying the area of maximum swelling (corresponds to Fig. 5-80 in original content).
Internervous Plane
There is no distinct internervous plane in this approach within the palm. Dissection proceeds through layers of soft tissue, requiring meticulous identification and protection of nerves and vessels.
Superficial Surgical Dissection
Skin and Subcutaneous Tissue
Incise the skin carefully; the line of the skin incision crosses the paths of the digital nerves. The initial incision should be made only through the skin and subcutaneous fat. The subcutaneous tissue of the palm is characterized by fat lobules separated by fibrous septa, which tether the skin to the palmar aponeurosis. This firm attachment often makes palpation of deep fluctuance challenging.
Identification and Protection of Neurovascular Structures
The digital nerves to the little and ring fingers run immediately under the palmar aponeurosis and cross the line of the skin incision. No part of the palmar aponeurosis should be incised transversely until these nerves have been dissected out fully (see Fig. 5-82). These common digital nerves and their proper digital branches lie in the subcutaneous fat, superficial to the palmar aponeurosis or immediately deep to it, making them highly vulnerable during a transverse incision.
*
Technique for Nerve Protection:
After incising the skin and subcutaneous fat, blunt dissection using fine scissors or a hemostat should be performed longitudinally, parallel to the anticipated course of the neurovascular bundles. The common digital nerves to the ring and little fingers, along with their accompanying digital arteries, are identified. They should be carefully visualized and retracted, typically distally or proximally, away from the planned entry point into the palmar aponeurosis. This step is paramount to prevent iatrogenic nerve injury, which can lead to permanent sensory deficits or painful neuromas.
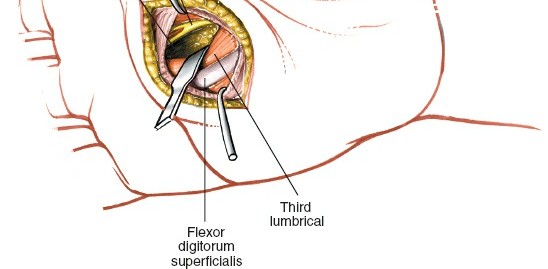
Figure 4: Intraoperative view demonstrating the meticulous blunt dissection required to identify and protect the digital nerves (e.g., to the little and ring fingers) that lie in close proximity to the proposed incision line, immediately beneath the palmar aponeurosis (corresponds to Fig. 5-82 in original content).
Entry into the Medial Midpalmar Space
Once the digital neurovascular bundles are safely identified and protected, the underlying palmar aponeurosis comes into view.
*
Incision of Palmar Fascia:
Open the palmar fascia by blunt dissection at the distal end of the wound. The palmar aponeurosis is incised longitudinally, parallel to the long axis of the hand, rather than transversely through the main body of the aponeurosis. This minimizes damage to the fibrous architecture. A small, carefully placed longitudinal incision through the aponeurosis is made.
*
Identification of Flexor Tendons:
Blunt dissection through the incised aponeurosis will reveal the long flexor tendon to the ring finger. This tendon, along with the flexor tendon to the little finger, serve as key anatomical landmarks.
*
Entering the Space:
Enter the medial midpalmar space by blunt dissection on the radial border of this tendon (Figs. 5-81 to 5-83). A curved hemostat or a blunt dissector is advanced carefully, aiming for the area of greatest fluctuance or presumed pus collection. The instrument is gently spread to open the fascial septa and gain access to the abscess cavity. Pus is often encountered at this stage. It is crucial to maintain blunt dissection to avoid injury to deeper structures, including the deep palmar arch and intrinsic muscles.
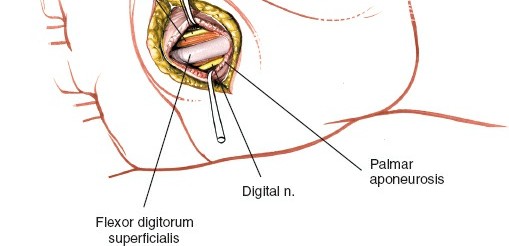
Figure 5: Surgical technique showing blunt dissection on the radial border of the flexor tendon to the ring finger, allowing safe entry into the medial midpalmar space (corresponds to Fig. 5-81 or 5-83 in original content).
