CONGENITAL DEFICIENCIES OF THE LONG BONES
The evaluation and management of congenital long bone deficiencies represent some of the most complex challenges in pediatric orthopedic surgery. The first highly structured, scientific approach to the taxonomy of congenital long bone deficiencies was devised by Frantz and O’Rahilly in 1961. Their widely utilized classification system fundamentally categorized deficiencies as either terminal or intercalary.
In terminal deficiencies, there is an amputation-like presentation with no anatomically normal body parts distal to the affected site. Conversely, in intercalary deficits, a middle segment of the limb is absent, but the distal segments remain present and often functional.
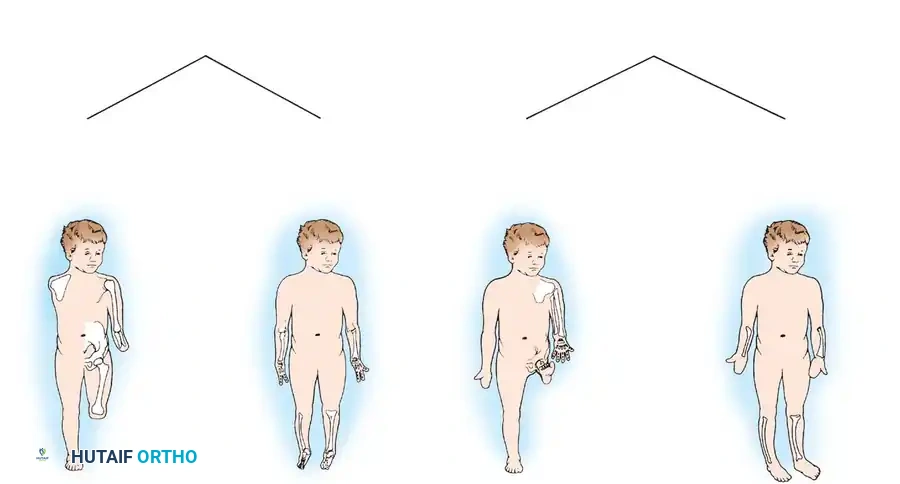
Terminal and intercalary deficiencies are further delineated as transverse or longitudinal. For instance, the complete absence of a hand at the wrist is classified as a terminal transverse deficiency. A complete hand presenting without a radius or ulna is an intercalary transverse deficiency. An example of a terminal longitudinal deficiency is fibular hemimelia, in which the lateral two rays of the foot are also missing. Fibular hemimelia in which the foot is entirely normal is classified as an intercalary longitudinal deficiency.
In 1964, Swanson, in conjunction with the American Society for Surgery of the Hand and the Federation of Societies for Surgery of the Hand, devised a more specific classification scheme based on embryological failures (e.g., failure of formation, failure of differentiation). Although this system was originally devised for upper extremity deficiencies, it is highly applicable to the lower extremities. Similar to its predecessors, the Swanson classification scheme aids significantly in taxonomy and academic communication, but it falls short in dictating specific treatment planning. Regardless of the taxonomic label, each child must be evaluated meticulously, as phenotypic expression is highly variable and no two children present exactly alike.

TIBIAL HEMIMELIA
Since the disorder was first described by Otto in 1941, tibial hemimelia has been known by a variety of names in the academic literature, including congenital longitudinal deficiency of the tibia, congenital dysplasia of the tibia, paraxial tibial hemimelia, tibial dysplasia, and congenital deficiency or absence of the tibia.
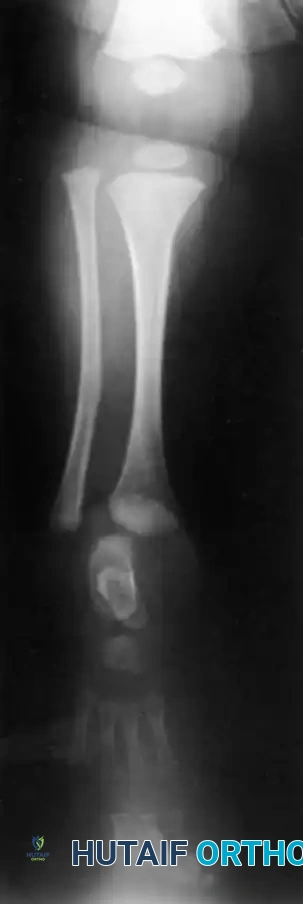
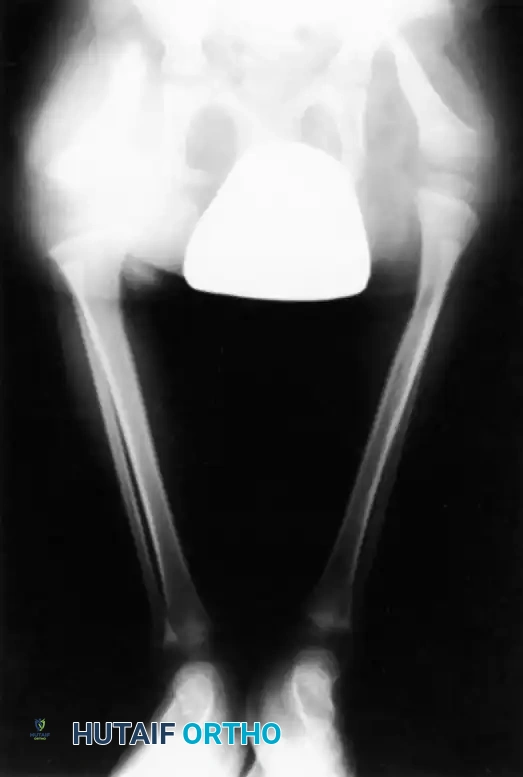
This condition represents a broad spectrum of deformities, ranging from the total absence of the tibia (the most severe form) to mild hypoplasia of the tibia (the least severe form). The incidence is exceedingly rare, estimated at one in 1 million live births, and the condition presents bilaterally in approximately 30% of affected patients.
Genetics and Pathoanatomy
Tibial hemimelia usually occurs sporadically, although familial cases demonstrating either autosomal dominant or recessive transmission patterns have been reported. At least four distinct genetic syndromes feature tibial hemimelia as a primary component:
* Werner Syndrome: Polydactyly–triphalangeal thumb syndrome.
* Tibial Hemimelia Diplopodia.
* Tibial Hemimelia–Split Hand/Foot Syndrome.
* Tibial Hemimelia–Micromelia–Trigonal Brachycephaly Syndrome.
Although the exact embryological insult remains unknown, Sweet and Lane described a murine model for tibial hemimelia in which the dominant mutation resides on the X chromosome.
