C2 Lateral Mass Fractures: A Comprehensive Academic Review of Diagnosis & Management
Key Takeaway
C2 lateral mass fractures affect the axis's superior articular facet, caused by axial compression and lateral bending. Management is typically non-operative, but surgery can address instability. Diagnosis demands advanced imaging to evaluate vertebral artery injury risk, C1-C2 biomechanics, and links to other cervical fractures.
Introduction and Epidemiology
Cervical spine fractures represent a significant portion of all spinal trauma, with C2 fractures accounting for approximately fifteen to twenty percent of these injuries. Among C2 fractures, those involving the lateral mass are distinct entities that frequently challenge diagnostic acumen and therapeutic decision-making. These fractures, involving the superior articular facet of the axis, are typically amenable to non-operative management but can necessitate surgical intervention in cases of instability, progressive deformity, or intractable pain.
Patients often present with axial neck pain, particularly exacerbated by motion, and limited range of motion, often without overt neurologic deficits, as noted in the seminal literature. The inherent stability of the C1-C2 segment, combined with the relatively large vertebral canal at this level, contributes to the low incidence of acute spinal cord injury associated with isolated C2 lateral mass fractures. However, the presence of these fractures mandates thorough evaluation for associated injuries, given the complex biomechanical interplay of the upper cervical spine.

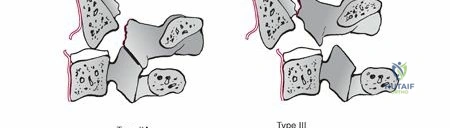
The mechanisms of injury are predominantly axial compression and lateral bending forces, frequently encountered in motor vehicle collisions, high-velocity falls, or direct blunt trauma to the cranium. A depression fracture of the C2 articular surface, signifying impaction from the occipital condyle and C1 lateral mass, is a common radiographic finding. These injuries can occur in isolation or, critically, in conjunction with other C2 fracture patterns, such as traumatic spondylolisthesis of C2 (Hangman fracture) or odontoid fractures, as well as C1 ring fractures. Early diagnosis, primarily facilitated by advanced cross-sectional imaging, is paramount for guiding appropriate management and preventing delayed complications such as post-traumatic arthrosis or basilar invagination.
Epidemiologically, upper cervical spine injuries demonstrate a bimodal distribution. High-energy trauma predominantly affects younger demographics, while low-energy falls are increasingly responsible for C2 fractures in the geriatric population due to underlying osteopenia and osteoporosis. The rising incidence of geriatric C2 fractures necessitates a nuanced approach, balancing the morbidities of prolonged immobilization against the physiologic burden of surgical stabilization.
Surgical Anatomy and Biomechanics
The axis is a complex vertebra, serving as a critical transitional segment between the atlantoaxial complex and the subaxial cervical spine. Its unique morphology, including the odontoid process, pars interarticularis, and prominent lateral masses, dictates its biomechanical function and susceptibility to specific fracture patterns.

The C2 lateral mass, specifically the superior articular facet, articulates with the inferior articular facet of C1, forming the lateral atlantoaxial joint. This joint, along with the median atlantoaxial joint, permits approximately fifty percent of total cervical rotation. The superior articular facet of C2 is typically convex in the sagittal plane and contributes to the highly congruent C1-C2 articulation. Inferiorly, the C2 inferior articular facet articulates with C3, forming a more typical subaxial cervical facet joint oriented at a forty-five-degree angle to the axial plane.
Key anatomical considerations for C2 lateral mass fractures include several critical neurovascular and ligamentous structures. The vertebral arteries traverse the transverse foramina of C1 and C2. Within C2, the vertebral artery typically runs anterior and lateral to the pars interarticularis, exhibiting a distinct lateral loop. Fractures extending into the transverse foramen or involving significant displacement pose a high risk of vertebral artery injury, including dissection, occlusion, or pseudoaneurysm formation. Preoperative vascular imaging is therefore an absolute necessity when surgical intervention is contemplated.