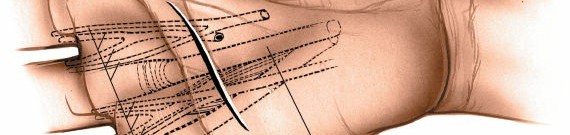
Figure 6: A deeper view of the dissection, illustrating the blunt instrument carefully probing the medial midpalmar space to break up loculations and ensure thorough drainage.
Exploration, Débridement, and Irrigation
Once the space is entered and pus is encountered:
*
Culture Acquisition:
A specimen of pus should be collected for Gram stain, aerobic and anaerobic cultures, and antibiotic sensitivities.
*
Thorough Exploration:
The abscess cavity must be thoroughly explored with a blunt instrument (e.g., a curved hemostat or finger if the incision allows). All loculations should be carefully broken down to ensure complete drainage. It is imperative to extend the exploration proximally towards the carpal tunnel and distally into the lumbrical canals to identify any potential spread of infection.
*
Débridement:
Necrotic or devitalized tissue within the abscess cavity should be débrided using fine instruments. Care must be taken to distinguish necrotic tissue from healthy structures.
*
Irrigation:
The entire space should be copiously irrigated with sterile saline solution until the effluent is clear. This helps to mechanically remove bacteria, inflammatory mediators, and debris.
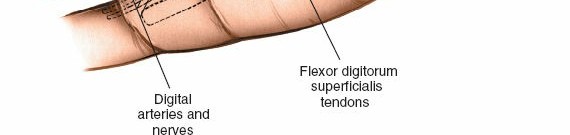
Figure 7: Intraoperative image showing thorough irrigation of the deep medial midpalmar space after initial pus evacuation, crucial for removing debris and reducing bacterial load.
Drainage Placement
- Drainage Strategy: After thorough irrigation, a drainage system is usually left in place to prevent re-accumulation of fluid and allow for continued egress of inflammatory exudate. A small Penrose drain is commonly used, passed through a separate stab incision (if feasible, but often through the main incision) and secured with a skin stitch. Alternatively, a small closed suction drain (e.g., a 1/8-inch Jackson-Pratt drain) can be used, especially for larger cavities. The drain should be positioned to exit dependently or at a site facilitating easy removal.
- Avoiding Tension: The drain should not be tightly packed into the wound, as this can impede drainage.
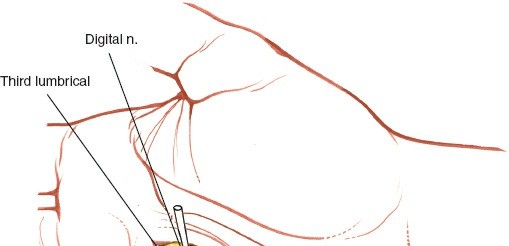
Figure 8: Placement of a Penrose drain within the drained medial midpalmar space, secured to the skin, ensuring continuous evacuation of exudate post-operatively.
Wound Closure
- Delayed Primary Closure: In most cases of acute purulent infections, the skin incision is left open or loosely approximated with a few sutures. This allows for continued drainage, reduces the risk of recurrent abscess formation, and allows for daily wound care. The wound can be secondarily closed after 3-5 days if there are no signs of ongoing infection and the wound appears clean.
- Secondary Intention: If the infection is extensive or the tissue is highly contaminated, the wound may be left open to heal by secondary intention, often packed with saline-moistened gauze.
- Dressings: A bulky, sterile dressing is applied, ensuring light compression without restricting digital motion. The hand is typically immobilized in a position of safety (wrist slightly extended, MP joints flexed 70 degrees, IP joints extended) if significant swelling or instability is present, but early motion is encouraged where appropriate.
Complications & Management
Despite meticulous surgical technique, complications can occur following drainage of medial midpalmar space infections. Recognition and prompt management are essential for optimal functional recovery.
