Introduction to Arthroscopic Shoulder Complications
Complications following diagnostic and operative shoulder arthroscopy are generally as uncommon as they are in other major joints; however, the unique anatomical constraints of the shoulder girdle present specific intraoperative challenges. The less frequent utilization of advanced shoulder arthroscopy by the average general orthopaedic surgeon may contribute to an increased incidence of complications.
Portal placement for shoulder arthroscopy is inherently more difficult than for knee arthroscopy. The shoulder provides fewer palpable bony landmarks, and the surgeon must traverse a significantly greater muscle mass. Furthermore, maneuvering the arthroscope and instruments is mechanically demanding because the glenohumeral joint and subacromial space are enveloped by thick, unyielding layers of the musculotendinous rotator cuff and dense capsular tissue. As new, advanced arthroscopic procedures are adopted by a broader range of surgeons, the steep learning curve associated with these techniques inevitably results in a transient increase in procedure failures and iatrogenic intraarticular damage.
Procedural Failure and Recurrent Instability
Currently, the most frequent complication of shoulder arthroscopy is the failure of the procedure itself, most notably recurrent instability or failure of tissue healing. Many potential arthroscopic complications can be entirely eliminated before the patient enters the operating room by thoroughly examining the root causes of historical recurrences and adhering to strict surgical indications.
Attempting to treat significant bony lesions (e.g., engaging Hill-Sachs lesions or glenoid bone loss exceeding 20%) and severe capsular hyperlaxity with standard arthroscopic soft-tissue techniques will almost certainly result in a high failure rate. However, in appropriately selected patients in whom an anatomic repair is achieved utilizing meticulous arthroscopic technique, the survivorship and chance of recurrent instability are highly comparable to traditional open techniques.
Conversely, iatrogenic overconstraint of the joint—leading to severe postoperative stiffness and secondary osteoarthritis—can usually be avoided by carefully evaluating the patient's baseline laxity and choosing appropriate candidates for interval closures and capsular plication procedures.
Clinical Pearl: The success of arthroscopic stabilization is dictated by patient selection. Unrecognized bipolar bone loss is the primary driver of arthroscopic Bankart failure. Always quantify glenoid and humeral bone loss on preoperative advanced imaging.
Neurological and Vascular Complications
Neurological injuries during shoulder arthroscopy typically occur secondary to excessive traction or direct mechanical pressure, and the vast majority are transient neuropraxias. However, direct iatrogenic transection or crush injuries can occur if the surgeon loses spatial awareness of the regional neurovascular anatomy.
Anatomic Vulnerabilities
Care in marking anatomic landmarks and precise portal positioning is critical.
* Suprascapular Nerve: This nerve is highly vulnerable as it resides less than 2 cm medial to the superior glenoid articular surface along the scapular neck.
* Axillary Nerve: The axillary nerve can be injured during inferior capsular releases or low portal placements, although it is generally 3 cm or more from standard posterior and anterior portals.
* Subclavian Vein: The subclavian vein lies within 1 cm of the 5-o’clock (low anterior) portal. To preserve the vein and the structural integrity of the subscapularis muscle, this portal should be established primarily as a percutaneous portal specifically for anchor placement, rather than a primary working portal.
Traction-Induced Neurological Injury
Although neurovascular structures are at direct risk with anterior portal placements that stray too medial or inferior, neurological complications result far more frequently from excessive traction on the operative extremity and improper patient positioning.
Paulos and Franklin reported an approximately 30% incidence of transient paresthesias in the upper extremity following shoulder arthroscopy utilizing longitudinal traction. Klein et al. reported a 10% combined incidence of transient paresthesias and palsies. In their cadaveric studies evaluating the strain on the brachial plexus under various traction loads and arm positions, they correlated plexus strain with intraarticular visibility.
The most common historical arthroscopic shoulder position—30 degrees of forward flexion and 70 degrees of abduction—produced the highest average strain on the brachial plexus. To maximize intraarticular visibility while minimizing strain to the brachial plexus, it is recommended to place the shoulder in either:
1. 45 degrees of forward flexion and 90 degrees of abduction.
