Introduction & Epidemiology
Articular cartilage, a highly specialized connective tissue, is critical for diarthrodial joint function, facilitating smooth, low-friction motion and distributing joint contact loads across the subchondral bone. Degeneration or traumatic injury to articular cartilage represents a significant clinical challenge in orthopedics, given its limited intrinsic capacity for repair. Focal chondral lesions and widespread osteoarthritis (OA) contribute substantially to morbidity, impacting patient quality of life and imposing a considerable socioeconomic burden.
Epidemiologically, symptomatic OA affects an estimated 15-20% of adults globally, with prevalence increasing with age, obesity, and prior joint trauma. Focal chondral defects, particularly in the knee, are frequently encountered in younger, active individuals, often associated with sports injuries or osteochondritis dissecans (OCD). The incidence of chondral lesions identified during knee arthroscopy ranges from 60-70%, though not all are symptomatic or require intervention. The progression of an untreated focal defect to widespread degenerative arthritis is a well-recognized pathway, underscoring the importance of early diagnosis and appropriate management strategies. Understanding the intricate structure and biomechanical properties of cartilage is paramount for effective diagnosis, treatment planning, and prognostic assessment in patients presenting with joint pain and dysfunction.
Surgical Anatomy & Biomechanics
Hyaline Cartilage Characteristics
Hyaline articular cartilage is a remarkably resilient tissue that covers the ends of articulating bones within synovial joints. Its unique composition and organization are responsible for its exceptional tribological and mechanical properties.
-
Articular Bearing Surface
- Hyaline cartilage provides a smooth, resilient bearing surface that facilitates frictionless movement between joint surfaces. It is composed primarily of chondrocytes (1-5% of tissue volume) embedded within an extensive extracellular matrix (ECM). The ECM largely consists of water (65-80% of wet weight), collagen (primarily type II, 10-30%), and proteoglycans (5-10%, mainly aggrecan).
- Chondrocytes are the sole cellular component, responsible for synthesizing and maintaining the ECM. Their metabolic activity is critical for cartilage health and is influenced by mechanical stimuli and nutrient availability.
-
The extracellular matrix is highly organized into distinct zones, each with specific collagen fiber orientations and proteoglycan content, optimized for resisting different mechanical stresses.
-
 -
 -
 -
Zonal Architecture:
- Superficial (Tangential) Zone: Outermost layer, composed of flattened chondrocytes and collagen fibers oriented parallel to the articular surface. This zone is crucial for resisting shear forces and providing a low-friction surface.
- Middle (Transitional) Zone: Larger, more spherical chondrocytes with collagen fibers arranged obliquely. This zone transitions biomechanically, offering resistance to both compressive and shear forces.
- Deep (Radial) Zone: Chondrocytes arranged in columns perpendicular to the joint surface, with thick, radially oriented collagen fibers. This zone is primarily responsible for resisting compressive loads.
- Calcified Cartilage Zone: Separated from the deep zone by the tidemark , this layer anchors the cartilage to the subchondral bone and facilitates the transfer of loads.
- Subchondral Bone: Provides structural support, vascularity (for the calcified layer and deeper cartilage), and mechanical coupling. Disruption of this interface, as seen in avascular necrosis or osteochondral lesions, significantly compromises cartilage integrity.
-
Decreases friction and distributes loads
- Articular cartilage's exceptional lubricity is critical for joint longevity. The coefficient of friction in a healthy human joint is remarkably low, ranging from 0.002 to 0.04, which is less than that of ice on ice. This ultralow friction is attributed to a complex interplay of fluid-film lubrication (hydrodynamic, elastohydrodynamic, weeping lubrication) and boundary lubrication mechanisms (e.g., lubricin, hyaluronic acid).
- Beyond lubrication, cartilage efficiently distributes compressive loads over a wide area, reducing peak stresses on the underlying subchondral bone. This viscoelastic property allows for deformation under load, followed by recovery upon unloading, a process vital for nutrient exchange.
-
Shock-absorbing cushion resists shear/compression.
- The viscoelastic nature of cartilage, conferred by its high water content and the interaction between its collagen and proteoglycan networks, enables it to absorb and dissipate energy from impact loads. Healthy articular cartilage can withstand impact loads up to 25 N/mm², protecting the subchondral bone from damaging forces. This shock-absorbing capacity is a critical function, preventing microfractures and bone remodeling under physiological stresses.
