Introduction & Epidemiology
Crystal deposition arthropathy (CDA) encompasses a group of inflammatory arthritides characterized by the intra-articular or periarticular deposition of microscopic crystals, triggering acute or chronic inflammatory responses. From an orthopedic perspective, understanding CDA is critical not only for accurate diagnosis and differential considerations but also for managing the chronic sequelae, diagnostic challenges, and, occasionally, direct surgical interventions required for severe complications.
The primary forms of CDA include:
*
Gout:
Caused by the deposition of monosodium urate (MSU) crystals, resulting from hyperuricemia. It is the most common inflammatory arthritis in adults, with a rising prevalence globally.
*
Calcium Pyrophosphate Deposition Disease (CPPD), often termed pseudogout:
Characterized by the deposition of calcium pyrophosphate dihydrate (CPPD) crystals. It is more prevalent in older adults and can mimic various arthritic conditions.
*
Calcium Apatite Deposition Disease (CADD):
Involves deposition of basic calcium phosphate crystals, often hydroxyapatite. It commonly presents as calcific periarthritis or Milwaukee shoulder syndrome.
*
Calcium Oxalate Deposition:
A rare form, typically seen in patients with end-stage renal disease on dialysis, involving calcium oxalate crystals.
The epidemiology of CDA shows a predilection for certain demographics and is strongly associated with metabolic comorbidities. Gout, for instance, is classically observed in men aged 40–60 years and postmenopausal women, with increasing prevalence linked to metabolic syndrome, obesity, renal impairment, and specific dietary patterns. CPPD also increases with age and can be associated with hyperparathyroidism, hemochromatosis, and hypomagnesemia. The orthopedic surgeon frequently encounters patients with undifferentiated arthralgias, acute monoarticular inflammation, or chronic destructive arthropathies where CDA must be high on the differential diagnosis. The implications extend from ruling out septic arthritis to managing mechanical complications of chronic crystal deposition.
Surgical Anatomy & Biomechanics
The specific anatomical sites affected by crystal deposition vary, influencing clinical presentation and potential orthopedic sequelae. Understanding these patterns is crucial for diagnosis and surgical planning.
Gout (Monosodium Urate)
- Predilection: Classically, the first metatarsophalangeal (MTP) joint (podagra) is involved, accounting for approximately 50% of initial attacks. However, other lower extremity joints (ankle, knee, midfoot) and less commonly upper extremity joints (wrist, elbow, fingers) can be affected.
-
Tophi Formation:
Chronic hyperuricemia leads to the formation of tophi – aggregates of MSU crystals surrounded by granulomatous inflammation. These can deposit in:
- Periarticular Soft Tissues: Bursae (olecranon, prepatellar), tendon sheaths (Achilles, patellar), and ligaments.
- Subcutaneous Tissues: Helix of the ear, eyelids, fingers, toes.
- Bone: Leading to characteristic "punched-out" erosions with sclerotic overhanging edges, often observed juxta-articularly.
- Kidneys: Urolithiasis and urate nephropathy represent a significant systemic manifestation.
-
Biomechanical Implications:
- Acute gouty flares cause severe synovitis, leading to effusions, pain, and restricted motion, often rendering the joint non-weight-bearing.
- Chronic tophaceous gout can lead to extensive joint destruction, cartilage erosion, and subchondral bone loss, mimicking severe degenerative or inflammatory arthritis.
- Tophi in tendons can cause tendinopathy, impingement, or rupture.
- Large tophi can cause nerve compression (e.g., carpal tunnel syndrome, tarsal tunnel syndrome) or skin ulceration due to mechanical stress.
Calcium Pyrophosphate Deposition Disease (CPPD)
- Predilection: Most commonly affects the knee, wrist, shoulder, and hips. The intervertebral discs and symphysis pubis can also be involved.
- Radiographic Hallmarks: Chondrocalcinosis – the characteristic calcification of articular cartilage (menisci, labra) and fibrocartilage (triangular fibrocartilage complex of the wrist).
-
Biomechanical Implications:
- Acute CPPD flares (pseudogout) result in severe synovitis, similar to gout, but often in larger joints.