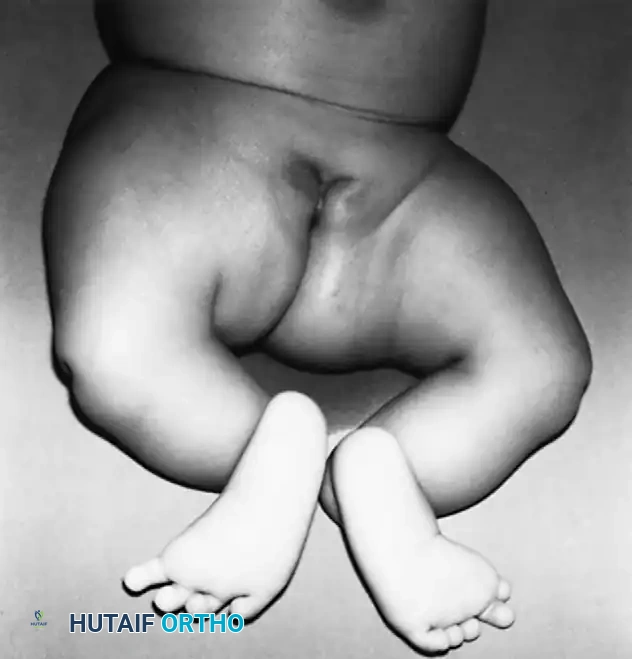
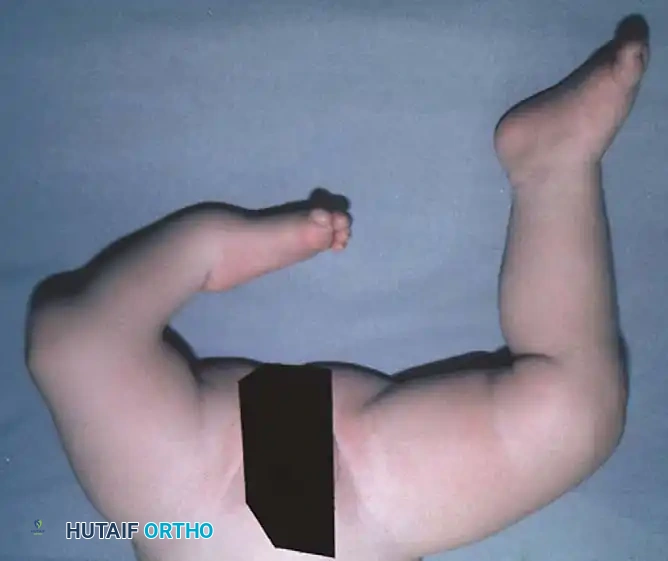
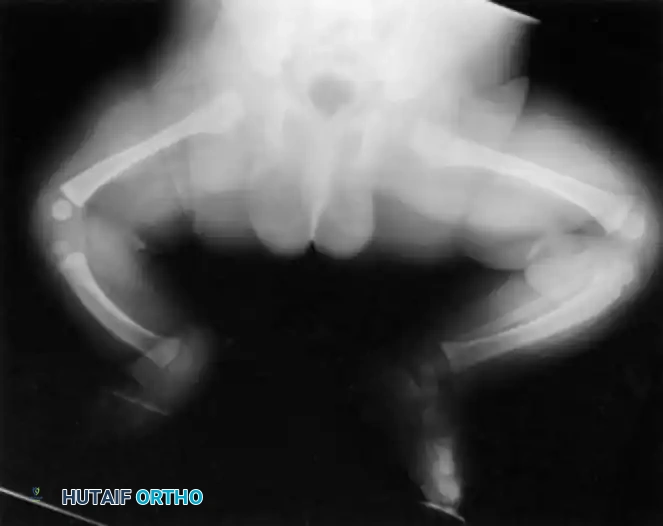
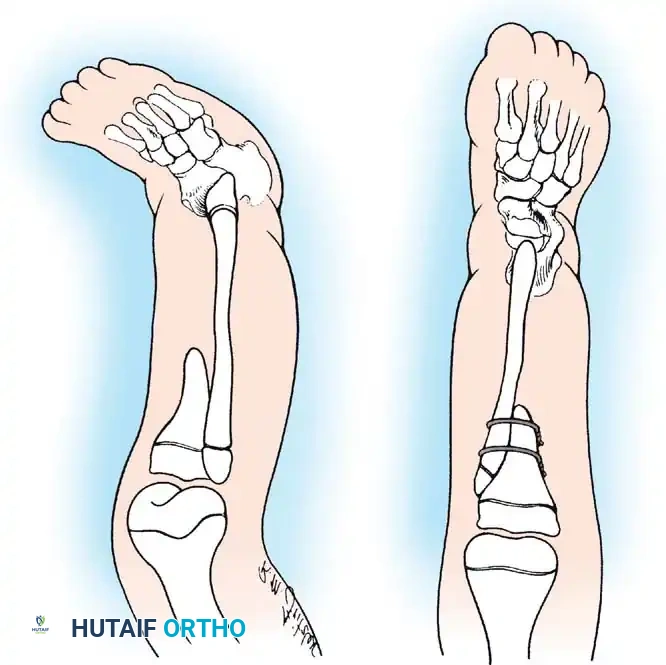
Clinically, the involved leg is significantly shortened. The fibular head is often palpable, particularly if it is proximally displaced into the popliteal fossa. The foot is typically held in severe, rigid equinovarus, and the hindfoot is profoundly stiff.
In older children, the proximal tibial anlage may be palpable upon deep clinical examination, even if it is not radiographically visible. The knee is generally flexed, and in more severe deformities, profound quadriceps insufficiency causes an absolute lack of active knee extension.
Clinical Pearl: Careful clinical evaluation of the quadriceps extensor mechanism is paramount. The presence or absence of active quadriceps function has the most significant prognostic value regarding the potential for successful surgical reconstruction of the knee joint.
Vascular anomalies are common. The anterior tibial artery is frequently absent, and the plantar arterial arch is incomplete. Similar vascular findings in severe clubfoot and fibular hemimelia suggest that reduced embryonic vascular flow may be a primary etiological factor. Associated anomalies generally correlate with the severity of the tibial deficiency.
Classification of Tibial Hemimelia
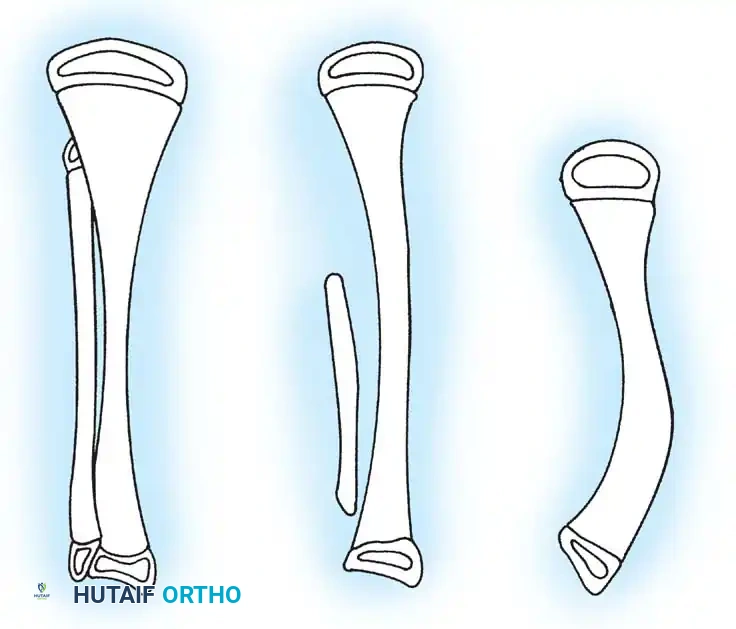
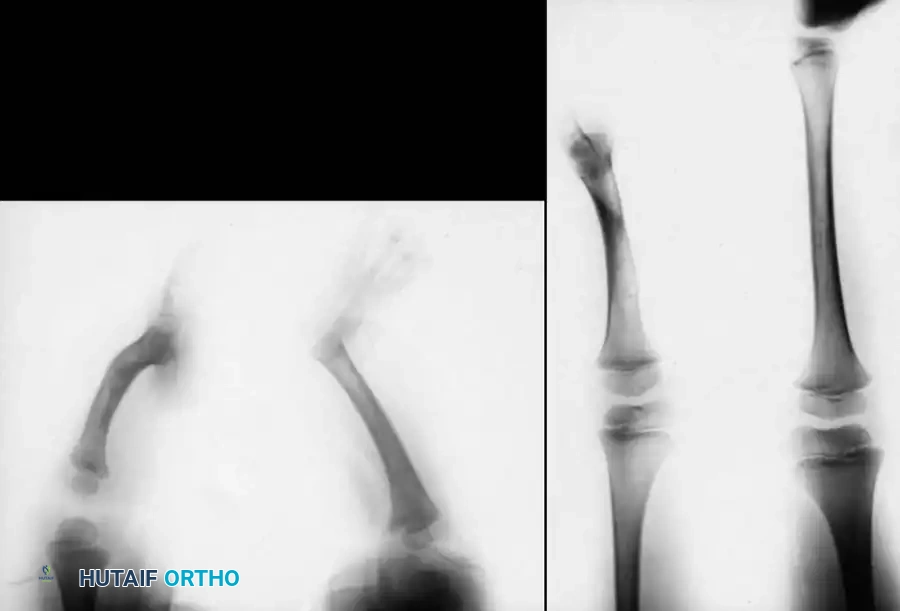
The most widely utilized and surgically relevant classification scheme for tibial hemimelia is that of Jones, Barnes, and Lloyd-Roberts. This system is based on early radiographic presentation and directly guides treatment recommendations.