2. 45 degrees of forward flexion and 0 degrees of abduction.
Surgical Warning: We typically place the shoulder in 30 to 70 degrees of abduction with a strictly controlled 10 to 13 lbs of traction. Exceeding 15 lbs of traction significantly increases the risk of traction neuropraxia without providing a proportional increase in joint visualization.
Suprascapular Nerve Decompression: Surgical Technique
In cases where the suprascapular nerve is entrapped at the suprascapular notch, an arthroscopic decompression may be indicated. This is an advanced technique requiring precise portal placement and a thorough understanding of the superior scapular anatomy.
Step-by-Step Approach:
1. Portal Placement: Establish standard viewing portals. The primary viewing portal is typically lateral, allowing a panoramic view of the suprascapular region. All other working devices are passed through a dedicated suprascapular nerve portal.
2. Identification: Carefully dissect the soft tissue overlying the suprascapular notch to identify the transverse scapular ligament. Extreme caution must be exercised to avoid the suprascapular artery, which typically runs superior to the ligament, while the nerve runs inferior to it.
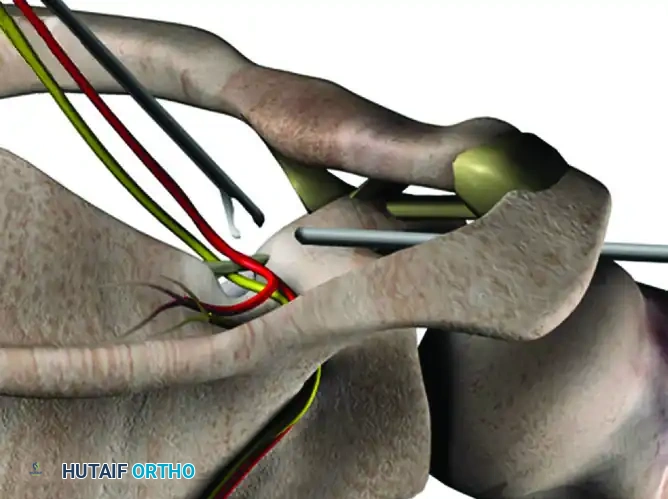
- Ligament Release: To perform the ligament release, establish a second working portal approximately 1.5 cm lateral to the suprascapular nerve portal to introduce arthroscopic scissors or a thermal ablation device.
- Transection: Carefully release the transverse scapular ligament, ensuring the underlying suprascapular nerve is protected during the transection.
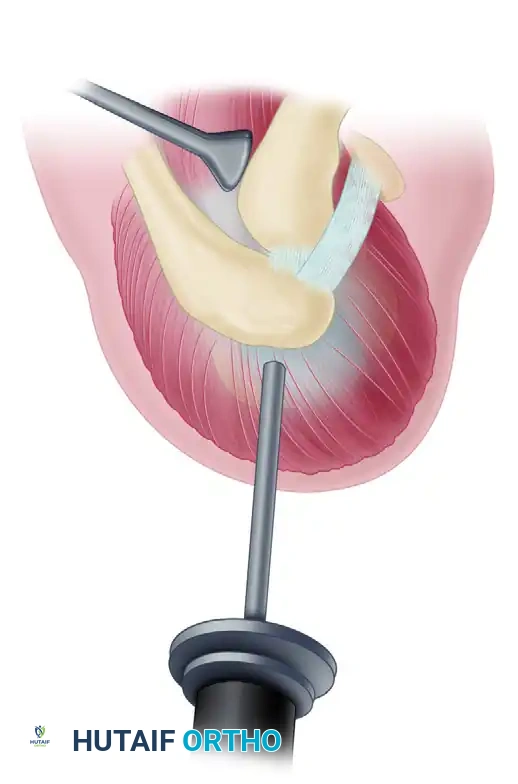
- Assessment and Notchplasty: After the release of the transverse scapular ligament, dynamically assess the decompression by gently manipulating the suprascapular nerve within the scapular notch using an arthroscopic probe. If there is residual compression of the nerve—usually resulting from bony hypertrophy or an ossified ligament within the suprascapular notch—perform a targeted notchplasty along the lateral border of the suprascapular notch utilizing an arthroscopic burr.