- Chronic CPPD can lead to a destructive arthropathy resembling osteoarthritis, sometimes more aggressive (e.g., Milwaukee shoulder with rapid rotator cuff tear and glenohumeral destruction).
- The presence of CPPD crystals within cartilage may compromise its structural integrity, predisposing to accelerated degeneration.
- Crystal deposits in ligaments or tendons can weaken these structures or cause impingement.
Calcium Apatite Deposition Disease (CADD)
- Predilection: Primarily affects the shoulder (rotator cuff tendinopathy, calcific periarthritis), hip (trochanteric bursitis), and wrist.
-
Biomechanical Implications:
- Acute attacks due to crystal shedding can be exquisitely painful, causing severe limitation of motion (e.g., acute calcific tendinitis of the shoulder).
- Chronic deposits can lead to tendon degeneration, impingement, and in severe cases, joint destruction, particularly in the shoulder ("Milwaukee shoulder").
General Biomechanical Considerations
- Joint Integrity: Crystal deposition directly damages articular cartilage and subchondral bone, altering load-bearing characteristics and increasing vulnerability to mechanical stress.
- Soft Tissue Envelope: Chronic inflammation and tophi can compromise the soft tissue envelope around joints, leading to skin atrophy, ulceration, and increased susceptibility to infection, which is crucial for surgical planning.
- Instability/Deformity: Significant bone erosion and soft tissue damage can lead to joint instability and progressive deformity, requiring reconstructive approaches.
Integrating anatomical and biomechanical understanding is essential for surgical decision-making, especially when considering debridement, tophi excision, or joint reconstruction.
Indications & Contraindications
Surgical intervention for crystal deposition arthropathy is typically reserved for complications of the disease, diagnostic dilemmas, or end-stage arthropathy, rather than as a primary treatment for crystal deposition itself. Medical management remains the cornerstone of care.
Indications for Surgical Intervention
-
Diagnostic Ambiguity:
- Atypical presentation of monoarthritis where septic arthritis cannot be definitively ruled out by aspiration/culture.
- Failure of aspiration to yield fluid for analysis.
- Biopsy of suspected tophi/deposits in unusual locations.
- Exclusion of other pathologies (e.g., tumor, infection).
-
Complications of Tophi (Gout):
- Nerve Compression: Tophi causing symptomatic compression neuropathy (e.g., carpal tunnel syndrome, tarsal tunnel syndrome, spinal canal stenosis).
- Skin Ulceration/Infection: Large, superficial tophi that have eroded through the skin, are recurrently infected, or are at high risk of ulceration.
- Functional Impairment/Pain: Tophi causing significant mechanical interference with joint movement, tendon excursion, or nerve function, leading to intractable pain or loss of function despite optimal medical management.
- Mass Effect/Cosmesis: Extremely large or disfiguring tophi, particularly on weight-bearing surfaces or cosmetically sensitive areas, that are resistant to medical therapy.
- Pathologic Fracture: Tophi compromising bone integrity leading to fracture.
-
End-Stage Arthropathy:
-
Severe joint destruction secondary to chronic crystal deposition leading to intractable pain, deformity, and functional loss, unresponsive to non-operative measures. This may necessitate:
- Arthroplasty: Total joint replacement (e.g., knee, hip, shoulder) for conditions mimicking severe osteoarthritis or inflammatory arthritis.
- Arthrodesis: Joint fusion for small joints or in cases where arthroplasty is contraindicated or technically unfeasible due to extensive bone loss or soft tissue compromise.
- Debridement/Synovectomy: For chronic synovitis and significant joint effusions with progressive cartilage damage.
-
Severe joint destruction secondary to chronic crystal deposition leading to intractable pain, deformity, and functional loss, unresponsive to non-operative measures. This may necessitate:
-
Infection:
- Septic arthritis coexisting with crystal deposition (a critical differential and a surgical emergency).
- Infected tophi or bursae.
- Secondary infection of operative sites.
-
Calcific Tendinopathy (CADD):
- Intractable pain and functional limitation from acute or chronic calcific tendinopathy (e.g., shoulder, hip), unresponsive to conservative management.
- Surgical removal of calcific deposits.