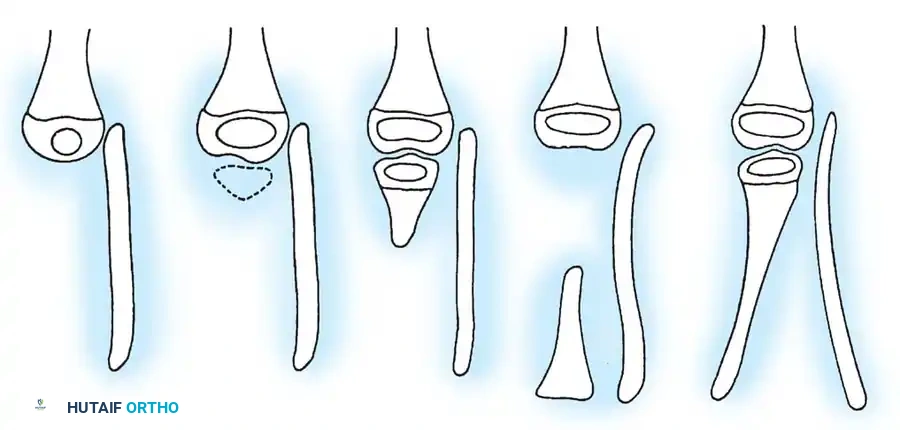
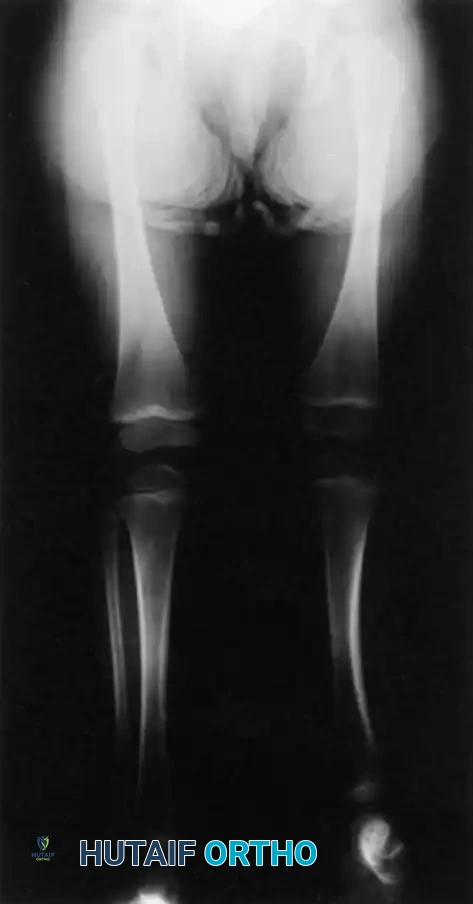
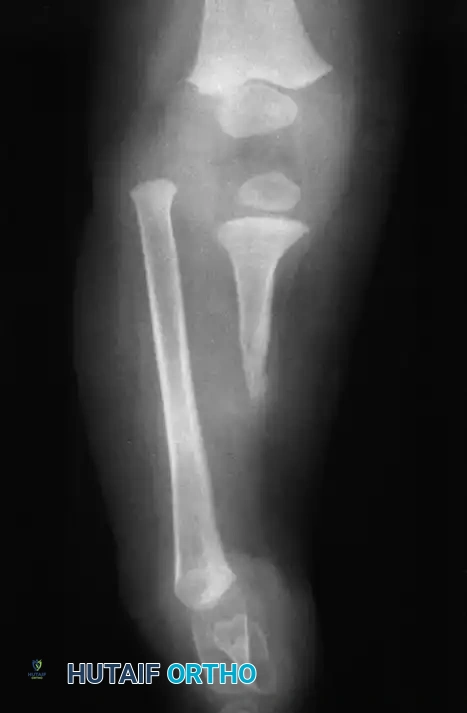
- Type 1A Deformity: There is complete radiographic absence of the tibia and a hypoplastic distal femoral epiphysis compared with the contralateral normal side.
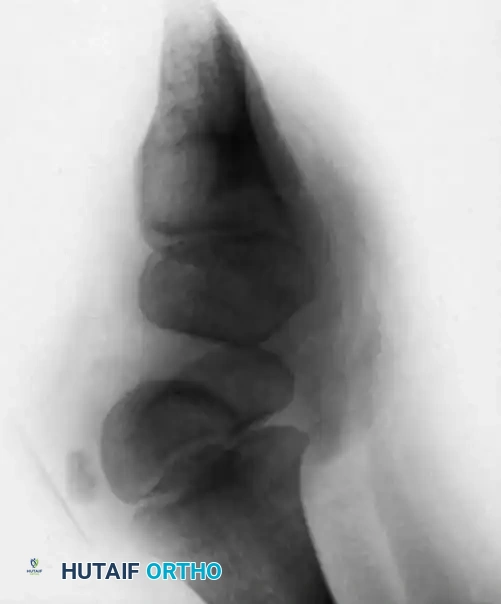
- Type 1B Deformity: There is no radiographic evidence of an ossified tibia; however, the distal femoral epiphysis appears more normal in size and shape. This distinction is critical because Type 1B deformities possess a proximal tibial cartilaginous anlage that can be expected to ossify over time. Modern imaging techniques, such as high-resolution ultrasound and MRI, are mandatory to identify this cartilaginous anlage.
- Type 2 Deformity: A proximal tibia of varying size is present and ossified at birth. The fibula is usually normal in size, but the fibular head is proximally dislocated.
- Type 3 Deformity: The proximal tibia is not radiographically visible, but the distal tibial epiphysis is present. This is an exceptionally rare variant. The distal femoral epiphysis is usually well-formed, but the upper end of the fibula is proximally dislocated, leading to profound knee instability.