-

-
Avascular, aneural, and alymphatic
- Mature articular cartilage is unique in its lack of direct blood supply, innervation, or lymphatic drainage. This avascular nature limits its intrinsic healing potential.
- Receives nutrients and oxygen primarily from the synovial fluid via diffusion. Mechanical loading and unloading cycles (joint motion) create hydrostatic pressure gradients that pump synovial fluid into and out of the cartilage matrix, facilitating nutrient exchange (e.g., glucose, amino acids) and waste removal. The deeper, calcified cartilage and subchondral bone receive some vascular supply from the underlying bone marrow, particularly important in the context of repair mechanisms like microfracture.
Joint Capsule and Synovium
The joint capsule, composed of an outer fibrous layer and an inner synovial membrane, encloses the synovial joint. The synovial membrane is a thin, highly vascularized tissue lining the non-articular surfaces of the joint. It is characterized by synoviocytes , primarily Type A (macrophage-like) and Type B (fibroblast-like) cells. Type B synoviocytes are responsible for producing synovial fluid , a viscous, clear ultrafiltrate of plasma rich in hyaluronic acid (HA) and lubricin. Synovial fluid acts as a lubricant, provides nutrition to the avascular articular cartilage, and removes metabolic waste products. Pathological changes in the synovium (e.g., synovitis, villonodular synovitis) can significantly impair joint function and cartilage health.

Relevant Pathologies
- Focal Chondral Defects: Isolated lesions resulting from acute trauma (e.g., direct impact, shear injury) or chronic repetitive microtrauma. These can range from partial-thickness chondromalacia to full-thickness defects exposing subchondral bone. Osteochondritis dissecans (OCD) is a specific type of focal lesion involving both cartilage and subchondral bone, often due to repetitive microtrauma or ischemia, typically affecting skeletally immature individuals.
- Osteoarthritis (OA): A degenerative joint disease characterized by progressive loss of articular cartilage, subchondral bone remodeling (sclerosis, osteophyte formation, cyst formation), and synovial inflammation. OA can be primary (idiopathic) or secondary (post-traumatic, developmental, inflammatory).
-
Avascular Necrosis (AVN) / Osteonecrosis: A condition resulting from the interruption of blood supply to the subchondral bone, leading to bone cell death. The necrotic bone weakens and collapses, causing secondary damage and delamination of the overlying articular cartilage. This is a common cause of secondary OA, particularly in the hip.

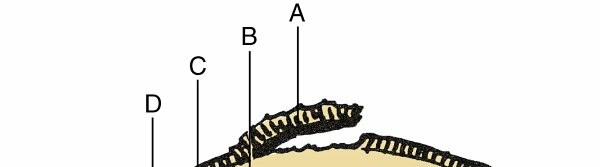
FIG. 1.25 Pathologic features of avascular necrosis. Illustration of articular cartilage (A) , necrotic bone (B) , reactive fibrous tissue (C) , hypertrophic bone (D) , and normal trabeculae (E) .
From Steinberg ME: The hip and its disorders, Philadelphia, 1991, Saunders, p 630. -
Inflammatory Arthropathies: Conditions such as rheumatoid arthritis, psoriatic arthritis, and spondyloarthropathies involve chronic synovial inflammation that can lead to enzymatic degradation and mechanical erosion of articular cartilage, often in a more diffuse pattern than focal traumatic lesions.

Indications & Contraindications
Accurate diagnosis and patient selection are paramount for successful outcomes in cartilage and joint preservation surgery. A thorough clinical evaluation combined with appropriate imaging is essential.
Diagnostic Modalities
- Clinical Examination: Assessment of pain, swelling, mechanical symptoms (locking, catching), range of motion, stability, and alignment.
-
Radiographs:
Weight-bearing anteroposterior, lateral, and specialized views (e.g., Rosenberg view for knee OA) to assess joint space narrowing, osteophytes, subchondral sclerosis, and bone alignment.