Contraindications for Surgical Intervention
- Acute Flare: Generally, elective surgery should be avoided during an acute flare of crystal arthropathy due to the high risk of exacerbating inflammation, poor wound healing, and post-operative complications.
- Uncontrolled Systemic Disease: Poorly controlled hyperuricemia, active systemic infection, or unstable medical comorbidities (e.g., severe cardiac, renal, or pulmonary disease) may preclude or delay elective surgery.
- Coagulopathy: Uncorrected bleeding diathesis.
- Insufficient Medical Optimization: Inadequate trial of conservative medical management for symptom control or reduction of crystal burden.
- Poor Surgical Candidate: Patients with significant risks for anesthesia or poor healing potential that outweigh the potential benefits of surgery.
- Lack of Clear Indication: Surgery solely for the presence of crystals without associated symptoms or complications is generally not indicated.
| Surgical Indication | Operative Management | Non-Operative Management |
|---|---|---|
| Diagnostic Ambiguity | Arthroscopy, Open Biopsy, Joint Aspiration for Culture | Serial clinical observation, empiric anti-inflammatory therapy |
| Symptomatic Tophi (Pain, Ulceration, Nerve Comp.) | Tophi Excision, Debridement | Urate-lowering therapy (ULT), anti-inflammatory agents, analgesics |
| End-Stage Joint Destruction | Arthroplasty, Arthrodesis, Synovectomy | Analgesics, physical therapy, bracing, intra-articular injections |
| Infected Tophi/Septic Joint | Incision & Drainage, Debridement, Synovectomy | Intravenous antibiotics (concurrently with surgical source control) |
| Intractable Calcific Tendinopathy (CADD) | Surgical Excision of Calcific Deposits | NSAIDs, physical therapy, corticosteroid injections, needling lavage |
| Pathologic Fracture | ORIF, Arthroplasty, Amputation | Immobilization, pain control (if surgery not feasible) |

Pre-Operative Planning & Patient Positioning
Careful pre-operative planning is paramount for patients undergoing surgery in the context of crystal deposition arthropathy, given the potential for systemic comorbidities, compromised local tissue, and post-operative flares.
Pre-Operative Planning
-
Comprehensive Medical Workup:
- Systemic Evaluation: Assess for comorbidities commonly associated with CDA, including renal insufficiency, hypertension, diabetes, hyperlipidemia, and cardiovascular disease.
- Inflammatory Control: Ensure the patient is not in an acute flare. If so, defer elective surgery until the inflammation is controlled, typically with anti-inflammatory medications.
- Urate-Lowering Therapy (ULT): For gout, discuss the continuation of ULT. Some protocols advocate for initiating or continuing colchicine perioperatively to prevent post-operative flares. Consult with a rheumatologist.
- Medication Review: Identify and manage medications that can exacerbate hyperuricemia (e.g., diuretics, aspirin) or interact with anesthetic agents.
-
Detailed Imaging:
- Plain Radiographs: Essential for evaluating joint destruction, erosions, chondrocalcinosis, and gross tophi burden.
- Ultrasound: Highly useful for detecting active synovitis, effusion, identification of crystals (double contour sign for gout, hyperechoic deposits for CPPD), and delineating soft tissue tophi not visible on X-ray. It can also guide aspiration and injection.
- Computed Tomography (CT): Provides detailed assessment of bone erosion, intraosseous tophi, joint destruction, and extent of soft tissue involvement, particularly for complex anatomy or when planning extensive debridement or arthroplasty.
- Magnetic Resonance Imaging (MRI): Best for evaluating soft tissue involvement, nerve compression, tendon integrity, and inflammatory changes, especially when diagnosing specific nerve compression syndromes or assessing rotator cuff pathology.
-
Microbiological and Histopathological Planning:
- Always consider the possibility of coexisting infection, especially with skin ulceration over tophi or in cases of acute monoarthritis. Pre-operative joint aspiration for Gram stain, cell count with differential, and crystal analysis, along with bacterial culture, is critical.
- Plan for intraoperative frozen section analysis and send all excised tissue for histopathology to confirm crystal type and rule out other diagnoses.