Table: Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence | Salvage Strategies |
|---|---|---|
| Neurovascular Injury | Rare (e.g., <1% for digital nerves with careful technique), but significant. | Immediate repair if identified intraoperatively. Delayed microsurgical repair or nerve grafting for persistent deficits. Neuroma excision for painful neuromas. |
| Incomplete Drainage/Recurrent Abscess | ~5-10% (higher with complex or loculated abscesses). | Re-exploration and drainage, typically through the same incision or an extended approach. Ensure all loculations are broken and proper drain placement. Adjust antibiotic regimen based on new cultures. |
| Persistent Infection/Spread | Can occur if primary drainage is inadequate or initial antibiotics are ineffective. | Aggressive débridement, culture-directed antibiotics, identification and drainage of contiguous spread (e.g., tenosynovitis, osteomyelitis, septic arthritis). Referral to infectious disease. |
| Scar Contracture/Adhesions | Common with prolonged inflammation or large incisions. | Early hand therapy including scar massage, silicone sheeting, splinting for contracture prevention. Surgical release of contractures if conservative measures fail. |
| Stiffness/Reduced ROM | Very common, especially involving flexor tendons. | Early and aggressive hand therapy focusing on active and passive range of motion. Dynamic splinting. Tenolysis if severe adhesions limit function post-infection resolution. |
| Chronic Pain | Variable, can be due to nerve injury, neuromas, or CRPS. | Pain management strategies, nerve blocks, physical therapy, CRPS management protocols. |
| Complex Regional Pain Syndrome (CRPS) | Low (e.g., 2-5% post-trauma/surgery), but debilitating. | Early recognition and multidisciplinary treatment: physical therapy, pain management (nerve blocks, pharmacotherapy), psychological support. |
| Osteomyelitis/Septic Arthritis | Rare, but possible if infection spreads to bone or joint. | Prolonged culture-directed antibiotics, surgical débridement of infected bone/joint, joint irrigation. Potential for arthrodesis or amputation in severe, refractory cases. |
Specific Management Considerations
- Nerve Injury: If a digital nerve is transected, immediate primary repair should be attempted if conditions allow (clean wound, minimal edema). If primary repair is not feasible or not recognized intraoperatively, delayed repair or nerve grafting may be necessary. Post-operative neurological assessment is crucial.
- Incomplete Drainage: Persistent pain, swelling, and fever post-operatively should raise suspicion for incomplete drainage. Ultrasound can confirm residual fluid. Revision surgery is often required.
- Scarring and Stiffness: Prevention is key through early mobilization and appropriate hand therapy. If significant contracture develops, surgical release (e.g., Z-plasty, flap coverage) may be indicated after the infection has completely resolved and tissues have matured.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is integral to restoring hand function and preventing long-term disability. The protocol is typically initiated promptly after surgery, often within 24-48 hours, depending on the wound status and the patient's pain level.
Early Phase (Days 1-7)
- Wound Care: Daily dressing changes, inspection for signs of infection (erythema, purulent discharge, increasing pain), and drain output monitoring. Saline irrigation or antiseptic washes as indicated.
- Antibiotics: Continued intravenous antibiotics, transitioning to oral antibiotics based on culture sensitivities and clinical improvement. The duration typically ranges from 7-14 days, but may be longer for severe infections or if osteomyelitis is identified.
- Edema Control: Elevation of the hand (above heart level), gentle compression dressings, and active range of motion (AROM) exercises for uninvolved joints.
- Pain Management: Oral analgesics, often including non-steroidal anti-inflammatory drugs (NSAIDs) and opioids for breakthrough pain.
-
Mobilization:
- Immobilization: If there is significant swelling, a dorsal blocking splint may be used initially to protect the flexor tendons and maintain the "position of safety" (wrist 20-30 degrees extension, MCPs 70 degrees flexion, IPs extended).
- Early Active Range of Motion: As soon as comfort allows, gentle active range of motion exercises for all uninvolved joints and, cautiously, for the digits affected by the infection. The goal is to prevent stiffness and tendon adhesions.
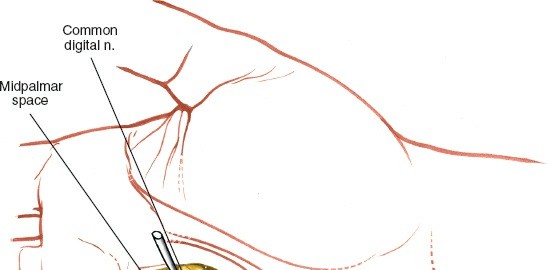
Figure 9: An example of a gentle active range of motion exercise for the hand, promoting early tendon gliding and preventing stiffness post-drainage.