Neurovascular Relationships
The C2 dorsal root ganglion and nerve root exit posterior to the C1-C2 lateral mass complex. The C2 nerve root is a significant contributor to the greater occipital nerve, making it uniquely vulnerable to compression or iatrogenic injury during posterior approaches. Severely displaced fractures or aggressive retraction can lead to postoperative occipital neuralgia. The decision to preserve or transect the C2 nerve root during posterior fixation remains a topic of debate, though transection is often utilized to achieve optimal visualization of the C1-C2 joint space and facilitate robust bone grafting.
Osteoligamentous Architecture
The C2 pars interarticularis connects the superior and inferior articular facets. While lateral mass fractures primarily involve the superior facet, fracture lines frequently propagate into the pars interarticularis or the pedicle, potentially compromising the structural integrity of the axis and rendering standard screw trajectories unsafe.
The C1-C2 segment is stabilized by a robust ligamentous complex, including the transverse atlantal ligament, alar ligaments, and apical ligament. The transverse atlantal ligament is the primary restraint against anterior translation of C1 on C2. While isolated C2 lateral mass fractures generally do not compromise the transverse atlantal ligament, associated injuries must be ruled out. The tectorial membrane, a cranial extension of the posterior longitudinal ligament, provides secondary stabilization against vertical translation and flexion. Understanding the integrity of these soft tissue restraints via magnetic resonance imaging is vital for determining the overall stability of the atlantoaxial complex following a lateral mass fracture.
Indications and Contraindications
The management of C2 lateral mass fractures hinges on a comprehensive assessment of fracture morphology, joint congruity, and overall atlantoaxial stability. The primary goal of treatment is to achieve a stable, painless spine while maximizing residual range of motion.
Non-operative management remains the gold standard for isolated, minimally displaced C2 lateral mass fractures. Patients with intact ligamentous structures, minimal articular depression, and no evidence of dynamic instability on upright radiographs are excellent candidates for external immobilization. Hard cervical orthoses are typically employed for a duration of six to twelve weeks, with serial radiographic monitoring to ensure maintenance of alignment.
Operative intervention is indicated when the fracture pattern inherently destabilizes the atlantoaxial or C2-C3 junction, or when non-operative management fails. Significant depression of the superior articular facet can lead to a highly incongruent joint, predisposing the patient to severe post-traumatic osteoarthritis, intractable mechanical neck pain, and progressive coronal or sagittal plane deformity. Furthermore, fractures that occur in tandem with other unstable injuries, such as a displaced odontoid fracture or a highly comminuted C1 ring fracture (floating lateral mass), necessitate rigid internal fixation.

Contraindications to surgical intervention must be carefully weighed. Active systemic or local infection is an absolute contraindication to instrumentation. Relative contraindications include severe medical comorbidities that preclude the safe administration of general anesthesia, particularly in the frail geriatric population. In cases of profound osteoporosis, traditional screw fixation may lack sufficient purchase, necessitating modified constructs, cement augmentation, or reliance on prolonged external immobilization despite an unstable fracture pattern.
| Indication Category | Non Operative Management | Operative Management |
|---|---|---|
| Fracture Displacement | Minimally displaced (< 2mm) | Significant displacement or depression (> 2mm) |
| Joint Congruity | Maintained C1-C2 articulation | Severe articular comminution or incongruity |
| Ligamentous Status | Intact transverse and alar ligaments | Disrupted ligamentous complex (MRI confirmed) |
| Associated Injuries | Isolated C2 lateral mass fracture | Concomitant unstable C1, Odontoid, or Hangman fractures |
| Clinical Presentation | Pain controlled, neurologically intact | Progressive deformity, intractable pain, neurologic deficit |
| Patient Factors | High surgical risk, poor bone quality | Medically optimized, adequate bone stock for fixation |
Pre Operative Planning and Patient Positioning
Meticulous preoperative planning is the cornerstone of successful upper cervical spine surgery. Standard anteroposterior, lateral, and open-mouth odontoid radiographs provide the initial assessment. However, high-resolution computed tomography with sagittal and coronal reconstructions is mandatory. Computed tomography allows for precise characterization of the fracture lines, assessment of articular depression, and measurement of the bony corridors available for screw placement.
