Compartment Syndrome and Volkmann Contracture: Surgical Management
Key Takeaway
Acute compartment syndrome is a surgical emergency characterized by elevated intracompartmental pressure that compromises capillary perfusion, leading to irreversible muscle and nerve ischemia. If left untreated, it progresses to Volkmann ischemic contracture, a devastating sequela marked by fibrotic muscle replacement and severe functional impairment. Prompt diagnosis via clinical examination and intracompartmental pressure monitoring, followed by emergent decompressive fasciotomy, is paramount to preserving limb viability and function.
Introduction and Historical Perspective
Acute compartment syndrome (ACS) of the upper extremity represents a catastrophic orthopedic emergency wherein elevated pressure within a closed osteofascial space compromises local capillary perfusion, leading to irreversible myoneural ischemia and necrosis. When left untreated or inadequately decompressed, this ischemic cascade culminates in Volkmann ischemic contracture—a devastating, permanent deformity characterized by the fibrotic replacement of necrotic muscle and nerve tissue.
The historical evolution of our understanding of this pathology is foundational to modern orthopedic principles. In 1881, Richard von Volkmann published his classic treatise attributing rapid-onset paralytic contractures to arterial insufficiency and muscle ischemia, which he erroneously believed were exclusively caused by extrinsic compression from tight bandages. This "extrinsic pressure" theory dominated the English literature for decades.
However, subsequent clinical observations dismantled this singular view. In 1909, Thomas documented 107 paralytic contractures, noting that several occurred following severe forearm contusions devoid of fractures, splints, or constrictive dressings. By 1914, Murphy astutely proposed that internal hemorrhage and effusion could elevate pressures within the unyielding deep fascial compartments of the forearm, obstructing venous return. Finally, in 1928, Jones synthesized these observations, concluding that Volkmann contracture could arise from pressure originating internally, externally, or a combination of both.
Today, the terminology surrounding this condition—acute, subacute, chronic exertional, and crush syndrome—can be complex. However, the fundamental paradigm remains: Compartment syndrome is the acute ischemic event, and Volkmann ischemic contracture is its chronic, fibrotic sequela.
Clinical Pearl:
Never assume that the absence of a fracture precludes the development of compartment syndrome. Severe soft tissue contusions, crush injuries, and prolonged limb compression can generate sufficient intracompartmental edema to initiate the ischemic cascade.
Pathophysiology and Biomechanics
The pathophysiology of compartment syndrome is driven by the ischemic-edema cycle (as famously depicted by Eaton and Green). Any event that either decreases the volume of a compartment (e.g., tight casts, localized external pressure) or increases the fluid volume within a compartment (e.g., hemorrhage, edema, reperfusion injury) can initiate the syndrome.
As intracompartmental pressure (ICP) rises, it eventually surpasses the intravascular pressure of the local venules and capillaries. This critical threshold reduces the arteriovenous pressure gradient, causing capillary collapse and impeding local blood flow. The resulting tissue hypoxia impairs cellular metabolism, leading to failure of the sodium-potassium pump, intracellular swelling, and further localized edema. This exacerbates the intracompartmental pressure, creating a vicious, self-propagating cycle of ischemia and necrosis.
Muscle tissue is highly sensitive to ischemia. Irreversible myoneural necrosis begins within 4 to 6 hours of absolute ischemia. Nerves exhibit altered conduction within 30 minutes and irreversible damage after 12 to 24 hours of sustained severe compression.
Osteofascial Anatomy of the Upper Extremity
A profound understanding of the compartmental anatomy is non-negotiable for the operating surgeon, as incomplete fascial release is the primary cause of fasciotomy failure.
The Forearm Compartments
The forearm comprises four distinct, yet interconnected, fascial compartments:
1. Superficial Volar Compartment: Contains the pronator teres (PT), flexor carpi radialis (FCR), palmaris longus (PL), flexor digitorum superficialis (FDS), and flexor carpi ulnaris (FCU).
2. Deep Volar Compartment: Contains the flexor digitorum profundus (FDP), flexor pollicis longus (FPL), and pronator quadratus (PQ). This compartment is the most frequently and severely affected in Volkmann contracture due to its proximity to the interosseous artery and its deep, unyielding boundaries.
3. Dorsal Compartment: Contains the extensor digitorum communis (EDC), extensor digiti minimi (EDM), extensor carpi ulnaris (ECU), and the deep outcropping muscles (abductor pollicis longus, extensor pollicis brevis, extensor pollicis longus, extensor indicis proprius).
4. Mobile Wad of Henry: Contains the brachioradialis (BR), extensor carpi radialis longus (ECRL), and extensor carpi radialis brevis (ECRB).