-
Surgical Site Preparation:
- Address any skin compromise (e.g., ulceration over tophi) pre-operatively with local wound care and antibiotics if infected.
- Ensure adequate hair removal and skin cleansing protocols are in place.
-
Patient Education:
- Inform the patient about the high risk of post-operative flare of their underlying crystal arthropathy.
- Discuss potential wound healing issues, infection risk, and the importance of long-term medical management.
Patient Positioning
Patient positioning is dictated by the anatomical site of involvement and the planned surgical procedure. General principles include:
*
Accessibility:
Ensure the surgical site is fully exposed and permits appropriate instrument access.
*
Stability:
Maintain stable positioning throughout the procedure, especially for prolonged cases.
*
Physiological Monitoring:
Allow for continuous access for anesthesia and monitoring.
*
Pressure Point Protection:
Meticulously pad all pressure points to prevent nerve injury or skin breakdown, particularly relevant in patients with compromised skin integrity or neuropathy.
*
Specific Examples:
*
Foot/Ankle Surgery (e.g., MTP joint tophi excision):
Supine position, often with a bump under the ipsilateral hip for internal rotation, or lateral decubitus.
*
Hand/Wrist Surgery (e.g., carpal tunnel release, digit tophi excision):
Supine with hand table.
*
Elbow Surgery (e.g., olecranon bursa tophi excision):
Supine with arm across chest or lateral decubitus.
*
Knee Surgery (e.g., arthroscopy, arthroplasty):
Supine.
*
Shoulder Surgery (e.g., calcific tendinopathy excision, arthroplasty):
Beach chair or lateral decubitus.
Detailed Surgical Approach / Technique
Surgical techniques for crystal deposition arthropathy are diverse, ranging from diagnostic procedures to complex reconstructive surgeries. The choice depends on the specific crystal type, anatomical location, extent of pathology, and clinical presentation.
1. Diagnostic Arthroscopy and Biopsy
- Indications: Atypical monoarthritis, ruling out septic arthritis, evaluation of joint destruction, obtaining synovial tissue for crystal analysis when aspiration is non-diagnostic.
- Approach: Standard arthroscopic portals relevant to the joint (e.g., knee, shoulder, wrist).
-
Technique:
- Diagnostic visualization of the joint to assess cartilage, synovium, and meniscal/ligamentous structures.
- Careful inspection for crystal deposits, often appearing as chalky, white, or yellowish material on the synovium, cartilage, or within the joint capsule.
- Synovial biopsy: Harvest multiple samples from inflamed or suspicious areas using biopsy forceps. Samples should be sent for Gram stain, aerobic/anaerobic cultures, fungal cultures, mycobacterial cultures, and histopathological examination (including specific fixation, e.g., absolute alcohol for MSU crystals, formalin for CPPD).
- Joint lavage: Thorough irrigation to remove loose crystals and inflammatory mediators, which can provide symptomatic relief.
- Pitfall: Crystals can be difficult to identify macroscopically during arthroscopy. A high index of suspicion and appropriate specimen handling are crucial.
2. Excision of Symptomatic Tophi (Gout)
- Indications: Nerve compression, skin ulceration, recurrent infection, intractable pain, significant functional impairment, or extreme disfigurement.
-
Principles:
- Skin Incision: Plan incisions to avoid thin, stretched skin over the tophus, which may have poor vascularity. Utilize existing skin creases or elliptical excisions if skin viability is compromised. Avoid direct incisions over pressure areas if possible.
- Dissection: Meticulous, careful dissection to avoid nerve damage (especially in the hand/foot), vessel injury, or damage to underlying tendons/ligaments. The tophaceous material is typically firm, chalky, and yellowish-white, and can be adherent to surrounding structures.
- Removal: Excision of the tophus should be as complete as possible without sacrificing critical anatomical structures. Use a curette, osteotome, or burr for bony erosions. Copious irrigation is essential to remove residual crystal fragments.
- Wound Closure: Primary closure is preferred, but may require local flaps or skin grafting if there is significant skin loss. Tension-free closure is critical due to compromised tissue. Drains may be considered for large cavities.