 -
Magnetic Resonance Imaging (MRI):
The gold standard for non-invasive evaluation of articular cartilage, menisci, ligaments, and subchondral bone. MRI can delineate cartilage lesion size, depth, and character, as well as identify associated pathology like bone marrow edema, osteophytes, or early avascular necrosis. Specific sequences (e.g., T2 mapping, dGEMRIC) can provide quantitative assessment of cartilage quality.
 - CT Arthrography: Useful for assessing cartilage defects in specific joints or when MRI is contraindicated, providing detailed bony morphology.
- Diagnostic Arthroscopy: The most accurate method for direct visualization and probing of articular cartilage lesions, allowing for definitive grading (e.g., ICRS classification) and assessment of associated intra-articular pathology.
Operative vs. Non-Operative Indications
The decision between operative and non-operative management is individualized, considering patient age, activity level, symptom severity, defect size/location, concurrent pathology, and overall joint health.
| Intervention Type | Indications | Contraindications |
|---|---|---|
| Non-Operative | Early osteoarthritis (OA) with mild symptoms, small stable partial-thickness defects, patients with significant medical comorbidities, lack of mechanical symptoms (locking, catching), initial management for most degenerative conditions. | Large, unstable defects; progressive symptoms despite conservative care; mechanical locking or instability; failure of conservative management; significant malalignment or ligamentous instability. |
| Operative | ||
| Chondroplasty / Debridement | Fibrillated or unstable partial-thickness cartilage flaps causing mechanical symptoms, limited full-thickness defects (<2 cm²) where stable margins are desired, often performed as an adjunctive procedure during arthroscopy for other pathology. | Full-thickness defects where regeneration is desired, widespread diffuse OA, presence of significant underlying bony pathology (e.g., severe subchondral sclerosis). |
| Bone Marrow Stimulation (Microfracture, Abrasion Arthroplasty) | ICRS Grade III-IV (full-thickness) focal chondral defects < 2-4 cm² in area, intact subchondral bone plate, absence of significant malalignment or instability, younger patients (<40-50 years). | Large defects (> 4 cm²); widespread severe OA; inflammatory arthropathy; significant subchondral bone loss or sclerosis; older patients with limited healing potential; significant malalignment or instability. |
| Osteochondral Autograft Transplantation (OATS / Mosaicplasty) | ICRS Grade III-IV focal defects (2-4 cm²) affecting weight-bearing areas, younger, active patients, viable subchondral bone, good donor site availability and health, absence of significant malalignment or instability. | Widespread OA; inflammatory arthropathy; patients >50-60 years; very large defects (> 4 cm²); poor quality or limited donor sites; significant malalignment or instability. |
| Osteochondral Allograft Transplantation (OCA) | Large ICRS Grade III-IV focal defects (> 4 cm²), salvage of failed primary repair procedures, OCD lesions unresponsive to other treatments, younger, active patients, viable subchondral bone. | Widespread severe OA; inflammatory arthropathy; active infection; significant malalignment or instability; patients with systemic diseases affecting graft incorporation. |
| Autologous Chondrocyte Implantation (ACI / MACI) | Large ICRS Grade III-IV focal defects (> 4 cm²), especially in younger, active patients, typically for defects unresponsive to microfracture, intact subchondral bone, absence of significant malalignment or instability. | Widespread severe OA; inflammatory arthropathy; patients >50-55 years; active infection; significant malalignment or instability; non-compliance with multi-stage protocol or extended rehabilitation. |
| Osteotomy (e.g., High Tibial Osteotomy - HTO, Distal Femoral Osteotomy - DFO) | Unicompartmental OA with mechanical axis malalignment (varus/valgus), often performed as a stand-alone procedure or in conjunction with cartilage repair to offload the damaged compartment, younger to middle-aged patients seeking joint preservation. | Tricompartmental OA; inflammatory arthropathy; severe obesity (BMI >35-40); significant preoperative stiffness or limited range of motion; inability to comply with prolonged non-weight-bearing. |
| Arthroscopy for Synovial/Capsular Pathology | Loose bodies causing mechanical symptoms, persistent synovitis unresponsive to conservative measures, symptomatic plicae, capsular contracture (arthrolysis), meniscal tears (often concurrent with chondral pathology). | Widespread severe OA as the sole indication; severe joint stiffness where arthrolysis may be insufficient or risky; active infection where primary debridement and drainage is required. |
General Contraindications for Cartilage Repair:
* Advanced tricompartmental osteoarthritis
* Active inflammatory arthritis or systemic rheumatologic disease
* Active local or systemic infection
* Uncorrected mechanical axis malalignment or significant ligamentous instability
* Severe obesity (BMI >35-40 kg/m²)
* Inability or unwillingness to comply with demanding post-operative rehabilitation protocols
* Co-morbidities precluding safe surgical intervention or rehabilitation (e.g., severe peripheral vascular disease, uncontrolled diabetes).