While the volar compartments are most commonly involved, the dorsal and mobile wad compartments can be affected in isolation or concomitantly. Clinically differentiating isolated deep versus superficial volar involvement is challenging; thus, complete decompression of all volar spaces is mandatory during surgery.
The Hand Compartments
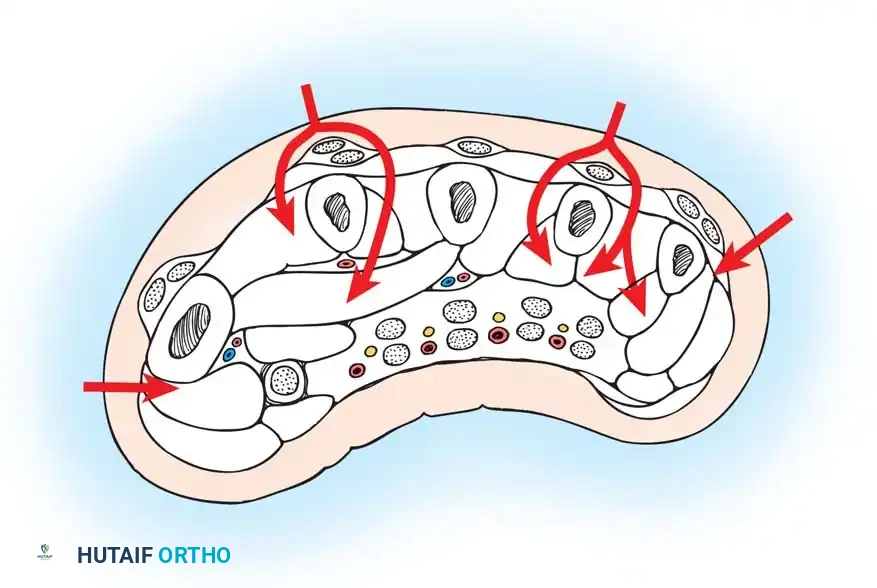
The intrinsic musculature of the hand is divided into multiple discrete compartments. Injection studies by Halpern and Mochizuki demonstrated that each interosseous muscle is enveloped by a tough, distinct fascial layer.

The hand contains the following compartments:
* Interosseous Compartments (10): Four dorsal and three volar interossei, each individually compartmentalized.
* Thenar Compartment: Contains the abductor pollicis brevis, flexor pollicis brevis, and opponens pollicis.
* Hypothenar Compartment: Contains the abductor digiti minimi, flexor digiti minimi brevis, and opponens digiti minimi.
* Adductor Compartment: Contains the adductor pollicis.
Furthermore, the neurovascular bundles of each digit are compartmentalized by Cleland's and Grayson's ligaments, rendering them vulnerable to microvascular compromise from excessive digital swelling.
Etiology
The etiology of upper extremity compartment syndrome is broad and encompasses any mechanism that alters the pressure-volume relationship within the fascia:
* Trauma: Fractures (especially supracondylar humerus and both-bone forearm fractures), crush injuries, and severe contusions.
* Bleeding Diatheses: Spontaneous or minor trauma-induced internal bleeding in patients with hemophilia or those on systemic anticoagulation.
* Iatrogenic: Intra-arterial injections of illicit drugs or sclerosing agents, extravasation of intravenous fluids, and intravenous regional anesthesia (Bier blocks) utilizing hypertonic saline.
* Vascular: Reperfusion injury following acute arterial occlusion repair.
* Environmental/Toxic: Snake envenomation, severe thermal or electrical burns (causing eschar formation that restricts expansion).
* Infectious: Severe deep space infections can elevate compartmental pressures, as noted by Schnall et al.
* Neonatal: Intrauterine malposition or strangulation of the extremity by the umbilical cord.
* Exertional: Chronic or recurrent exertional compartment syndromes, though rare, have been reported in the first dorsal interosseous muscle and the volar forearm of elite athletes.
Clinical Diagnosis
The diagnosis of acute compartment syndrome is primarily clinical, demanding a high index of suspicion. The classic "6 Ps" (Pain, Paresthesia, Pallor, Paralysis, Pulselessness, Poikilothermia) are often cited, but relying on them can be a fatal diagnostic error.
Surgical Warning:
Pulselessness and Pallor are late, end-stage signs. Capillary perfusion pressure is compromised long before arterial systolic pressure is occluded. If you wait for the radial pulse to disappear, irreversible muscle necrosis has already occurred.
Early Clinical Signs:
1. Pain out of proportion to the injury: This is the hallmark symptom. The pain is typically described as deep, unremitting, and poorly localized.