-
Specific Examples:
- Foot (e.g., 1st MTP joint, interphalangeal joints): Dorsal or plantar longitudinal incisions. Identify and protect neurovascular bundles.
- Hand/Wrist: Incisions parallel to major neurovascular structures. For carpal tunnel release, standard volar approach.
- Olecranon Bursa: Elliptical incision over the bursa, careful dissection to remove the bursa and any contained tophi, avoiding the ulnar nerve.
- Post-operative: Anti-inflammatory prophylaxis (colchicine) should be considered to prevent post-operative gout flares.

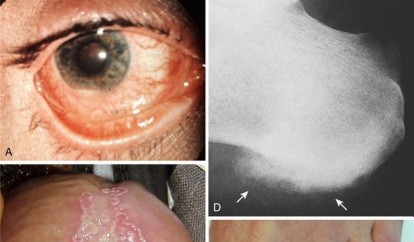
Extensive tophaceous gout involving the foot, demonstrating soft tissue swelling and bone destruction prior to surgical debridement.
3. Excision of Calcific Deposits (CADD)
- Indications: Intractable pain from calcific tendinopathy (e.g., shoulder, hip) unresponsive to conservative measures.
- Approach: Open or arthroscopic. Arthroscopic excision is favored for shoulder calcific tendinopathy.
-
Technique (Arthroscopic Shoulder):
- Standard posterior and anterior portals.
- Identification of the calcific deposit within the rotator cuff tendon (commonly supraspinatus).
- Incision of the tendon fibers overlying the deposit.
- Mechanical debridement/excision of the calcium using a shaver, rasp, or curette. The material is often pasty or chalky.
- Thorough lavage.
- Assessment of the remaining rotator cuff integrity; repair if a significant tear is created or uncovered.
- Pitfall: Incomplete removal can lead to persistent symptoms. Over-aggressive debridement can weaken the tendon.
4. Arthroplasty and Arthrodesis for End-Stage Arthropathy
- Indications: Severe, irreversible joint destruction, pain, and functional loss from chronic crystal deposition, mimicking end-stage osteoarthritis or inflammatory arthritis.
-
Challenges:
- Soft Tissue Envelope: Chronic inflammation, tophi, and prior skin ulceration can compromise the soft tissue coverage, increasing infection and wound healing risks.
- Bone Loss: Extensive erosions and cyst formation can lead to significant bone stock deficiency, complicating implant fixation.
- Recurrence: Continued crystal deposition can occur around implants, potentially leading to prosthesis loosening or further inflammatory responses.
- Infection Risk: Patients with CDA often have comorbidities, and compromised skin/soft tissue further elevates infection risk.
- Approach: Standard approaches for total joint arthroplasty (e.g., total knee arthroplasty, total hip arthroplasty, total ankle arthroplasty) or arthrodesis.
-
Technique:
- Meticulous soft tissue handling.
- Careful debridement of synovium and any obvious crystal deposits.
- Address bone defects with grafting or specialized components.
- Consider prophylactic antibiotics specific for high-risk patients.
- For arthrodesis, ensure robust fixation to achieve fusion in a functional position.
- Post-operative: Aggressive medical management of the underlying crystal disorder is crucial to protect the implant.
Complications & Management
Surgical intervention in patients with crystal deposition arthropathy carries unique risks beyond those of standard orthopedic procedures, largely due to the inflammatory nature of the disease, systemic comorbidities, and compromised local tissue environment.
1. Acute Post-Operative Flare
- Incidence: Very common (up to 30-50%) following surgery in patients with gout or CPPD, often triggered by surgical stress, tissue damage, and changes in fluid status.
- Symptoms: Intense pain, swelling, erythema, and warmth at or around the surgical site, mimicking infection.
-
Management:
- Prevention: Prophylactic colchicine (e.g., 0.6 mg BID) initiated pre-operatively and continued for several weeks post-operatively is highly effective, especially for gout. NSAIDs can also be used.
- Treatment: Aggressive anti-inflammatory therapy with NSAIDs (if not contraindicated), colchicine, or corticosteroids (oral or intramuscular) as soon as a flare is recognized and infection is ruled out. Intra-articular corticosteroids can be considered for localized flares in the absence of infection.