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is critical for optimizing outcomes in cartilage and joint preservation surgery.
-
Imaging Review and Assessment:
- Thorough review of all radiographs (weight-bearing, stress views), MRI, and potentially CT scans.
- Precisely define the size, depth, and location of the chondral defect(s).
- Assess the status of the subchondral bone (e.g., edema, sclerosis, cysts, AVN).
- Identify any associated pathology: meniscal tears, ligamentous instability, osteophytes, malalignment. For osteotomies, precise templating using full-length weight-bearing radiographs is essential to calculate the required correction angle and plan hardware placement.
-

-
Patient Education and Expectation Management:
- Discuss the nature of the lesion, the chosen surgical technique, potential outcomes (including the possibility of fibrocartilage formation rather than hyaline cartilage), expected recovery timeline, and the demanding nature of post-operative rehabilitation.
- Address the possibility of revision surgery or progression to arthroplasty in the long term.
-
Anesthesia Consultation:
- Discuss options for general or regional anesthesia.
- Plan for multimodal perioperative pain management.
-
Patient Positioning:
- The specific position (supine, lateral, prone) depends on the joint being addressed and the surgical approach.
- For knee procedures, a supine position on a standard operating table is typical, often with a lateral post and foot support for stability and joint flexion/extension. A tourniquet is routinely applied to facilitate a bloodless field, particularly for open procedures or complex arthroscopic interventions.
- For hip arthroscopy, specialized traction tables and perineal posts are required to create joint distraction.
- Adequate padding of bony prominences and neurovascular structures is essential to prevent iatrogenic injury.
-
Prophylaxis:
- Antibiotics: Administer intravenous prophylactic antibiotics (e.g., cefazolin) within 60 minutes of incision.
- Venous Thromboembolism (VTE): Assess patient-specific risk factors for DVT/PE and institute appropriate mechanical (e.g., sequential compression devices) and/or pharmacological prophylaxis.
Detailed Surgical Approach / Technique
The surgical management of cartilage lesions is highly variable, ranging from simple debridement to complex cell-based therapies, often performed arthroscopically or through mini-open approaches.
General Principles of Arthroscopy
Arthroscopy is the primary modality for evaluating and treating many chondral lesions.
*
Portal Placement:
Careful planning of standard and accessory portals to allow comprehensive visualization, instrumentation, and access to the lesion.
*
Fluid Management:
Use of continuous inflow/outflow to maintain joint distension and clear visualization.
*
Diagnostic Arthroscopy:
A systematic survey of the entire joint to identify the primary chondral lesion, assess its ICRS grade (0-IV), measure its dimensions, and identify any concomitant pathology (e.g., meniscal tears, ligamentous instability, loose bodies, synovitis).
Chondroplasty / Debridement
- Indications: Primarily for unstable, fibrillated, or partially detached cartilage flaps that cause mechanical symptoms but do not represent a full-thickness defect suitable for restorative procedures.
- Technique: Arthroscopic debridement using motorized shavers, burrs, or hand instruments to remove unstable cartilage and create stable, perpendicular edges around the lesion. The goal is symptom relief, not cartilage regeneration. Care must be taken to avoid damage to healthy adjacent cartilage.
Bone Marrow Stimulation (Microfracture, Abrasion Arthroplasty)
- Principle: To penetrate the subchondral bone plate, allowing mesenchymal stem cells (MSCs) and growth factors from the bone marrow to form a "superclot" within the defect. This clot then differentiates into fibrocartilaginous repair tissue.
-
Technique (Microfracture):
- Lesion Preparation: Debride all damaged or unstable cartilage to expose the underlying subchondral bone. Create stable, perpendicular walls around the defect. Remove any calcified cartilage layer, leaving a clean, firm subchondral bone bed.