2. Pain with passive stretch: Passive extension of the fingers (stretching the ischemic volar flexors) elicits excruciating pain. This is the most sensitive early clinical finding.
3. Palpable tenseness: The affected compartment feels "wood-like" or rigidly tense.
Objective Intracompartmental Pressure (ICP) Monitoring:
In obtunded, intubated, or polytrauma patients where clinical examination is unreliable, objective ICP measurement is mandatory.
* Absolute Pressure: Historically, an absolute pressure > 30 mm Hg was an indication for fasciotomy.
* Delta Pressure ($\Delta$P): The current gold standard is the $\Delta$P, calculated as the Diastolic Blood Pressure minus the Compartment Pressure ($\Delta$P = DBP - ICP). A $\Delta$P of less than 30 mm Hg indicates inadequate capillary perfusion and is an absolute indication for emergent fasciotomy.
Surgical Management: Acute Compartment Syndrome
The definitive treatment for acute compartment syndrome is emergent decompressive fasciotomy. There is no role for conservative management once the diagnosis is confirmed.
Forearm Fasciotomy Technique
A complete forearm fasciotomy requires decompression of all four compartments. The volar approach is the most critical.
Volar Approach (Extended Henry):
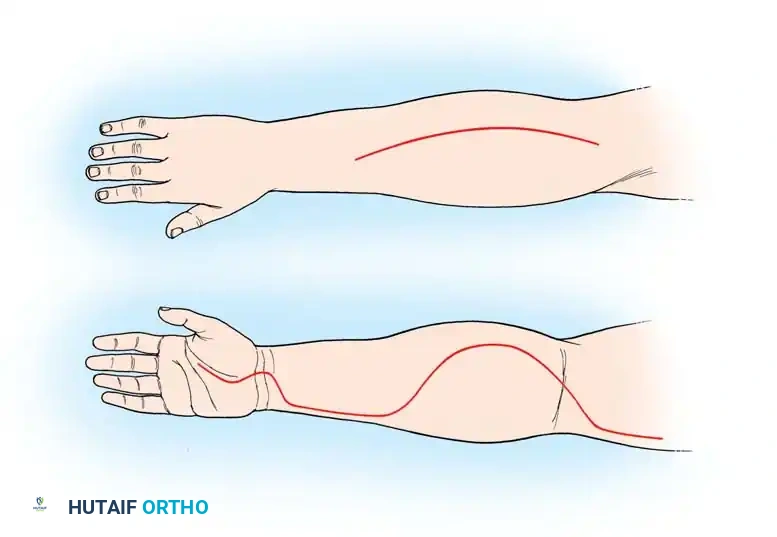
1. Incision: Begin proximal to the antecubital fossa, medial to the biceps tendon. Extend the incision distally in a curvilinear fashion over the volar forearm, crossing the wrist crease obliquely (to prevent flexion contractures), and extending into the palm along the thenar crease.
2. Superficial Decompression: Incise the superficial fascia of the forearm, exposing the superficial flexor mass (FCR, FCU, FDS).
3. Carpal Tunnel Release: Extend the fascial release distally to divide the transverse carpal ligament, decompressing the median nerve and the distal extent of the flexor tendons.
4. Deep Decompression: Retract the superficial flexors (FDS) and the neurovascular bundle to expose the deep compartment. Incise the fascia overlying the FDP and FPL.
5. Epimysiotomy: If the muscle bellies remain tight after fascial release, perform longitudinal epimysiotomies (incising the muscle sheath) to allow the muscle fibers to expand fully.
6. Mobile Wad Decompression: The mobile wad can often be decompressed through the proximal extent of the volar incision by undermining the skin radially.
Dorsal Approach:
If the dorsal compartment remains tense after volar release, a separate dorsal incision is required.
1. Incision: Make a longitudinal incision over the dorsal forearm, from the lateral epicondyle directed toward the Lister tubercle.
2. Decompression: Incise the dorsal fascia to release the EDC and deep outcropping muscles.
Hand Fasciotomy Technique
Decompression of the hand intrinsic muscles requires a meticulous approach to avoid iatrogenic injury to the extensor tendons and dorsal sensory nerves.
- Dorsal Interossei: Make two longitudinal dorsal incisions. The first is placed over the index metacarpal (decompressing the 1st and 2nd dorsal interossei). The second is placed over the ring metacarpal (decompressing the 3rd and 4th dorsal interossei).
- Volar Interossei: Through the same dorsal incisions, blunt dissection is carried volar to the transverse metacarpal ligament to decompress the volar interossei.