Computed tomography angiography is critical for evaluating the vertebral arteries. The surgeon must identify the dominance of the vertebral arteries, their exact course through the C2 transverse foramen, and any anomalous anatomy, such as a medialized vertebral artery or a high-riding vertebral artery. If the vertebral artery is dominant on the side of the planned surgical approach, or if the fracture extends into the transverse foramen, alternative fixation strategies must be considered to mitigate the risk of catastrophic vascular injury. Magnetic resonance imaging is utilized to evaluate the integrity of the transverse atlantal ligament, the tectorial membrane, and the spinal cord, particularly if the patient exhibits neurological deficits.

Patient Positioning and Setup
Surgical intervention for C2 lateral mass fractures is predominantly performed via a posterior approach. The patient is positioned prone on a radiolucent Jackson table or similar operative frame. The head is secured using a Mayfield skull clamp or Gardner-Wells tongs. Positioning the head and neck is a critical step; the neck should be in a neutral or slightly extended position to optimize the sagittal alignment of the cervical spine. Over-extension must be avoided to prevent narrowing of the spinal canal and potential cord compression.
A military tuck positioning of the chin is often employed to facilitate access to the upper cervical spine. The shoulders are gently taped down to allow for adequate lateral fluoroscopic visualization of the lower cervical spine, although this is less critical for isolated C1-C2 procedures.
Intraoperative neuromonitoring, including somatosensory evoked potentials and motor evoked potentials, is highly recommended to monitor spinal cord integrity during positioning, reduction maneuvers, and instrumentation. Once positioned, biplanar fluoroscopy or intraoperative cone-beam computed tomography is brought into the field to confirm acceptable alignment and to plan the surgical trajectories. The sterile field is prepped widely to allow for extension of the incision or harvesting of autologous bone graft if necessary.
Detailed Surgical Approach and Technique
The standard approach for posterior C1-C2 fixation is a midline longitudinal incision extending from the external occipital protuberance to the spinous process of C3 or C4, depending on the required exposure. The dissection proceeds through the subcutaneous tissue to the avascular midline raphe (ligamentum nuchae). Adherence to the strict midline plane minimizes bleeding and preserves the paraspinal musculature.
Subperiosteal dissection is performed to expose the posterior arch of C1, the spinous process and laminae of C2, and the lateral masses of both vertebrae. Dissection on the C1 arch must not extend more than one and a half centimeters laterally from the midline to avoid injury to the vertebral artery as it courses over the superior aspect of the C1 arch (the sulcus arteriosus). At the C2 level, dissection is carried laterally to expose the pars interarticularis and the medial aspect of the lateral mass. Venous bleeding from the epidural plexus surrounding the C2 nerve root is common and can be controlled with bipolar electrocautery and hemostatic agents.

Instrumentation Strategies
The choice of fixation depends heavily on the specific fracture morphology and the patient's anatomy as determined by preoperative imaging.
C1 Lateral Mass Screws:
The entry point for C1 lateral mass screws is typically at the junction of the posterior arch and the lateral mass, slightly medial to the center of the lateral mass. The trajectory is directed ten to fifteen degrees medially to direct the screw toward the anterior tubercle of C1, avoiding the vertebral canal, and slightly cephalad to avoid the C1-C2 joint space. The C2 nerve root often needs to be mobilized inferiorly or transected to achieve adequate visualization of the entry point and the C1-C2 joint.