2. Wound Complications
- Incidence: Higher in CDA patients, especially those with chronic tophaceous gout. Skin overlying tophi may be thin, poorly vascularized, or previously ulcerated.
-
Types:
- Dehiscence: Wound breakdown due to tissue fragility or excessive tension.
- Delayed Healing: Slow epithelialization, persistent drainage.
- Skin Necrosis: Particularly over superficial tophi where vascularity is compromised.
- Fistula Formation: Persistent sinus tracts from retained tophaceous material or infection.
-
Management:
- Prevention: Meticulous surgical technique, tension-free closure, careful pre-operative skin assessment, judicious use of drains.
- Treatment: Local wound care, serial debridement, antibiotics if infected. May require secondary closure, local flaps, or skin grafting for extensive defects.
3. Infection
- Incidence: Increased risk due to compromised skin integrity, chronic inflammation, systemic comorbidities, and immunosuppressive medications often used to treat CDA. Coexisting septic arthritis must always be considered in an acutely inflamed joint.
- Symptoms: Fever, chills, persistent or worsening pain, purulent drainage, erythema extending beyond the surgical site.
-
Management:
- Prevention: Strict aseptic technique, perioperative prophylactic antibiotics, careful wound management.
- Treatment: Prompt surgical debridement, irrigation, drainage, and appropriate broad-spectrum intravenous antibiotics tailored to culture results. Revision surgery or implant removal may be necessary in arthroplasty cases.
4. Nerve Injury
- Incidence: Risk during tophi excision, particularly in areas with superficial nerves (e.g., ulnar nerve at the elbow, digital nerves in the hand/foot, common peroneal nerve at the knee).
- Symptoms: Numbness, paresthesias, motor weakness in the distribution of the affected nerve.
-
Management:
- Prevention: Thorough pre-operative anatomical knowledge, careful surgical dissection, magnification.
- Treatment: Depends on the severity of injury (neuropraxia, axonotmesis, neurotmesis). Observation for neurapraxia. Surgical exploration and repair for transection.
5. Recurrence of Crystal Deposition / Tophi
- Incidence: High if the underlying metabolic disorder (e.g., hyperuricemia) is not effectively managed post-operatively.
- Symptoms: Re-accumulation of crystals, leading to pain, swelling, and potential re-emergence of complications.
-
Management:
- Prevention: Strict adherence to long-term medical therapy (e.g., ULT for gout) and regular monitoring of crystal levels.
- Treatment: Often medical. Repeat surgery only for specific complications as outlined in the indications.
6. Joint Stiffness / Reduced Range of Motion
- Incidence: Common after any joint surgery, exacerbated by prolonged inflammation or immobilization.
-
Management:
- Prevention: Early, controlled post-operative rehabilitation, judicious use of continuous passive motion (CPM) or splinting.
- Treatment: Physical therapy, occupational therapy, manipulation under anesthesia (if appropriate), or tenolysis/arthrolysis in recalcitrant cases.
| Complication | Incidence | Salvage Strategies |
|---|---|---|
| Acute Post-Operative Flare | Up to 50% (Gout), common (CPPD) | NSAIDs, Colchicine, Systemic Corticosteroids, Intra-articular Corticosteroids (if infection ruled out) |
| Wound Dehiscence/Poor Healing | Variable, higher in chronic tophaceous gout | Local wound care, Serial debridement, Secondary closure, Local flaps, Skin grafts |
| Surgical Site Infection | Elevated risk compared to general orthopedics | Prompt surgical debridement, Irrigation, IV Antibiotics (culture-directed), Implant removal (if arthroplasty) |
| Nerve Injury | Dependent on location and procedure | Observation for neurapraxia, Surgical exploration and repair for severe injury, Nerve grafting |
| Recurrence of Crystal Deposition | High if underlying metabolic disorder unmanaged | Optimize long-term medical therapy (e.g., ULT), Repeat surgical debridement for symptomatic recurrence |
| Joint Stiffness | Common after joint surgery | Aggressive physical therapy, Occupational therapy, Manipulation Under Anesthesia, Arthrolysis/Tenolysis |

Post-operative appearance following excision of large tophaceous deposits from the dorsal foot. Note the meticulous closure despite significant tissue loss.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation following surgery for crystal deposition arthropathy must be individualized, considering the specific surgical procedure, the patient's overall health, and the need to manage the underlying inflammatory condition. The primary goals are to optimize functional recovery, prevent stiffness, manage pain, and crucially, prevent recurrence or exacerbation of the crystal-induced inflammation.