- Thenar and Adductor Compartments: Make a longitudinal incision along the radial border of the first metacarpal to release the thenar fascia. The adductor pollicis can be decompressed through the first dorsal incision or a separate palmar incision.
- Hypothenar Compartment: Make a longitudinal incision along the ulnar border of the fifth metacarpal.
Postoperative Protocol for Acute Fasciotomy
- Wound Management: The wounds are never closed primarily. They are dressed with non-adherent dressings or, preferably, a Negative Pressure Wound Therapy (NPWT/VAC) device set to -75 to -125 mmHg to manage exudate and reduce edema.
- Splinting: The limb is splinted in a functional position (wrist extended 20 degrees, MCP joints flexed 70 degrees, IP joints fully extended) to prevent contractures.
- Second Look: The patient is returned to the operating room at 48-72 hours for a "second look" debridement of any definitively necrotic tissue.
- Closure: Delayed primary closure is attempted once swelling subsides (usually 5-7 days). If closure is not possible without tension, split-thickness skin grafting is required.
Management of Established Volkmann Contracture
If compartment syndrome is missed or inadequately treated, the necrotic muscle is gradually replaced by dense, unyielding fibrous scar tissue, resulting in Volkmann ischemic contracture. The classic posture is elbow flexion, forearm pronation, wrist flexion, metacarpophalangeal (MCP) joint extension, and interphalangeal (IP) joint flexion.
The infarct geometry is typically ellipsoid, with the most profound necrosis occurring at the deepest aspect of the forearm—specifically the middle third of the FDP and FPL muscles, which lie directly against the interosseous membrane.
Classification (Tsuge)
- Mild: Localized to the deep flexors (usually FDP of the middle and ring fingers). Presents with flexion contractures of 2-3 digits.
- Moderate: Involves both deep and superficial flexors, often with median and ulnar nerve sensory deficits. Classic claw hand deformity.
- Severe: Involves all flexors, extensors, and profound neurological deficits. The limb is often a non-functional, insensate appendage.
Surgical Reconstruction: Muscle Sliding Operations
For established, moderate Volkmann contracture where some functional muscle excursion remains, a muscle sliding operation (Page-Scaglietti-Gosset technique) is indicated to correct the flexion deformity while preserving grip strength.
Surgical Steps for Flexor Muscle Slide:
1. Approach: An extensive volar ulnar incision is utilized, extending from the medial epicondyle down to the wrist.
2. Neurolysis: The ulnar and median nerves are meticulously identified, neurolysed, and freed from the surrounding fibrotic scar bed.
3. Origin Release: The common flexor origin is sharply detached from the medial epicondyle.
4. Distal Advancement: The flexor muscle mass (PT, FCR, FCU, FDS, FDP) is elevated subperiosteally from the ulna, interosseous membrane, and radius.
5. Correction: The entire muscle mass is allowed to slide distally (typically 2 to 4 cm). This distal migration releases the tension on the tendons, allowing the wrist and digits to extend.
6. Fixation: The muscle mass is not sutured back to bone; it is allowed to heal via secondary fibrosis in its new, lengthened position.
Surgical Pitfall:
During a muscle slide, extreme care must be taken not to devascularize the muscle mass. The primary blood supply from the ulnar and anterior interosseous arteries must be identified and protected during the subperiosteal elevation.
Salvage Procedures for Severe Contracture
In severe cases where the flexor musculature is entirely fibrotic and non-contractile, muscle sliding is futile. Management shifts to salvage operations:
* Infarct Excision: Complete excision of the fibrotic muscle mass to decompress the median and ulnar nerves.
* Tendon Transfers: Utilizing expendable, innervated muscles (e.g., BR, ECRL) transferred to the FDP and FPL tendons to restore basic grip and pinch.
* Free Functioning Muscle Transfer (FFMT): In the most severe cases, microvascular transfer of the gracilis muscle, innervated by a fascicle of the median or ulnar nerve, may be required to restore active digital flexion.
* Arthrodesis: Wrist arthrodesis may be necessary to place the hand in a functional position if dynamic reconstruction is impossible.
Conclusion
Compartment syndrome of the upper extremity is a high-stakes clinical entity that demands rapid recognition and decisive surgical intervention. The transition from acute ischemia to the irreversible fibrosis of Volkmann contracture represents a profound failure of early diagnosis. By mastering the intricate osteofascial anatomy, utilizing objective pressure monitoring when clinical signs are equivocal, and executing meticulous, wide-awake decompressive fasciotomies, the orthopedic surgeon can halt the ischemic cascade and preserve the functional integrity of the limb.
You Might Also Like