C2 Pedicle Screws:
C2 pedicle screws offer robust biomechanical fixation. The entry point is located in the cranial and medial quadrant of the C2 lateral mass. The trajectory is approximately twenty to twenty-five degrees medial and twenty degrees cephalad, following the anatomical axis of the pedicle. This trajectory requires meticulous preoperative planning to ensure the pedicle is large enough to accommodate the screw and to avoid breaching the medial wall (spinal canal) or the lateral wall (vertebral artery foramen).
C2 Pars Screws:
In cases where a high-riding vertebral artery precludes pedicle screw placement, or if the fracture pattern involves the pedicle, C2 pars screws are a viable alternative. The entry point is slightly more cranial and medial compared to the pedicle screw. The trajectory is directed parallel to the sagittal plane and parallel to the medial border of the pars interarticularis. While biomechanically slightly inferior to pedicle screws, pars screws carry a lower risk of vertebral artery injury.
C2 Translaminar Screws:
Translaminar screws provide an excellent salvage option or primary fixation method when both pedicle and pars anatomy are compromised. The entry point is at the junction of the spinous process and the lamina on the contralateral side. The trajectory proceeds through the cancellous core of the lamina toward the base of the ipsilateral spinous process. This technique eliminates the risk to the vertebral artery but requires an intact posterior arch of C2.
Reduction and Arthrodesis
Once the screws are placed, reduction of the C2 lateral mass fracture can be attempted. This is often achieved indirectly through the application of rods and the use of compression or distraction forces across the C1-C2 construct. Direct manipulation of the depressed lateral mass fragment is rarely performed due to the risk of exacerbating bleeding or causing neurologic injury.
Following instrumentation and alignment confirmation, meticulous arthrodesis technique is paramount. The posterior aspect of the C1-C2 joint can be decorticated. The C2 nerve root is often sacrificed to allow for thorough decortication of the articular surfaces and packing of the joint space with autologous bone graft (typically harvested from the iliac crest or local bone) or allograft. Additional bone graft is placed over the decorticated posterior arches of C1 and C2. The wound is then closed in layers over a subfascial drain.
Complications and Management
Surgical management of C2 lateral mass fractures is technically demanding and carries a distinct profile of potential complications. Anticipation, early recognition, and algorithmic management are critical for optimizing patient outcomes.

Vertebral artery injury is the most dreaded intraoperative complication. It can occur during exposure, drilling, or screw insertion, particularly if the anatomy is distorted by the fracture or if a high-riding vertebral artery is present. If arterial bleeding is encountered, immediate control is required. Tamponade with hemostatic agents and bone wax is the first-line response. If bleeding is controlled, the screw can sometimes be placed to act as a plug. Postoperatively, immediate angiography is mandatory to assess for pseudoaneurysm, dissection, or occlusion, and endovascular coiling or stenting may be required.
Occipital neuralgia resulting from C2 nerve root manipulation or entrapment is a frequent postoperative complaint. While transection of the nerve root during surgery reliably prevents entrapment neuropathy, it results in permanent numbness in the occipital distribution, which most patients tolerate well. If the root is preserved, meticulous hemostasis and avoidance of excessive retraction are necessary. Postoperative neuralgia is typically managed with neuropathic pain modulators (gabapentin, pregabalin) and occasionally local nerve blocks.
| Complication | Estimated Incidence | Prevention and Salvage Strategies |
|---|---|---|
| Vertebral Artery Injury | 1% - 4% | Preoperative CTA, use of navigation, alternative trajectories (translaminar). Salvage: Tamponade, screw plugging, endovascular coiling. |
| C2 Occipital Neuralgia | 10% - 20% (if preserved) | Gentle retraction, bipolar hemostasis. Consider planned transection. Salvage: Neuropathic medications, occipital nerve blocks. |
| Dural Tear / CSF Leak | 2% - 5% | Meticulous subperiosteal dissection, avoidance of aggressive decortication near the canal. Salvage: Primary repair, dural sealants, lumbar drain. |
| Hardware Failure / Pullout | 2% - 6% | Assessment of bone density, bicortical purchase where appropriate, rigid postoperative orthosis. Salvage: Revision instrumentation, extension of fusion construct. |
| Pseudarthrosis (Nonunion) | 3% - 8% | Thorough joint decortication, generous autograft application, rigid fixation. Salvage: Revision surgery with bone grafting, optimization of metabolic factors. |
| Surgical Site Infection | 1% - 3% | Strict sterile technique, prophylactic antibiotics, minimization of dead space. Salvage: Irrigation and debridement, targeted intravenous antibiotics, hardware retention if stable. |
Hardware failure and pseudarthrosis are ongoing risks, particularly in patients with poor bone quality or those who are non-compliant with postoperative immobilization protocols. Meticulous preparation of the fusion bed and the use of robust autologous bone graft are the best preventative measures.