- Type 4 Deformity: The tibia is shortened, and there is proximal migration of the fibula with distal tibiofibular diastasis. This rare deformity is also referred to as congenital diastasis of the ankle joint. The distal tibial epiphysis may be entirely absent.
SURGICAL MANAGEMENT PRINCIPLES
As with all congenital lower limb deficiencies, the ultimate goal of treatment is to provide a functional, stable limb that is equal in length to the normal limb, allowing for energy-efficient ambulation. The specific surgical intervention depends heavily on the radiographic classification, the presence of a cartilaginous anlage, and the clinical integrity of the extensor mechanism.
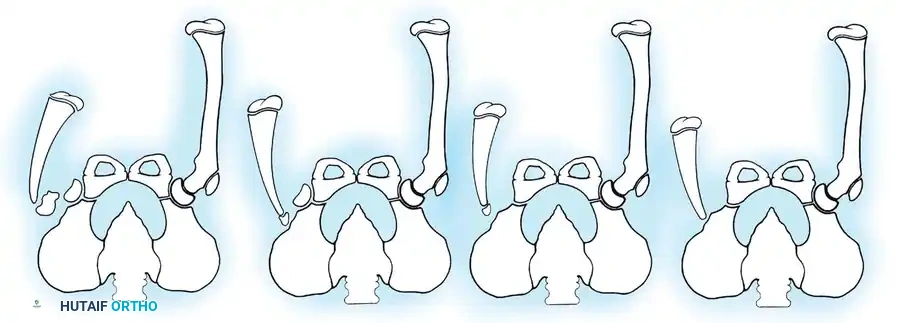
Management of Type 1A Deformities
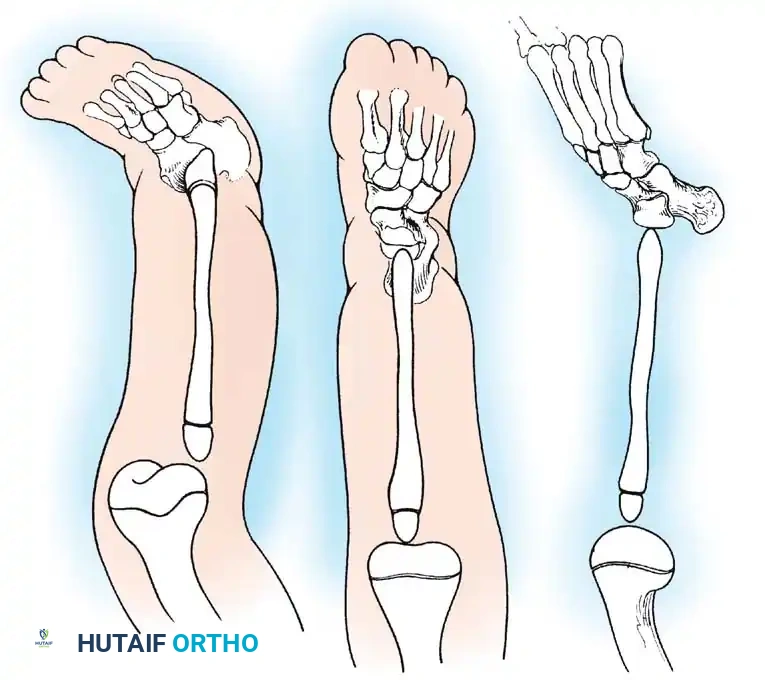
For severe Type 1A deficiencies, amputation and prosthetic rehabilitation remain the most practical and reliable means of treatment. The two historical options are knee disarticulation and complex knee reconstruction (the Brown procedure).
Knee Disarticulation:
The most effective and predictable option is knee disarticulation followed by fitting with an above-knee prosthesis. This provides a definitive solution with a single operation. Knee disarticulation is vastly preferred over a trans-femoral (above-knee) amputation because diaphyseal amputations in young children frequently result in severe skin complications secondary to bony stump overgrowth. Because the ultimate femoral growth is often diminished in these patients, the end result of a knee disarticulation functions biomechanically like an above-knee amputation level. Children treated with early disarticulation are almost uniformly active, highly functional prosthetic users.
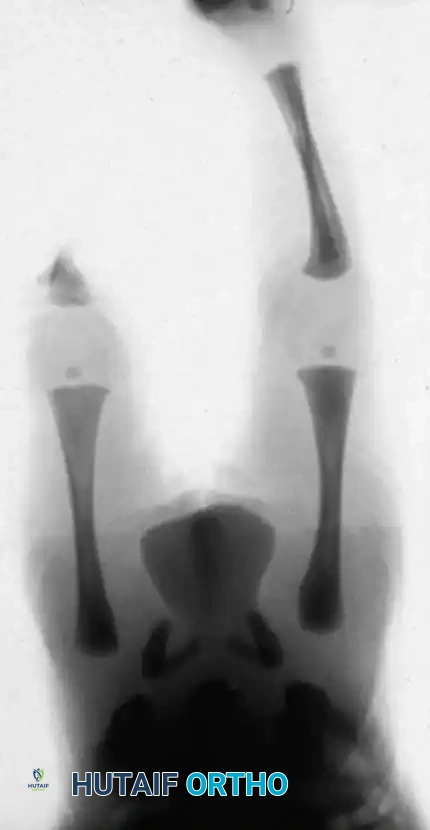
The Brown Procedure (Historical Context):
Historically, Brown described a reconstruction for Type 1A tibial hemimelia in which the fibula was surgically centralized and transferred into the intercondylar notch of the femur to create a neo-tibia.
Surgical Warning: The Brown procedure is rarely performed today. Subsequent long-term outcome studies have reported failure rates ranging from 50% to 100%. Patients frequently require secondary revision procedures or eventual amputation due to progressive knee flexion contractures, severe ligamentous instability, and insufficient quadriceps strength.
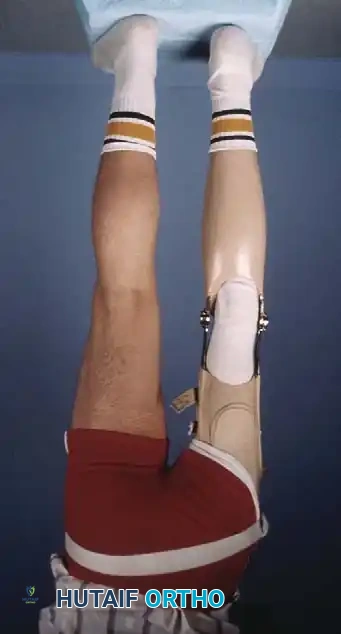
Management of Type 1B and Type 2 Deformities
In Type 1B and Type 2 deformities, a functional knee joint exists (or will develop from the anlage), and knee disarticulation is contraindicated provided the quadriceps mechanism is functional.