- Closure: Thoroughly irrigate the space and close the portals with an absorbable subcutaneous suture.
Infection and Postarthroscopic Glenohumeral Chondrolysis
Infection
Deep joint infection should be an exceedingly infrequent complication in shoulder arthroscopy due to the limited surgical exposure, the rich vascularity of the shoulder girdle, and the massive dilutional effect of the continuous irrigating solution. However, infections can and do occur, almost exclusively due to violations of sterile technique. Careful skin preparation and meticulous draping are required to minimize fluid leakage and prevent contamination through saturated drapes communicating with unsterile areas.
Chondrolysis
Postarthroscopic glenohumeral chondrolysis (PAGCL) is a severely debilitating complication characterized by the rapid, irreversible destruction of articular cartilage. The primary, evidence-based risk factor for PAGCL is the postarthroscopic intraarticular infusion of local anesthetics (specifically bupivacaine with or without epinephrine) via continuous pain pumps. This risk is exponentially magnified in young patients and when prominent intraarticular suture anchors are utilized.
Surgical Warning: The intraarticular use of continuous local anesthetic pain pumps is strictly contraindicated in shoulder arthroscopy due to the high risk of catastrophic chondrolysis.
Systemic Complications: Thromboembolism and Fluid Retention
Thromboembolic Events
While historically considered rare in upper extremity surgery, thromboembolic events have been increasingly reported in patients undergoing shoulder arthroscopy. Kuremsky et al. reported a 0.31% prevalence of venous thromboembolism (VTE) in patients following shoulder arthroscopy. Deep vein thrombosis (DVT) can occur in either the ipsilateral upper extremity or the lower extremities. Alarmingly, in their series, four of six patients with VTE developed a pulmonary embolism (PE), and three of six had identifiable preoperative risk factors. The routine use of lower extremity sequential compression devices (SCDs) intraoperatively is highly beneficial and strongly recommended, particularly if patient-specific risk factors are evident.
Fluid Retention and Extravasation
Lo and Burkhart reported systemic fluid retention and weight gain as a common, although usually transient, complication of shoulder arthroscopy. In a prospective study of 53 patients, the average weight gain caused by arthroscopic fluid extravasation was 4.2 ± 3.8 lbs (range: 0 to 14.5 lbs). While these fluid retention problems typically resolve rapidly in the postoperative period through diuresis, massive fluid extravasation can be a potential, life-threatening problem in a cardiac-compromised patient or can lead to airway compromise if fluid tracks into the neck. Strict monitoring of inflow/outflow fluid discrepancies and maintaining pump pressures below 50 mm Hg are essential safety measures.
Biomechanics of Arthroscopic Knot Tying and Suture Management
As previously stated, the failure of the procedure is the most common complication. Earlier stabilization techniques utilizing transglenoid sutures yielded unacceptable failure rates ranging from 4% to 40%. To prevent suture loosening, knot slippage, and subsequent recurrent instability, appropriate tension must be placed on the soft tissues, and meticulous, biomechanically sound intraarticular knot-tying techniques must be employed.
The arthroscopic surgeon must be highly skilled in the tying of two basic knot categories: the nonsliding knot and the sliding knot.
* Nonsliding Knots: Can be used for any application and are essential when the suture does not slide freely through the anchor eyelet or the soft tissue.
* Sliding Knots: A superior choice when the suture slides easily, allowing for excellent tissue approximation and tensioning before locking the knot.
The Revo Knot (Nonsliding)
A variety of arthroscopic knots can be utilized, but the surgeon’s knot, or Revo knot, remains the gold standard for a secure, nonsliding knot. It provides a tight suture loop with virtually no slippage when tied correctly.