Post Operative Rehabilitation Protocols
The immediate postoperative phase focuses on neurologic monitoring, pain control, and early mobilization. Patients are typically monitored in an intensive care or step-down unit for the first twenty-four hours. A rigid cervical orthosis, such as a Miami J or Aspen collar, is routinely applied immediately postoperatively. The duration of orthosis wear depends on the bone quality, the rigidity of the fixation construct, and the surgeon's preference, but typically ranges from six to twelve weeks.
Early mobilization is encouraged to prevent the complications of prolonged bed rest, including deep vein thrombosis, pulmonary embolism, and atelectasis. Physical therapy is initiated on postoperative day one, focusing on safe transfers, ambulation, and activities of daily living while maintaining spinal precautions. Active range of motion of the cervical spine is strictly prohibited during the initial healing phase.
Radiographic follow-up is critical to assess the maintenance of alignment and the progression of arthrodesis. Standard anteroposterior and lateral radiographs are typically obtained at two weeks, six weeks, three months, six months, and one year postoperatively. Computed tomography may be utilized at the six-month or one-year mark to definitively confirm osseous bridging and continuous trabecular bone formation across the fusion mass.
Once clinical and radiographic evidence of fusion is established, the cervical orthosis is gradually weaned. At this stage, a supervised physical therapy program is instituted to restore cervical musculature strength and improve residual range of motion. Isometric strengthening exercises are introduced initially, followed by progressive active range of motion exercises. Patients must be counseled that some permanent loss of cervical rotation is inevitable following a C1-C2 arthrodesis, typically amounting to a fifty percent reduction in axial rotation.
Summary of Key Literature and Guidelines
The management of upper cervical spine trauma has evolved significantly over the past several decades, driven by advancements in biomechanical understanding and spinal instrumentation. The seminal work by Harms and Melcher revolutionized the surgical management of atlantoaxial instability by introducing the C1 lateral mass and C2 pedicle screw construct, which provided superior biomechanical rigidity compared to traditional posterior wiring techniques (e.g., Brooks-Jenkins or Gallie techniques).
The AOSpine Upper Cervical Injury Classification System provides a comprehensive framework for categorizing these complex fractures, aiding in standardized communication and treatment algorithms. Within this system, lateral mass fractures are evaluated based on their morphology and the integrity of the tension band mechanisms.
Guidelines published by neurosurgical and orthopedic spine societies emphasize the necessity of advanced imaging. The utilization of computed tomography angiography for the assessment of vertebral artery anatomy prior to any upper cervical instrumentation is universally recommended as a standard of care.
Literature comparing operative versus non-operative management of isolated C2 lateral mass fractures generally supports conservative care for minimally displaced injuries. Studies have demonstrated high rates of union and favorable clinical outcomes with rigid external immobilization. However, for fractures with significant articular depression or associated instability, surgical stabilization has been shown to significantly reduce the incidence of late deformity, nonunion, and post-traumatic arthrosis. The decision-making process remains highly individualized, requiring the surgeon to synthesize radiographic data, patient-specific physiologic factors, and a thorough understanding of upper cervical biomechanics to achieve optimal outcomes.
Clinical & Radiographic Imaging