- Microfracture Holes: Using an awl or K-wire, create multiple small holes (3-4 mm apart, 2-4 mm deep) perpendicular to the bone surface, penetrating the subchondral bone plate. The goal is to induce bleeding without compromising the structural integrity of the subchondral bone. Release of red marrow indicates successful penetration.
-

- Abrasion Arthroplasty: Utilizes a motorized burr to abrade the subchondral bone to a bleeding surface, similar in principle but often considered more aggressive and less controlled than microfracture.
Osteochondral Autograft Transplantation (OATS / Mosaicplasty)
- Principle: To transfer healthy hyaline cartilage and subchondral bone plugs from a non-weight-bearing donor site to the chondral defect. This is a one-stage procedure providing immediate hyaline cartilage coverage.
-
Technique:
- Lesion Preparation: Core out the recipient lesion using a specialized coring reamer, creating a perfectly cylindrical defect with perpendicular walls.
- Donor Harvest: Identify a suitable non-weight-bearing donor site (e.g., superolateral trochlea or intercondylar notch of the knee). Harvest one or more osteochondral plugs of matched diameter and depth using a precision harvesting system.
- Graft Implantation: Insert the harvested plug(s) into the recipient site(s) with the articular surface flush with the surrounding native cartilage. Impaction is usually performed by hand or with specific instruments to ensure a secure press-fit. Multiple small plugs (mosaicplasty) can be used to cover larger defects.
-

Osteochondral Allograft Transplantation (OCA)
- Principle: Similar to OATS, but uses cadaveric donor tissue, allowing for larger defects to be addressed without donor site morbidity. Grafts can be fresh (viable chondrocytes) or cryopreserved.
- Technique: Recipient site preparation can involve creating a single large cylindrical defect or a custom-fit geometric defect using specialized milling guides. The allograft is then sized, shaped, and press-fit or fixed into place.
Autologous Chondrocyte Implantation (ACI / MACI)
- Principle: A two-stage cell-based therapy aimed at regenerating hyaline-like cartilage.
- Stage 1 (Biopsy): Arthroscopic harvest of 200-300 mg of healthy articular cartilage from a non-weight-bearing area. Chondrocytes are then isolated and expanded in vitro.
-
Stage 2 (Implantation):
- ACI: An open or arthroscopic approach exposes the defect. The lesion is debrided, and a periosteal patch or synthetic collagen membrane is sewn over the defect, creating a sealed compartment. The cultured chondrocytes are then injected beneath this membrane.
- MACI (Matrix-induced ACI): The expanded chondrocytes are seeded onto a biocompatible collagen membrane or scaffold ex vivo. This cell-seeded matrix is then trimmed to fit the defect and implanted, typically with fibrin glue or sutures, avoiding the need for a watertight seal. This is technically less demanding than traditional ACI.
Osteotomy for Load Redistribution
- Principle: To correct mechanical axis malalignment in a joint (e.g., varus or valgus deformity in the knee) to shift weight-bearing load from a damaged compartment to a healthier one, thereby slowing cartilage degeneration or promoting healing of cartilage repair procedures.
-
Technique (High Tibial Osteotomy - HTO):
- Planning: Preoperative full-length weight-bearing radiographs are used to determine the exact amount of angular correction needed to achieve neutral or slight overcorrection (e.g., 62-65% Fujisawa point for valgus correction).
- Approach: Medial approach for opening wedge HTO or lateral approach for closing wedge HTO. The tibia is osteotomized distal to the tibial tuberosity, preserving the posterior cortex for stability.
- Correction: The osteotomy is opened (or closed) to the planned angle.
- Fixation: A specialized plate and screws (e.g., TomoFix plate) are used to stabilize the osteotomy. Bone graft (autograft, allograft, or synthetic) may be used in opening wedge osteotomies.
-

- Distal Femoral Osteotomy (DFO): Performed for lateral compartment OA with valgus deformity, typically via a lateral approach.
Adjunctive Procedures
Often, cartilage repair is combined with other interventions to optimize joint mechanics and stability, such as:
* Meniscal repair or transplantation.
* Ligament reconstruction (e.g., ACL reconstruction).
* Loose body removal.
* Synovectomy.