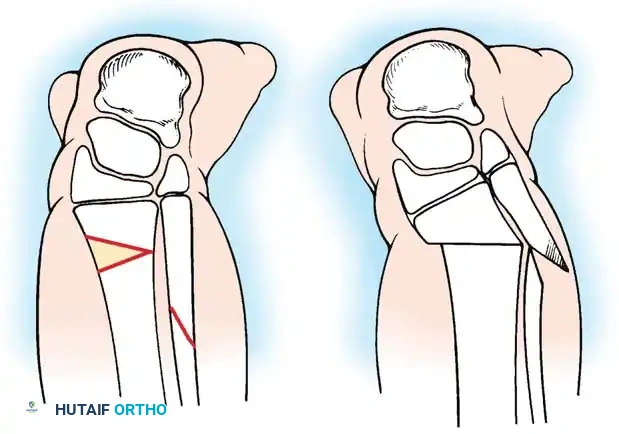
The treatment of choice is a proximal tibiofibular synostosis combined with a Syme amputation or complex distal reconstruction. Creating a synostosis between the fibula and the proximal tibia creates a uniform, in-line, weight-bearing mechanical axis. If the fibula is not surgically transferred and fused to the tibia, a peculiar, curved, hypertrophied fibula develops, causing a severe secondary deformity. Fusing the fibula underneath the tibia encourages its hypertrophy in a mechanically advantageous alignment.
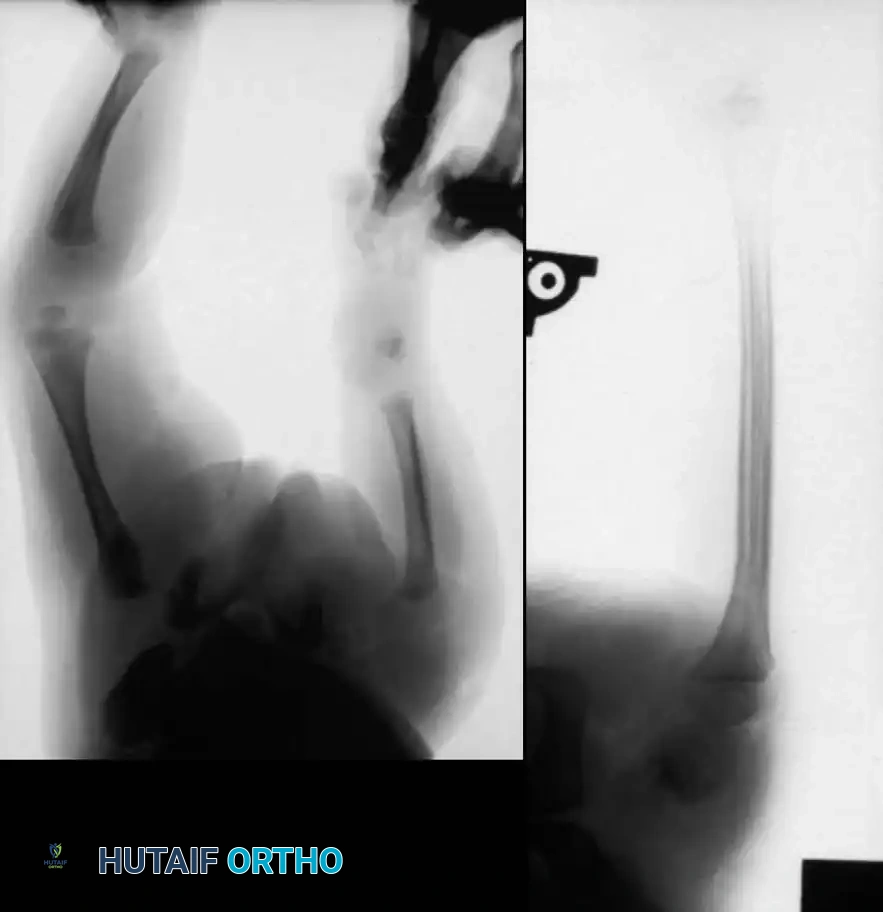
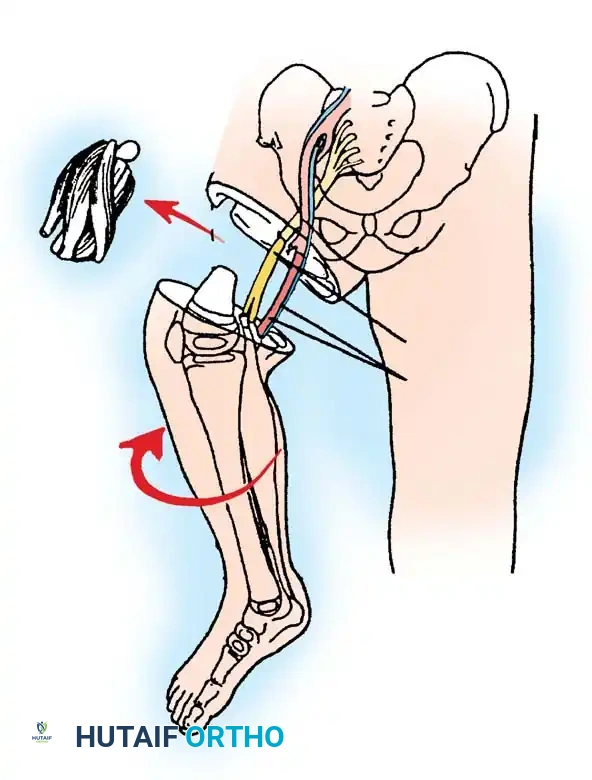
SURGICAL TECHNIQUES
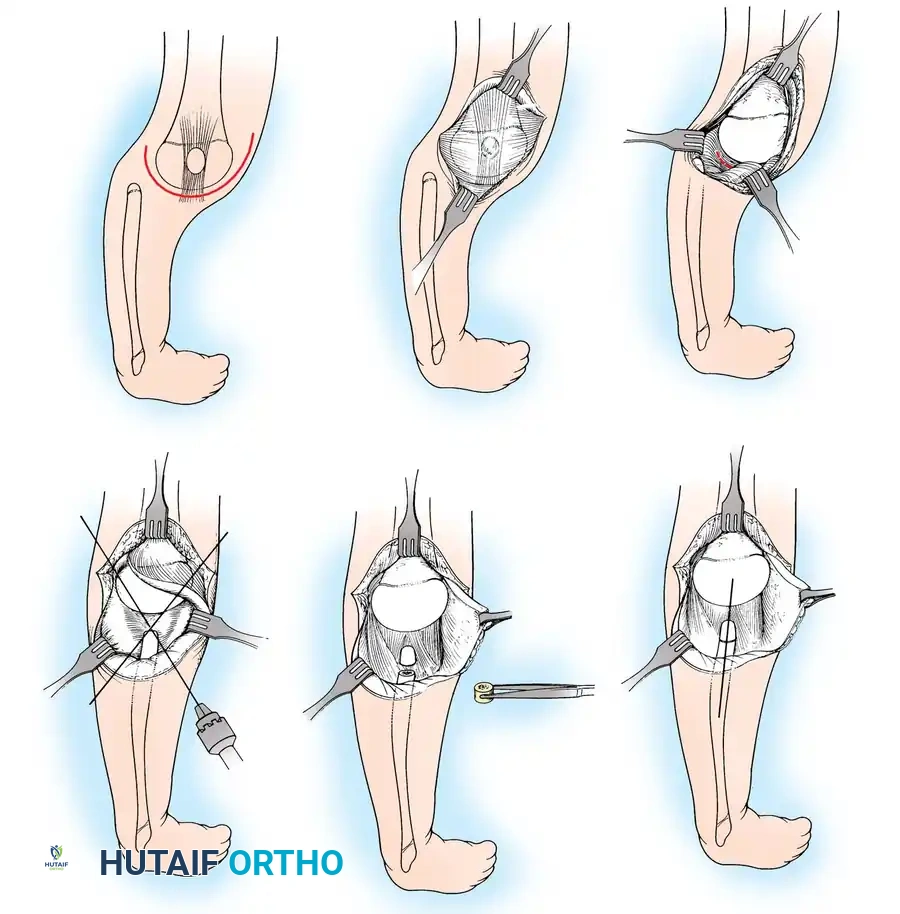
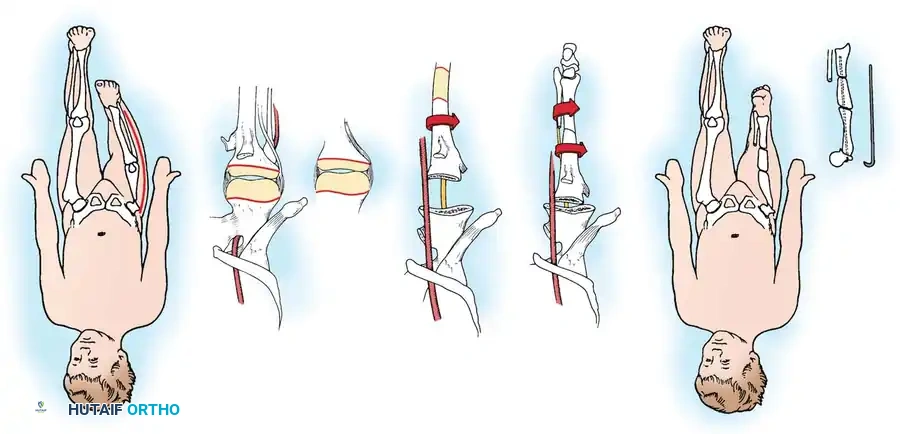
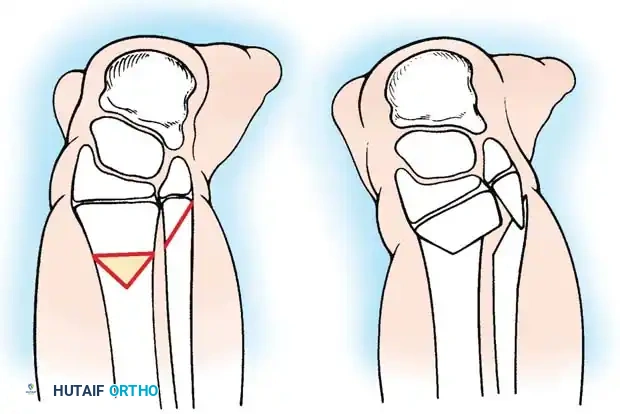
Lateral Release, Medial Plication, and Patellar Tendon Transfer
(Technique 26-25: Beaty; Modified from Gao et al. and Langenskiöld)
This procedure is indicated for congenital patellar dislocations and severe extensor mechanism malalignment often seen in conjunction with congenital long bone deficiencies. The goal is to centralize the extensor vector and stabilize the patella within the trochlear groove.
Patient Positioning and Preparation:
1. Place the patient supine on a radiolucent operating table.
2. Apply a well-padded pneumatic tourniquet to the proximal thigh.
3. Prep and drape the limb in a standard sterile fashion, allowing for full, unhindered range of motion of the knee.
Surgical Approach:
1. Make a midline incision extending from the distal aspect of the femur to the tibial tuberosity.
* Note: Because a midline surgical incision over the patella tends to heal with proliferative scarring in children, an anterolateral incision may be utilized so that the resulting scar is not under direct pressure against the patella during kneeling.
2. Perform a full-thickness skin dissection over the patella to expose the medial and lateral aspects of the knee joint and the quadriceps femoris muscle. The incision must be long enough to expose a sufficient portion of the quadriceps muscle for realignment. In an infant with severe congenital patellar dislocation, this incision may need to extend halfway up the thigh.