General Principles
- Pain Management: Aggressive pain control is essential to facilitate early mobilization. This includes multimodal analgesia, regional blocks, and appropriate oral medications.
- Inflammation Control: Continue medical management of the underlying crystal arthropathy. Prophylactic colchicine or NSAIDs should be maintained to prevent post-operative flares. Close monitoring for signs of flare is paramount.
- Wound Care: Meticulous wound care is critical, especially given potential for compromised skin. Monitor for signs of infection, dehiscence, or delayed healing.
- Early Mobilization (as appropriate): Depending on the procedure, controlled early motion helps prevent stiffness, adhesion formation, and deep vein thrombosis.
- Protection of Surgical Site: Protect the surgical area from undue stress, particularly for procedures involving bone or tendon repair, using appropriate splints, braces, or limited weight-bearing protocols.
Specific Rehabilitation Considerations
A. After Tophi Excision or Calcific Deposit Removal
-
Initial Phase (0-2 weeks):
- Elevation and Edema Control: Consistent elevation of the limb to reduce swelling.
- Wound Care: Dressing changes, monitor for infection.
- Immobilization: May require a splint or cast for comfort and protection, especially if extensive soft tissue dissection or bone work was performed. However, aim for minimal immobilization to avoid stiffness.
- Gentle Range of Motion (ROM): Commence passive or gentle active-assisted ROM of adjacent, uninvolved joints.
-
Intermediate Phase (2-6 weeks):
- Active ROM: Progress to active ROM of the involved joint(s) as wound healing permits.
- Scar Management: Gentle massage and stretching to prevent scar contracture.
- Strengthening: Begin isometric exercises, progressing to light resistance as tolerated.
- Weight-bearing: Gradual progression to full weight-bearing, if applicable, based on surgical site and stability.
-
Advanced Phase (6+ weeks):
- Functional Training: Focus on regaining full strength, endurance, and functional activities specific to the patient's needs.
- Return to Activity: Gradual return to sports or high-impact activities, ensuring no residual pain or instability.
B. After Diagnostic Arthroscopy/Synovectomy
-
Initial Phase (0-1 week):
- Pain and Edema Control: Ice, elevation, analgesics.
- Early ROM: Gentle active and passive ROM exercises to prevent stiffness.
- Weight-bearing: Typically immediate weight-bearing as tolerated for lower extremity joints.
-
Intermediate/Advanced Phases:
- Focus on restoring full ROM, strength, and proprioception. Full recovery is usually quicker than with more invasive procedures.
C. After Arthroplasty or Arthrodesis for End-Stage Arthropathy
- Protocols: Generally follow standard post-arthroplasty or arthrodesis protocols for the specific joint (e.g., total knee, hip, ankle arthroplasty).
-
Key Adaptations:
- Wound Vigilance: Enhanced vigilance for wound complications and infection due to the patient's underlying condition and potential comorbidities.
- Flare Management: Prophylactic anti-inflammatory medications and swift management of any post-operative flares are critical to protect the implant and facilitate rehabilitation.
- Bone Stock/Soft Tissue Concerns: Rehabilitation may be modified if significant bone loss or soft tissue compromise impacted implant stability or repair integrity.
- Long-Term Medical Management: Emphasize the absolute necessity of ongoing and optimized medical management of the crystal deposition disease to prevent recurrence around the implant or in other joints.
Summary of Key Literature / Guidelines
The literature concerning crystal deposition arthropathy predominantly focuses on medical diagnosis and management, with surgical interventions primarily addressing complications. As academic orthopedic surgeons, our understanding must integrate both rheumatologic guidelines and orthopedic surgical principles.
1. American College of Rheumatology (ACR) Guidelines
- The ACR provides comprehensive guidelines for the management of gout and CPPD.