Complications & Management
Complications associated with cartilage and joint preservation surgeries can range from general surgical risks to procedure-specific failures. Early recognition and appropriate management are crucial for salvage and optimizing patient outcomes.
| Complication | Incidence (Approx.) | Salvage / Management Strategies |
|---|---|---|
| General Surgical Complications | ||
| Infection (Superficial/Deep) | <1% for arthroscopy; 1-2% for open procedures (e.g., ACI, osteotomy) | Superficial: Oral antibiotics, local wound care. Deep: Urgent surgical debridement, irrigation, cultures, targeted intravenous antibiotics; hardware removal if necessary. In severe chronic cases, a two-stage revision may be required. |
| Deep Vein Thrombosis (DVT) / Pulmonary Embolism (PE) | 0.5-2%, varies with procedure and patient risk factors | DVT: Anticoagulation (oral or subcutaneous). PE: Urgent systemic anticoagulation, respiratory support; consider inferior vena cava (IVC) filter for recurrent PE or contraindication to anticoagulation. Prolonged prophylactic anticoagulation for high-risk patients. |
| Nerve / Vascular Injury | <0.1-0.5%, site-dependent (e.g., peroneal nerve with HTO, saphenous nerve with medial knee arthrotomy) | Immediate surgical exploration and repair; neurolysis; close monitoring of perfusion and neurological status; appropriate bracing or splinting. |
| Hematoma / Seroma | 1-5% | Aspiration under sterile conditions; compression dressings; observation; surgical drainage if persistent or symptomatic. |
| Cartilage-Specific Complications | ||
| Graft Failure / Delamination (OATS, OCA, ACI) | 10-30% reported, depends on patient factors, defect size, and technique | Early failure: Revision surgery (microfracture, repeat OATS/ACI/OCA, allograft). Late failure/progression of OA: Symptomatic management, osteotomy for load redistribution, or ultimately, joint arthroplasty as salvage. |
| Donor Site Morbidity (OATS) | 5-15% (e.g., pain, fracture, avascular necrosis, graft harvest site OA) | Symptomatic management with NSAIDs, physical therapy; activity modification. Rarely, bone grafting or surgical intervention for persistent pain or structural failure. |
| Persistent Pain / Stiffness | 10-25%, often related to underlying OA progression, arthrofibrosis, or insufficient rehabilitation | Aggressive physical therapy, pain management strategies (analgesics, injections); arthroscopic lysis of adhesions (arthrolysis); manipulation under anesthesia for severe stiffness. |
| Progression of Osteoarthritis | Variable, long-term risk after any cartilage procedure | Non-operative management, activity modification, weight loss, intra-articular injections; revision surgery if focal treatable lesions develop; eventually, total joint arthroplasty. |
| Hypertrophy / Overgrowth (ACI) | 5-10%, particularly with periosteal patch ACI | Arthroscopic debridement or shaving of the hypertrophic tissue to restore smooth articular surface and improve joint mechanics. |
| Subchondral Bone Edema / Cyst Formation | Variable, often seen on post-op MRI | Symptomatic management; protected weight-bearing. Bone grafting may be considered if extensive or causing mechanical instability. |
| Osteotomy-Specific Complications | ||
| Nonunion / Malunion | 5-10% (HTO) | Revision osteotomy with bone grafting and stable internal fixation; consider conversion to arthroplasty for persistent symptoms or severe malunion. |
| Hardware Failure (Plate/Screws) | 5-10% | Hardware removal and/or revision fixation, often combined with bone grafting if nonunion is present. |
| Correction Loss | <5% | Close radiographic follow-up. Revision osteotomy if significant. |
| Patella Baja (HTO) | 5-10% | Physiotherapy for quadriceps strengthening and patellar mobilization. Surgical intervention (e.g., tibial tubercle osteotomy with distalization) if severe and symptomatic. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is as critical as the surgical technique itself for the success of cartilage repair and joint preservation procedures. Protocols vary significantly based on the specific procedure, lesion size, joint involved, and patient factors, but share common principles.
General Principles
- Protection of the Repair/Reconstruction: Initially, protection from excessive loading or stress is paramount to allow for tissue healing and integration. This often involves periods of non-weight-bearing or protected weight-bearing.