Lateral Release:
3. Release the vastus lateralis from its most proximal muscle origin in the quadriceps femoris down to the level of the joint.
4. This step frequently requires the release of the iliotibial band laterally down to the intermuscular septum to eliminate all lateral tethering forces.
5. Optional: Occasionally, the rectus femoris is severely contracted and must be dissected and lengthened via a formal Z-plasty.
Medial Plication and Transfer:
6. Incise the vastus medialis obliquus (VMO) from its origin proximally and distally from the patella, the medial capsule, and the patellar tendon.
7. Manually reduce the patella into the femoral trochlear groove.
8. Reattach the VMO laterally and distally to the patellar tendon and medial retinaculum to securely lock the patella within the femoral groove.
Intraoperative Assessment:
9. Once the initial distal suture is placed, move the knee through a gentle, full range of motion to assess the reduction and tracking of the patella.
* Pitfall: If the tension on the VMO is too tight, it will restrict flexion. Remove the suture and transfer the muscle slightly more proximally.
* Pitfall: If the tension is too lax, the patella will subluxate laterally. Attach the VMO further distally and laterally.
10. In cases of extreme instability, the gracilis or semitendinosus tendon must be divided at the musculotendinous junction and transferred into the patella to act as a dynamic checkrein. The VMO is then sutured to the remaining retinaculum.
Closure and Postoperative Protocol:
11. Continue the repair of the VMO proximally and distally, ensuring a watertight capsular closure.
12. Perform a final range of motion check to ensure normal tracking during full flexion and extension.
13. Deflate the tourniquet and obtain meticulous hemostasis with electrocautery.
14. Insert a deep suction drain, and close the subcutaneous tissue and skin in layers.
15. Apply a well-molded long leg cast with the knee positioned in 30 degrees of flexion to protect the medial repair. The cast is typically maintained for 6 weeks before initiating progressive rehabilitation.