Intermediate Phase (Weeks 1-4)
- Progressive Mobilization: Increase the intensity and frequency of AROM exercises. Begin passive range of motion (PROM) exercises, cautiously, as tolerated.
- Scar Management: Once the wound is closed (either primarily or secondarily), begin scar massage with moisturizing cream to soften the scar tissue and prevent adhesions. Silicone gel sheets or scar pads can be beneficial.
- Strengthening: Introduce light strengthening exercises with therapeutic putty or soft resistance bands, focusing on grip strength and fine motor coordination, as tolerated.
- Desensitization: If nerve irritation or hypersensitivity is present, begin desensitization techniques.
Late Phase (Weeks 4 onwards)
- Advanced Strengthening: Progress to more challenging strengthening exercises.
- Functional Activities: Incorporate activities that mimic daily living tasks and work-specific demands.
- Dynamic Splinting: If joint contractures are developing, dynamic or static progressive splinting may be employed to regain lost range of motion.
- Return to Activity: Gradually return to full activity based on pain, strength, and range of motion. Close communication with occupational or physical therapists is vital.
The duration and specific components of rehabilitation will be individualized based on the severity of the initial infection, the extent of surgical débridement, the patient's response to treatment, and the presence of any complications. Regular assessment by a hand therapist is crucial for optimizing functional recovery.
Summary of Key Literature / Guidelines
The management of deep hand infections, including those of the medial midpalmar space, adheres to principles established in general orthopaedic and plastic surgery literature. While specific randomized controlled trials focusing solely on medial midpalmar space drainage are rare due to the relatively lower incidence of isolated cases, several key guidelines and meta-analyses inform best practices:
- Prompt Surgical Drainage: Consensus remains that established deep space abscesses require prompt surgical drainage, débridement, and irrigation to prevent spread of infection, preserve function, and avoid systemic complications. This aligns with the principles outlined by such authors as Neviaser and Louis, who emphasized the importance of adequate decompression.
- Antibiotic Selection: Initial empiric broad-spectrum intravenous antibiotics covering common skin flora (S. aureus, Streptococcus species) are standard, with particular attention to MRSA coverage in relevant populations. Culture-directed antibiotic therapy based on intraoperative samples is crucial for optimizing outcomes and minimizing resistance. The Infectious Diseases Society of America (IDSA) guidelines for skin and soft tissue infections provide comprehensive recommendations for antibiotic choices.
- Imaging Utility: The literature supports the use of ultrasound as the primary imaging modality for diagnosing and localizing deep space abscesses, especially in guiding aspiration. MRI is reserved for complex cases or when osteomyelitis is suspected, offering superior soft tissue and bone detail.
- Surgical Technique: Emphasis on meticulous surgical technique to protect vital neurovascular structures, particularly the digital nerves, is paramount. The described transverse incision in the palm, with careful subcutaneous dissection and blunt entry into the fascial spaces, is a well-established and accepted method for accessing these deep spaces. Authors like Henry and Hunter have extensively detailed safe surgical approaches to the hand.
- Post-Operative Management: The importance of aggressive post-operative hand therapy, including early mobilization, scar management, and edema control, is consistently highlighted across the literature to prevent stiffness and optimize functional recovery.
- Complication Management: Prevention and early recognition of complications such as incomplete drainage, nerve injury, and spread of infection are stressed. Revision surgery and prolonged, targeted antibiotic regimens are standard for persistent or recurrent infections. Management of post-infectious stiffness and contractures often requires a multidisciplinary approach involving hand surgeons and therapists.
In summary, current practice for medial midpalmar space infections is guided by principles of early diagnosis, aggressive surgical intervention with careful attention to anatomy, appropriate antibiotic therapy, and comprehensive post-operative rehabilitation. Adherence to these guidelines minimizes morbidity and maximizes the potential for excellent functional outcomes.