-
Gout:
ACR guidelines emphasize:
- Acute Flare Management: NSAIDs, colchicine, or corticosteroids as first-line. Combination therapy may be necessary for severe flares.
- Urate-Lowering Therapy (ULT): Allopurinol and febuxostat are xanthine oxidase inhibitors, while probenecid is a uricosuric agent. Pegloticase is reserved for refractory chronic gout. ULT is indicated for patients with tophi, frequent flares, CKD stage 2 or higher, or a history of urolithiasis.
- Target Serum Urate: Below 6 mg/dL, or below 5 mg/dL for severe gout.
- Lifestyle Modifications: Dietary advice (avoid purine-rich foods, alcohol, sugary drinks), weight loss.
-
CPPD:
ACR guidelines acknowledge the diverse clinical presentations of CPPD and emphasize:
- Acute Pseudogout Flare Management: NSAIDs, colchicine, or corticosteroids. Joint aspiration with crystal analysis is diagnostic and therapeutic.
- Chronic CPPD Arthropathy: Management is similar to osteoarthritis, focusing on pain control, physical therapy, and functional improvement. No specific crystal-dissolving therapy exists.
- Associated Conditions: Screen for underlying metabolic conditions such as hemochromatosis, hyperparathyroidism, and hypomagnesemia.
2. Orthopedic Literature on Surgical Management
-
Tophi Excision:
Literature supports surgical excision of symptomatic tophi for nerve compression, skin breakdown, infection, or severe functional impairment. Studies emphasize meticulous technique, aggressive debridement, and the importance of post-operative gout prophylaxis to prevent flares. Outcomes are generally favorable for symptom relief, but recurrence is possible if hyperuricemia is uncontrolled.
- Reference: Kumar, A., et al. "Surgical management of chronic tophaceous gout: a systematic review." Journal of Clinical Rheumatology , 2017.
-
Arthroplasty in CDA:
Total joint arthroplasty for end-stage arthropathy secondary to gout or CPPD can provide significant pain relief and functional improvement. However, studies highlight:
- Increased risk of perioperative complications, including infection, wound healing issues, and post-operative flares.
- The necessity of aggressive medical management of the underlying crystal disorder to improve implant longevity and reduce complication rates.
- The importance of preoperative evaluation of the soft tissue envelope and bone stock.
- Reference: Simsek, D., et al. "Total joint arthroplasty in gouty arthritis: a systematic review." International Orthopaedics , 2018.
-
Calcific Tendinopathy:
Surgical removal of calcific deposits, particularly in the shoulder, is well-established for patients with persistent symptoms unresponsive to conservative measures. Arthroscopic techniques have demonstrated good clinical outcomes with minimal morbidity.
- Reference: Loew, M., et al. "Clinical results of arthroscopic removal of calcifying deposits in the shoulder." Arthroscopy: The Journal of Arthroscopic and Related Surgery , 2003.
- Diagnostic Role: Joint aspiration and synovial biopsy remain critical diagnostic tools, particularly to differentiate crystal arthropathy from septic arthritis, an orthopedic emergency. The proper handling of synovial fluid (polarizing microscopy for crystals) and biopsy specimens is paramount.

Histopathology image depicting calcium pyrophosphate dihydrate (CPPD) crystals within articular cartilage, a hallmark of CPPD disease (hematoxylin and eosin stain).
3. Multidisciplinary Approach
- Consensus across specialties emphasizes a multidisciplinary approach involving rheumatologists, nephrologists, primary care physicians, and orthopedic surgeons for optimal management of CDA, especially in complex cases or those requiring surgical intervention. This ensures comprehensive medical optimization, prevention of systemic complications, and coordinated post-operative care.
In conclusion, while surgical intervention for crystal deposition arthropathy is typically reserved for complications or end-stage disease, the orthopedic surgeon plays a vital role in diagnosis, managing specific complications (tophi, tendinopathy, nerve compression), and performing reconstructive procedures when joints are severely damaged. A thorough understanding of the pathophysiology, a robust pre-operative workup, meticulous surgical technique, and vigilant post-operative care are essential for achieving favorable outcomes.
Clinical & Radiographic Imaging