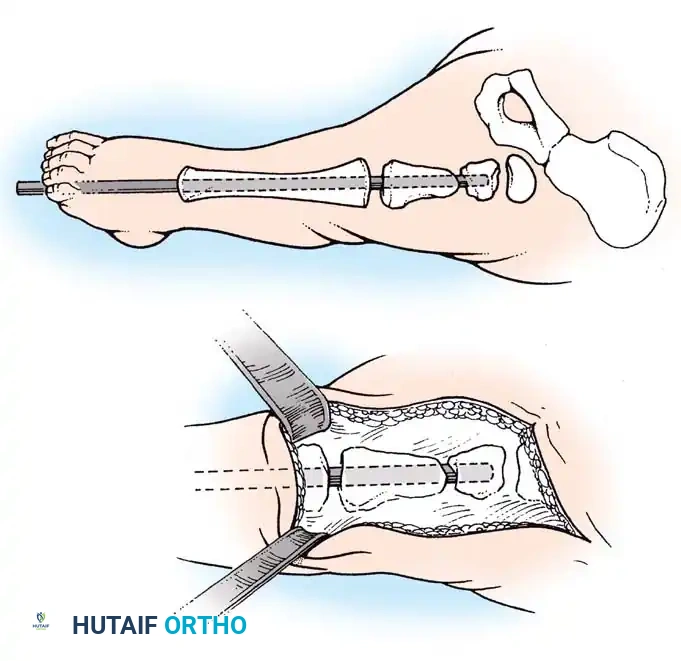
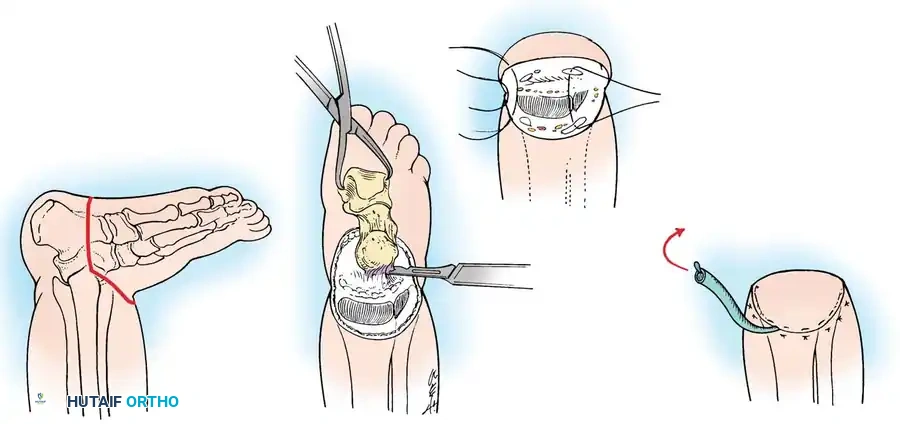
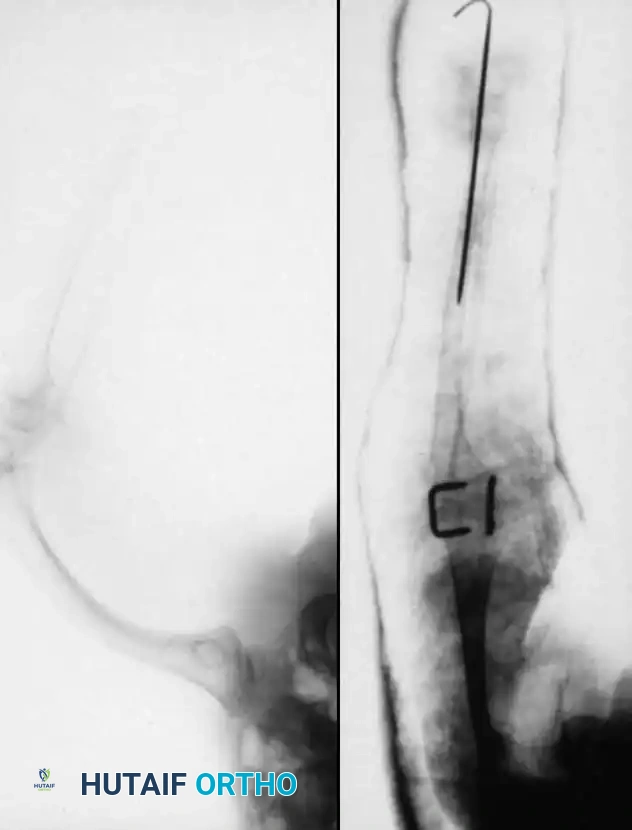
Proximal Tibiofibular Synostosis
For Type 1B and Type 2 tibial hemimelia, centralizing the mechanical axis is critical. The fibula must be brought under the mechanical axis of the femur or the existing proximal tibial anlage.
Technique Overview:
1. A lateral or anterolateral approach is utilized to expose the proximal fibula and the tibial anlage.
2. The proximal fibula is carefully osteotomized or mobilized, taking extreme care to protect the common peroneal nerve, which often has an anomalous course in these patients.
3. The cartilaginous tibial anlage is split or prepared to receive the proximal fibula.
4. The fibula is centralized beneath the tibia/anlage and fixed using smooth Kirschner wires or a small fragment plate, depending on the child's age and bone quality.
5. Bone graft (autograft or allograft) is packed around the synostosis site to promote rapid union.
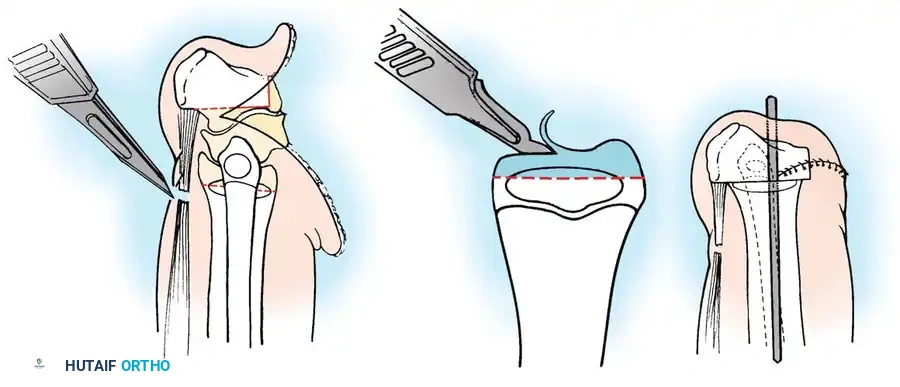
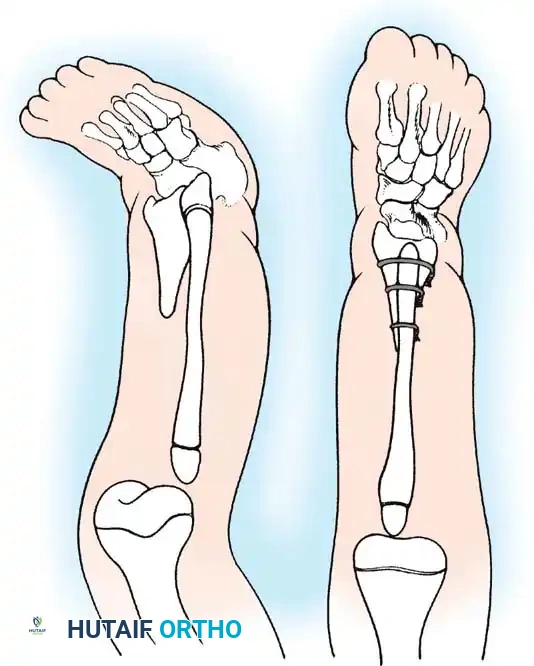
By fusing the fibula to the tibia, the surgeon prevents the progressive lateral bowing and hypertrophy of the fibula that inevitably occurs if it is left in its native, eccentric position. This synostosis provides a robust, centralized bony column that can later be lengthened or fitted with a prosthesis following a distal Syme amputation.
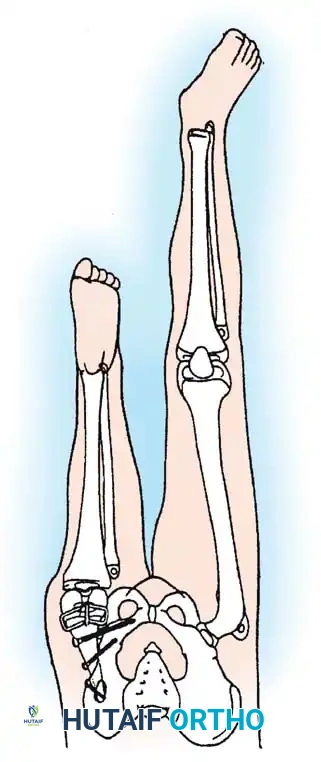
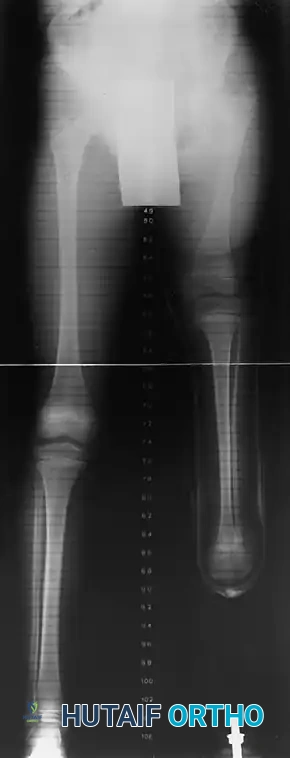
POSTOPERATIVE REHABILITATION AND PROSTHETICS
The rehabilitation phase following surgical intervention for tibial hemimelia is lifelong. For patients undergoing knee disarticulation (Type 1A), prosthetic fitting should begin as soon as the surgical wound is completely healed, typically around 6 to 8 weeks postoperatively. Early fitting is crucial to integrate the prosthesis into the child's developing body image and to facilitate normal motor milestone acquisition (e.g., pulling to stand, cruising).