- Controlled Range of Motion (ROM): Early, controlled motion is generally encouraged to facilitate nutrient diffusion into cartilage (for avascular tissues), prevent adhesion formation, and optimize cellular metabolism. Continuous Passive Motion (CPM) machines are frequently utilized for specific procedures like ACI or microfracture.
- Gradual Weight-Bearing Progression: Weight-bearing is progressively increased according to the tissue's healing capacity and the surgical construct's stability.
- Strength Training: Progressive strengthening of surrounding musculature is vital for joint stability, load absorption, and functional recovery.
- Proprioception and Neuromuscular Control: Retraining balance and coordination is essential for restoring dynamic joint stability and preventing re-injury.
- Functional Training: Activity-specific drills are introduced gradually to prepare the patient for a safe return to activities of daily living, work, and sport.
Specific Protocols (Examples for Knee)
-
Chondroplasty / Debridement:
- Weight Bearing: Immediate full weight-bearing as tolerated.
- ROM: Immediate full ROM as tolerated.
- Rehab: Focus on pain control, swelling management, and early return to activity.
- Return to Sport: Typically 2-6 weeks, once pain-free and full function restored.
-
Microfracture / Bone Marrow Stimulation:
-
Weight Bearing:
- Non-weight bearing (NWB): 4-6 weeks for weight-bearing lesions (e.g., femoral condyle, tibia).
- Toe-touch weight bearing (TTWB): If non-weight-bearing is difficult.
- Progression: Gradual increase to partial weight-bearing (PWB) and then full weight-bearing (FWB) over the next 4-8 weeks, often in a brace.
-
ROM:
- CPM: Initiated immediately post-op for 6-8 hours/day for 6 weeks (e.g., 0-40 degrees initially, progressing to full).
- Active ROM: Gentle, pain-free active and passive ROM exercises.
- Strengthening: Isometrics initially, progressing to gentle closed-chain exercises, avoiding high-impact or deep flexion for several months.
- Return to Sport: Gradual return, typically 6-12 months, with progressive impact loading. Full return to competitive sports often after 12 months.
-
Weight Bearing:
-
Osteochondral Autograft/Allograft Transplantation (OATS/OCA):
-
Weight Bearing:
- Protected weight-bearing (PWB) or NWB: 6-8 weeks for weight-bearing lesions, often in a hinged knee brace.
- Progression: Gradual increase to FWB over 3-4 months.
- ROM: Earlier and more aggressive ROM than microfracture for some protocols, particularly with stable grafts. CPM may be used, but generally less emphasized.
- Strengthening: Similar to microfracture but potentially more aggressive in initial phases, while still protecting the graft.
- Return to Sport: Typically 6-9 months, depending on graft integration and functional recovery.
-
Weight Bearing:
-
Autologous Chondrocyte Implantation (ACI / MACI):
-
Weight Bearing:
- Strict NWB: 6-8 weeks for weight-bearing lesions.
- TTWB/PWB: Initiated after 6-8 weeks, progressing slowly to FWB over 3-6 months.
- Bracing: Hinged knee brace used for protection during weight-bearing phases.
-
ROM:
- CPM: Essential, typically 6-8 hours/day for 6-8 weeks, gradually increasing ROM (e.g., 0-30 degrees initially, progressing over weeks).
- Active ROM: Gradual, pain-free progression.
- Strengthening: Gentle isometrics, then low-load, high-repetition exercises. Avoid deep flexion, impact, and resisted open-chain exercises for several months.
- Return to Sport: Very gradual return, typically 12-18 months, with a slow progression from non-impact to impact activities.
-
Weight Bearing:
-
Osteotomy (e.g., HTO):
-
Weight Bearing:
- Protected weight-bearing (TTWB/PWB) in a brace: 6-12 weeks, until radiographic evidence of initial union.
- Progression: FWB once union is confirmed, typically 3-4 months.
- ROM: Early, protected ROM is often encouraged, but caution is advised to avoid stress on the osteotomy site.
- Strengthening: Focus on quadriceps and hamstring strengthening, initially isometric, then progressive closed-chain exercises.
- Return to Sport: Varies, but typically 6-12 months, after full union, strength, and confidence are regained.