Revo Knot Technique:
1. Place the knot pusher on the designated post strand.
2. Throw an underhand loop (first half-hitch) around the post, and advance the loop into the operative cavity, alternating tension on each strand until the first half-hitch is tightened down on the tissue to be opposed.
3. Withdraw the knot pusher while maintaining tension on the post strand, and throw another underhand half-hitch around the same post. Push it into the joint until the knot has seated.
4. Maintain tension on the post strand, and this time throw an overhand hitch around the same post and push it down into place. Tension the knot by past-pointing—accomplished by passing the knot pusher beyond the knot sequence and applying tension on both strands while holding the pusher in position beyond the knot.
5. The knot pusher is then switched to the second limb, which is designated as the new post.
6. Throw an underhand half-hitch around the new post and tension.
7. Switch the knot pusher back to the original post, throw an overhand hitch, and tension. Cut the tails of the suture 3 to 4 mm from the knot.
Locking-Sliding Knots
According to comprehensive biomechanical studies by Lo and Burkhart evaluating loop and knot security, locking-sliding knots such as the Duncan loop, SMC knot, and Roeder knot are highly secure only when backed up by three reversing half-hitches placed on alternating posts.
When tying arthroscopic intraarticular knots, the initial knot should be either a slipknot with two half-hitches thrown in the same direction over the same post, or a fisherman’s knot (Duncan loop). The initial slipknot is then secured by three reversing half-hitched throws over alternating posts.
Principles of Secure Knot Tying:
The post suture must be held under strict tension while the knot pusher is used to tease the slipknot down securely. The hitch suture is pushed to the side and past the point of the knot to tie the throw securely. The post changes from one suture strand to the other with each throw of the suture. This may also be achieved by alternating tension on the two suture ends; the suture tail under tension serves as the post suture.
Loutzenheiser's Rules for Arthroscopic Knots:
1. Always switch the post: This creates a locking mechanism that prevents the knot from backing out.
2. Past-point all the way down: The knot pusher must advance the suture without obstruction to ensure the knot is seated flush against the tissue.
3. Avoid excessive tension: No excessive tension should be placed on the soft tissue being sutured, as this leads to tissue necrosis and failure ("cheese-wiring"). Tension can be relieved by decreasing arm traction, internally rotating the shoulder, and placing a tension suture or grasper on the soft tissue while tying the knot.
Suture Material Considerations
The choice of suture material impacts both handling and failure rates. Absorbable monofilament Polydioxanone (PDS) maintains 40% to 50% of its tensile strength at 6 weeks but can elongate up to 30% when placed under continuous tension. Although PDS is easier for passing through tissue and tying knots, it slips and elongates much easier than modern ultra-high-molecular-weight polyethylene (UHMWPE) nonabsorbable sutures. Therefore, PDS should be reserved primarily for capsular plication techniques and as a shuttle relay to assist in passing permanent anchor sutures through the capsule, rather than as the primary load-bearing fixation.
Postoperative Protocols and Rehabilitation
Following uncomplicated arthroscopic procedures, or specific interventions such as a suprascapular nerve decompression, patients are typically discharged on the day of surgery.
For isolated nerve decompressions, patients are instructed to wear a standard sling for the first 48 to 72 hours strictly for comfort, as there is no structural repair requiring protection. Pendulum exercises and active range of motion are encouraged beginning on the first postoperative day, and patients are permitted to progress their activity without restrictions as tolerated. Routine clinical follow-up should be scheduled at 6 weeks and 6 months to ensure the resolution of preoperative neurological symptoms and the absence of delayed complications.
For structural repairs (e.g., Bankart, rotator cuff), postoperative protocols must be rigidly tailored to protect the specific biomechanical construct while preventing iatrogenic stiffness, balancing the delicate timeline of biologic healing with the restoration of functional kinematics. As surgical skill levels, implant technology, and our understanding of glenohumeral pathoanatomy have increased, the failure rates of arthroscopic shoulder procedures have successfully come into line with, and in many cases surpassed, traditional open techniques.