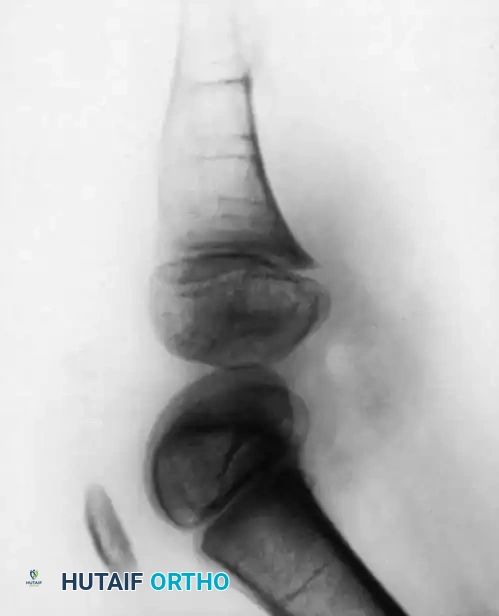
For patients undergoing tibiofibular synostosis and distal amputation (Type 1B/2), the limb is protected in a cast until radiographic union of the synostosis is confirmed. Once healed, the patient is fitted with a specialized prosthesis that accommodates the reconstructed limb. Because these limbs often lack normal longitudinal growth, the patient will require frequent prosthetic adjustments and may eventually be a candidate for limb lengthening procedures (e.g., Ilizarov or Taylor Spatial Frame) as they approach skeletal maturity.
COMPLICATIONS AND PITFALLS
The surgical management of congenital long bone deficiencies is fraught with potential complications.
- **
Associated Surgical & Radiographic Imaging
📚 Medical References
- Aitken GT: Amputation as a treatment for certain lower-extremity congenital anomalies, J Bone Joint Surg 41A:1267, 1959.
- Aitken GT: Tibial hemimelia: a symposium on selected lower limb anomalies: surgical and prosthetic management, Washington, DC, 1971, National Academy of Sciences. Aitken GT: The child amputee: an overview, Orthop Clin North Am 3:447, 1972.
- Aitken GT: Congenital lower limb defi ciencies, Instr Course Lect 24:81, 1975.
- Bose K: Congenital diastasis of the inferior tibiofi bular joint: report of a case, J Bone Joint Surg 58A:886, 1976.
- Brown FW: Construction of a knee joint in congenital total absence of the tibia (paraxial hemimelia tibia): a preliminary report, J Bone Joint Surg 47A:695, 1965.
- Brown FW, Pohnert WH: Construction of a knee joint in meromelia tibia (congenital absence of the tibia): a fi fteen-year follow-up study, J Bone Joint Surg 54A:1333, 1972.
- Christini P, Levy EJ, Facanha FA, et al: Fibular transfer for congenital absence of the tibia, J Pediatr Orthop 13:378, 1993.
- de Sanctis N, Razzano E, Scognamiglio R, et al: Tibial agenesis: a new rationale in management of type II—report of three cases with long-term follow-up, J Pediatr Orthop 10:198, 1990.
- Epps CH Jr, Tooms RE, Edholm CD, et al: Failure of centralization of the fi bula for congenital longitudinal defi ciency of the tibia, J Bone Joint Surg 73A:858, 1991.
- Fernandez-Palazzi F, Bendahan J, Rivas S: Congenital defi ciency of the tibia: a report on 22 cases, J Pediatr Orthop B 7:298, 1998.
- Garbarino JL, Clancy M, Harcke HT, et al: Congenital diastasis of the inferior tibiofi bular joint: a review of the literature and report of two cases, J Pediatr Orthop 5:573, 1985.
- Grissom LE, Harcke HT, Kumar SJ: Sonography in the management of tibial hemimelia, Clin Orthop Relat Res 251:266, 1990.
- Herring JA, Lloyd-Roberts G: Instructional case: management of tibial dysplasia, J Pediatr Orthop 1:339, 1981.
- Igou RA Jr, Kruger LM: Fibula dimelia in association with ipsilateral proximal focal femoral defi ciency, tibial defi ciency, and polydactyly: a case report, Clin Orthop Relat Res 258:237, 1990.
- Jayakumar SS, Eilert RE: Fibular transfer for congenital absence of the tibia, Clin Orthop Relat Res 139:97, 1979.
- Jones E, Barnes J, Lloyd-Roberts GC: Congenital aplasia and dysplasia of the tibia with intact fi bula: classifi cation and management, J Bone Joint Surg 60B:31, 1978.
- Kalamchi A, Dawe RV: Congenital defi ciency of the tibia, J Bone Joint Surg 67B:581, 1985.
- Kumar A, Kruger LM: Fibular dimelia with defi ciency of the tibia, J Pediatr Orthop 13:203, 1993.
- Lamb DW, Wynne-Davies R, Whitmore JM: Five-fi ngered hand associated with partial or complete tibial absence and pre-axial polydactyly: a kindred of 15 affected individuals in fi ve generations, J Bone Joint Surg 65B:60, 1983.
- Loder RT, Herring JA: Fibular transfer for congenital absence of the tibia: a reassessment, J Pediatr Orthop 7:8, 1987.
- Miller LS, Armstrong PF: The morbid anatomy of congenital defi ciency of the tibia and its relevance to treatment, Foot Ankle 3:396, 1992.
- Otto AW: Monstorum sexeentorum descripto anatomica sumptibus , Breslau, 1841, Ferdinandi Hirt. Putti V: Operative lengthening of the femur, Surg Gynecol Obstet 58:318, 1934.
- Schoenecker PL, Capelli AM, Millar EA, et al: Congenital longitudinal defi ciency of the tibia, J Bone Joint Surg 71A:278, 1989.
- Simmons ED, Ginsburg GM, Hall JE: Brown’s procedure for congenital absence of the tibia revisited, J Pediatr Orthop 16:85, 1996.
- Sweet HO, Lane PW: X-linked polydactyly (Xpl): a new mutation in the mouse, J Hered 71:207, 1980.
- Wada A, Fujii T, Takamura K, et al: Limb salvage treatment for congenital defi ciency of the tibia, J Pediatr Orthop 26:226, 2006.
- Williams L, Wientroub S, Getty CJ, et al: Tibial dysplasia: a study of the anatomy, J Bone Joint Surg 65B:157, 1983.
- Fibular Hemimelia Achterman C, Kalamchi A: Congenital defi ciency of the fi bula, J Bone Joint Surg 61B:133, 1979.
- Aldegheri R: Distraction osteogenesis for lengthening of the tibia in patients who have