-
Weight Bearing:
Role of Physical Therapy
A well-structured physical therapy program, guided by an experienced therapist, is indispensable for all cartilage repair and joint preservation procedures. Close communication between the surgeon and therapist is paramount to ensure adherence to the protocol and to address any emerging issues. Long-term follow-up and activity modification are often required to maintain joint health and longevity.
Summary of Key Literature / Guidelines
The field of cartilage repair and joint preservation is continuously evolving, with ongoing research refining techniques and expanding our understanding of outcomes. Several key organizations and publications provide foundational knowledge and clinical guidance.
- International Cartilage Repair Society (ICRS): The ICRS is a leading organization promoting research and education in cartilage biology and repair. Their ICRS Cartilage Repair Registry collects prospective data on various cartilage procedures, contributing to evidence-based guidelines. The ICRS Classification System for articular cartilage lesions is widely adopted, providing a standardized method for describing defect morphology and severity. ICRS guidelines emphasize patient selection, defect characteristics, and the importance of addressing concomitant pathology (e.g., meniscal tears, ligamentous instability, malalignment) for optimal outcomes.
- American Academy of Orthopaedic Surgeons (AAOS) Clinical Practice Guidelines: The AAOS periodically publishes clinical practice guidelines for conditions such as osteoarthritis of the knee, providing evidence-based recommendations for both non-operative and operative treatments. These guidelines help inform decision-making, particularly regarding the appropriate timing and selection of surgical interventions.
-
Level I Evidence for Cartilage Repair:
- Microfracture: While effective for small defects in younger patients, long-term outcomes often show degeneration of the fibrocartilaginous repair tissue, with inferior results compared to hyaline cartilage restoration techniques for larger defects.
- OATS/OCA: Demonstrated good to excellent short- to mid-term outcomes for focal lesions, particularly in younger, active individuals. Advantages include immediate hyaline cartilage coverage. However, donor site morbidity (for OATS) and challenges with allograft matching and incorporation remain considerations.
- ACI/MACI: Considered superior to microfracture for large (typically >4 cm²) focal chondral defects, especially in younger patients. Studies have shown more hyaline-like repair tissue and improved long-term functional outcomes compared to microfracture, though requiring a two-stage procedure and longer rehabilitation. MACI has largely replaced traditional ACI due to technical advantages.
- Osteotomies: High Tibial Osteotomy (HTO) and Distal Femoral Osteotomy (DFO) have demonstrated efficacy in delaying total joint arthroplasty in appropriately selected patients with unicompartmental OA and malalignment. Long-term survivorship rates for HTO vary, with 10-year survivorship ranging from 60-80%.
-
Outcomes:
- Overall, reported success rates for cartilage repair procedures are highly variable depending on the definition of success, follow-up duration, and patient/lesion characteristics. Functional scores (e.g., IKDC, Lysholm, KOOS) generally improve post-operatively.
- Long-term follow-up studies emphasize the importance of patient age, BMI, lesion size, presence of underlying OA, and adherence to rehabilitation protocols as key prognostic factors.
-
Emerging Technologies and Future Directions:
- Biologics: The use of platelet-rich plasma (PRP) and bone marrow aspirate concentrate (BMAC) as adjunctive therapies to enhance cartilage repair has gained traction, although robust Level I evidence for their routine use is still accumulating.
- Scaffold-Based Regeneration: Advancements in biomaterial science are leading to sophisticated scaffolds designed to mimic native cartilage architecture and promote more hyaline-like regeneration, often combined with stem cells or growth factors.
- Synthetic Cartilage Implants: Novel synthetic implants are being developed as potential resurfacing options for focal lesions, offering off-the-shelf availability without donor site morbidity.
- Tissue Engineering and Gene Therapy: Ongoing research explores direct chondrogenesis using genetically modified cells or growth factor delivery systems to achieve superior and more durable cartilage repair. Personalized medicine approaches, leveraging patient-specific biological responses and biomechanical profiles, represent the frontier of this field.
The comprehensive understanding of cartilage biology, biomechanics, and the evolving landscape of surgical techniques and rehabilitative strategies is essential for orthopedic surgeons dedicated to joint preservation and restoration. Continued adherence to evidence-based practices and participation in research initiatives will drive further improvements in patient care.
Clinical & Radiographic Imaging